Effects of Transcranial Pulse Stimulation (TPS) on Adults with Symptoms of Depression—A Pilot Randomized Controlled Trial
Abstract
1. Introduction
1.1. Conventional Treatment Approach towards Depression in Psychiatry
1.2. Non-Intrusive Brain Stimulation (NIBS)
1.3. How Does TPS Work on the Human Brain?
1.4. Biological Mechanism of TPS
1.5. Clinical Effects of TPS
1.6. Past Research on TPS
1.7. Research Gap
1.8. Objectives of This Study
2. Materials and Methods
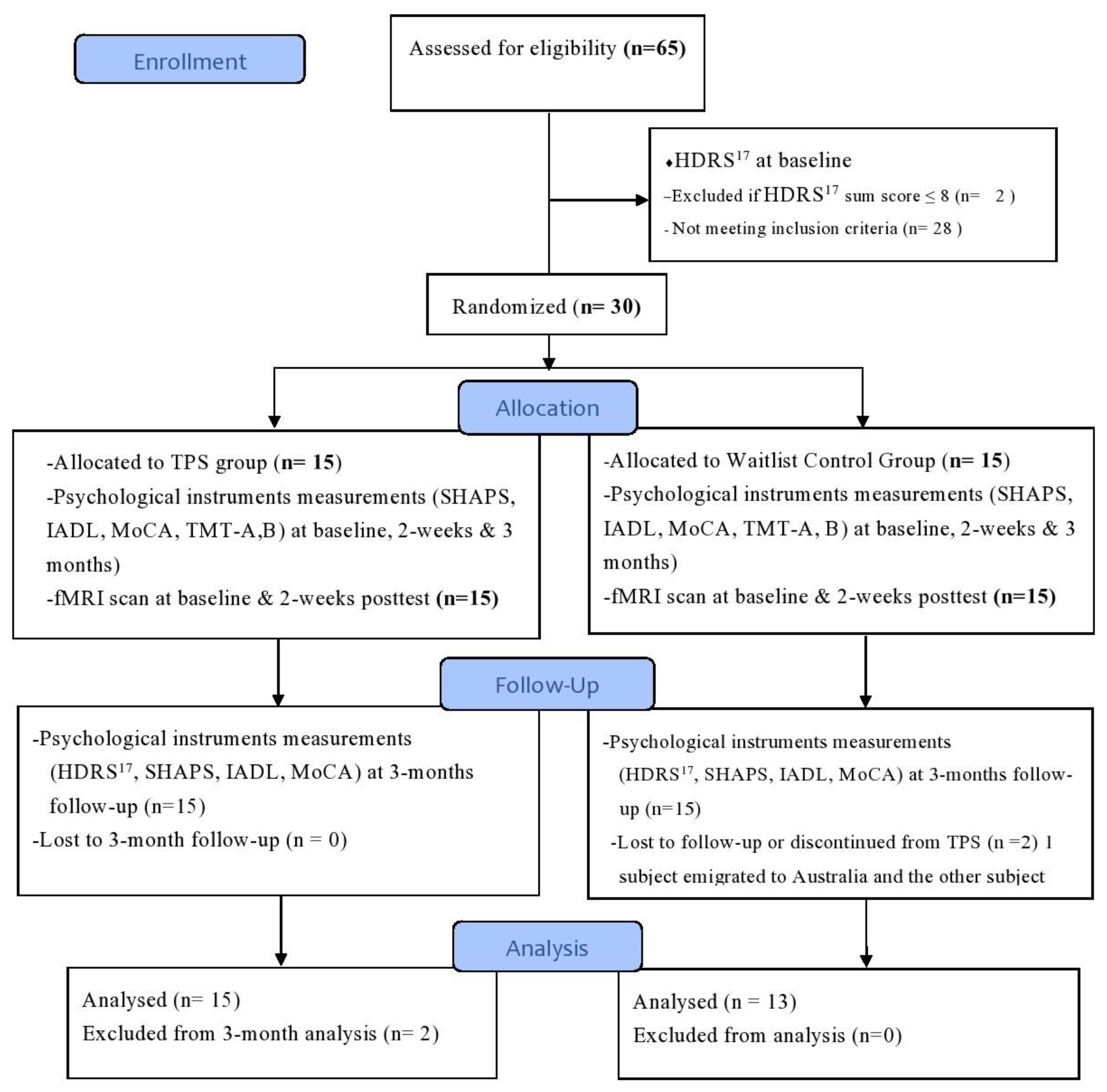
2.1. Study Design
2.2. Sample Size Estimation
2.3. Subject Recruitment
2.4. Subjects
2.5. Randomization, Allocation, and Masking
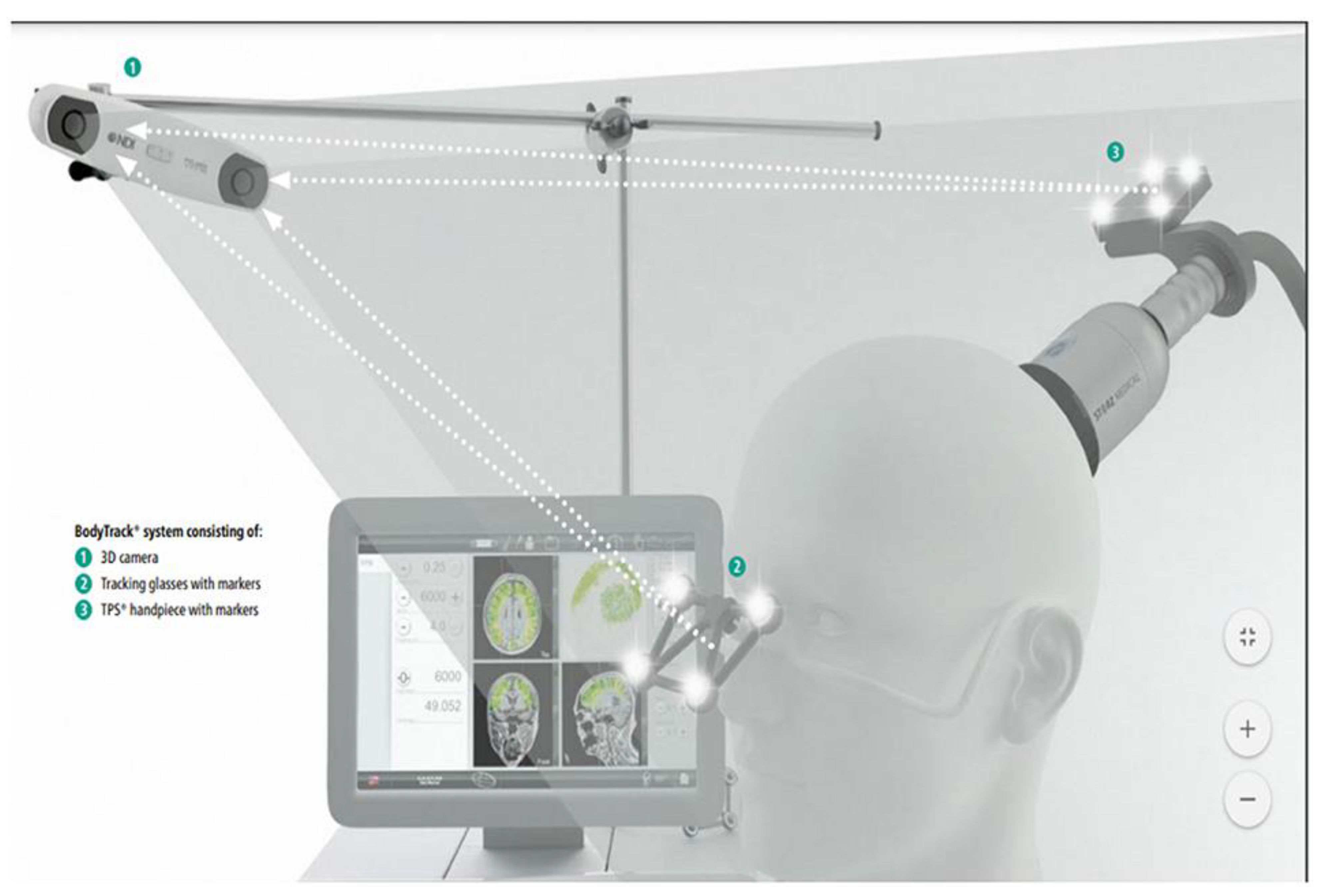
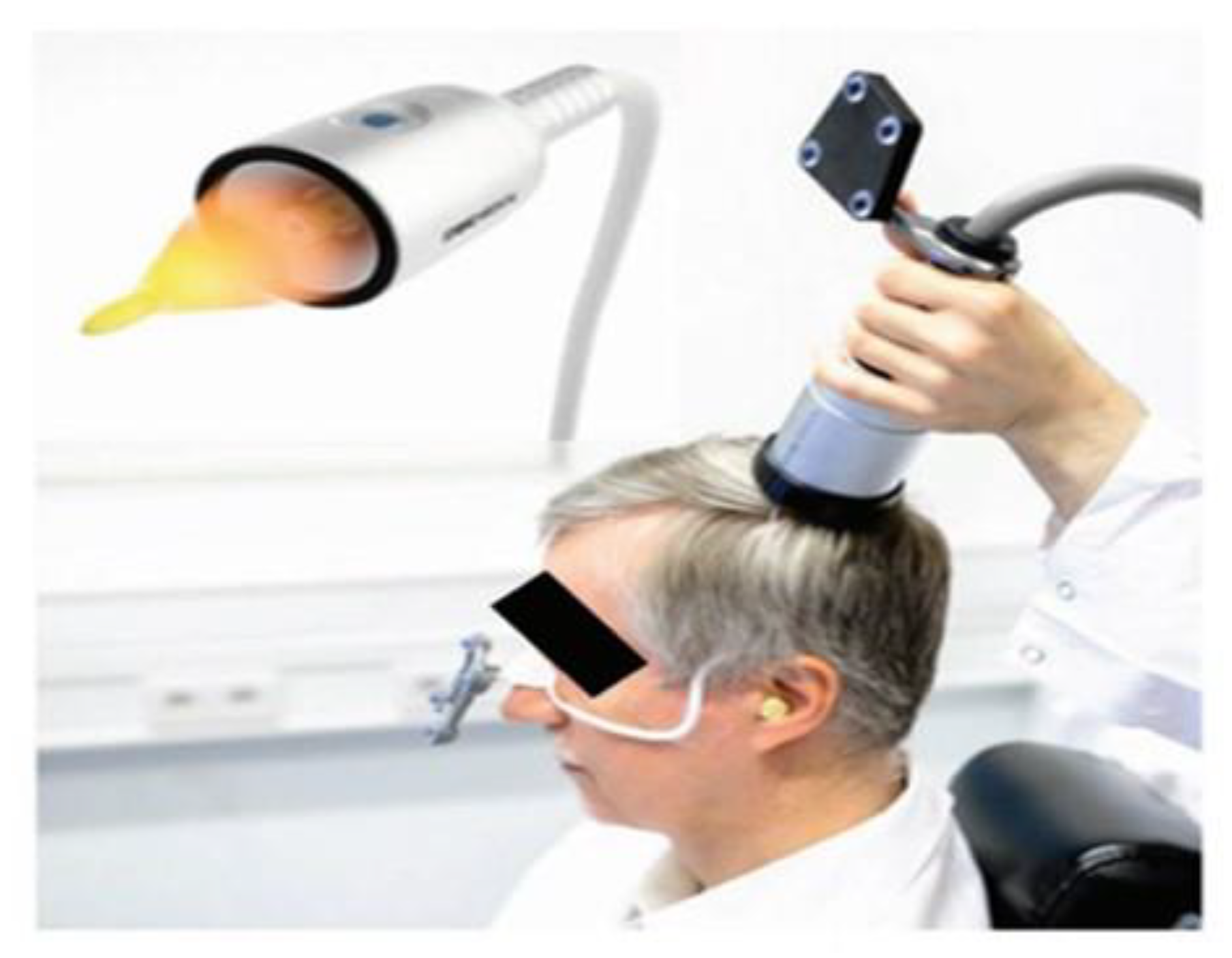
2.6. Intervention (Transcranial Pulse Stimulation)
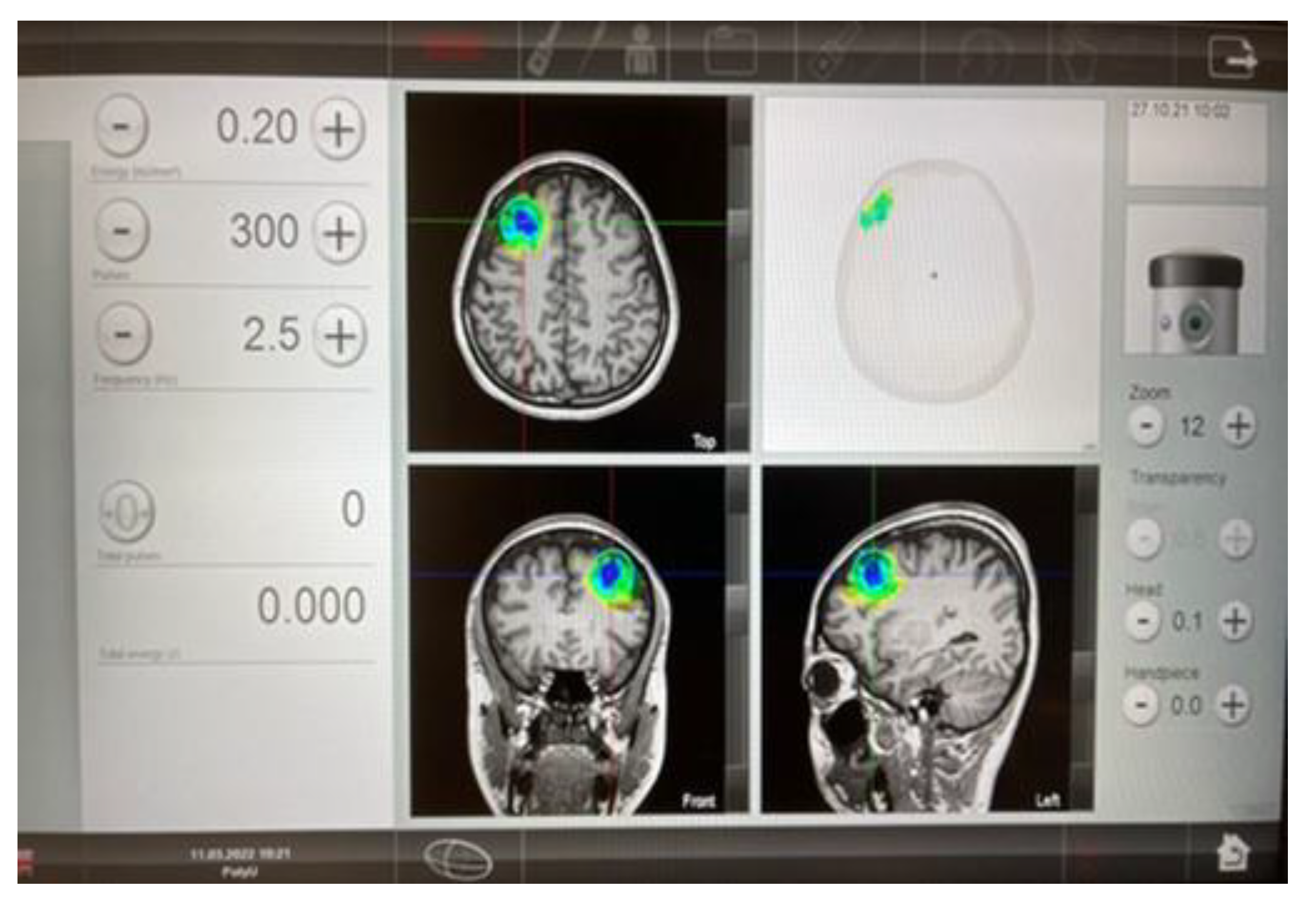
2.6.1. TPS Procedures
2.6.2. Intervention Dose
2.7. Ethical Considerations
2.8. Baseline Assessments
2.9. Outcome Measurements
2.10. Safety Issues, Adverse Effects, and Risk Indicators of TPS
2.11. Statistical Analysis
3. Results
3.1. Sociodemographic Characteristics between the TPS Group and the Waitlist Control (WC) Group
3.2. Effect of the TPS Intervention on Depression
3.3. Differences in Secondary Outcomes between Baseline and Post-Test
3.4. Differences in Primary and Secondary Outcomes between Baseline, Post-Test, and at the 3-Month Follow-Up
4. Discussion
4.1. Cognition
4.2. Anhedonia
4.3. Instrumental Activities of Daily Living (IADL), Working Memory, and Executive Function
4.4. Limitations of This Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fong, B.Y.F.; Wong, M.C.S.; Law, V.T.S.; Lo, M.F.; Ng, T.K.C.; Yee, H.H.L.; Leung, T.C.H.; Ho, P.W.T. Relationships between Physical and Social Behavioural Changes and the Mental Status of Homebound Residents in Hong Kong During the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2020, 17, 6653. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.P.H.; Hui, B.P.H.; Wan, E.Y.F. Depression and Anxiety in Hong Kong during COVID-19. Int. J. Environ. Res. Public Health 2020, 17, 3740. [Google Scholar] [CrossRef] [PubMed]
- Cheung, T.; Lam, S.C.; Lee, P.H.; Xiang, Y.T.; Yip, P.S.F. The International Research Collaboration on COVID-19 Global Imperative of Suicidal Ideation in 10 Countries Amid the COVID-19 Pandemic. Front. Psychiatry 2020, 11, 588781. [Google Scholar] [CrossRef] [PubMed]
- Hamasha Sd Ahmed, A.; Ali, H.; Cole, A.; Panos, G. Major Stressors and Coping Strategies of Frontline Nursing Staff during the Outbreak of Coronavirus Disease 2020 (COVID-19) in Alabama. J. Multidiscip. Healthc. 2021, 14, 2057–2068. [Google Scholar]
- Shreffler, J.; Petrey, J.; Huecker, M. The Impact of COVID-19 on Healthcare Worker Wellness: A Scoping Review. West J. Emerg. Med. 2020, 21, 1059–1066. [Google Scholar] [CrossRef]
- Robb, C.E.; de Jager, C.A.; Ahmadi-Abhari, S.; Giannakopoulou, P.; Udeh-Momoh, C.; McKeand, J.; Price, G.; Car, J.; Majeed, A.; Ward, H.; et al. Associations of Social Isolation with Anxiety and Depression During the Early COVID-19 Pandemic: A Survey of Older Adults in London, UK. Front. Psychiatry 2020, 11, 591120. [Google Scholar] [CrossRef]
- Xiang, Y.-T.; Yang, Y.; Li, W.; Zhang, L.; Zhang, Q.; Cheung, T.; Ng, C.H. Timely mental health care for the 2019 novel coronavirus outbreak is urgently needed. Lancet Psychiatry 2020, 7, 228–229. [Google Scholar] [CrossRef] [PubMed]
- Djillani, A.; Pietri, M.; Mazella, J.; Heurteaux, C.; Borsotto, M. Fighting against depression with TREK-1 blockers: Past and future. A focus on spadin. Pharmacol. Ther. 2019, 194, 185–198. [Google Scholar] [CrossRef]
- Rocha, R.B.; Dondossola, E.R.; Grande, A.J.; Colonetti, T.; Ceretta, L.B.; Passos, I.C.; Quevedo, J.; da Rosa, M.I. Increased BDNF levels after electroconvulsive therapy in patients with major depressive disorder: A meta-analysis study. J. Psychiatr. Res. 2016, 83, 47–53. [Google Scholar] [CrossRef]
- Cheng, P.; Luik, A.I.; Fellman-Couture, C.; Peterson, E.; Joseph, C.L.; Tallent, G.; Tran, K.M.; Ahmedani, B.K.; Roehrs, T.; Roth, T.; et al. Efficacy of digital CBT for insomnia to reduce depression across demographic groups: A randomized trial. Psychol. Med. 2019, 49, 491–500. [Google Scholar] [CrossRef]
- Loo, C.K.; Husain, M.M.; McDonald, W.M.; Aaronson, S.; O’Reardon, J.P.; Alonzo, A.; Weickert, C.S.; Martin, D.M.; McClintock, S.M.; Mohan, A.; et al. International randomized-controlled trial of transcranial Direct Current Stimu-lation in depression. Brain Stimul. 2018, 11, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Stubbeman, W.F.; Zarrabi, B.; Bastea, S.; Rosenbaum, K.; Ragland, V.E.; Khairkhah, R. Transcranial magnetic stimulation treatment of depression using 20 hertz theta burst pulse parameters. Brain Stimul. 2018, 11, e8. [Google Scholar] [CrossRef]
- Fitzgerald, P.B.; Hoy, K.E.; Reynolds, J.; Singh, A.; Gunewardene, R.; Slack, C.; Ibrahim, S.; Daskalakis, Z.J. A pragmatic randomized controlled trial exploring the relationship between pulse number and response to repetitive transcranial magnetic stimulation treatment in depression. Brain Stimul. 2020, 13, 145–152. [Google Scholar] [CrossRef]
- Lee, H.J.; Kim, S.M.; Kwon, J.Y. Repetitive transcranial magnetic stimulation treatment for peripartum depression: Systematic review & meta-analysis. BMC Pregnancy Childbirth 2021, 21, 118. [Google Scholar] [CrossRef]
- Beisteiner, R.; Matt, E.; Fan, C.; Baldysiak, H.; Schönfeld, M.; Philippi Novak, T.; Amini, A.; Aslan, T.; Reinecke, R.; Lehrner, J.; et al. Transcranial Pulse Stimulation with Ultrasound in Alzheimer’s Disease—A New Navigated Focal Brain Therapy. Adv. Sci. 2020, 7, 1902583. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.Z.; Wong, J.Y.H.; Luk, T.T.; Wai, A.K.C.; Lam, T.H.; Wang, M.P. Mental health crisis under COVID-19 pandemic in Hong Kong, China. Int. J. Infect. Dis. 2020, 100, 431–433. [Google Scholar] [CrossRef] [PubMed]
- Minjoli, S.; Saturnino, G.B.; Blicher, J.U.; Stagg, C.J.; Siebner, H.R.; Antunes, A.; Thielscher, A. The impact of large structural brain changes in chronic stroke patients on the electric field caused by transcranial brain stimulation. NeuroImage: Clin. 2017, 15, 106–117. [Google Scholar] [CrossRef]
- Spagnolo, P.A.; Wang, H.; Srivanitchapoom, P.; Schwandt, M.; Heilig, M.; Hallett, M. Lack of Target Engagement Following Low-Frequency Deep Transcranial Magnetic Stimulation of the Anterior Insula. Neuromodulation Technol. Neural Interface 2019, 22, 877–883. [Google Scholar] [CrossRef]
- Legon, W.; Ai, L.; Bansal, P.; Mueller, J.K. Neuromodulation with single-element transcranial focused ultrasound in human thalamus. Hum. Brain Mapp. 2018, 39, 1995–2006. [Google Scholar] [CrossRef]
- Hatanaka, K.; Ito, K.; Shindo, T.; Kagaya, Y.; Ogata, T.; Eguchi, K.; Kurosawa, R.; Shimokawa, H. Molecular mechanisms of the angiogenic effects of low-energy shock wave therapy: Roles of mechanotransduction. Am. J. Physiol. Physiol. 2016, 311, C378–C385. [Google Scholar] [CrossRef]
- Matsuda, M.; Kanno, H.; Sugaya, T.; Yamaya, S.; Yahata, K.; Handa, K.; Shindo, T.; Shimokawa, H.; Ozawa, H.; Itoi, E. Low-energy extracorporeal shock wave therapy promotes BDNF expression and im-proves functional recovery after spinal cord injury in rats. Exp. Neurol. 2020, 328, 113251. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Ning, H.; Reed-Maldonado, A.B.; Zhou, J.; Ruan, Y.; Zhou, T.; Wang, H.S.; Oh, B.S.; Banie, L.; Lin, G.; et al. Low-Intensity Extracorporeal Shock Wave Therapy Enhances Brain-Derived Neurotrophic Factor Expression through PERK/ATF4 Signaling Pathway. Int. J. Mol. Sci. 2017, 18, 433. [Google Scholar] [CrossRef] [PubMed]
- Ingber, D.E. Cellular mechanotransduction, putting all the pieces together again. FASEB J. 2006, 20, 811–827. [Google Scholar] [CrossRef] [PubMed]
- D’Agostino, M.C.; Craig, K.; Tibalt, E.; Respizzi, S. Shock wave as biological therapeutic tool, From mechanical stimulation to recovery and healing, through mechanotransduction. Int. J. Surg. 2015, 24, 147–153. [Google Scholar] [CrossRef]
- Zhang, J.; Kang, N.; Yu, X.; Ma, Y.; Pang, X. Radial Extracorporeal Shock Wave Therapy Enhances the Proliferation and Differen-tiation of Neural Stem Cells by Notch, PI3K/AKT, and Wnt/β-catenin Signaling. Sci. Rep. 2017, 7, 15321. [Google Scholar] [CrossRef]
- Mariotto, S.; Cavalieri, E.; Amelio, E.; Ciampa, A.R.; de Prati, A.C.; Marlinghaus, E.; Russo, S.; Suzuki, H. Extracorporeal shock waves: From lithotripsy to anti-inflammatory action by NO production. Nitric Oxide 2005, 12, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Monteleone, P.; Serritella, C.; Martiadis, V.; Maj, M. Decreased levels of serum brain-derived neurotrophic factor in both depressed and euthymic patients with unipolar depression and in euthymic patients with bipolar I and II disorders. Bipolar Disord. 2008, 10, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Taliaz, D.; Stall, N.; Dar, D.E.; Zangen, A. Knockdown of brain-derived neurotrophic factor in specific brain sites precipitates behaviors associated with depression and reduces neurogenesis. Mol. Psychiatry 2010, 15, 80–92. [Google Scholar] [CrossRef]
- Legon, W.; Sato, T.F.; Opitz, A.; Mueller, J.; Barbour, A.; Williams, A.; Tyler, W.J. Transcranial focused ultrasound modulates the activity of primary somatosensory cortex in humans. Nat. Neurosci. 2014, 17, 322–329. [Google Scholar] [CrossRef]
- Lohse-Busch, H.; Reime, U.; Falland, R. Symptomatic treatment of unresponsive wakefulness syndrome with transcranially focused extracorporeal shock waves. Neurorehabilitation 2014, 35, 235–244. [Google Scholar] [CrossRef]
- Beisteiner, R.; Lozano, A.M. Transcranial Ultrasound Innovations Ready for Broad Clinical Application. Adv. Sci. 2020, 7, 2002026. [Google Scholar] [CrossRef] [PubMed]
- Bragazzi, N.L. The COVID-19 Pandemic Seen from a Syndemic Perspective: The LGBTQIA2SP+ Community. Infect. Dis. Rep. 2021, 13, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement, Updated Guidelines for Reporting Parallel Group Randomized Trials. Open Med. 2010, 115, 1063–1070. [Google Scholar] [CrossRef]
- Patterson, B.; Boyle, M.H.; Kivlenieks, M.; van Ameringen, M. The use of waitlists as control conditions in anxiety disorders re-search. J. Psychiatr. Res. 2016, 83, 112–120. [Google Scholar] [CrossRef]
- Grischott, T. The Shiny Balancer—Software and imbalance criteria for optimally balanced treatment allocation in small RCTs and cRCTs. BMC Med. Res. Methodol. 2018, 18, 108. [Google Scholar] [CrossRef]
- Grimm, S.; Beck, J.; Schuepbach, D.; Hell, D.; Boesiger, P.; Bermpohl, F.; Niehaus, L.; Boeker, H.; Northoff, G. Imbalance between Left and Right Dorsolateral Prefrontal Cortex in Major Depression Is Linked to Negative Emotional Judgment, An fMRI Study in Severe Major Depressive Disorder. Biol. Psychiatry 2008, 63, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Sheng, J.; Li, B.; Zhang, X. Recent Advances in Non-invasive Brain Stimulation for Major Depressive Disorder. Front. Hum. Neurosci. 2017, 11, 526. [Google Scholar] [CrossRef]
- Hamilton, M. Development of a Rating Scale for Primary Depressive Illness. Br. J. Soc. Clin. Psychol. 1967, 6, 278–296. [Google Scholar] [CrossRef]
- Liu, W.; Wang, L.; Zhu, Y.; Li, M.; Chan, R.C.K. Clinical utility of the Snaith-Hamilton-Pleasure scale in the Chinese settings. BMC Psychiatry 2012, 12, 184. [Google Scholar] [CrossRef]
- Tong, A.Y.; Man, D.W. The Validation of the Hong Kong Chinese Version of the Lawton Instrumental Activities of Daily Living Scale for Institutionalized Elderly Persons. OTJR Occup. Particip. Health 2002, 22, 132–142. [Google Scholar] [CrossRef]
- Yeung, P.Y.; Wong, L.L.; Chan, C.C.; Yung, C.Y.; Leung, L.J.; Tam, Y.Y.; Nin Tang, L.; Li, H.S.; Lau, M.L. Montreal Cognitive Assessment—Single Cutoff Achieves Screening Purpose. Neuropsychiatr. Dis. Treat. 2020, 16, 2681–2687. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Routledge: New York, NY, USA, 1988. [Google Scholar]
- Begemann, M.J.; Brand, B.A.; Ćurčić-Blake, B.; Aleman, A.; Sommer, I.E. Efficacy of non-invasive brain stimulation on cognitive functioning in brain disorders, a meta-analysis. Psychol. Med. 2020, 50, 2465–2486. [Google Scholar] [CrossRef] [PubMed]
- Höflich, A.; Michenthaler, P.; Kasper, S.; Lanzenberger, R. Circuit Mechanisms of Reward, Anhedonia, and Depression. Int. J. Neuropsychopharmacol. 2019, 22, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, A.M.; Kang, J.W.D.; Gobin, A.P.; Tirrell, E.; Kokdere, F.; Carpenter, L.L. Effects of transcranial magnetic stimulation on an-hedonia in treatment resistant major depressive disorder. Brain Behav. 2021, 11, e2329. [Google Scholar] [CrossRef]
- Gonda, X.; Pompili, M.; Serafini, G.; Carvalho, A.F.; Rihmer, Z.; Dome, P. The role of cognitive dysfunction in the symptoms and remission from depression. Ann. Gen. Psychiatry 2015, 14, 27. [Google Scholar] [CrossRef] [PubMed]
- Knight, M.J.; Baune, B.T. Psychosocial Dysfunction in Major Depressive Disorder—Rationale, Design, and Characteristics of the Cognitive and Emotional Recovery Training Program for Depression (CERT-D). Front. Psychiatry 2017, 8, 280. [Google Scholar] [CrossRef]
- Vinckier, F.; Gourion, D.; Mouchabac, S. Anhedonia predicts poor psychosocial functioning, Results from a large cohort of pa-tients treated for major depressive disorder by general practitioners. Eur. Psychiatry 2017, 44, 1–8. [Google Scholar] [CrossRef]
- Liu, Y.; Yin, M.; Luo, J.; Huang, L.; Zhang, S.; Pan, C.; Hu, X. Effects of transcranial magnetic stimulation on the performance of the activities of daily living and attention function after stroke: A randomized controlled trial. Clin. Rehabil. 2020, 34, 1465–1473. [Google Scholar] [CrossRef]
- Popescu, T.; Pernet, C.; Beisteiner, R. Transcranial ultrasound pulse stimulation reduces cortical atrophy in Alzheimer’s patients, A follow-up study. Alzheimers Dement. 2021, 7, e12121. [Google Scholar] [CrossRef]
| IG (n = 15) | WC (n = 15) | ||
|---|---|---|---|
| Mean (SD)/n (%) | p | ||
| Age | 38.8 (15.0) | 34.3 (16.5) | 0.44 |
| Gender | >0.99 | ||
| Male | 4 (27) | 4 (27) | |
| Female | 11 (73) | 11 (73) | |
| Living with family members | 2.53 (1.19) | 2.8 (1.47) | 0.59 |
| Education level | 0.1 | ||
| Elementary or below | 1 (7) | 1 (7) | |
| High school | 0 (0) | 4 (27) | |
| University or above | 14 (93) | 10 (67) | |
| Marital status | 0.5 | ||
| Single | 6 (40) | 8 (53) | |
| In a relationship | 3 (20) | 1 (7) | |
| Married | 5 (33) | 6 (40) | |
| Divorced/separated | 1 (7) | 0 (0) | |
| Widowed | 0 (0) | 0 (0) | |
| Occupation | 0.41 | ||
| Administrative/clerical staff | 1 (7) | 0 (0) | |
| Managerial staff | 1 (7) | 0 (0) | |
| Casual worker | 0 (0) | 1 (7) | |
| Students (full-time) | 5 (33) | 6 (40) | |
| Housewife | 0 (0) | 2 (13) | |
| Licensed professionals | 3 (20) | 1 (7) | |
| Retirees | 0 (0) | 1 (7) | |
| Unemployed | 5 (33) | 4 (27) | |
| Income (HKD) | 0.16 | ||
| ≤20,000 | 4 (27) | 8 (53) | |
| >20,000–49,999 | 4 (27) | 5 (33) | |
| >50,000–79,999 | 4 (27) | 2 (13) | |
| ≥80,000 | 3 (20) | 0 (0) | |
| Chronic illness | >0.99 | ||
| Yes | 1 (7) | 1 (7) | |
| No | 14 (93) | 14 (93) | |
| Psychiatric history (personal) | >0.99 | ||
| Yes | 15 (100) | 15 (100) | |
| No | 0 (0) | 0 (0) | |
| Duration of having major depressive disorder (in months) | 98 (113) | 48.4 (38.3) | 0.12 |
| Currently taking prescribed antidepressants | 0.08 | ||
| Yes | 9 (60) | 14 (93) | |
| No | 6 (40) | 1 (7) | |
| Duration of taking prescribed antidepressants (in months) | 33.5 (48.5) | 39.1 (34.8) | 0.72 |
| Time Points | Intervention (n = 15) | Control (n = 15) | |||
|---|---|---|---|---|---|
| Mean (SD) | Mean Difference | p | d | ||
| Pre-test | 25.73 (9.45) | 21.60 (8.70) | 4.13 | 0.15 | |
| Post-test | 13.20 (7.24) | 19.80 (6.89) | −6.60 | 0.02 | −0.93 |
| Secondary Outcomes | Baseline | Post-Test | Mean Difference | p | d |
|---|---|---|---|---|---|
| Cognition | 26.03 (3.74) | 28.7 (1.97) | 2.64 | 0.003 | 0.88 |
| Trail making test-A | 11.22 (8.35) | 8.31 (6.05) | −2.91 | <0.001 | −0.40 |
| Trail making test-B | 35.9 (25.40) | 33.7 (23.7) | −2.15 | 0.07 | −0.09 |
| IADL | 23.0 (5.42) | 24.8 (3.46) | 1.83 | <0.001 | 0.40 |
| Anhedonia | 20.8 (9.02) | 14.8 (7.75) | −6.0 | <0.001 | −0.79 |
| DS_Forward | 11.9 (2.40) | 12.8 (1.91) | 0.93 | 0.003 | 0.43 |
| DS_Backward | 8.03 (2.67) | 10.2 (3.44) | 2.14 | <0.001 | 0.69 |
| Baseline (T0) | Post Test (T1) | 3-Month Follow-Up (T2) | Post Hoc Test | Effect Size | |||
|---|---|---|---|---|---|---|---|
| n | Mean (SD) | p | d (T2-T0) | ||||
| HDRS-17 | 28 | 20.9 (7.73) | 12.1 (8.03) | 11.0 (6.86) | <0.001 | T0 > T1 = T2 | −1.35 |
| Global cognition | 28 | 26.0 (3.74) | 28.7 (1.97) | 29.4 (1.42) | <0.001 | T0 < T1 = T2 | 1.20 |
| Trail making test-A | 28 | 11.2 (8.35) | 8.31 (6.05) | 7.04 (3.89) | <0.001 | T0 > T1 = T2 | −0.64 |
| Trail making test-B | 28 | 35.9 (25.4) | 33.7 (23.7) | 7.78 (4.74) | <0.001 | T0 = T1 > T2 | −1.10 |
| IADL | 28 | 23.0 (5.42) | 24.8 (3.46) | 25.9 (2.24) | <0.001 | T0 < T1 = T2 | 0.70 |
| SHAPS | 28 | 20.8 (9.02) | 14.8 (7.75) | 13.2 (9.66) | <0.001 | T0 > T1 = T2 | −0.81 |
| DS_Forward | 28 | 11.9 (2.40) | 12.8 (1.91) | 13.0 (1.58) | 0.02 | T0 < T1 = T2 | 0.54 |
| DS_Backward | 28 | 8.03 (2.67) | 10.2 (3.44) | 10.9 (2.62) | <0.001 | T0 < T1 = T2 | 1.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheung, T.; Li, T.M.H.; Ho, Y.S.; Kranz, G.; Fong, K.N.K.; Leung, S.F.; Lam, S.C.; Yeung, W.F.; Lam, J.Y.T.; Fong, K.H.; et al. Effects of Transcranial Pulse Stimulation (TPS) on Adults with Symptoms of Depression—A Pilot Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2023, 20, 2333. https://doi.org/10.3390/ijerph20032333
Cheung T, Li TMH, Ho YS, Kranz G, Fong KNK, Leung SF, Lam SC, Yeung WF, Lam JYT, Fong KH, et al. Effects of Transcranial Pulse Stimulation (TPS) on Adults with Symptoms of Depression—A Pilot Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2023; 20(3):2333. https://doi.org/10.3390/ijerph20032333
Chicago/Turabian StyleCheung, Teris, Tim Man Ho Li, Yuen Shan Ho, Georg Kranz, Kenneth N. K. Fong, Sau Fong Leung, Simon Ching Lam, Wing Fai Yeung, Joyce Yuen Ting Lam, Kwan Hin Fong, and et al. 2023. "Effects of Transcranial Pulse Stimulation (TPS) on Adults with Symptoms of Depression—A Pilot Randomized Controlled Trial" International Journal of Environmental Research and Public Health 20, no. 3: 2333. https://doi.org/10.3390/ijerph20032333
APA StyleCheung, T., Li, T. M. H., Ho, Y. S., Kranz, G., Fong, K. N. K., Leung, S. F., Lam, S. C., Yeung, W. F., Lam, J. Y. T., Fong, K. H., Beisteiner, R., Xiang, Y.-T., & Cheng, C. P. W. (2023). Effects of Transcranial Pulse Stimulation (TPS) on Adults with Symptoms of Depression—A Pilot Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 20(3), 2333. https://doi.org/10.3390/ijerph20032333








