Machine Learning-Based Prediction of Heatwave-Related Hospitalizations: A Case Study in Matam, Senegal
Abstract
1. Introduction
2. Materials and Methods
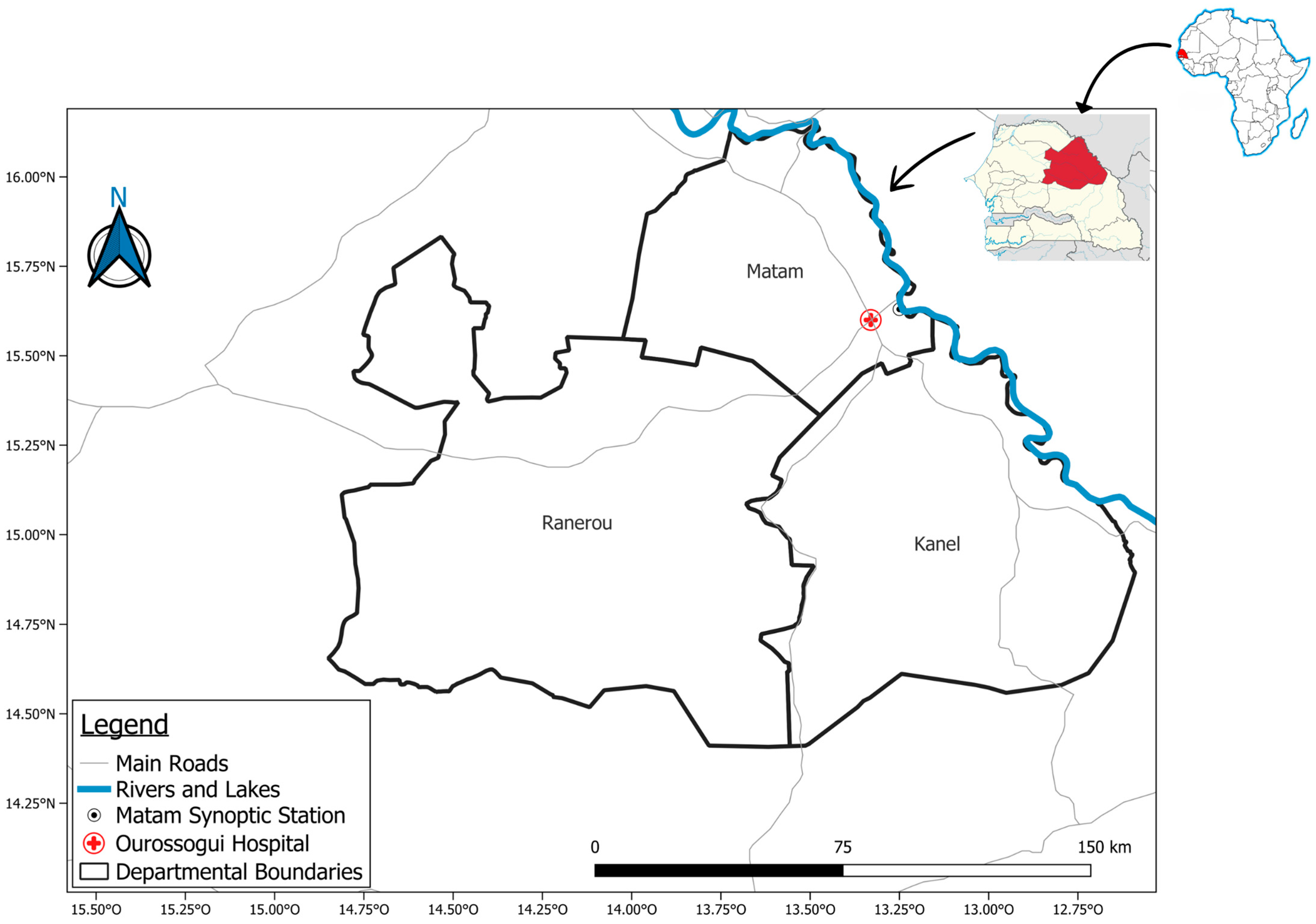
2.1. Study Area
2.2. Data
- Climate data from the Matam meteorological station, established in 1918.
- Health data, collected from Ourossogui Regional Hospital, the region’s largest healthcare facility, receives the highest number of patients and the most severe cases.
2.2.1. Climate Data
2.2.2. Health Data
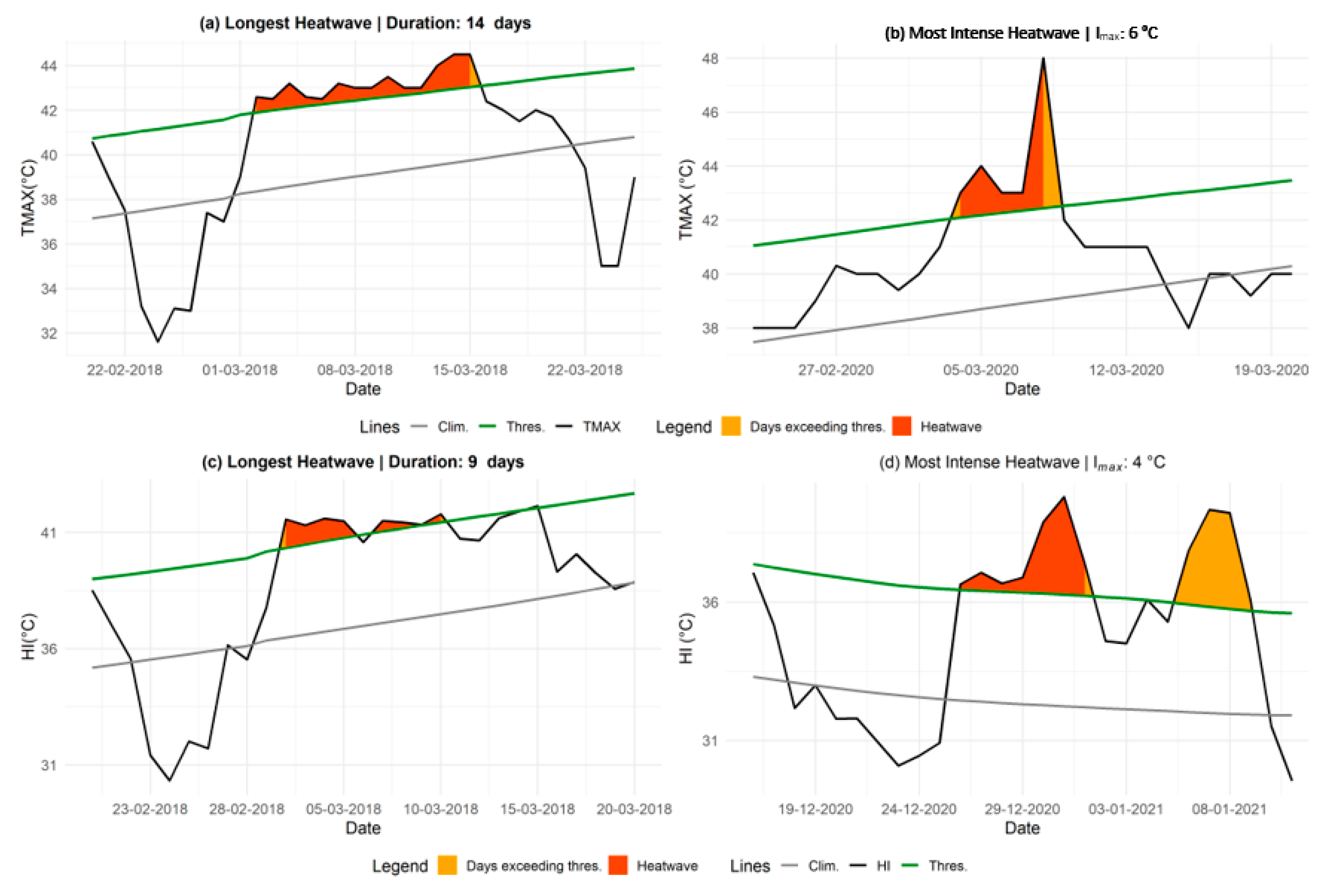
2.3. Detection and Characterization of Heatwaves
2.3.1. Identification of Heatwaves
2.3.2. Heatwave Characterization Variables
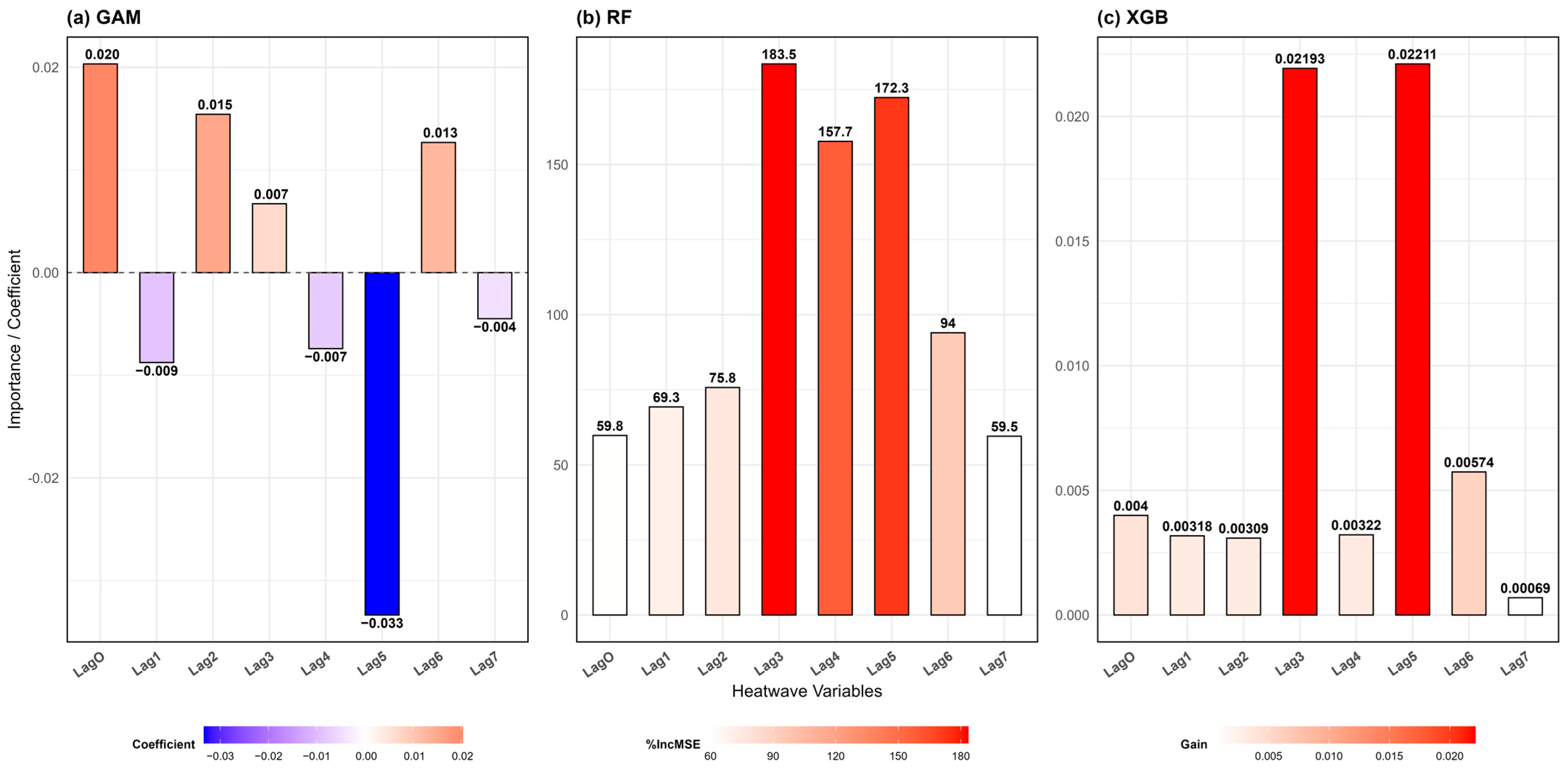
2.4. Selection of Explicit Variables
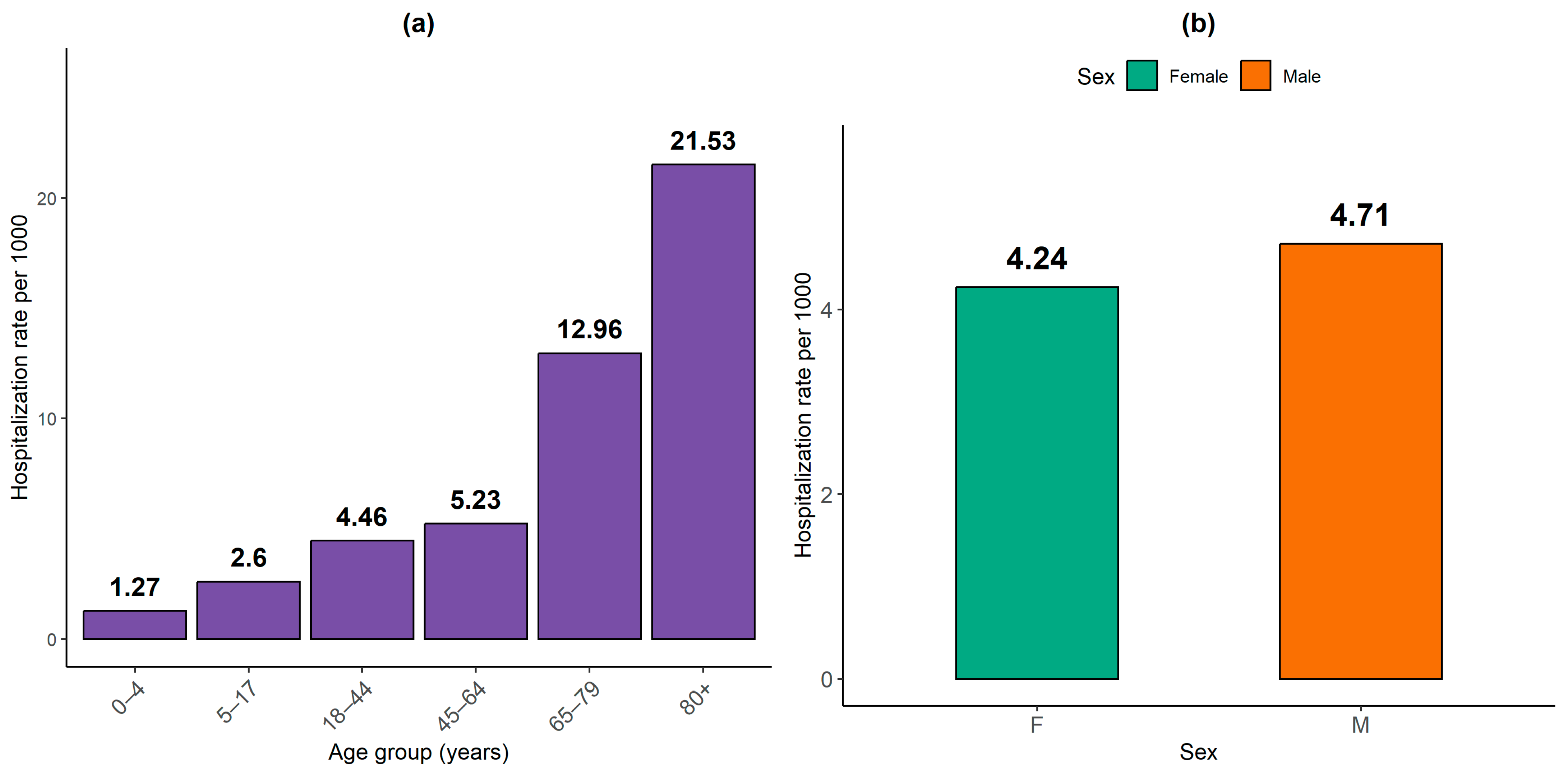
- Differences in Hospital Attendance: Although men generally visit healthcare facilities more frequently, heatwaves lead to an increase in female consultations, suggesting higher vulnerability among women;
- At-Risk Groups: Infants and dependent elderly individuals, despite benefiting from family protection, remain the most vulnerable to the effects of heatwaves.
- Number of female patients consulted per day;
- Number of infants (0–4 years) consulted per day;
- Number of fragile seniors (65–79 years) consulted per day;
- Number of very elderly and dependent seniors (80+ years) consulted per day.
2.5. Optimization and Evaluation Method of Models
- Training set (80%): Used to fit and train the models;
- Test set (20%): Used to evaluate model performance.
- Coefficient of Determination (R2): Measures the proportion of variance in the observed data explained by the model [79].
- Root Mean Square Error (RMSE): Indicates the dispersion of prediction errors, heavily penalizing large errors [80].
- Mean Absolute Error (MAE): Computes the average magnitude of absolute differences between predicted and observed values, offering a direct interpretation of average error magnitude [81].
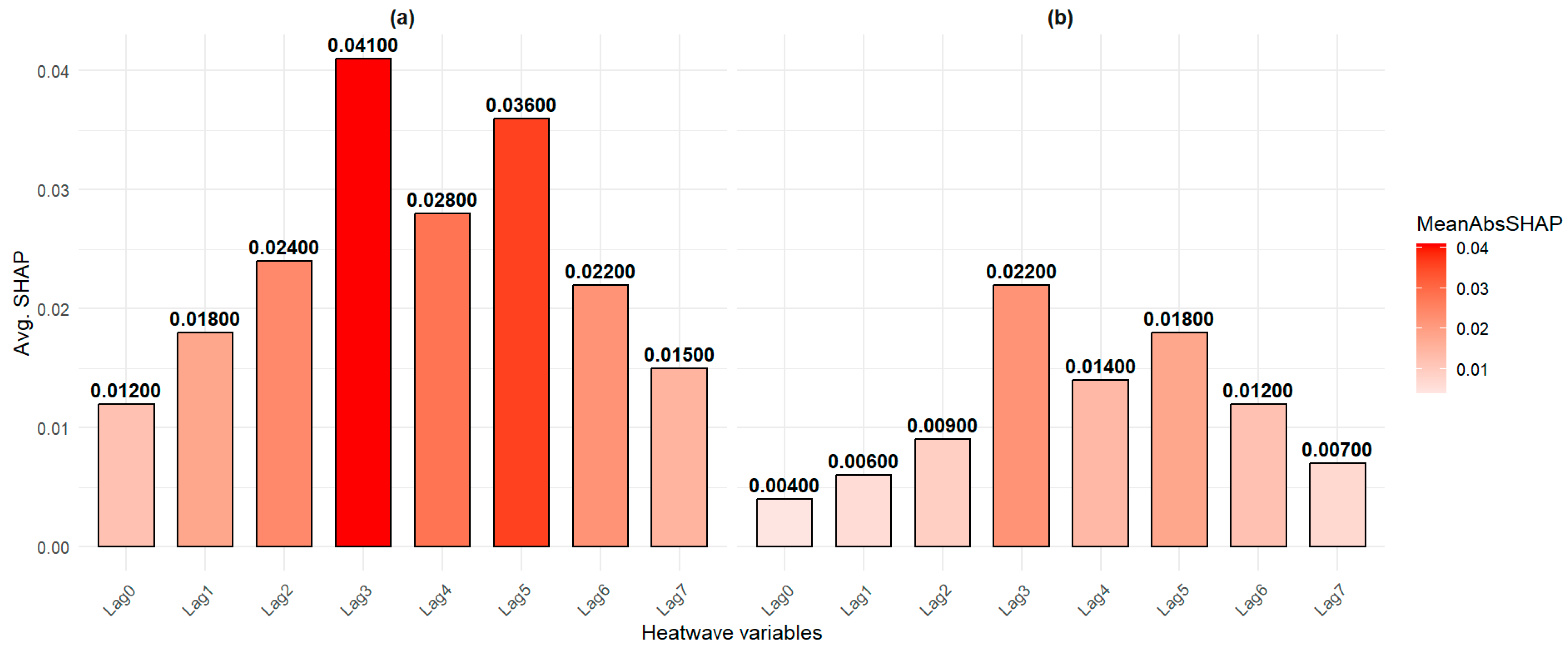
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Barry, J. The Politics of Actually Existing Unsustainability: Human Flourishing in a Climate-Changed, Carbon Constrained World; Oxford University Press: Cary, NC, USA, 2012. [Google Scholar]
- Harper, C.; Snowden, M. Environment and Society: Human Perspectives on Environmental Issues; Routledge: Oxfordshire, UK, 2017. [Google Scholar]
- IPCC Climate Change 2021: The Physical Science Basis|Climate Change 2021: The Physical Science Basis. Available online: https://www.ipcc.ch/report/ar6/wg1/ (accessed on 17 February 2025).
- WHO Heat and Health. Available online: https://www.who.int/fr/news-room/fact-sheets/detail/climate-change-heat-and-health (accessed on 17 February 2025).
- Amegah, A.K.; Rezza, G.; Jaakkola, J.J.K. Temperature-Related Morbidity and Mortality in Sub-Saharan Africa: A Systematic Review of the Empirical Evidence. Environ. Int. 2016, 91, 133–149. [Google Scholar] [CrossRef] [PubMed]
- Ayal, D.Y. Climate Change and Human Heat Stress Exposure in Sub-Saharan Africa. CABI Rev. 2021, 16, PAVSNNR202116049. [Google Scholar] [CrossRef]
- Giannini, A. Mechanisms of Climate Change in the Semiarid African Sahel: The Local View. J. Clim. 2010, 23, 743–756. [Google Scholar] [CrossRef]
- Kanu, I.A.; Ndubisi, E.J. Climate Change In Africa. Sci. Res. 2020, 13, 48–55. [Google Scholar]
- Sissoko, K.; van Keulen, H.; Verhagen, J.; Tekken, V.; Battaglini, A. Agriculture, Livelihoods and Climate Change in the West African Sahel. Reg. Environ. Change 2011, 11, 119–125. [Google Scholar] [CrossRef]
- Sylla, M.B.; Nikiema, P.M.; Gibba, P.; Kebe, I.; Klutse, N.A.B. Climate Change over West Africa: Recent Trends and Future Projections. In Adaptation to Climate Change and Variability in Rural West Africa; Yaro, J.A., Hesselberg, J., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 25–40. ISBN 978-3-319-31497-6. [Google Scholar]
- Acosta, D.; Barrow, A.; Mahamadou, I.S.; Assuncao, V.S.; Edwards, M.E.; McKune, S.L. Climate Change and Health in the Sahel: A Systematic Review. R. Soc. Open Sci. 2024, 11, 231602. [Google Scholar] [CrossRef]
- Sy, I.; Cissé, B.; Ndao, B.; Touré, M.; Aziz Diouf, A.; Adama Sarr, M.; Ndiaye, O. Heat Waves and Health Impacts in the Northern Part of Senegal: Implementation of an Early Warning System to Support Health National Adaptation Plan (HNAP). In Proceedings of the EGU General Assembly 2024, Vienna, Austria, 14–19 April 2024. [Google Scholar] [CrossRef]
- Alho, A.M.; Oliveira, A.P.; Viegas, S.; Nogueira, P. Effect of Heatwaves on Daily Hospital Admissions in Portugal, 2000–2018: An Observational Study. Lancet Planet. Health 2024, 8, e318–e326. [Google Scholar] [CrossRef]
- Clark, A.; Grineski, S.; Curtis, D.S.; Cheung, E.S.L. Identifying Groups At-Risk to Extreme Heat: Intersections of Age, Race/Ethnicity, and Socioeconomic Status. Environ. Int. 2024, 191, 108988. [Google Scholar] [CrossRef]
- Sy, I.; Cissé, B.; Ndao, B.; Touré, M.; Diouf, A.A.; Sarr, M.A.; Ndiaye, O.; Ndiaye, Y.; Badiane, D.; Lalou, R.; et al. Heat Waves and Health Risks in the Northern Part of Senegal: Analysing the Distribution of Temperature-Related Diseases and Associated Risk Factors. Environ. Sci. Pollut. Res. 2022, 29, 83365–83377. [Google Scholar] [CrossRef]
- Cheng, J.; Xu, Z.; Bambrick, H.; Prescott, V.; Wang, N.; Zhang, Y.; Su, H.; Tong, S.; Hu, W. Cardiorespiratory Effects of Heatwaves: A Systematic Review and Meta-Analysis of Global Epidemiological Evidence. Environ. Res. 2019, 177, 108610. [Google Scholar] [CrossRef]
- Hopp, S.; Dominici, F.; Bobb, J.F. Medical Diagnoses of Heat Wave-Related Hospital Admissions in Older Adults. Prev. Med. 2018, 110, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Berry, P.; Ebi, K.L.; Enright, P. Developing Health System Resilience for the Climate Crisis. In Handbook of Health System Resilience; Edward Elgar Publishing: Cheltenham, UK, 2024; pp. 307–328. [Google Scholar]
- Muhammad, A.; Qureshi, A.Z.; Farhan, M.; Oduoye, M.O.; Shehzad, F.; Imran, M. Emergency Trauma Care: Pakistan’s Preparedness amidst the Growing Impact of Rapid Climate Change. Int. J. Surg. 2024, 110, 2532–2534. [Google Scholar] [CrossRef] [PubMed]
- Iriti, M.; Vitalini, S. Climate Change, Natural Disasters, Armed Conflicts and Migrations at the Crossroads between Food and Nutrition Insecurity and Undernourishment. Funct. Food Sci. 2025, 5, 1–5. [Google Scholar] [CrossRef]
- Kurebwa, J.; Kurebwa, N.Y. Climate Change and Health Resilient Systems: Strengthening Public Health in a Changing World. In Managing the Health Risks of Climate Change; IGI Global Scientific Publishing: Hershey, PA, USA, 2025; pp. 33–56. [Google Scholar]
- Thiaw, W.M.; Bekele, E.; Diouf, S.N.; Dewitt, D.G.; Ndiaye, O.; Ndiaye, M.K.N.; Ndiaye, P.N.; Diene, N.; Diouf, M.; Diaw, A.; et al. Toward Experimental Heat–Health Early Warning in Africa. Bull. Am. Meteorol. Soc. 2022, 103, E1843–E1860. [Google Scholar] [CrossRef]
- D’Ippoliti, D.; Michelozzi, P.; Marino, C.; de’Donato, F.; Menne, B.; Katsouyanni, K.; Kirchmayer, U.; Analitis, A.; Medina-Ramón, M.; Paldy, A.; et al. The Impact of Heat Waves on Mortality in 9 European Cities: Results from the EuroHEAT Project. Environ. Health 2010, 9, 37. [Google Scholar] [CrossRef]
- Anderson, B.G.; Bell, M.L. Weather-Related Mortality: How Heat, Cold, and Heat Waves Affect Mortality in the United States. Epidemiology 2009, 20, 205–213. [Google Scholar] [CrossRef]
- Gasparrini, A.; Guo, Y.; Hashizume, M.; Lavigne, E.; Zanobetti, A.; Schwartz, J.; Tobias, A.; Tong, S.; Rocklöv, J.; Forsberg, B.; et al. Mortality Risk Attributable to High and Low Ambient Temperature: A Multicountry Observational Study. Lancet 2015, 386, 369–375. [Google Scholar] [CrossRef]
- Nhamo, G.; Chapungu, L.; Mutanda, G.W. Trends and Impacts of Climate-Induced Extreme Weather Events in South Africa (1920–2023). Environ. Dev. 2025, 55, 101183. [Google Scholar] [CrossRef]
- Gamble, J.L.; Hurley, B.J.; Schultz, P.A.; Jaglom, W.S.; Krishnan, N.; Harris, M. Climate Change and Older Americans: State of the Science. Environ. Health Perspect. 2013, 121, 15–22. [Google Scholar] [CrossRef]
- Li, M.; Gu, S.; Bi, P.; Yang, J.; Liu, Q. Heat Waves and Morbidity: Current Knowledge and Further Direction-A Comprehensive Literature Review. Int. J. Environ. Res. Public Health 2015, 12, 5256–5283. [Google Scholar] [CrossRef]
- Narocki, C. Heatwaves as an Occupational Hazard: The Impact of Heat and Heatwaves on Workers’ Health, Safety and Wellbeing and on Social Inequalities; Report, European Trade Union Institute: Brussels, Belgium, 2021. [Google Scholar]
- Mayrhuber, E.A.-S.; Dückers, M.L.A.; Wallner, P.; Arnberger, A.; Allex, B.; Wiesböck, L.; Wanka, A.; Kolland, F.; Eder, R.; Hutter, H.-P.; et al. Vulnerability to Heatwaves and Implications for Public Health Interventions—A Scoping Review. Environ. Res. 2018, 166, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, S.; Hsu, S.; Ragunathan, S.; Lindsay, J. The Impact of Climate Change Related Extreme Weather Events on People with Pre-Existing Disabilities and Chronic Conditions: A Scoping Review. Disabil. Rehabil. 2023, 45, 4338–4358. [Google Scholar] [CrossRef] [PubMed]
- Steadman, R.G. The Assessment of Sultriness. Part I: A Temperature-Humidity Index Based on Human Physiology and Clothing Science. J. Appl. Meteorol. Climatol. 1979, 18, 861–873. [Google Scholar] [CrossRef]
- Wills-Karp, M. Climate Change-Associated Health Impacts: A Way Forward. Front. Sci. 2024, 2, 1395850. [Google Scholar] [CrossRef]
- Marcucci, S.; Verhulst, S. Reimagining the Policy Cycle in the Age of Artificial Intelligence; SSRN, 2025. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5137557 (accessed on 15 June 2025).
- Masselot, P.; Chebana, F.; Campagna, C.; Lavigne, É.; Ouarda, T.B.M.J.; Gosselin, P. Machine Learning Approaches to Identify Thresholds in a Heat-Health Warning System Context. J. R. Stat. Soc. Ser. A Stat. Soc. 2021, 184, 1326–1346. [Google Scholar] [CrossRef]
- Huang, R.; Xiao, Y.; Li, S.; Li, J.; Weng, W.; Shao, Q.; Zhang, J.; Zhang, Y.; Yang, L.; Huang, C.; et al. A Novel Framework for Dynamic and Quantitative Mapping of Damage Severity Due to Compound Drought–Heatwave Impacts on Tea Plantations, Integrating Sentinel-2 and UAV Images. Comput. Electron. Agric. 2025, 228, 109688. [Google Scholar] [CrossRef]
- Galves, V.L.; Cataldi, M.; Souza, J. Developing a Multivariate System for Predicting and Mitigating the Health Effects ofHeat Waves in Niterói, Rio de Janeiro. In Proceedings of the EGU General Assembly 2024, Vienna, Austria, 14–19 April 2024. [Google Scholar] [CrossRef]
- Ke, D.; Takahashi, K.; Takakura, J.; Takara, K.; Kamranzad, B. Effects of Heatwave Features on Machine-Learning-Based Heat-Related Ambulance Calls Prediction Models in Japan. Sci. Total Environ. 2023, 873, 162283. [Google Scholar] [CrossRef]
- Aerens, N. Etude des Vulnérabilités Sanitaires aux Evénements Météorologiques Extrêmes et Elaboration de Seuils D’alerte Pour le Québec. 2022. Available online: https://www.ouranos.ca/fr/projets-publications/etude-des-vulnerabilites-sanitaires-aux-evenements-meteorologiques-extremes-et (accessed on 15 June 2025).
- Sambou, M.-J.G.; Janicot, S.; Pohl, B.; Badiane, D.; Dieng, A.L.; Gaye, A. Heat Wave Occurrences over Senegal during Spring: Regionalization and Synoptic Patterns. Int. J. Climatol. 2020, 40, 440–457. [Google Scholar] [CrossRef]
- Diouf, S.; Sambou, M.-J.G.; Deme, A.; Fall, P.; Gueye, D.; Mignot, J.; Janicot, S. Dust Content Modulation and Spring Heat Waves in Senegal (2003–2022). Atmosphere 2024, 15, 1413. [Google Scholar] [CrossRef]
- Sagna, P.; Ndiaye, O.; Diop, C.; Niang, A.D.; Sambou, P.C. Les variations récentes du climat constatées au Sénégal sont-elles en phase avec les descriptions données par les scénarios du GIEC? Pollut. Atmosphérique Clim. Santé Société 2016, 277. [Google Scholar] [CrossRef]
- Sow, M.; Gaye, D. Categorization and Multi-Criteria Analysis of Heat Wave Vulnerability in Senegal. J. Water Clim. Change 2024, 15, 5382–5396. [Google Scholar] [CrossRef]
- Bodian, A.; Diop, L.; Panthou, G.; Dacosta, H.; Deme, A.; Dezetter, A.; Ndiaye, P.M.; Diouf, I.; Vischel, T. Recent Trend in Hydroclimatic Conditions in the Senegal River Basin. Water 2020, 12, 436. [Google Scholar] [CrossRef]
- ANSD Cinquième Recensement Général de la Population et de l’Habitat (RGPH-5). 2023. Available online: https://www.ansd.sn/recensement/rgph-5-2023 (accessed on 28 February 2025).
- Schwingshackl, C.; Sillmann, J.; Vicedo-Cabrera, A.M.; Sandstad, M.; Aunan, K. Heat Stress Indicators in CMIP6: Estimating Future Trends and Exceedances of Impact-Relevant Thresholds. Earth’s Future 2021, 9, e2020EF001885. [Google Scholar] [CrossRef]
- Guigma, K.H. Heat Waves in the West African Sahel: Nature, Drivers and Predictabilty. PhD Thesis, University of Sussex, Brighton, UK, 2021. [Google Scholar]
- Perkins, S.E.; Alexander, L.V. On the Measurement of Heat Waves. J. Clim. 2013, 26, 4500–4517. [Google Scholar] [CrossRef]
- Rothfusz, L.P.; Headquarters, N.S.R. The Heat Index Equation (or, More than You Ever Wanted to Know about Heat Index). Fort Worth Tex. Natl. Ocean. Atmos. Adm. Natl. Weather Serv. Off. Meteorol. 1990, 9023, 640. [Google Scholar]
- Kaynakli, O.; Mutlu, M.; Atmaca, I.; Kilic, M. Investigation of Humidity Effects on the Thermal Comfort and Heat Balance of the Body. In Progress in Exergy, Energy, and the Environment; Dincer, I., Midilli, A., Kucuk, H., Eds.; Springer International Publishing: Cham, Switzerland, 2014; pp. 421–434. ISBN 978-3-319-04681-5. [Google Scholar]
- Zhang, J.; You, Q. Avoidable Heat Risk under Scenarios of Carbon Neutrality by Mid-Century. Sci. Total Environ. 2023, 892, 164679. [Google Scholar] [CrossRef]
- Wolkoff, P. Indoor Air Humidity, Air Quality, and Health—An Overview. Int. J. Hyg. Environ. Health 2018, 221, 376–390. [Google Scholar] [CrossRef]
- Verdaguer-Codina, J.; Martin, D.E.; Pujol-Amat, P.; Ruiz, A.; Prat, J.A. Climatic Heat Stress Studies at the Barcelona Olympic Games, 1992. Sports Med. Train. Rehabil. 1995, 6, 167–192. [Google Scholar] [CrossRef]
- Zhang, Y.; Paparoditis, E.; Politis, D.N. Simultaneous Statistical Inference for Second Order Parameters of Time Series under Weak Conditions. Ann. Stat. 2024, 52, 2375–2399. [Google Scholar] [CrossRef]
- Gasparrini, A.; Armstrong, B.; Kovats, S.; Wilkinson, P. The Effect of High Temperatures on Cause-Specific Mortality in England and Wales. Occup Environ. Med. 2012, 69, 56–61. [Google Scholar] [CrossRef]
- McCubbin, A.J.; Irwin, C.G.; Costa, R.J.S. Nourishing Physical Productivity and Performance On a Warming Planet—Challenges and Nutritional Strategies to Mitigate Exertional Heat Stress. Curr. Nutr. Rep. 2024, 13, 399–411. [Google Scholar] [CrossRef] [PubMed]
- Thongprayoon, C.; Qureshi, F.; Petnak, T.; Cheungpasitporn, W.; Chewcharat, A.; Cato, L.D.; Boonpheng, B.; Bathini, T.; Hansrivijit, P.; Vallabhajosyula, S.; et al. Impact of Acute Kidney Injury on Outcomes of Hospitalizations for Heat Stroke in the United States. Diseases 2020, 8, 28. [Google Scholar] [CrossRef] [PubMed]
- Pantavou, K.; Kotroni, V.; Kyros, G.; Lagouvardos, K. Thermal Bioclimate in Greece Based on the Universal Thermal Climate Index (UTCI) and Insights into 2021 and 2023 Heatwaves. Theor. Appl. Clim. 2024, 155, 6661–6675. [Google Scholar] [CrossRef]
- Marchand, M.; Gin, K. The Cardiovascular System in Heat Stroke. CJC Open 2022, 4, 158–163. [Google Scholar] [CrossRef]
- Deng, X.; Zhao, L.; Xiao, C.; Dai, R.; Xu, Q.; Yao, Y.; Liang, C.; Yao, L.; He, D. Heatstroke Characteristics and Meteorological Conditions in Hefei, China: Thresholds and Driving Factors. BMC Public Health 2025, 25, 352. [Google Scholar] [CrossRef]
- Rowland, S.T.; Boehme, A.K.; Rush, J.; Just, A.C.; Kioumourtzoglou, M.-A. Can Ultra Short-Term Changes in Ambient Temperature Trigger Myocardial Infarction? Environ. Int. 2020, 143, 105910. [Google Scholar] [CrossRef]
- Kuźma, Ł.; Kurasz, A.; Niwińska, M.; Zalewska-Adamiec, M.; Bachórzewska-Gajewska, H.; Dobrzycki, S. Does Climate Change Affect the Chronobiological Trends in the Occurrence of Acute Coronary Syndrome. Arch. Med. Sci. 2021, 17, 1–9. [Google Scholar] [CrossRef]
- Vermandele, F.; Sasaki, M.; Winkler, G.; Dam, H.G.; Madeira, D.; Calosi, P. When the Going Gets Tough, the Females Get Going: Sex-Specific Physiological Responses to Simultaneous Exposure to Hypoxia and Marine Heatwave Events in a Ubiquitous Copepod. Glob. Change Biol. 2024, 30, e17553. [Google Scholar] [CrossRef]
- Smoll, L.I. Sirenian Conservation Physiology: An Integrated Approach to Evaluate the Health of Dugongs and Manatees. Ph.D Thesis, The University of Queensland, Brisbane, Australia, 2024. Available online: https://espace.library.uq.edu.au/view/UQ:abb2936 (accessed on 15 June 2025).
- McGeehin, M.A.; Mirabelli, M. The Potential Impacts of Climate Variability and Change on Temperature-Related Morbidity and Mortality in the United States. Environ. Health Perspect. 2001, 109, 185–189. [Google Scholar] [CrossRef]
- Ioannou, L.G.; Mantzios, K.; Tsoutsoubi, L.; Panagiotaki, Z.; Kapnia, A.K.; Ciuha, U.; Nybo, L.; Flouris, A.D.; Mekjavic, I.B. Effect of a Simulated Heat Wave on Physiological Strain and Labour Productivity. Int. J. Environ. Res. Public Health 2021, 18, 3011. [Google Scholar] [CrossRef]
- Bobb, J.F.; Obermeyer, Z.; Wang, Y.; Dominici, F. Cause-Specific Risk of Hospital Admission Related to Extreme Heat in Older Adults. JAMA 2014, 312, 2659–2667. [Google Scholar] [CrossRef] [PubMed]
- Sawka, M.N.; Leon, L.R.; Montain, S.J.; Sonna, L.A. Integrated Physiological Mechanisms of Exercise Performance, Adaptation, and Maladaptation to Heat Stress. Compr. Physiol. 2011, 1, 1883–1928. [Google Scholar] [CrossRef] [PubMed]
- Cheshire, W.P. Thermoregulatory Disorders and Illness Related to Heat and Cold Stress. Auton. Neurosci. 2016, 196, 91–104. [Google Scholar] [CrossRef]
- Hanna, E.G.; Tait, P.W. Limitations to Thermoregulation and Acclimatization Challenge Human Adaptation to Global Warming. Int. J. Environ. Res. Public Health 2015, 12, 8034–8074. [Google Scholar] [CrossRef]
- Woodman, R.J.; Mayner, L. The Usefulness of Maximum Daily Temperatures versus Defined Heatwave Periods in Assessing the Impact of Extreme Heat on ED Admissions for Chronic Conditions. Int. J. Stat. Med. Res. 2016, 5, 81. [Google Scholar] [CrossRef]
- Vandal, G. Impact de Facteurs de Vulnérabilité à l’échelle de Secteurs Géographiques Sur La Morbidité et Mortalité Associés à La Chaleur Dans La Région de l’Estrie. PhD Thesis, Université de Sherbrooke, Sherbrooke, QC, Canada, 2022. [Google Scholar]
- Hughes, F.; Parsons, L.; Levy, J.H.; Shindell, D.; Alhanti, B.; Ohnuma, T.; Kasibhatla, P.; Montgomery, H.; Krishnamoorthy, V. Impact of Wildfire Smoke on Acute Illness. Anesthesiology 2024, 141, 779–789. [Google Scholar] [CrossRef]
- Alonso, L.; Renard, F. A Comparative Study of the Physiological and Socio-Economic Vulnerabilities to Heat Waves of the Population of the Metropolis of Lyon (France) in a Climate Change Context. Int. J. Environ. Res. Public Health 2020, 17, 1004. [Google Scholar] [CrossRef]
- Arsad, F.S.; Hod, R.; Ahmad, N.; Ismail, R.; Mohamed, N.; Baharom, M.; Osman, Y.; Radi, M.F.M.; Tangang, F. The Impact of Heatwaves on Mortality and Morbidity and the Associated Vulnerability Factors: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 16356. [Google Scholar] [CrossRef]
- Kollanus, V.; Tiittanen, P.; Lanki, T. Mortality Risk Related to Heatwaves in Finland—Factors Affecting Vulnerability. Environ. Res. 2021, 201, 111503. [Google Scholar] [CrossRef]
- Trahan, A.; Walshe, R.; Mehta, V. Extreme Heat, Gender, and Access to Preparedness Measures: An Analysis of the Heatwave Early Warning System in Ahmedabad, India. Int. J. Disaster Risk Reduct. 2023, 99, 104080. [Google Scholar] [CrossRef]
- Tibshirani, R. Regression Shrinkage and Selection via the Lasso. J. R. Stat. Soc. Ser. B Stat. Methodol. 1996, 58, 267–288. [Google Scholar] [CrossRef]
- Pearson, K.; Henrici, O.M.F.E., VII. Mathematical Contributions to the Theory of Evolution.—III. Regression, Heredity, and Panmixia. Philos. Trans. R. Soc. London. Ser. A Contain. Pap. A Math. Or Phys. Character 1997, 187, 253–318. [Google Scholar] [CrossRef]
- Gauss, C.-F. Theoria Combinationis Observationum Erroribus Minimis Obnoxiae; Henricus Dieterich: Göttingen, Germany, 1823. [Google Scholar]
- Gibbs, J.W. Elementary Principles in Statistical Mechanics: Developed with Especial Reference to the Rational Foundations of Thermodynamics; C. Scribner’s Sons: New York, NY, USA, 1902. [Google Scholar]
- Nagelkerke, N.J. A Note on a General Definition of the Coefficient of Determination. Biometrika 1991, 78, 691–692. [Google Scholar] [CrossRef]
- Willmott, C.J.; Matsuura, K. Advantages of the Mean Absolute Error (MAE) over the Root Mean Square Error (RMSE) in Assessing Average Model Performance. Clim. Res. 2005, 30, 79–82. [Google Scholar] [CrossRef]
- Willmott, C.J.; Robeson, S.M.; Matsuura, K. A Refined Index of Model Performance. Int. J. Climatol. 2012, 32, 2088–2094. [Google Scholar] [CrossRef]
- Grunkemeier, G.L.; Wu, Y. Bootstrap Resampling Methods: Something for Nothing? Ann. Thorac. Surg. 2004, 77, 1142–1144. [Google Scholar] [CrossRef]
- Martin, M.A. On Bootstrap Iteration for Coverage Correction in Confidence Intervals. J. Am. Stat. Assoc. 1990, 85, 1105–1118. [Google Scholar] [CrossRef]
- Chan, K.Y.F.; Lee, S.M.S. An Exact Iterated Bootstrap Algorithm for Small-Sample Bias Reduction. Comput. Stat. Data Anal. 2001, 36, 1–13. [Google Scholar] [CrossRef]
- Peng, Y.; Nagata, M.H. An Empirical Overview of Nonlinearity and Overfitting in Machine Learning Using COVID-19 Data. Chaos Solitons Fractals 2020, 139, 110055. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Chen, T.; Guestrin, C. XGBoost: A Scalable Tree Boosting System. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13 August 2016; ACM: Sydney, Australia; pp. 785–794. [Google Scholar]
- Hastie, T.; Tibshirani, R. Generalized Additive Models. Stat. Sci. 1986, 1, 297–310. [Google Scholar] [CrossRef]
- Wood, S.N. Stable and Efficient Multiple Smoothing Parameter Estimation for Generalized Additive Models. J. Am. Stat. Assoc. 2004, 99, 673–686. [Google Scholar] [CrossRef]
- Ibrahim, D.; Mongare, J. Modelling Claim Frequency with Spatial Effects for Accurate Insurance Premium Cost Calculations. Indones. J. Appl. Math. Stat. 2024, 1, 65–70. [Google Scholar]
- Kamoljitprapa, P.; Leelasilapasart, P. Nonlinear Models for Influenza Patients for Different Age Groups in Thailand. In Proceedings of the 2024 9th International Conference on Information and Education Innovations, Verbania Italy, 12 April 2024; ACM: Verbania, Italy, 2024; pp. 109–112. [Google Scholar]
- Lundberg, S.M.; Lee, S.-I. A Unified Approach to Interpreting Model Predictions. In Proceedings of the Advances in Neural Information Processing Systems, Long Beach, CA, USA, 4–9 December 2017; Curran Associates, Inc.: Sydney, Australia, 2017; Volume 30. [Google Scholar]
- Kenny, G.P.; Yardley, J.; Brown, C.; Sigal, R.J.; Jay, O. Heat Stress in Older Individuals and Patients with Common Chronic Diseases. CMAJ 2010, 182, 1053–1060. [Google Scholar] [CrossRef]


| Variable | Formula | Unit | Description |
|---|---|---|---|
| HW | HW = 1 if Index > 90thP for at least 3 days, otherwise 0 | - | Presence of a heatwave. Indicates whether the thermal index exceeds the threshold for at least three consecutive days. |
| D | days | Total duration of the heatwave in consecutive days. | |
| °C | Average excess temperature index above the threshold during the event. | ||
| °C | Maximum excess value reached by the temperature index during the heatwave. | ||
| °C2 | Daily variability of excess temperatures. | ||
| °C | Total heat accumulation during the event. | ||
| °C/day | Rate of temperature increase until the heatwave peak. | ||
| °C/day | Rate of temperature decrease after the heatwave peak. |
| Error Metrics | Formula | Interpretation |
|---|---|---|
| R2 | Higher values (closer to 1) indicate better model fit. | |
| RMSE | Lower = better performance | |
| MAE |
| Model | R2 | RMSE | MAE | Parameters |
|---|---|---|---|---|
| GAM | [0.33; 0.48] | [1.22; 1.5] | [0.89; 0.98] | Family: Poisson |
| RF | [0.51; 0.72] | [0.91; 1.38] | [0.74; 0.89] | Trees: 5000; Optimal number of variables (mtry): 3 |
| XGB | [0.46; 0.72] | [0.91; 1.46] | [0.74; 0.9] | Trees: 100; Max Depth: 3; Learning Rate: 0.05; Gamma: 0; Colsample bytree: 0.8; Subsample: 0.8; Min Child Weight: 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toure, M.; Sy, I.; Diouf, I.; Gueye, O.; Bekele, E.; Bhuiyan, M.A.E.; Sambou, M.J.; Ndiaye, P.N.; Thiaw, W.M.; Badiane, D.; et al. Machine Learning-Based Prediction of Heatwave-Related Hospitalizations: A Case Study in Matam, Senegal. Int. J. Environ. Res. Public Health 2025, 22, 1349. https://doi.org/10.3390/ijerph22091349
Toure M, Sy I, Diouf I, Gueye O, Bekele E, Bhuiyan MAE, Sambou MJ, Ndiaye PN, Thiaw WM, Badiane D, et al. Machine Learning-Based Prediction of Heatwave-Related Hospitalizations: A Case Study in Matam, Senegal. International Journal of Environmental Research and Public Health. 2025; 22(9):1349. https://doi.org/10.3390/ijerph22091349
Chicago/Turabian StyleToure, Mory, Ibrahima Sy, Ibrahima Diouf, Ousmane Gueye, Endalkachew Bekele, Md Abul Ehsan Bhuiyan, Marie Jeanne Sambou, Papa Ngor Ndiaye, Wassila Mamadou Thiaw, Daouda Badiane, and et al. 2025. "Machine Learning-Based Prediction of Heatwave-Related Hospitalizations: A Case Study in Matam, Senegal" International Journal of Environmental Research and Public Health 22, no. 9: 1349. https://doi.org/10.3390/ijerph22091349
APA StyleToure, M., Sy, I., Diouf, I., Gueye, O., Bekele, E., Bhuiyan, M. A. E., Sambou, M. J., Ndiaye, P. N., Thiaw, W. M., Badiane, D., Diongue-Niang, A., Gaye, A. T., Ndiaye, O., & Faye, A. (2025). Machine Learning-Based Prediction of Heatwave-Related Hospitalizations: A Case Study in Matam, Senegal. International Journal of Environmental Research and Public Health, 22(9), 1349. https://doi.org/10.3390/ijerph22091349








