Design of a Computer Model for the Identification of Adolescent Swimmers at Risk of Low BMD
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Study Design
2.2. Anthropometric and Bone Measurements
2.3. Evaluation of Pubertal Stage
2.4. Fitness Assessment
2.5. Performance and Questionnaires
2.6. Statistical Analysis
Decision Tree Modeling
3. Results
3.1. Participant Characteristics
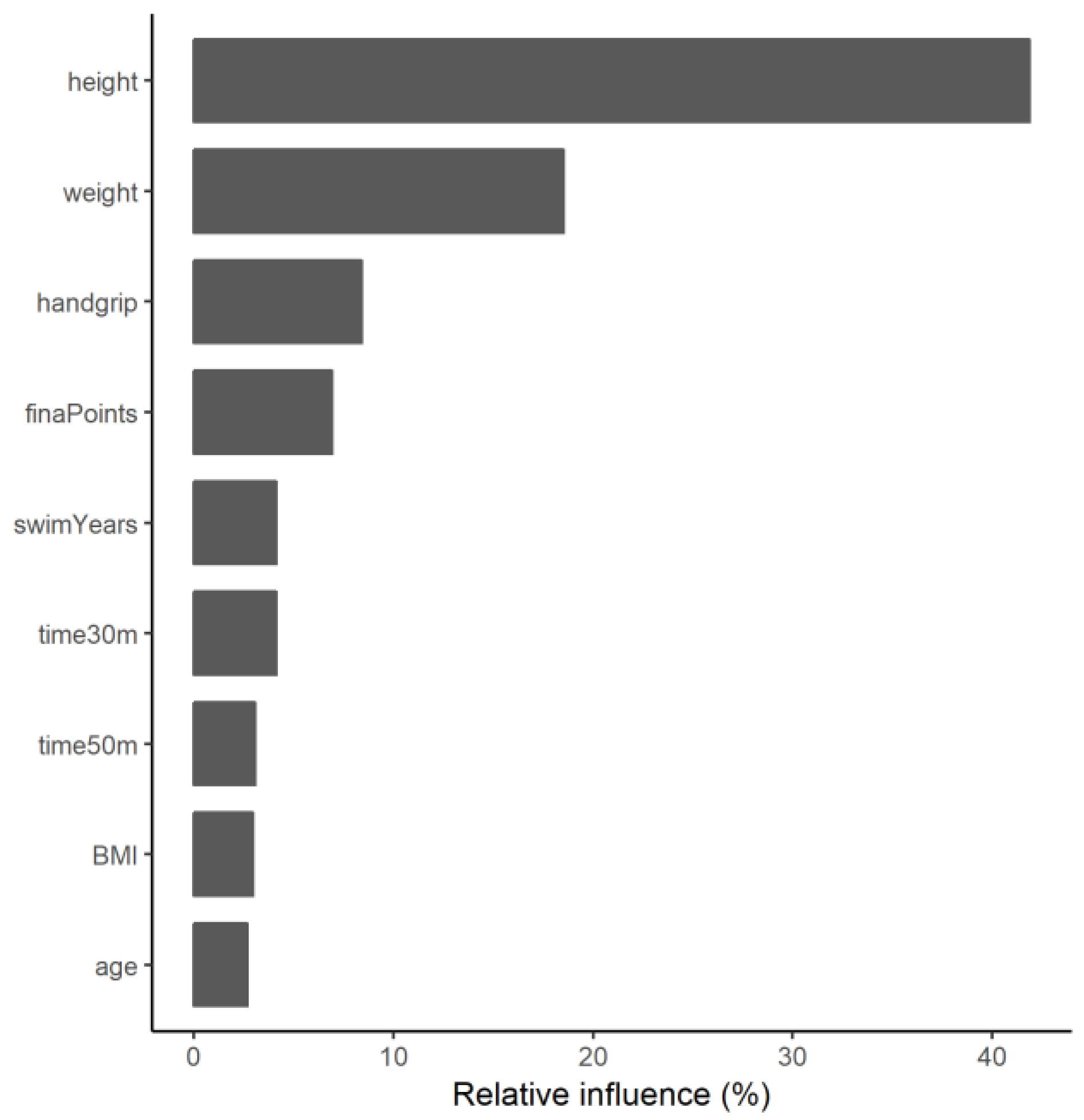
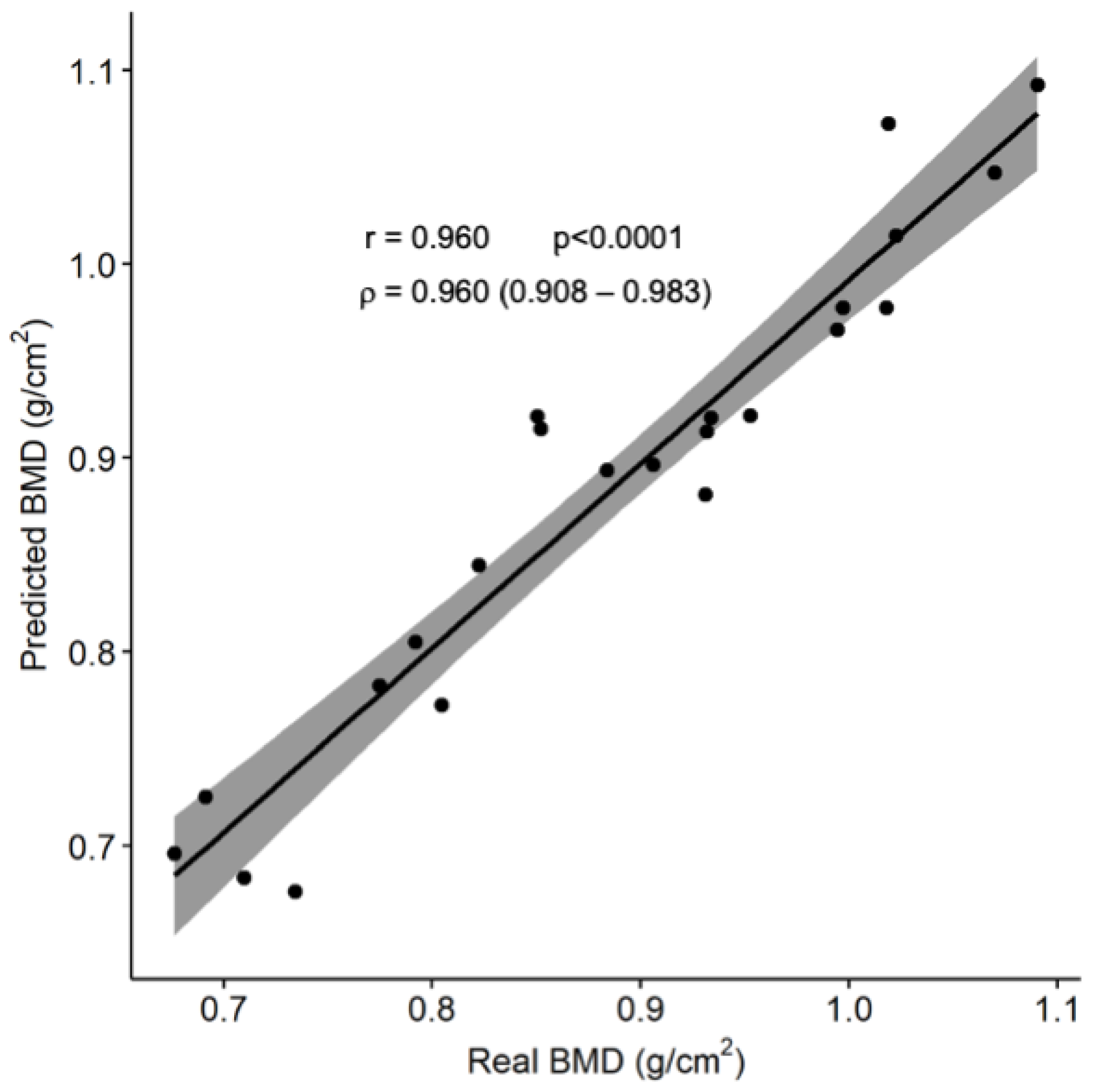
3.2. Gradient-Boosting Machine Regression Tree
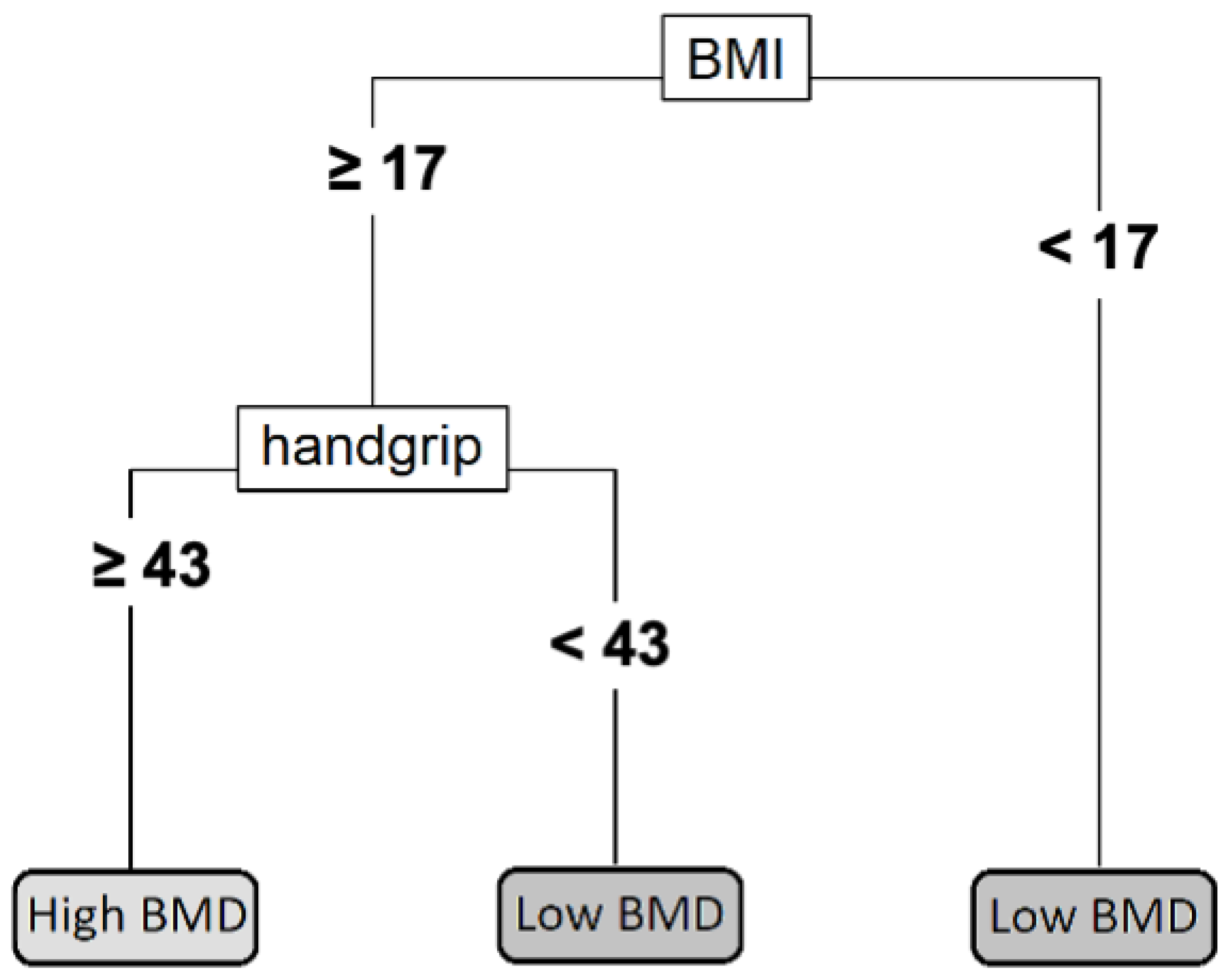
3.3. Individual Decision Tree
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anonymous. Consensus Development Conference: Diagnosis, Prophylaxis, and Treatment of Osteoporosis. Am. J. Med. 1993, 94, 646–650. [Google Scholar] [CrossRef]
- Hernlund, E.; Svedbom, A.; Ivergård, M.; Compston, J.; Cooper, C.; Stenmark, J.; McCloskey, E.V.; Jönsson, B.; Kanis, J.A. Osteoporosis in the European Union: Medical Management, Epidemiology and Economic Burden. A Report Prepared in Collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch. Osteoporos. 2013, 8, 136. [Google Scholar] [CrossRef]
- Daly, R.M.; Petit, M.A. (Eds.) Optimizing Bone Mass and Strength; Karger: Basel, Switzerland, 2007; ISBN 978-3-8055-8275-9. [Google Scholar]
- Bliuc, D.; Nguyen, N.D.; Milch, V.E.; Nguyen, T.V.; Eisman, J.A.; Center, J.R. Mortality Risk Associated with Low-Trauma Osteoporotic Fracture and Subsequent Fracture in Men and Women. JAMA 2009, 301, 513–521. [Google Scholar] [CrossRef]
- Cooper, C.; Cole, Z.A.; Holroyd, C.R.; Earl, S.C.; Harvey, N.C.; Dennison, E.M.; Melton, L.J.; Cummings, S.R.; Kanis, J.A. Secular Trends in the Incidence of Hip and Other Osteoporotic Fractures. Osteoporos. Int. 2011, 22, 1277–1288. [Google Scholar] [CrossRef]
- Hayawi, L.M.; Graham, I.D.; Tugwell, P.; Abdelrazeq, S.Y. Screening for Osteoporosis: A Systematic Assessment of the Quality and Content of Clinical Practice Guidelines, Using the AGREE II Instrument and the IOM Standards for Trustworthy Guidelines. PLoS ONE 2018, 13, e0208251. [Google Scholar] [CrossRef]
- Baxter-Jones, A.D.; Faulkner, R.A.; Forwood, M.R.; Mirwald, R.L.; Bailey, D.A. Bone Mineral Accrual from 8 to 30 Years of Age: An Estimation of Peak Bone Mass. J. Bone Miner. Res. 2011, 26, 1729–1739. [Google Scholar] [CrossRef]
- Bonjour, J.P.; Chevalley, T.; Ferrari, S.; Rizzoli, R. The Importance and Relevance of Peak Bone Mass in the Prevalence of Osteoporosis. Salud Publica Mex. 2009, 51 (Suppl. 1), S5–S17. [Google Scholar] [CrossRef]
- Weaver, C.M.; Gordon, C.M.; Janz, K.F.; Kalkwarf, H.J.; Lappe, J.M.; Lewis, R.; O’Karma, M.; Wallace, T.C.; Zemel, B.S. The National Osteoporosis Foundation’s Position Statement on Peak Bone Mass Development and Lifestyle Factors: A Systematic Review and Implementation Recommendations. Osteoporos. Int. 2016, 27, 1281–1386. [Google Scholar] [CrossRef]
- Gruodyte, R.; Jürimäe, J.; Saar, M.; Jürimäe, T. The Relationships among Bone Health, Insulin-like Growth Factor-1 and Sex Hormones in Adolescent Female Athletes. J. Bone Miner. Metab. 2010, 28, 306–313. [Google Scholar] [CrossRef]
- Gómez-Bruton, A.; Matute-Llorente, Á.; González-Agüero, A.; Casajús, J.A.; Vicente-Rodríguez, G. Plyometric Exercise and Bone Health in Children and Adolescents: A Systematic Review. World J. Pediatr. 2017, 13, 112–121. [Google Scholar] [CrossRef]
- Holroyd, C.; Harvey, N.; Dennison, E.; Cooper, C. Epigenetic Influences in the Developmental Origins of Osteoporosis. Osteoporos. Int. 2012, 23, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Bruton, A.; Montero-Marín, J.; González-Agüero, A.; Gómez-Cabello, A.; García-Campayo, J.; Moreno, L.A.; Casajús, J.A.; Vicente-Rodríguez, G. Swimming and Peak Bone Mineral Density: A Systematic Review and Meta-Analysis. J. Sport. Sci. 2018, 36, 365–377. [Google Scholar] [CrossRef]
- Agostinete, R.R.; Maillane-Vanegas, S.; Lynch, K.R.; Turi-Lynch, B.; Coelho-e-Silva, M.J.; Campos, E.Z.; Cayres, S.U.; Fernandes, R.A. The Impact of Training Load on Bone Mineral Density of Adolescent Swimmers: A Structural Equation Modeling Approach. Pediatr. Exerc. Sci. 2017, 29, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Buttazzoni, C.; Rosengren, B.; Tveit, M.; Landin, L.; Nilsson, J.Å.; Karlsson, M. Preterm Children Born Small for Gestational Age Are at Risk for Low Adult Bone Mass. Calcif. Tissue Int. 2016, 98, 105–113. [Google Scholar] [CrossRef]
- Frost, M.; Petersen, I.; Andersen, T.L.; Langdahl, B.L.; Buhl, T.; Christiansen, L.; Brixen, K.; Christensen, K. Birth Weight and Adult Bone Metabolism Are Unrelated: Results from Birth Weight-Discordant Monozygotic Twins. J. Bone Miner. Res. 2013, 28, 2561–2569. [Google Scholar] [CrossRef]
- Cosman, F.; de Beur, S.J.; LeBoff, M.S.; Lewiecki, E.M.; Tanner, B.; Randall, S.; Lindsay, R. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Osteoporos. Int. 2014, 25, 2359–2381. [Google Scholar] [CrossRef]
- Dell, R.; Greene, D. Is Osteoporosis Disease Management Cost Effective? Curr. Osteoporos. Rep. 2010, 8, 49–55. [Google Scholar] [CrossRef]
- Nelson, H.D.; Haney, E.M.; Dana, T.; Bougatsos, C.; Chou, R. Screening for Osteoporosis: An Update for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2010, 153, 99–111. [Google Scholar] [CrossRef]
- Gomez-Bruton, A.; Gonzalez-Aguero, A.; Casajus, J.A.; Vicente-Rodriguez, G. Swimming Training Repercussion on Metabolic and Structural Bone Development; Benefits of the Incorporation of Whole Body Vibration or Pilometric Training; the RENACIMIENTO Project. Nutr. Hosp. 2014, 30, 399–409. [Google Scholar] [CrossRef]
- Gracia-Marco, L.; Ortega, F.B.; Jimenez-Pavon, D.; Rodriguez, G.; Castillo, M.J.; Vicente-Rodriguez, G.; Moreno, L.A.; Jiménez-Pavón, D.; Rodríguez, G.; Castillo, M.J.; et al. Adiposity and Bone Health in Spanish Adolescents. The HELENA Study. Osteoporos. Int. 2012, 23, 937–947. [Google Scholar] [CrossRef]
- Zemel, B.S.; Kalkwarf, H.J.; Gilsanz, V.; Lappe, J.M.; Oberfield, S.; Shepherd, J.A.; Frederick, M.M.; Huang, X.; Lu, M.; Mahboubi, S.; et al. Revised Reference Curves for Bone Mineral Content and Areal Bone Mineral Density According to Age and Sex for Black and Non-Black Children: Results of the Bone Mineral Density in Childhood Study. J. Clin. Endocrinol. Metab. 2011, 96, 3160–3169. [Google Scholar] [CrossRef] [PubMed]
- Crabtree, N.J.; Arabi, A.; Bachrach, L.K.; Fewtrell, M.; El-Hajj Fuleihan, G.; Kecskemethy, H.H.; Jaworski, M.; Gordon, C.M. Dual-Energy X-ray Absorptiometry Interpretation and Reporting in Children and Adolescents: The Revised 2013 ISCD Pediatric Official Positions. J. Clin. Densitom. 2014, 17, 225–242. [Google Scholar] [CrossRef] [PubMed]
- Tanner, J.M.; Whitehouse, R.H.; Takaishi, M. Standards from Birth to Maturity for Height, Weight, Height Velocity, and Weight Velocity: British Children, 1965. I. Arch. Dis. Child. 1966, 41, 454–471. [Google Scholar] [CrossRef] [PubMed]
- Leone, M.; Comtois, A. Validity and reliability of self-assessment of sexual maturity in elite adolescent athletes. J. Sport. Med. Phys. Fit. 2007, 47, 361–365. [Google Scholar]
- Léger, L.A.; Lambert, J. A Maximal Multistage 20-m Shuttle Run Test to Predict VO2 Max. Eur. J. Appl. Physiol. Occup. Physiol. 1982, 49, 1–12. [Google Scholar] [CrossRef]
- Barr, S.I. Associations of Social and Demographic Variables with Calcium Intakes of High School Students. J. Am. Diet. Assoc. 1994, 94, 260–269. [Google Scholar] [CrossRef]
- Almárcegui, C.J.; Huybrechts, I.; Bruton, A.G.; Llorente, Á.M.; Agüero, A.G.; Cabello, A.G.; Moreno, L.A.; Casajús, J.A.; Rodríguez, G.V. Validity of a Food-Frequency Questionnaire for Estimating Calcium Intake in Adolescent Swimmers. Nutr. Hosp. 2015, 32, 1773–1779. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Therneau, T.; Atkinson, B. Rpart: Recursive Partitioning and Regression Trees. 2021. Available online: https://cran.r-project.org/package=rpart (accessed on 15 January 2023).
- Greenwell, B.; Boehmke, B.; Cunningham, J.; Developers, G. Gbm: Generalized Boosted Regression Models. 2021. Available online: https://github.com/gbm-developers/gbm (accessed on 15 January 2023).
- Friedman, J.; Hastie, T.; Tibshirani, R. Additive Logistic Regression: A Statistical View of Boosting (with Discussion and a Rejoinder by the Authors). Ann. Stat. 2000, 28, 337–407. [Google Scholar] [CrossRef]
- Natekin, A.; Knoll, A. Gradient Boosting Machines, a Tutorial. Front. Neurorobot. 2013, 7, 21. [Google Scholar] [CrossRef]
- Myles, A.J.; Feudale, R.N.; Liu, Y.; Woody, N.A.; Brown, S.D. An Introduction to Decision Tree Modeling. J. Chemom. 2004, 18, 275–285. [Google Scholar] [CrossRef]
- Ortega, F.B.; Silventoinen, K.; Tynelius, P.; Rasmussen, F. Muscular Strength in Male Adolescents and Premature Death: Cohort Study of One Million Participants. BMJ 2012, 345, e7279. [Google Scholar] [CrossRef]
- Gomez-Bruton, A.; Matute-Llorente, A.; Pardos-Mainer, E.; Gonzalez-Aguero, A.; Gomez-Cabello, A.; Casajus, J.A.; Vicente-Rodriguez, G. Factors Affecting Children and Adolescents 50 Meter Performance in Freestyle Swimming—PubMed. J. Sport. Med. Phys. Fit. 2016, 56, 1439–1447. [Google Scholar]
- Crandall, C.J.; Larson, J.; Gourlay, M.L.; Donaldson, M.G.; LaCroix, A.; Cauley, J.A.; Wactawski-Wende, J.; Gass, M.L.; Robbins, J.A.; Watts, N.B.; et al. Osteoporosis Screening in Postmenopausal Women 50 to 64 Years Old: Comparison of US Preventive Services Task Force Strategy and Two Traditional Strategies in the Women’s Health Initiative. J. Bone Miner. Res. 2014, 29, 1661–1666. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; McCloskey, E.V.; Johansson, H.; Oden, A.; Ström, O.; Borgström, F. Development and Use of FRAX in Osteoporosis. Osteoporos. Int. 2010, 21 (Suppl. 2), 407–413. [Google Scholar] [CrossRef] [PubMed]
- Lydick, E.; Cook, K.; Turpin, J.; Melton, M.; Stine, R.; Byrnes, C. Development and Validation of a Simple Questionnaire to Facilitate Identification of Women Likely to Have Low Bone Density—PubMed. Am. J. Manag. Care 1998, 4, 37–48. [Google Scholar]
- Koh, L.K.H.; Ben Sedrine, W.; Torralba, T.P.; Kung, A.; Fujiwara, S.; Chan, S.P.; Huang, Q.R.; Rajatanavin, R.; Tsai, K.S.; Park, H.M.; et al. A Simple Tool to Identify Asian Women at Increased Risk of Osteoporosis. Osteoporos. Int. 2001, 12, 699–705. [Google Scholar] [CrossRef]
- O’Donnell, S.; Osteoporosis Surveillance Expert Working Group. Screening, Prevention and Management of Osteoporosis among Canadian Adults. Health Promot. Chronic Dis. Prev. Can. 2018, 38, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, W.T.; Newman, E.D.; Blosky, M.A.; Stewart, W.F.; Wood, G.C. Improving Detection and Treatment of Osteoporosis: Redesigning Care Using the Electronic Medical Record and Shared Medical Appointments. Osteoporos. Int. 2009, 20, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Geusens, P.; Dumitrescu, B.; Van Geel, T.; Van Helden, S.; Vanhoof, J.; Dinant, G.J. Impact of Systematic Implementation of a Clinical Case Finding Strategy on Diagnosis and Therapy of Postmenopausal Osteoporosis. J. Bone Miner. Res. 2008, 23, 812–818. [Google Scholar] [CrossRef]
| Variables | Overall (n = 78) | Training (n = 55) | Testing (n = 23) |
|---|---|---|---|
| General characteristics and anthropometric variables | |||
| Sex (male/female) | 40/38 | 26/29 | 14/9 |
| Tanner stage (I/II/III/IV/V) | 2/18/16/38/7 | 1/10/13/27/4 | 1/6/3/11/2 |
| Age (years) | 14.3 ± 1.9 | 14.3 ± 1.8 | 14.3 ± 2.2 |
| Height (cm) | 163.8 ± 12.0 | 163.1 ± 11.3 | 165.4 ± 13.5 |
| Weight (kg) | 54.3 ± 12.1 | 53.3 ± 11.2 | 56.7 ± 13.9 |
| BMI (kg/m2) | 20.0 ± 2.5 | 19.8 ± 2.3 | 20.4 ± 2.8 |
| Bone mineral variables | |||
| Subtotal BMC (g) | 1388 ± 416 | 1350 ± 392 | 1481 ± 465 |
| Subtotal BMD (g/cm2) | 0.867 ± 0.113 | 0.858 ± 0.108 | 0.889 ± 0.123 |
| Total hip BMC (g) | 29.9 ± 9.1 | 28.9 ± 8.2 | 32.1 ± 10.6 |
| Total hip BMD (g/cm2) | 0.888 ± 0.133 | 0.879 ± 0.136 | 0.907 ± 0.126 |
| Variables | Overall (n = 78) | Low BMD (n = 24) | Not Low BMD (n = 54) |
|---|---|---|---|
| Fitness | |||
| Handgrip strength (kg) | 53.5 ± 17.3 | 46.3 ± 16.1 | 56.7 ± 17.0 * |
| Long jump (cm) | 185.6 ± 30.4 | 174.3 ± 31.8 | 191.0 ± 28.5 * |
| 30-m run (s) | 5.21 ± 0.48 | 5.33 ± 0.42 | 5.15 ± 0.50 |
| VO2max (mL/kg·min) | 50.4 ± 5.3 | 49.3 ± 5.0 | 51.0 ± 5.3 |
| Training and performance | |||
| Weekly training (h) | 10.0 ± 2.1 | 9.8 ± 2.1 | 10.0 ± 2.1 |
| Swimming history (years) | 7.9 ± 2.9 | 7.6 ± 2.7 | 8.0 ± 3.0 |
| 50-m swim (s) | 31.2 ± 3.5 | 32.8 ± 4.4 | 30.5 ± 2.8 * |
| FINA points | 354 ± 84 | 317 ± 82 | 370 ± 80 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marin-Puyalto, J.; Gomez-Cabello, A.; Gomez-Bruton, A.; Matute-Llorente, A.; Castillo-Bernad, S.; Lozano-Berges, G.; Gonzalez-Agüero, A.; Casajus, J.A.; Vicente-Rodriguez, G. Design of a Computer Model for the Identification of Adolescent Swimmers at Risk of Low BMD. Int. J. Environ. Res. Public Health 2023, 20, 3454. https://doi.org/10.3390/ijerph20043454
Marin-Puyalto J, Gomez-Cabello A, Gomez-Bruton A, Matute-Llorente A, Castillo-Bernad S, Lozano-Berges G, Gonzalez-Agüero A, Casajus JA, Vicente-Rodriguez G. Design of a Computer Model for the Identification of Adolescent Swimmers at Risk of Low BMD. International Journal of Environmental Research and Public Health. 2023; 20(4):3454. https://doi.org/10.3390/ijerph20043454
Chicago/Turabian StyleMarin-Puyalto, Jorge, Alba Gomez-Cabello, Alejandro Gomez-Bruton, Angel Matute-Llorente, Sergio Castillo-Bernad, Gabriel Lozano-Berges, Alejandro Gonzalez-Agüero, Jose A. Casajus, and German Vicente-Rodriguez. 2023. "Design of a Computer Model for the Identification of Adolescent Swimmers at Risk of Low BMD" International Journal of Environmental Research and Public Health 20, no. 4: 3454. https://doi.org/10.3390/ijerph20043454
APA StyleMarin-Puyalto, J., Gomez-Cabello, A., Gomez-Bruton, A., Matute-Llorente, A., Castillo-Bernad, S., Lozano-Berges, G., Gonzalez-Agüero, A., Casajus, J. A., & Vicente-Rodriguez, G. (2023). Design of a Computer Model for the Identification of Adolescent Swimmers at Risk of Low BMD. International Journal of Environmental Research and Public Health, 20(4), 3454. https://doi.org/10.3390/ijerph20043454










