Pregnancy in Patients with Pulmonary Arterial Hypertension in Light of New ESC Guidelines on Pulmonary Hypertension
Abstract
1. Introduction
2. Risk Factors
3. Birth Control
4. Termination of Pregnancy
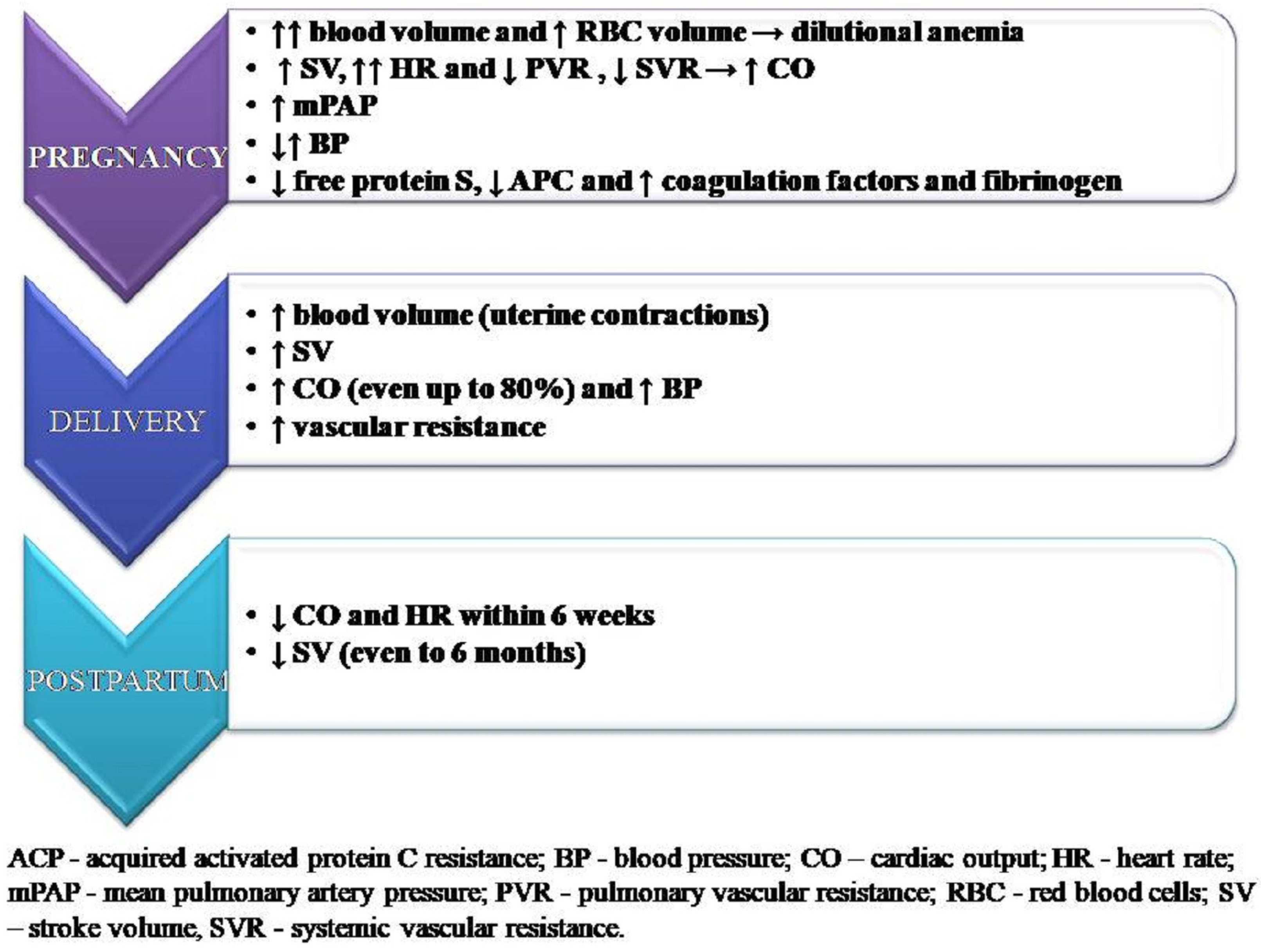
5. Hemodynamics Changes in Pregnancy
6. Hemodynamics Changes during Delivery
7. Pharmacotherapy during Pregnancy
8. Delivery and Puerperium
9. Our Center’s Recommendations for Motherhood
10. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Humbert, M.; Kovacs, G.; Hoeper, M.; Badagliacca, R.; Berger, R.; Brida, M.; Carlsen, J.; Coats, A.; Escribano-Subias, P.; Ferrari, P.; et al. ESC/ERS Scientific Document Group, 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: Developed by the task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). Endorsed by the International Society for Heart and Lung Transplantation (ISHLT) and the European Reference Network on rare respiratory diseases (ERN-LUNG). Eur. Heart J. 2022, 43, ehac237. [Google Scholar] [CrossRef]
- Regitz-Zagrosek, V.; Roos-Hesselink, J.W.; Bauersachs, J.; Blomström-Lundqvist, C.; Cífková, R.; De Bonis, M.; Iung, B.; Johnson, M.R.; Kintscher, U.; Kranke, P.; et al. 2018 ESC Guidelines for the management of cardiovascular diseases during pregnancy. Eur. Heart J. 2018, 39, 3165–3241. [Google Scholar] [CrossRef] [PubMed]
- Weiss, B.M.; Zemp, L.; Seifert, B.; Hess, O.M. Outcome of pulmonary vascular disease in pregnancy: A systematic overview from 1978 through 1996. J. Am. Coll.Cardiol. 1998, 31, 1650–1657. [Google Scholar] [CrossRef] [PubMed]
- Low, T.T.; Guron, N.; Ducas, R.; Yamamura, K.; Charla, P.; Granton, J.; Silversides, C.K. Pulmonary arterial hypertension in pregnancy-a systematic review of outcomes in the modern era. Pulm. Circ. 2021, 11, 20458940211013671. [Google Scholar] [CrossRef]
- Duan, R.; Xu, X.; Wang, X.; Yu, H.; You, Y.; Liu, X.; Xing, A.; Zhou, R.; Xi, M. Pregnancy outcome in women with Eisenmenger’s syndrome: A case series from west China. BMC PregnancyChildbirth 2016, 16, 356. [Google Scholar] [CrossRef] [PubMed]
- Bédard, E.; Dimopoulos, K.; Gatzoulis, M.A. Has there been any progress made on pregnancy outcomes among women with pulmonary arterial hypertension? Eur. Heart J. 2009, 30, 256–265. [Google Scholar] [CrossRef]
- Jaïs, X.; Olsson, K.M.; Barbera, J.A.; Blanco, I.; Torbicki, A.; Peacock, A.; Vizza, C.D.; Macdonald, P.; Humbert, M.; Hoeper, M.M. Pregnancy outcomes in pulmonary arterial hypertension in the modern management era. Eur. Respir. J. 2012, 40, 881–885. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Wang, J.; Zhang, X.; Zhuang, Q.; Wang, R.; Shen, J.; Lin, J. Incidence and long-term outcomes of pregnant women complicated with pulmonary arterial hypertension during different pregnancies: A prospective cohort study from China. Int. J.Cardiol. 2021, 326, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhang, H.; Xu, X.; Wang, Y.; Gu, H.; Zhang, J. Risk factors for perinatal cardiac complications in pregnancy with pulmonary hypertension. Pregnancy Hypertens 2018, 12, 207–213. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Zhang, J.; Zhang, D.; Li, J.; Zhao, Y.; Liu, K.; Ma, X.; Bai, C.; Gu, H.; et al. Maternal and fetal outcomes of pregnant women with pulmonary arterial hypertension associated with congenital heart disease in Beijing, China: A retrospective study. Pulm. Circ. 2022, 12, e12079. [Google Scholar] [CrossRef]
- Morrell, N.W.; Aldred, M.A.; Chung, W.K.; Elliott, C.G.; Nichols, W.C.; Soubrier, F.; Trembath, R.C.; Loyd, J.E. Genetics and genomics of pulmonary arterial hypertension. Eur. Respir. J. 2019, 53, 1801899. [Google Scholar] [CrossRef]
- Cipolletta, S.; Ravasio, G.; Bussotti, M. Sexual and Reproductive Health in Women with Pulmonary Hypertension: A Qualitative Study. Arch. Sex Behav. 2022, 51, 1647–1657. [Google Scholar] [CrossRef] [PubMed]
- Division of Reproductive Health; National Center for Chronic Disease Prevention and Health Promotion; Centers for Disease Control and Prevention (CDC). U.S. Selected Practice Recommendations for Contraceptive Use, 2013: Adapted from the World Health Organization selected practice recommendations for contraceptive use, 2nd edition. MMWR Recomm. Rep. 2013, 62, 1–60. [Google Scholar]
- Lindley, K.J.; Madden, T.; Cahill, A.G.; Ludbrook, P.A.; Billadello, J.J. Contraceptive Use and Unintended Pregnancy in Women with Congenital Heart Disease. Obs. Gynecol. 2015, 126, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Hill, W.; Holy, R.; Traiger, G. EXPRESS: Intimacy, Contraception, and Pregnancy Prevention in Patients with Pulmonary Arterial Hypertension: Are We Counseling Our Patients? Pulm. Circ. 2018, 10, 2045894018785259. [Google Scholar] [CrossRef]
- Thorne, S.; Nelson-Piercy, C.; MacGregor, A.; Gibbs, S.; Crowhurst, J.; Panay, N.; Rosenthal, E.; Walker, F.; Williams, D.; de Swiet, M.; et al. Pregnancy and contraception in heart disease and pulmonary arterial hypertension. J. Fam.Plann. Reprod. Health Care. 2006, 32, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Weiss, B.M.; Hess, O.M. Pulmonary vascular disease and pregnancy: Current controversies, management strategies, and perspectives. Eur. Heart J. 2000, 21, 104–115. [Google Scholar] [CrossRef]
- Satoh, H.; Masuda, Y.; Izuta, S.; Yaku, H.; Obara, H. Pregnant patient with primary pulmonary hypertension: General anesthesia and extracorporeal membrane oxygenation support for termination of pregnancy. Anesthesiology 2002, 97, 1638–1640. [Google Scholar] [CrossRef]
- Pritchard, J.A. Changes in The Blood Volume during Pregnancy and Delivery. Anesthesiology 1965, 26, 393–399. [Google Scholar] [CrossRef]
- Katz, R.; Karliner, J.S.; Resnik, R. Effects of a natural volume overload state (pregnancy) on left ventricular performance in normal human subjects. Circulation 1978, 58, 434–441. [Google Scholar] [CrossRef]
- Mesa, A.; Jessurun, C.; Hernandez, A.; Adam, K.; Brown, D.; Vaughn, W.K.; Wilansky, S. Left ventricular diastolic function in normal human pregnancy. Circulation 1999, 99, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.L.; Cotton, D.B.; Lee, W.; Bishop, C.; Hill, T.; Southwick, J.; Pivarnik, J.; Spillman, T.; DeVore, G.R.; Phelan, J.; et al. Central hemodynamic assessment of normal term pregnancy. Am. J.Obs. Gynecol. 1989, 161, 1439–1442. [Google Scholar] [CrossRef] [PubMed]
- Meah, V.L.; Cockcroft, J.R.; Backx, K.; Shave, R.; Stöhr, E.J. Cardiac output and related haemodynamics during pregnancy: A series of meta-analyses. Heart 2016, 102, 518–526. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.L.; Zhang, J. Analysis of hemodynamics and outcomes of pregnant women with congenital heart disease-pulmonary arterial hypertension. Zhonghua Fu Chan Ke Za Zhi 2019, 54, 438–444. (In Chinese) [Google Scholar] [CrossRef] [PubMed]
- Shotan, A.; Ostrzega, E.; Mehra, A.; Johnson, J.V.; Elkayam, U. Incidence of arrhythmias in normal pregnancy and relation to palpitations, dizziness, and syncope. Am. J. Cardiol. 1997, 79, 1061–1064. [Google Scholar] [CrossRef]
- Hermida, R.C.; Ayala, D.E.; Mojón, A.; Fernández, J.R. Time-qualified reference values for ambulatory blood pressure monitoring in pregnancy. Hypertension 2001, 38, 746–752. [Google Scholar] [CrossRef]
- MacGillivray, I.; Rose, G.A.; Rowe, B. Blood pressure survey in pregnancy. Clin. Sci. 1969, 37, 395–407. [Google Scholar]
- Hellgren, M. Hemostasis during normal pregnancy and puerperium. Semin. Thromb.Hemost. 2003, 29, 125–130. [Google Scholar] [CrossRef]
- Hsu, C.H.; Gomberg-Maitland, M.; Glassner, C.; Chen, J.H. The management of pregnancy and pregnancy-related medical conditions in pulmonary arterial hypertension patients. Int. J. Clin. Pr. Suppl. 2011, 172, 6–14. [Google Scholar] [CrossRef]
- European Society of Gynecology (ESG); Association for European Paediatric Cardiology (AEPC); German Society for Gender Medicine (DGesGM); Regitz-Zagrosek, V.; Blomstrom Lundqvist, C.; Borghi, C.; Cifkova, R.; Ferreira, R.; Foidart, J.M.; Gibbs, J.S.; et al. ESC Guidelines on the management of cardiovascular diseases during pregnancy: The Task Force on the Management of Cardiovascular Diseases during Pregnancy of the European Society of Cardiology (ESC). Eur. Heart J. 2011, 32, 3147–3197. [Google Scholar] [CrossRef]
- Phoophiboon, V.; Pachinburavan, M.; Ruamsap, N.; Sanguanwong, N.; Jaimchariyatam, N. Critical care management of pulmonary arterial hypertension in pregnancy: The pre-, peri- and post-partum stages. Acute Crit. Care. 2021, 36, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.; Robson, S.C. Adaptation of the maternal heart in pregnancy. Br. Heart J. 1992, 68, 540–543. [Google Scholar] [CrossRef] [PubMed]
- Amlodipine Besylate FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2011%2F019787s047lbl.pdf&usg=AOvVaw1bzMQHdiUF6xhlQiY8LC-F (accessed on 1 October 2022).
- Nifedipine FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2013%2F018482s051lbl.pdf&usg=AOvVaw2WX4ugufcP-r-RQ5Tqy3q1 (accessed on 1 October 2022).
- Diltiazem Hydrochloride FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2014%2F018602s065lbl.pdf&usg=AOvVaw0Ss3cAa0aLtUylctHK_d0- (accessed on 1 October 2022).
- Sildenafil FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2011%2F020895s036lbl.pdf&usg=AOvVaw1KhihEPiWQc92y4b5UNdAX (accessed on 1 October 2022).
- Tadalafil FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2011%2F021368s20s21lbl.pdf&usg=AOvVaw3rkxXrjRh_TgClRzvaDHMG (accessed on 1 October 2022).
- Ambrisentan FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2018%2F022081s040lbl.pdf&usg=AOvVaw0rJrBJyCisN8ne0Wdt0Fqx (accessed on 1 October 2022).
- Bosentan FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2017%2F209279s000lbl.pdf&usg=AOvVaw0dHdxlL3CNDJAdTdFH0aE_ (accessed on 1 October 2022).
- Macitentan FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2019%2F204410s017lbl.pdf&usg=AOvVaw0OoTrfi1RqokR3Sk0Qqja7 (accessed on 1 October 2022).
- Iloprost FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2009%2F021779s009lbl.pdf&usg=AOvVaw1awsKukjMNz-H2Q92KXtWn (accessed on 1 October 2022).
- Epoprostenol FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2012%2F022260s005lbl.pdf&usg=AOvVaw2gz1ScpY2hWcVn7tTSFHLb (accessed on 1 October 2022).
- Treprostinil FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2018%2F021272s026lbl.pdf&usg=AOvVaw3JItWhOOWwneX63ULEgTy3 (accessed on 1 October 2022).
- Oral Treprostinil FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2013%2F203496s000lbl.pdf&usg=AOvVaw21R5jakNqFrm0ztVggVWOV (accessed on 1 October 2022).
- Inhaled Treprostinil FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2009%2F022387lbl.pdf&usg=AOvVaw3KQxEGFHRaZEn0wkRv_FyS (accessed on 1 October 2022).
- Selexipag FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2015%2F207947s000lbl.pdf&usg=AOvVaw3uiiH0ih6RoxmJqVsRGJIQ (accessed on 1 October 2022).
- Riocuguat FDA Label. Available online: https://www.accessdata.fda.gov%2Fdrugsatfda_docs%2Flabel%2F2017%2F204819s006lbl.pdf&usg=AOvVaw2dYaZayTn1WQNm9qm7RLdE (accessed on 1 October 2022).
- Wolla, C.D.; Matos, J.R.; Wineland, R. Anesthetic Management of Severe Pulmonary Hypertension in Pregnancy. J. Cardiothorac. Vasc. Anesth. 2022, 36, 542–545. [Google Scholar] [CrossRef]
- Smith, J.S.; Mueller, J.; Daniels, C.J. Pulmonary arterial hypertension in the setting of pregnancy: A case series and standard treatment approach. Lung 2012, 190, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Olsson, K.M.; Channick, R. Pregnancy in pulmonary arterial hypertension. Eur. Respir. Rev. 2016, 25, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Z.; Fernandes, T.M.; Kim, N.H.; Poch, D.S.; Kerr, K.M.; Lombardi, S.; Melber, D.; Kelly, T.; Papamatheakis, D.G. Pregnancy and pulmonary arterial hypertension: A case series and literature review. Am. J.Obs. Gynecol. MFM 2021, 3, 100358. [Google Scholar] [CrossRef]
- Wollein, U.; Schech, B.; Hardt, J.; Schramek, N. Determination and quantitation of sildenafil and its major metabolite in the breast milk of a lactating woman. J. Pharm. Biomed. Anal. 2016, 120, 100–105. [Google Scholar] [CrossRef]
- Hemnes, A.R.; Kiely, D.G.; Cockrill, B.A.; Safdar, Z.; Wilson, V.J.; Al Hazmi, M.; Preston, I.R.; MacLean, M.R.; Lahm, T. Statement on pregnancy in pulmonary hypertension from the Pulmonary Vascular Research Institute. Pulm. Circ. 2015, 5, 435–465. [Google Scholar] [CrossRef]
- Olsson, K.M.; Jais, X. Birth control and pregnancy management in pulmonary hypertension. Semin. Respir. Crit. Care Med. 2013, 34, 681–688. [Google Scholar] [CrossRef]
- Uebing, A.; Steer, P.J.; Yentis, S.M.; Gatzoulis, M.A. Pregnancy and congenital heart disease. BMJ 2006, 332, 401–406. [Google Scholar] [CrossRef]
- Liu, S.; Liston, R.M.; Joseph, K.S.; Heaman, M.; Sauve, R.; Kramer, M.S.; Maternal Health Study Group of the Canadian Perinatal Surveillance System. Maternal mortality and severe morbidity associated with low-risk planned cesarean delivery versus planned vaginal delivery at term. CMAJ 2007, 176, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Safdar, Z. Pulmonary arterial hypertension in pregnant women. Adv. Respir. Dis. 2013, 7, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.L.; Landau, R.; Viktorsdottir, O.; Banayan, J.; Grant, T.; Bateman, B.; Smiley, R.; Reitman, E. Pulmonary Hypertension in Pregnancy: A Report of 49 Cases at Four Tertiary North American Sites. Obs. Gynecol. 2017, 129, 511–520. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, J.; Zhou, X.; Xu, X.; Ye, Q.; Ou, Q.; Li, Y.; Huang, J. Perioperative Management of Pregnant Women with Idiopathic Pulmonary Arterial Hypertension: An Observational Case Series Study from China. J.Cardiothorac.Vasc.Anesth. 2018, 32, 2547–2559. [Google Scholar] [CrossRef]
- Thomas, J.S.; Koh, S.H.; Cooper, G.M. Haemodynamic effects of oxytocin given as i.v. bolus or infusion on women undergoing Caesarean section. Br. J.Anaesth. 2007, 98, 116–119. [Google Scholar] [CrossRef]
- Ballard, W., 3rd; Dixon, B.; McEvoy, C.A.; Verma, A.K. Pulmonary Arterial Hypertension in Pregnancy. Cardiol. Clin. 2021, 39, 109–118. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, H.; Li, H.; Liu, Y.; Bai, Q. Association of hemodynamics during caesarean section, outcomes after caesarean section and hormone changes with different anesthesia in pregnant women with severe pulmonary arterial hypertension. Exp. Med. 2018, 15, 4731–4736. [Google Scholar] [CrossRef] [PubMed]
- Parissis, J.T.; Paraskevaidis, I.; Bistola, V.; Farmakis, D.; Panou, F.; Kourea, K.; Nikolaou, M.; Filippatos, G.; Kremastinos, D. Effects of levosimendan on right ventricular function in patients with advanced heart failure. Am. J.Cardiol. 2006, 11, 1489–1492. [Google Scholar] [CrossRef]
- Eichhorn, E.J.; Konstam, M.A.; Weiland, D.S.; Roberts, D.J.; Martin, T.T.; Stransky, N.B.; Salem, D.N. Differential effects of milrinone and dobutamine on right ventricular preload, afterload and systolic performance in congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am. J.Cardiol. 1987, 60, 1329–1333. [Google Scholar] [CrossRef]
- Overgaard, C.B.; Dzavík, V. Inotropes and vasopressors: Review of physiology and clinical use in cardiovascular disease. Circulation 2008, 118, 1047–1056. [Google Scholar] [CrossRef]
- Holmes, C.L.; Landry, D.W.; Granton, J.T. Science Review: Vasopressin and the cardiovascular system part 2—Clinical physiology. Crit. Care. 2004, 8, 15–23. [Google Scholar] [CrossRef]
- Anjum, H.; Surani, S. Pulmonary Hypertension in Pregnancy: A Review. Medicina 2021, 57, 259. [Google Scholar] [CrossRef]
- Coz Yataco, A.; Aguinaga Meza, M.; Buch, K.P.; Disselkamp, M.A. Hospital and intensive care unit management of decompensated pulmonary hypertension and right ventricular failure. Heart Fail. Rev. 2016, 21, 323–346. [Google Scholar] [CrossRef]
- Abid Memon, H.; Safdar, Z.; Goodarzi, A. Use of Extracorporeal Membrane Oxygenation in Postpartum Management of a Patient with Pulmonary Arterial Hypertension. Case Rep.Pulmonol. 2018, 2018, 7031731. [Google Scholar] [CrossRef]
- Vitulo, P.; Beretta, M.; Martucci, G.; Hernandez Baravoglia, C.M.; Romano, G.; Bertani, A.; Martino, L.; Callari, A.; Panarello, G.; Pilato, M.; et al. Challenge of Pregnancy in Patients with Pre-Capillary Pulmonary Hypertension: Veno-Arterial Extracorporeal Membrane Oxygenation as an Innovative Support for Delivery. J.Cardiothorac.Vasc.Anesth. 2017, 31, 2152–2155. [Google Scholar] [CrossRef]
- Svetlichnaya, J.; Janmohammed, M.; De Marco, T. Special Situations in Pulmonary Hypertension: Pregnancy and Right Ventricular Failure. Cardiol. Clin. 2016, 34, 473–487. [Google Scholar] [CrossRef]
- Ye, J.; Chen, J.Y.; Xu, N.; Wu, B.; Wang, Z.P.; Xu, H.Y.; Ma, J.Q. Bilateral lung transplantation after caesarean section in pregnancy with severe pulmonary arterial hypertension: A case report. Medicine 2019, 98, e18109. [Google Scholar] [CrossRef]
- Corbach, N.; Berlier, C.; Lichtblau, M.; Schwarz, E.I.; Gautschi, F.; Groth, A.; Schüpbach, R.; Krähenmann, F.; Saxer, S.; Ulrich, S. Favorable Pregnancy Outcomes in Women with Well-Controlled Pulmonary Arterial Hypertension. Front. Med. 2021, 8, 689764. [Google Scholar] [CrossRef]
- Wojciuk, M.; Dziecioł-Anikiej, Z.; Kaniewska, K.; Ciołkiewicz, M.; Moskal-Jasinska, D.; Kuryliszyn-Moskal, A. Exercise Training as a Non-Pharmacological Therapy for Patients with Pulmonary Arterial Hypertension: Home-Based Rehabilitation Program and Training Recommendations. J. Clin. Med. 2022, 11, 6932. [Google Scholar] [CrossRef]

| Medication Category | Generic Name | FDA Pregnancy Category | Recommendation during Pregnancy | Recommendation during Lactation |
|---|---|---|---|---|
| Calcium channelblockers | Amlodipine | C | No adequate studies in pregnant women. Benefit vs. risk. | Unknown presence in human milk. Breastfeeding not recommended. |
| Felodipine | C | No adequate studies in pregnant women. Benefit vs. risk. | Unknown presence in human milk. Benefit vs. risk | |
| Nifedipine | C | No adequate studies in pregnant women. Benefit vs. risk. | Present in breast milk. Benefit vs. risk. | |
| Diltiazem | C | No adequate studies in pregnant women. Benefit vs. risk. | Present in breast milk, probably in approximate serum levels. Breastfeeding not recommended. | |
| PDE5i | Sildenafil | B | No adequate studies in pregnant women. Not indicated for use. | Present in human milk. |
| Tadalafil | B | No adequate studies in pregnant women. Not indicated for use. | Unknown presence in human milk. Not indicated for use. | |
| ERA | Ambrisentan | X | Contraindicated. | Unknown presence in human milk. Breastfeeding not recommended. |
| Bosentan | X | Contraindicated. | Unknown presence in human milk. Breastfeeding not recommended. | |
| Macitentan | X | Contraindicated. | Unknown presence in human milk. Breastfeeding not recommended. | |
| Prostacyclin | Iloprost | C | Benefit vs. risk. | Unknown presence in human milk. Breastfeeding not recommended. |
| Epoprostenol | B | No adequate studies in pregnant women. Used only if clearly needed. | Unknown presence in human milk, use with caution. | |
| Treprostinil | Parental—B | No adequate studies in pregnant women. | Unknown presence in human milk. | |
| Oral—C | Benefit vs. risk. | Breastfeeding not recommended. | ||
| Inhaled—B | No adequate studies in pregnant women. | Unknown presence in human milk, use with caution. | ||
| Prostacyclinanalogue | Selexipag | Not assigned | Limited data. | Unknown presence in human milk. Breastfeeding not recommended. |
| Guanylatecyclasestimulator | Riociguat | X | Contraindicated. | Unknown presence in human milk. Breastfeeding not recommended. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barańska-Pawełczak, K.; Wojciechowska, C.; Jacheć, W. Pregnancy in Patients with Pulmonary Arterial Hypertension in Light of New ESC Guidelines on Pulmonary Hypertension. Int. J. Environ. Res. Public Health 2023, 20, 4625. https://doi.org/10.3390/ijerph20054625
Barańska-Pawełczak K, Wojciechowska C, Jacheć W. Pregnancy in Patients with Pulmonary Arterial Hypertension in Light of New ESC Guidelines on Pulmonary Hypertension. International Journal of Environmental Research and Public Health. 2023; 20(5):4625. https://doi.org/10.3390/ijerph20054625
Chicago/Turabian StyleBarańska-Pawełczak, Karolina, Celina Wojciechowska, and Wojciech Jacheć. 2023. "Pregnancy in Patients with Pulmonary Arterial Hypertension in Light of New ESC Guidelines on Pulmonary Hypertension" International Journal of Environmental Research and Public Health 20, no. 5: 4625. https://doi.org/10.3390/ijerph20054625
APA StyleBarańska-Pawełczak, K., Wojciechowska, C., & Jacheć, W. (2023). Pregnancy in Patients with Pulmonary Arterial Hypertension in Light of New ESC Guidelines on Pulmonary Hypertension. International Journal of Environmental Research and Public Health, 20(5), 4625. https://doi.org/10.3390/ijerph20054625





