Tobacco Smoke Exposure According to Location of Home Smoking in Israel: Findings from the Project Zero Exposure Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Samples and Procedures
2.2. Eligibility Criteria
2.3. Ethics and Registration
2.4. Measures
2.4.1. Primary Outcome
2.4.2. Independent Measures
2.5. Statistical Analyses
2.5.1. Analyses, Smoking Families vs. Nonsmoking Families (Study 1 vs. Study 2)
2.5.2. TSE by Location of Home Smoking and Other Explanatory Variables (Study 1)
3. Results
3.1. Table 1 Describes the Sample Characteristics of Participating Families for Both Studies
| Study 1: Smoking Households (n = 159) | Study 2: Non-Smoking Households (n = 20) | ||||
|---|---|---|---|---|---|
| n | % | n | % | ||
| Geographic Region | Tel Aviv | 63 | 39.6 | 6 | 30 |
| Central Region | 50 | 31.4 | 12 | 60 | |
| Jerusalem | 7 | 4.4 | 1 | 5 | |
| Haifa | 7 | 4.4 | 0 | 0 | |
| South | 30 | 18.9 | 1 | 5 | |
| Judaea and Samaria | 2 | 1.3 | 0 | 0 | |
| Child sex | Female | 82 | 51.6 | 8 | 40.0 |
| Male | 77 | 48.4 | 12 | 60.0 | |
| Child age (months) | Mean (SD) | 154 | 37.0 (23.3) | 20 | 29.4 (19.9) |
| Smoking parent | Neither | 0 | 0.0 | 20 | 100.0 |
| Mother Only | 27 | 17.0 | 0 | 0.0 | |
| Father Only | 61 | 38.4 | 0 | 0.0 | |
| Both Father and Mother | 71 | 44.7 | 0 | 0.0 | |
| Parents’ nationality | Both Israeli born | 102 | 64.2 | 17 | 85.0 |
| Other | 57 | 35.8 | 3 | 15.0 | |
| Mother’s education | Junior/High school | 26 | 16.7 | 1 | 5.6 |
| Some post-high school | 24 | 15.4 | 1 | 5.6 | |
| Attended/completed university | 106 | 68.0 | 16 | 88.9 | |
| Father’s education | Junior/High school | 45 | 29.8 | 0 | 0.0 |
| Some post-high school | 37 | 24.5 | 1 | 5.6 | |
| Attended/completed university | 69 | 45.7 | 17 | 94.4 | |
| Income a | Lower | 36 | 23.5 | 2 | 10.0 |
| Average | 49 | 32.0 | 3 | 15.0 | |
| Above | 68 | 44.4 | 15 | 75.0 | |
| Neighborhood SES b | Mean (SD) | 149 | 13.2 (3.4) | 19 | 14.4 (2.2) |
3.2. Characteristics of Smoking and Non-Smoking Families
3.3. Laboratory Batch Results
3.4. Child TSE in Smoking vs. Nonsmoking Families
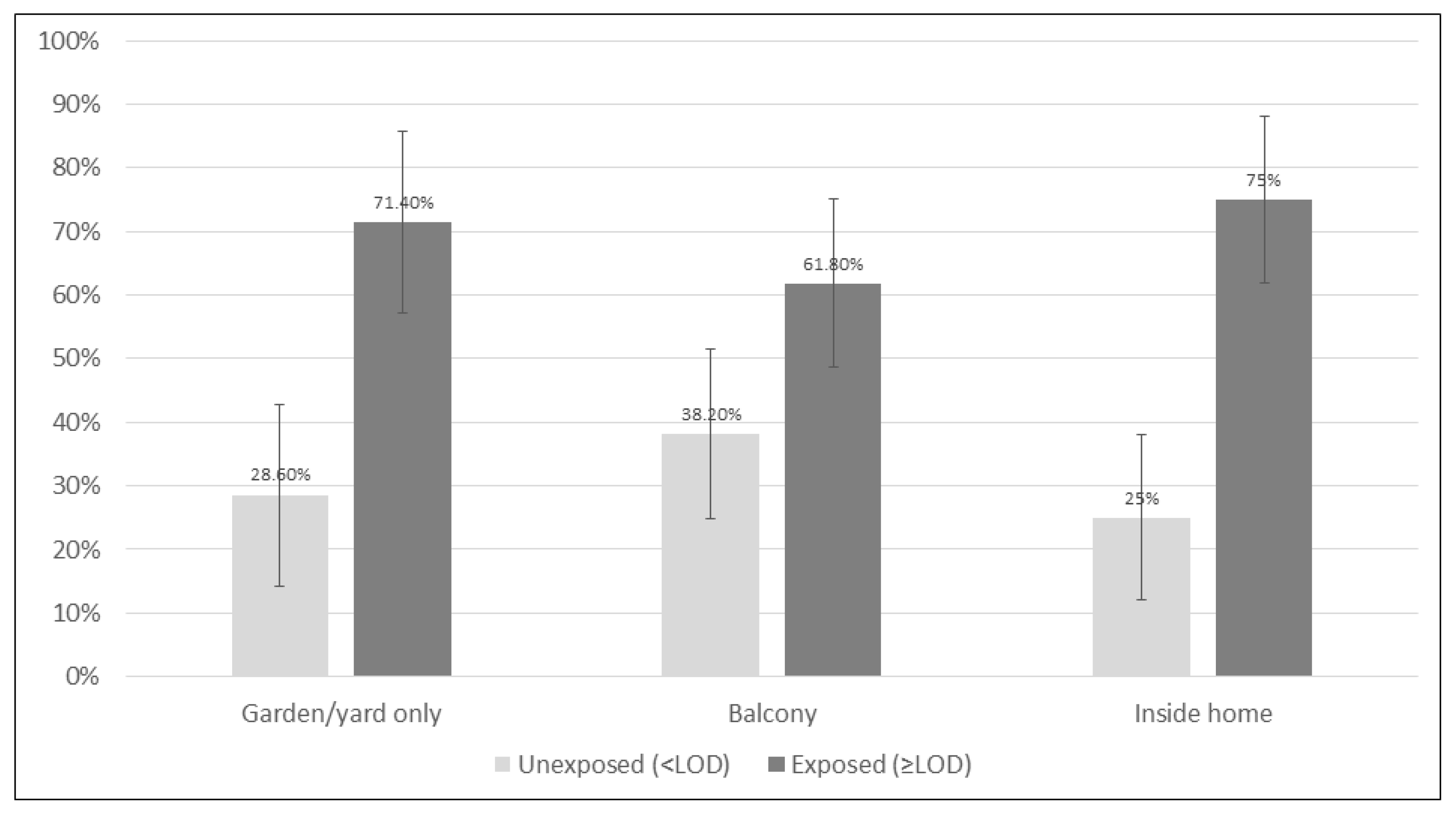
3.5. The Association between Child TSE and Location in Smoking Families
3.6. The Association between Child TSE and Other Variables in Smoking Families
4. Discussion
Public Health Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Tobacco; WHO: Geneva, Switzerland, 2022; Available online: https://www.who.int/news-room/fact-sheets/detail/tobacco#:~:text=Tobacco%20kills%20more%20than%208,%2D%20and%20middle%2Dincome%20countries (accessed on 9 February 2023).
- US Dept of Health & Human Services. The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General. 2006. Available online: http://www.surgeongeneral.gov/library/secondhandsmoke (accessed on 15 September 2014).
- Centers for Disease Control and Prevention. Health Effects of Secondhand Smoke; CDC: Atlanta, GA, USA, 2021. Available online: https://www.cdc.gov/tobacco/data_statistics/fact_sheets/secondhand_smoke/health_effects/index.htm#:~:text=They%20also%20have%20fluid%20in,in%20ear%20tubes%20for%20drainage.&text=Secondhand%20smoke%20can%20trigger%20an,a%20child%27s%20life%20in%20danger (accessed on 9 February 2023).
- World Health Organization. Tobacco Control to Improve Child Health and Development: Thematic Brief; WHO Policy Brief: Geneva, Switzerland, 2021; Available online: https://www.who.int/publications/i/item/9789240022218 (accessed on 9 February 2023).
- Mbulo, L.; Palipudi, K.M.; Andes, L.; Morton, J.; Bashir, R.; Fouad, H.; Ramanandraibe, N.; Caixeta, R.; Dias, R.C.; Wijnhoven, T.M. Secondhand smoke exposure at home among one billion children in 21 countries: Findings from the Global Adult Tobacco Survey (GATS). Tob. Control 2016, 25, e95–e100. [Google Scholar] [CrossRef] [PubMed]
- Hyland, A.; Barnoya, J.; Corral, J.E. Smoke-free air policies: Past, present and future. Tob. Control 2012, 21, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Barnoya, J.; Navas-Acien, A. Protecting the world from secondhand tobacco smoke exposure: Where do we stand and where do we go from here? Nicotine Tob. Res. 2012, 15, 789–804. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO FCTC Guidelines for Implementation of Article 8; WHO: Geneva, Switzerland, 2017; Available online: https://fctc.who.int/publications/m/item/protection-from-exposure-to-tobacco-smoke (accessed on 9 February 2023).
- Centers for Disease Control and Prevention. Smokefree Policies Improve Health; CDC: Atlanta, GA, USA, 2021. Available online: https://www.cdc.gov/tobacco/data_statistics/fact_sheets/secondhand_smoke/protection/improve_health/index.htm (accessed on 9 February 2023).
- World Health Organization. WHO Report on the Global Tobacco Epidemic, 2009: Implementing Smoke-Free Environments; WHO: Geneva, Switzerland, 2009; Available online: https://www.who.int/publications/i/item/9789241563918 (accessed on 9 February 2023).
- King, B.A.; Dube, S.R.; Homa, D.M. Peer Reviewed: Smoke-Free Rules and Secondhand Smoke Exposure in Homes and Vehicles Among US Adults, 2009–2010. Prev. Chronic Dis. 2013, 10, E79. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.-p.; Suen, Y.-n.; Wong, B.Y.-m.; Li, W.H.-c.; Koh, D.S.-q.; Lam, T.-h.; Chan, S.S.-c. Paternal smoking and maternal protective behaviors at home on infant’s saliva cotinine levels. Pediatr. Res. 2018, 83, 936–942. [Google Scholar] [CrossRef]
- Rosen, L.J.; Lev, E.; Guttman, N.; Tillinger, E.; Rosenblat, S.; Zucker, D.M.; Myers, V. Parental Perceptions and Misconceptions of Child Tobacco Smoke Exposure. Nicotine Tob. Res. 2018, 20, 1369–1377. [Google Scholar] [CrossRef]
- Myers, V.; Lev, E.; Guttman, N.; Tillinger, E.; Rosen, L. “I can’t stand it… but I do it sometimes” parental smoking around children: Practices, beliefs, and conflicts–a qualitative study. BMC Public Health 2020, 20, 693. [Google Scholar] [CrossRef]
- Matt, G.; Quintana, P.; Hovell, M.; Bernert, J.; Song, S.; Novianti, N.; Juarez, T.; Floro, J.; Gehrman, C.; Garcia, M. Households contaminated by environmental tobacco smoke: Sources of infant exposures. Tob. Control 2004, 13, 29–37. [Google Scholar] [CrossRef]
- Yamakawa, M.; Yorifuji, T.; Kato, T.; Tsuda, T.; Doi, H. Maternal smoking location at home and hospitalization for respiratory tract infections among children in Japan. Arch. Environ. Occup. Health 2017, 72, 343–350. [Google Scholar] [CrossRef]
- Johansson, A.; Hermansson, G.r.; Ludvigsson, J. How should parents protect their children from environmental tobacco-smoke exposure in the home? Pediatrics 2004, 113, e291–e295. [Google Scholar] [CrossRef]
- Jurado, D.; Muñoz, C.; Luna, J.D.D.; Fernández-Crehuet, M. Environmental tobacco smoke exposure in children: Parental perception of smokiness at home and other factors associated with urinary cotinine in preschool children. J. Expo. Sci. Environ. Epidemiol. 2004, 14, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Ulbricht, S.; Unger, F.; Groß, S.; Nauck, M.; Meyer, C.; John, U. Factors associated with secondhand smoke exposure prevalence and secondhand smoke level of children living with parental smokers: A cross sectional study. J. Commun. Health 2015, 40, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Al-Delaimy, W.K.; Crane, J.; Woodward, A. Passive smoking in children: Effect of avoidance strategies at home as measured by hair nicotine levels. Arch. Environ. Health Int. J. 2001, 56, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Bahçeciler, N.N.; Barlan, I.B.; Nuhoĝlu, Y.; BaŞaran, M.M. Parental smoking behavior and the urinary cotinine levels of asthmatic children. J. Asthma 1999, 36, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Berman, T.; Barnett-Itzhaki, Z.; Axelrod, R.; Keinan-Boker, L.; Shimony, T.; Goldsmith, R.; Göen, T.; Geva, H.; Rosen, L. Socioeconomic inequalities in exposure to environmental tobacco smoke in children in Israel. Environ. Int. 2018, 121, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.; Zucker, D.; Guttman, N.; Brown, N.; Bitan, M.; Rule, A.; Berkovitch, M.; Myers, V. Protecting children from tobacco smoke exposure: A randomized controlled trial of Project Zero Exposure. Nicotine Tob. Res. 2021, 23, 2003–2012. [Google Scholar] [CrossRef]
- Myers, V.; Rosen, L.J.; Zucker, D.M.; Shiloh, S. Parental perceptions of children’s exposure to tobacco smoke and parental smoking behaviour. Int. J. Environ. Res. Public Health 2020, 17, 3397. [Google Scholar] [CrossRef]
- Myers, V.; Shiloh, S.; Zucker, D.M.; Rosen, L.J. Changing Exposure Perceptions: A Randomized Controlled Trial of an Intervention with Smoking Parents. Int. J. Environ. Res. Public Health 2020, 17, 3349. [Google Scholar] [CrossRef]
- Myers, V. Development and Testing of a New Construct to Quantify Parental Perceptions of Child Exposure to Tobacco Smoke. PhD Thesis, Tel Aviv University, Tel Aviv-Yafo, Israel, 2019. [Google Scholar]
- NAÁMAT. Naámat—Movement of Working Women and Volunteers. Available online: https://www.naamat.org.il/english/ (accessed on 4 August 2019).
- Florescu, A.; Ferrence, R.; Einarson, T.; Selby, P.; Soldin, O.; Koren, G. Methods for Quantification of Exposure to Cigarette Smoking and Environmental Tobacco Smoke: Focus on Developmental Toxicology. Ther. Drug Monit. 2009, 31, 14–30. [Google Scholar] [CrossRef]
- Rosen, L.J.; Tillinger, E.; Guttman, N.; Rosenblat, S.; Zucker, D.; Stillman, F.; Myers, V. Parental receptivity to child biomarker testing for tobacco smoke exposure: A qualitative study. Patient Educ. Couns. 2015, 98, 1439–1445. [Google Scholar] [CrossRef]
- Kim, S.R.; Wipfli, H.; Avila-Tang, E.; Samet, J.M.; Breysse, P.N. Method validation for measurement of hair nicotine level in nonsmokers. Biomed. Chromatogr. 2009, 23, 273–279. [Google Scholar] [CrossRef]
- Israel Central Bureau of Statistics. Data on the Israeli Households Income, Expenditure and Durable Goods Ownership Findings from the Household Expenditure Survey 2016. Available online: www.cbs.gov.il (accessed on 9 February 2023).
- Wilson, K.M.; Klein, J.D.; Blumkin, A.K.; Gottlieb, M.; Winickoff, J.P. Tobacco-smoke exposure in children who live in multiunit housing. Pediatrics 2011, 127, 85–92. [Google Scholar] [CrossRef]
- Jacob, P., III.; Benowitz, N.L.; Destaillats, H.; Gundel, L.; Hang, B.; Martins-Green, M.; Matt, G.E.; Quintana, P.J.; Samet, J.M.; Schick, S.F. Thirdhand smoke: New evidence, challenges, and future directions. Chem. Res. Toxicol. 2017, 30, 270–294. [Google Scholar] [CrossRef]
- Matt, G.E.; Quintana, P.J.; Zakarian, J.M.; Fortmann, A.L.; Chatfield, D.A.; Hoh, E.; Uribe, A.M.; Hovell, M.F. When smokers move out and non-smokers move in: Residential thirdhand smoke pollution and exposure. Tob. Control 2011, 20, e1. [Google Scholar] [CrossRef] [PubMed]
- Semple, S.; Latif, N. How long does secondhand smoke remain in household air: Analysis of PM 2.5 data from smokers’ homes. Nicotine Tob. Res. 2014, 16, 1365–1370. [Google Scholar] [CrossRef] [PubMed]
- Gee, I.L.; Semple, S.; Watson, A.; Crossfield, A. Nearly 85% of tobacco smoke is invisible—A confirmation of previous claims. Tob. Control 2013, 22, 429. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, M.I.; Curtis, J.A.; Vearrier, D. The perception of odor is not a surrogate marker for chemical exposure: A review of factors influencing human odor perception. Clin. Toxicol. 2013, 51, 70–76. [Google Scholar] [CrossRef]
- Northrup, T.F.; Khan, A.M.; Jacob, P.; Benowitz, N.L.; Hoh, E.; Hovell, M.F.; Matt, G.E.; Stotts, A.L. Thirdhand smoke contamination in hospital settings: Assessing exposure risk for vulnerable paediatric patients. Tob. Control 2016, 25, 619–623. [Google Scholar] [CrossRef]
- Cameron, M.; Brennan, E.; Durkin, S.; Borland, R.; Travers, M.J.; Hyland, A.; Spittal, M.J.; Wakefield, M.A. Secondhand smoke exposure (PM 2.5) in outdoor dining areas and its correlates. Tob. Control 2010, 19, 19–23. [Google Scholar] [CrossRef]
- López, M.J.; Fernández, E.; Gorini, G.; Moshammer, H.; Polanska, K.; Clancy, L.; Dautzenberg, B.; Delrieu, A.; Invernizzi, G.; Muñoz, G. Exposure to secondhand smoke in terraces and other outdoor areas of hospitality venues in eight European countries. PLoS ONE 2012, 7, e42130. [Google Scholar] [CrossRef]
- Sureda, X.; Fernández, E.; López, M.J.; Nebot, M. Secondhand tobacco smoke exposure in open and semi-open settings: A systematic review. Environ. Health Perspect. 2013, 121, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Brennan, E.; Cameron, M.; Warne, C.; Durkin, S.; Borland, R.; Travers, M.J.; Hyland, A.; Wakefield, M.A. Secondhand smoke drift: Examining the influence of indoor smoking bans on indoor and outdoor air quality at pubs and bars. Nicotine Tob. Res. 2010, 12, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.; Wilson, N. Smoking outdoors at pubs and bars: Is it a problem? An air quality study. NZ Med. J. 2011, 124, 27–37. [Google Scholar]
- Israel Central Bureau of Statistics. Statistical Abstract of Israel 2014—No. 2.21; Central Bureau of Statistics: Jerusalem, Israel, 2015. [Google Scholar]
- Jordaan, E.R.; Ehrlich, R.I.; Potter, P. Environmental tobacco smoke exposure in children: Household and community determinants. Arch. Environ. Health Int. J. 1999, 54, 319–327. [Google Scholar] [CrossRef]
- Theitler, N.; Rosen, L. Tobacco Smoke Penetration into Residential Homes in Israel: A Cross-Sectional Survey. M.P.H. Thesis, Tel Aviv University, Tel Aviv-Yafo, Israel, 2019. [Google Scholar]
- Hwang, J.; Lee, K. Determination of outdoor tobacco smoke exposure by distance from a smoking source. Nicotine Tob. Res. 2014, 16, 478–484. [Google Scholar] [CrossRef]
- Rosen, L.; Kislev, S.; Bar-Zeev, Y.; Levine, H. Historic tobacco legislation in Israel: A moment to celebrate. Isr. J. Health Policy Res. 2020, 9, 1–7. [Google Scholar] [CrossRef]
- QS207Tobacco: Treating Dependence; National Institute for Clinical Excellence: London, UK, 2022.
- Health Scotland. A Guide to Smoking Cessation 2010. Addendum on Tobacco Harm Reduction. 2014. Available online: https://www.healthscotland.scot/media/1094/5062_guide-to-smoking-cessation-hr-addendum-cr.pdf (accessed on 9 February 2023).
- Siegel, J. Cabinet Approves Creation of Unit to Fight Tobacco Use. Available online: http://www.jpost.com/HealthAndSci-Tech/Health/Article.aspx?id=222712 (accessed on 9 June 2011).
| Odds Ratio | 95% CI | p-Value | ||
|---|---|---|---|---|
| Model 1 (Bivariable) n = 141 | Location | 0.34 | ||
| GYO a vs. indoors | 0.83 | 0.32–2.17 | ||
| Balcony vs. indoors | 0.54 | 0.23–1.29 | ||
| GYO a vs. balcony | 1.54 | 0.65–3.66 | ||
| Batch | Not estimable | 0.66 | ||
| Model 2 (Multivariable) n = 115 | Location | 0.65 | ||
| GYO a vs. indoors | 1.26 | 0.38–3.92 | ||
| Balcony vs. indoors | 0.74 | 0.25–2.16 | ||
| GYO a vs. balcony | 1.65 | 0.56–4.88 | ||
| Batch | Not included | |||
| Child Age (months) | 0.98 | 0.96–1.00 | 0.06 | |
| Child Sex | 0.50 | |||
| Girl vs. Boy | 0.73 | 0.28–1.86 | ||
| Parental Combined CPD b | 1.03 | 0.97–1.08 | 0.33 | |
| Parental Smokers | 0.65 | |||
| Both parents vs. one parent | 0.81 | 0.31–2.07 | ||
| Parental Nationality | 0.29 | |||
| Both Israeli born vs. not both Israeli born | 0.58 | 0.22–1.58 | ||
| Maternal Education c | 0.86 | |||
| Not academic vs. academic | 1.11 | 0.37–3.30 | ||
| Paternal Education c | 0.001 | |||
| Not academic vs. academic | 5.96 | 2.00–17.81 | ||
| Monthly household income | 0.20 | |||
| Average or below average vs. above average | 0.51 | 0.18–1.42 | ||
| Neighborhood SES | 1.05 | 0.90–1.21 | 0.53 |
| n, Model R2 | Least Squared Mean/Beta [SE] | p-Value | ||
|---|---|---|---|---|
| Model 3 (Bivariable, controlling for batch) | 141, 0.32 | |||
| Location | 0.46 | |||
| GYO a | −2.27 | |||
| Balcony | −2.49 | |||
| Indoors | −2.11 | |||
| Batch | Range:−0.63,−3.86 | <0.001 | ||
| Model 4 (Multivariable) | 115, 0.43 | Location | 0.87 | |
| GYO a | −2.42 | |||
| LSM Balcony | −2.56 | |||
| LSM Indoors | −2.41 | |||
| Batch | Range: −5.06–−0.78 | <0.001 | ||
| Child Age (months) | −0.008 [0.006] | 0.21 | ||
| Child Sex | 0.22 | |||
| Girl | −2.63 | |||
| Boy | −2.29 | |||
| Parental Combined CPD b | 0.03 [0.016] | 0.04 | ||
| Parental Smokers | 0.30 | |||
| Both parents smoke | −2.31 | |||
| One parent smokes | −2.62 | |||
| Parental Nationality | 0.47 | |||
| Both Israeli born | −2.36 | |||
| Not both Israeli born | −2.57 | |||
| Maternal Education c | 0.42 | |||
| Not Academic | −2.59 | |||
| Academic | −2.34 | |||
| Paternal Education c | 0.002 | |||
| Not academic | −1.97 | |||
| Academic | −2.96 | |||
| Monthly household income | 0.67 | |||
| Average or below average | −2.53 | |||
| Above average | −2.40 | |||
| Neighborhood SES | 0.004 [0.045] | 0.92 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosen, L.J.; Zucker, D.M.; Gravely, S.; Bitan, M.; Rule, A.M.; Myers, V. Tobacco Smoke Exposure According to Location of Home Smoking in Israel: Findings from the Project Zero Exposure Study. Int. J. Environ. Res. Public Health 2023, 20, 3523. https://doi.org/10.3390/ijerph20043523
Rosen LJ, Zucker DM, Gravely S, Bitan M, Rule AM, Myers V. Tobacco Smoke Exposure According to Location of Home Smoking in Israel: Findings from the Project Zero Exposure Study. International Journal of Environmental Research and Public Health. 2023; 20(4):3523. https://doi.org/10.3390/ijerph20043523
Chicago/Turabian StyleRosen, Laura J., David M. Zucker, Shannon Gravely, Michal Bitan, Ana M. Rule, and Vicki Myers. 2023. "Tobacco Smoke Exposure According to Location of Home Smoking in Israel: Findings from the Project Zero Exposure Study" International Journal of Environmental Research and Public Health 20, no. 4: 3523. https://doi.org/10.3390/ijerph20043523
APA StyleRosen, L. J., Zucker, D. M., Gravely, S., Bitan, M., Rule, A. M., & Myers, V. (2023). Tobacco Smoke Exposure According to Location of Home Smoking in Israel: Findings from the Project Zero Exposure Study. International Journal of Environmental Research and Public Health, 20(4), 3523. https://doi.org/10.3390/ijerph20043523








