Associations of Anxiety, Insomnia, and Physical Activity during the COVID-19 Pandemic
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Outcome Assessments
2.2.1. Insomnia Severity Index
2.2.2. Generalized Anxiety Disorder-7
2.2.3. International Physical Activity Questionnaire
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ng, M.A.; Naranjo, A.; Schlotzhauer, A.E.; Shoss, M.K.; Kartvelishvili, N.; Bartek, M.; Ingraham, K.; Rodriguez, A.; Schneider, S.K.; Silverlieb-Seltzer, L.; et al. Has the COVID-19 Pandemic Accelerated the Future of Work or Changed Its Course? Implications for Research and Practice. Int. J. Environ. Res. Public Health 2021, 18, 10199. [Google Scholar] [CrossRef] [PubMed]
- Ahn, T. Reduction of Working Time: Does It Lead to a Healthy Lifestyle? Health Econ. 2016, 25, 969–983. [Google Scholar] [CrossRef] [PubMed]
- McDowell, C.P.; Herring, M.P.; Lansing, J.; Brower, C.S.; Meyer, J.D. Associations Between Employment Changes and Mental Health: US Data From During the COVID-19 Pandemic. Front. Psychol. 2021, 12, 631510. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US) Committee on Sleep Medicine and Research; Colten, H.R.; Altevogt, B.M. (Eds.) Chapter 3: Extent and Health Consequences of Chronic Sleep Loss and Sleep Disorders. In Sleep Disorders and Sleep Deprivation: An Unmet Public Health Problem; National Academies Press (US): Washington, DC, USA, 2006. [Google Scholar]
- Baron, K.G.; Reid, K.J.; Zee, P.C. Exercise to improve sleep in insomnia: Exploration of the bidirectional effects. J. Clin. Sleep Med. 2013, 9, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.J.; Webb, T.L.; Martyn-St James, M.; Rowse, G.; Weich, S. Improving sleep quality leads to better mental health: A meta-analysis of randomised controlled trials. Sleep Med. Rev. 2021, 60, 101556. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Mendoza, J.; Vgontzas, A.N. Insomnia and its impact on physical and mental health. Curr. Psychiatry Rep. 2013, 15, 418. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.O.; Roth, T.; Breslau, N. The association of insomnia with anxiety disorders and depression: Exploration of the direction of risk. J. Psychiatr. Res. 2006, 40, 700–708. [Google Scholar] [CrossRef] [PubMed]
- Youngstedt, S.D.; O’Connor, P.J.; Dishman, R.K. The effects of acute exercise on sleep: A quantitative synthesis. Sleep 1997, 20, 203–214. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Generalized Anxiety Disorder. In Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar] [CrossRef]
- Xiao, H.; Zhang, Y.; Kong, D.; Li, S.; Yang, N. The Effects of Social Support on Sleep Quality of Medical Staff Treating Patients with Coronavirus Disease 2019 (COVID-19) in January and February 2020 in China. Med. Sci. Monit. 2020, 26, e923549. [Google Scholar] [CrossRef]
- COVID-19 Mental Disorders Collaborators. Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. Lancet 2021, 398, 1700–1712. [Google Scholar] [CrossRef]
- Kang, H.-J.; Bae, K.-Y.; Kim, S.-W.; Shin, H.-Y.; Shin, I.-S.; Yoon, J.-S.; Kim, J.-M. Impact of Anxiety and Depression on Physical Health Condition and Disability in an Elderly Korean Population. Psychiatry Investig. 2017, 14, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, M.; Favieri, F.; Tambelli, R.; Forte, G. The enemy who sealed the world: Effects quarantine due to the COVID-19 on sleep quality, anxiety, and psychological distress in the Italian population. Sleep Med. 2020, 75, 12–20. [Google Scholar] [CrossRef]
- Martínez-de-Quel, Ó.; Suárez-Iglesias, D.; López-Flores, M.; Pérez, C.A. Physical activity, dietary habits and sleep quality before and during COVID-19 lockdown: A longitudinal study. Appetite 2021, 158, 105019. [Google Scholar] [CrossRef] [PubMed]
- Jahrami, H.A.; Alhaj, O.A.; Humood, A.M.; Alenezi, A.F.; Fekih-Romdhane, F.; AlRasheed, M.M.; Saif, Z.Q.; Bragazzi, N.L.; Pandi-Perumal, S.R.; BaHammam, A.S.; et al. Sleep disturbances during the COVID-19 pandemic: A systematic review, meta-analysis, and meta-regression. Sleep Med. Rev. 2022, 62, 101591. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Insomnia Disorder. In Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar] [CrossRef]
- Puccinelli, P.J.; da Costa, T.S.; Seffrin, A.; de Lira, C.A.B.; Vancini, R.L.; Nikolaidis, P.T.; Knechtle, B.; Rosemann, T.; Hill, L.; Andrade, M.S. Reduced level of physical activity during COVID-19 pandemic is associated with depression and anxiety levels: An internet-based survey. BMC Public Health 2021, 21, 425. [Google Scholar] [CrossRef]
- Steeves, J.A.; Tudor-Locke, C.; Murphy, R.A.; King, G.A.; Fitzhugh, E.C.; Bassett, D.R.; Van Domelen, D.; Schuna, J.M., Jr.; Harris, T.B. Daily Physical Activity by Occupational Classification in US Adults: NHANES 2005–2006. J. Phys. Act. Health 2018, 15, 900–911. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.E.; Nicol, C.W.; Bredin, S.S. Health benefits of physical activity: The evidence. CMAJ 2006, 174, 801–809. [Google Scholar] [CrossRef] [PubMed]
- CDC. Inactivity Prevalence Maps. Available online: https://www.cdc.gov/physicalactivity/data/inactivity-prevalence-maps/index.html#overall (accessed on 5 January 2023).
- Dubinina, E.; Korostovtseva, L.S.; Rotar, O.; Amelina, V.; Boyarinova, M.; Bochkarev, M.; Shashkova, T.; Baranova, E.; Libis, R.; Duplyakov, D.; et al. Physical Activity Is Associated With Sleep Quality: Results of the ESSE-RF Epidemiological Study. Original Research. Front. Psychol. 2021, 12, 705212. [Google Scholar] [CrossRef]
- Kline, C.E. The Bidirectional Relationship between Exercise and Sleep: Implications for Exercise Adherence and Sleep Improvement. Am. J. Lifestyle Med. 2014, 8, 375–379. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Morin, C.M.; Belleville, G.; Bélanger, L.; Ivers, H. The Insomnia Severity Index: Psychometric indicators to detect insomnia cases and evaluate treatment response. Sleep 2011, 34, 601–608. [Google Scholar] [CrossRef]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.W.; Löwe, B. A Brief Measure for Assessing Generalized Anxiety Disorder: The GAD-7. Arch. Intern. Med. 2006, 166, 1092–1097. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.L.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Riebe, D.; Ehrman, J.K.; Liguori, G.; Magal, M.; American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Wolters Kluwer: Philadelphia, PA, USA, 2018. [Google Scholar]
- IPAQ Analysis. 2022. Available online: https://www.physio-pedia.com/images/c/c7/Quidelines_for_interpreting_the_IPAQ.pdf (accessed on 5 January 2023).
- Hamaker, E.L.; Kuiper, R.M.; Grasman, R.P. A critique of the cross-lagged panel model. Psychol. Methods 2015, 20, 102–116. [Google Scholar] [CrossRef] [PubMed]
- Allison, P.D.; Williams, R.; Moral-Benito, E. Maximum Likelihood for Cross-lagged Panel Models with Fixed Effects. Socius 2017, 3, 2378023117710578. [Google Scholar] [CrossRef]
- Williams, R.; Allison, P.D.; Moral-Benito, E. Linear Dynamic Panel-data Estimation Using Maximum Likelihood and Structural Equation Modeling. Stata J. 2018, 18, 293–326. [Google Scholar] [CrossRef]
- Hu, L.-t.; Bentler, P.M. Fit Indices in Covariance Structure Modeling: Sensitivity to Underparameterized Model Misspecification. Psychol. Methods 1998, 3, 424–453. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; LaMonte, M.J.; Lee, I.-M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Harvey, A.G. A cognitive model of insomnia. Behav. Res. Ther. 2002, 40, 869–893. [Google Scholar] [CrossRef]
- Schuch, F.B.; Bulzing, R.A.; Meyer, J.; López-Sánchez, G.F.; Grabovac, I.; Willeit, P.; Vancampfort, D.; Caperchione, C.M.; Sadarangani, K.P.; Werneck, A.O.; et al. Moderate to vigorous physical activity and sedentary behavior changes in self-isolating adults during the COVID-19 pandemic in Brazil: A cross-sectional survey exploring correlates. Sport Sci. Health 2022, 18, 155–163. [Google Scholar] [CrossRef]
- Park, A.H.; Zhong, S.; Yang, H.; Jeong, J.; Lee, C. Impact of COVID-19 on physical activity: A rapid review. J. Glob. Health 2022, 12, 05003. [Google Scholar] [CrossRef]
- Stockwell, S.; Trott, M.; Tully, M.; Shin, J.; Barnett, Y.; Butler, L.; McDermott, D.; Schuch, F.; Smith, L. Changes in physical activity and sedentary behaviours from before to during the COVID-19 pandemic lockdown: A systematic review. BMJ Open Sport Exerc. Med. 2021, 7, e000960. [Google Scholar] [CrossRef]
- Morin, C.M.; Bjorvatn, B.; Chung, F.; Holzinger, B.; Partinen, M.; Penzel, T.; Ivers, H.; Wing, Y.K.; Chan, N.Y.; Merikanto, I.; et al. Insomnia, anxiety, and depression during the COVID-19 pandemic: An international collaborative study. Sleep Med. 2021, 87, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Kokou-Kpolou, C.K.; Megalakaki, O.; Laimou, D.; Kousouri, M. Insomnia during COVID-19 pandemic and lockdown: Prevalence, severity, and associated risk factors in French population. Psychiatry Res. 2020, 290, 113128. [Google Scholar] [CrossRef]
- Li, Y.; Qin, Q.; Sun, Q.; Sanford, L.D.; Vgontzas, A.N.; Tang, X. Insomnia and psychological reactions during the COVID-19 outbreak in China. J. Clin. Sleep Med. 2020, 16, 1417–1418. [Google Scholar] [CrossRef]
- Sharma, M.; Aggarwal, S.; Madaan, P.; Saini, L.; Bhutani, M. Impact of COVID-19 pandemic on sleep in children and adolescents: A systematic review and meta-analysis. Sleep Med. 2021, 84, 259–267. [Google Scholar] [CrossRef]
- Korman, M.; Tkachev, V.; Reis, C.; Komada, Y.; Kitamura, S.; Gubin, D.; Kumar, V.; Roenneberg, T. COVID-19-mandated social restrictions unveil the impact of social time pressure on sleep and body clock. Sci. Rep. 2020, 10, 22225. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.P., Jr.; Linton, S.K.; Withrow, D.; Casiraghi, L.; Lanza, S.M.; de la Iglesia, H.; Vetter, C.; Depner, C.M. Sleep in university students prior to and during COVID-19 Stay-at-Home orders. Curr. Biol. 2020, 30, R797–R798. [Google Scholar] [CrossRef]
- McDowell, C.P.; Dishman, R.K.; Gordon, B.R.; Herring, M.P. Physical Activity and Anxiety: A Systematic Review and Meta-analysis of Prospective Cohort Studies. Am. J. Prev. Med. 2019, 57, 545–556. [Google Scholar] [CrossRef]
- Hamilton, M.T.; Hamilton, D.G.; Zderic, T.W. Role of low energy expenditure and sitting in obesity, metabolic syndrome, type 2 diabetes, and cardiovascular disease. Diabetes 2007, 56, 2655–2667. [Google Scholar] [CrossRef]
- Ekelund, U.; Tarp, J.; Fagerland, M.W.; Johannessen, J.S.; Hansen, B.H.; Jefferis, B.J.; Whincup, P.H.; Diaz, K.M.; Hooker, S.; Howard, V.J.; et al. Joint associations of accelero-meter measured physical activity and sedentary time with all-cause mortality: A harmonised meta-analysis in more than 44 000 middle-aged and older individuals. Br. J. Sports Med. 2020, 54, 1499–1506. [Google Scholar] [CrossRef] [PubMed]
- Bourdier, P.; Simon, C.; Bessesen, D.H.; Blanc, S.; Bergouignan, A. The role of physical activity in the regulation of body weight: The overlooked contribution of light physical activity and sedentary behaviors. Obes. Rev. 2023, 24, e13528. [Google Scholar] [CrossRef] [PubMed]
- Santabárbara, J.; Lasheras, I.; Lipnicki, D.M.; Bueno-Notivol, J.; Pérez-Moreno, M.; López-Antón, R.; De la Cámara, C.; Lobo, A.; Gracia-García, P. Prevalence of anxiety in the COVID-19 pandemic: An updated meta-analysis of community-based studies. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 109, 110207. [Google Scholar] [CrossRef] [PubMed]
| Variable | Mean + SD or N (%) | |
|---|---|---|
| Age | 43.85 ± 15.85 (range, 19–87) | |
| BMI | 27.7 ± 8.1 | |
| Sex/gender | Male | 95 (47%) |
| Female | 103 (50%) | |
| Transgender Male | 3 (1.5%) | |
| Transgender Female | 3 (1.5%) | |
| Education | <12th grade | 2 (1%) |
| High school graduation or GED | 7 (3.5%) | |
| Some college or associate’s (2-year) degree | 42 (21%) | |
| Bachelor’s (4-year) degree | 75 (37%) | |
| Graduate degree or more | 75 (37%) | |
| Has COVID-19 affected your work/occupation? | Yes | 130 (64%) |
| No | 55 (27%) | |
| Household Income | <USD 10,000 | 10 (5%) |
| USD 10,000-USD 25,000 | 20 (10%) | |
| USD 26,000-USD 50,000 | 59 (29%) | |
| USD 51,000-USD 75,000 | 23 (11%) | |
| USD 76,000-USD 100,000 | 27 (13%) | |
| >USD 101,000 | 62 (30%) | |
| Marital Status | Single, never married | 58 (27%) |
| Married | 111 (54%) | |
| Separated | 4 (2%) | |
| Divorced | 21 (10%) | |
| Widowed | 4 (2%) | |
| Other | 3 (1.5%) | |
| Race/Ethnicity | Hispanic or Latino | 28 (14%) |
| Not Hispanic or Latino | 173 (85%) | |
| American Indian/Alaskan Native | 7 (3%) | |
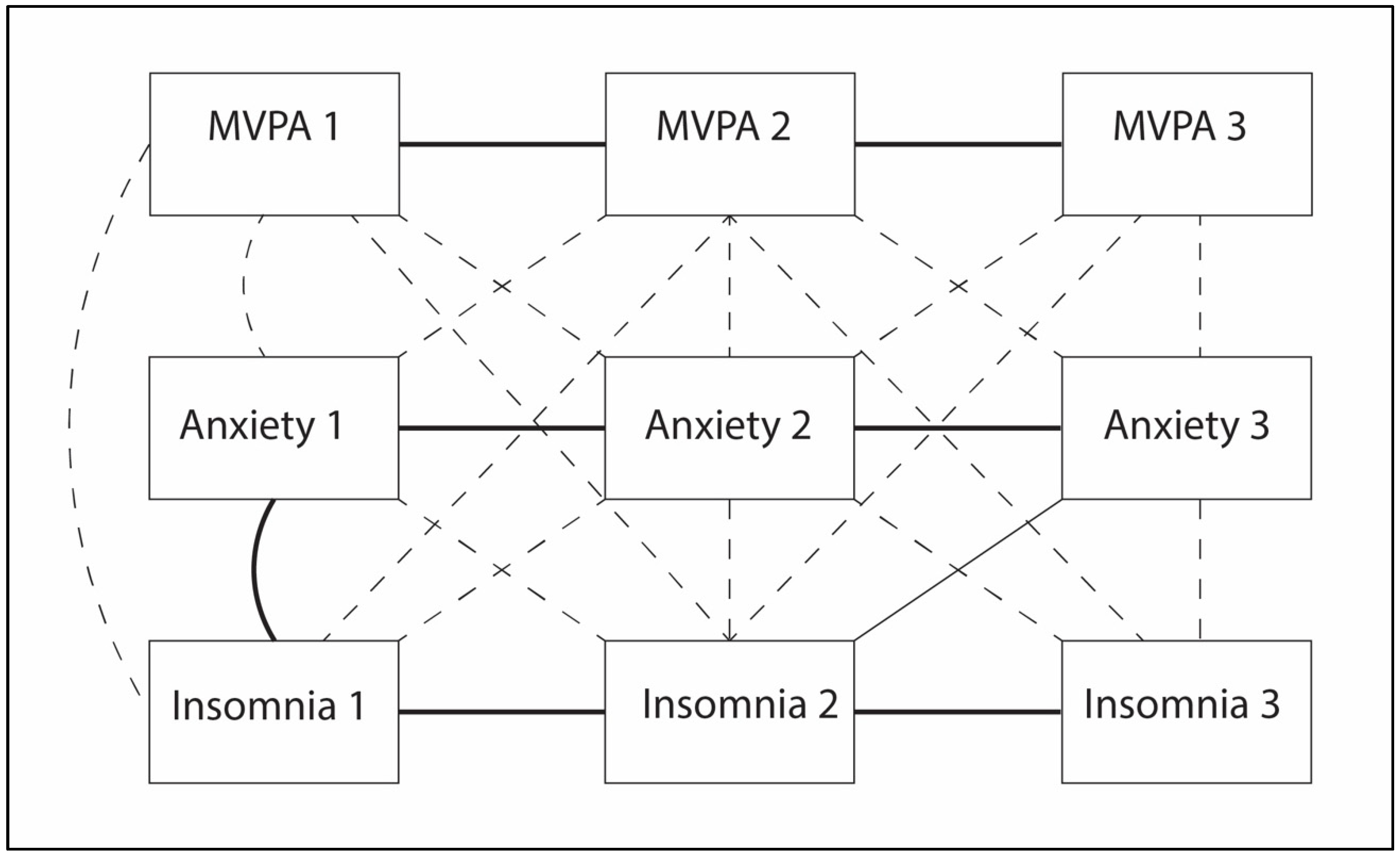
| Variable 1 | Variable 2 | β-Coefficient | 95% CI | p-Value |
|---|---|---|---|---|
| Structural | ||||
| MVPA 2 | MVPA 1 | 0.45 *** | (0.23–0.67) | <0.0001 |
| Anxiety 1 | −27.72 | (−89.78–34.33) | 0.38 | |
| Insomnia 1 | 14.47 | (−35.51–64.45) | 0.57 | |
| Anxiety 2 | MVPA 1 | −0.0005 | (−0.001–0.0001) | 0.11 |
| Anxiety 1 | 0.61 *** | (0.48–0.74) | <0.0001 | |
| Insomnia 1 | 0.05 | (−0.06–0.15) | 0.37 | |
| Insomnia 2 | MVPA 1 | −0.0003 | (−0.0009–0.0004) | 0.42 |
| Anxiety 1 | −0.06 | (−0.20–0.08) | 0.40 | |
| Insomnia 1 | 0.74 *** | (0.63–0.86) | <0.0001 | |
| MVPA 3 | MVPA 2 | 0.59 *** | (0.41–0.76) | <0.0001 |
| Anxiety 2 | 51.96 | (−2.89–106.81) | 0.06 | |
| Insomnia 2 | 12.25 | (−30.00–54.49) | 0.57 | |
| Anxiety 3 | MVPA 2 | 0.0002 | (−0.0002–0.0007) | 0.31 |
| Anxiety 2 | 0.54 *** | (0.41–0.70) | <0.0001 | |
| Insomnia 2 | 0.16 ** | (0.07–0.267) | 0.001 | |
| Insomnia 3 | MVPA 2 | 0.0001 | (−0.0004–0.0007) | 0.63 |
| Anxiety 2 | 0.10 | (−0.05–0.26) | 0.20 | |
| Insomnia 2 | 0.67 *** | (0.55–0.79) | <0.0001 | |
| Covariates | ||||
| MVPA 1 | Anxiety 1 | −8.27 | (−1273.01–1256.47) | 0.98 |
| MVPA 1 | Insomnia 1 | −951.24 | (−2618.46–715.98) | 0.26 |
| Anxiety 1 | Insomnia 1 * | 15.87 *** | (11.26–20.49) | <0.0001 |
| Questionnaire | N | Avg ± sd | Range | |
|---|---|---|---|---|
| Insomnia Severity Index (ISI) | Time point 1 | 201 | 8.5 ± 6.1 | 0–25 |
| Time point 2 | 169 | 7.9 ± 5.6 | 0–28 | |
| Time point 3 | 143 | 7.0 ± 5.5 | 0–25 | |
| MVPA (MET-min/week) | Time point 1 | 94 | 1978.4 ± 1324.8 | 49.5–4998 |
| Time point 2 | 98 | 2045.9 ± 1382.0 | 49.5–4982 | |
| Time point 3 | 91 | 1986.3 ± 1245.9 | 33–4776 | |
| Generalized Anxiety Disorder Scale (GAD-7) | Time point 1 | 199 | 5.5 ± 4.9 | 0–21 |
| Time point 2 | 169 | 5.2 ± 4.8 | 0–21 | |
| Time point 3 | 143 | 4.1 ± 4.2 | 0–21 |
| Outcome | β-Coefficient | 95% CI | p-Value | |
|---|---|---|---|---|
| Anxiety | Lag Anxiety | 0.56 *** | 0.42–0.69 | <0.001 |
| Lag Insomnia | 0.11 * | 0.02–0.20 | 0.014 | |
| MVPA | Lag MVPA | 0.63 | −1.26–2.53 | 0.51 |
| Lag Anxiety | 30.11 | −2445.62–2505.84 | 0.98 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tobin, S.Y.; Halliday, T.M.; Shoaf, K.; Burns, R.D.; Baron, K.G. Associations of Anxiety, Insomnia, and Physical Activity during the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2024, 21, 428. https://doi.org/10.3390/ijerph21040428
Tobin SY, Halliday TM, Shoaf K, Burns RD, Baron KG. Associations of Anxiety, Insomnia, and Physical Activity during the COVID-19 Pandemic. International Journal of Environmental Research and Public Health. 2024; 21(4):428. https://doi.org/10.3390/ijerph21040428
Chicago/Turabian StyleTobin, Selene Y., Tanya M. Halliday, Kimberley Shoaf, Ryan D. Burns, and Kelly G. Baron. 2024. "Associations of Anxiety, Insomnia, and Physical Activity during the COVID-19 Pandemic" International Journal of Environmental Research and Public Health 21, no. 4: 428. https://doi.org/10.3390/ijerph21040428
APA StyleTobin, S. Y., Halliday, T. M., Shoaf, K., Burns, R. D., & Baron, K. G. (2024). Associations of Anxiety, Insomnia, and Physical Activity during the COVID-19 Pandemic. International Journal of Environmental Research and Public Health, 21(4), 428. https://doi.org/10.3390/ijerph21040428








