The Bidirectional Relationship between Chronic Kidney Disease and Hyperuricemia: Evidence from a Population-Based Prospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Data Collection
2.3. Study Outcomes and Related Definitions
2.4. Covariates
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Analysis I: Associations between CKD, eGFR at Baseline, and Dynamic Changes of eGFR with New-Onset HUA
3.3. Analysis II: Associations between HUA, sUA at Baseline, and Dynamic Changes of sUA with New-Onset CKD
3.4. Analysis III: Cross-Lagged Panel Analysis between sUA and eGFR
4. Discuss
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013, 3 (Suppl. 1), 1–150. [Google Scholar]
- Gansevoort, R.T.; Correa-Rotter, R.; Hemmelgarn, B.R.; Jafar, T.H.; Heerspink, H.J.L.; Mann, J.F.; Matsushita, K.; Wen, C.P. Chronic kidney disease and cardiovascular risk: Epidemiology, mechanisms, and prevention. Lancet 2013, 382, 339–352. [Google Scholar] [CrossRef]
- Drueke, T.B.; Floege, J. Cardiovascular complications of chronic kidney disease: Pioneering studies. Kidney Int. 2020, 98, 522–526. [Google Scholar] [CrossRef]
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafat, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Bikbov, B.; Purcell, C.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Jafar, T.H.; Nitsch, D.; Neuen, B.L.; Perkovic, V. Chronic kidney disease. Lancet 2021, 398, 786–802. [Google Scholar] [CrossRef]
- Johnson, R.J.; Bakris, G.L.; Borghi, C.; Chonchol, M.B.; Feldman, D.; Lanaspa, M.A.; Merriman, T.R.; Moe, O.W.; Mount, D.B.; Stahl, E. Hyperuricemia, Acute and Chronic Kidney Disease, Hypertension, and Cardiovascular Disease: Report of a Scientific Workshop Organized by the National Kidney Foundation. Am. J. Kidney Dis. 2018, 71, 851–865. [Google Scholar] [CrossRef]
- Chen, W.Y.; Fu, Y.P.; Zhou, M. The bidirectional relationship between metabolic syndrome and hyperuricemia in China: A longitudinal study from CHARLS. Endocrine 2022, 76, 62–69. [Google Scholar] [CrossRef]
- Cao, X.; Wu, L.; Chen, Z. The association between elevated serum uric acid level and an increased risk of renal function decline in a health checkup cohort in China. Int. Urol. Nephrol. 2018, 50, 517–525. [Google Scholar] [CrossRef]
- Li, L.; Yang, C.; Zhao, Y.; Zeng, X.; Liu, F.; Fu, P. Is hyperuricemia an independent risk factor for new-onset chronic kidney disease?: A systematic review and meta-analysis based on observational cohort studies. BMC Nephrol. 2014, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sah, O.S.P.; Qing, Y.X. Associations between hyperuricemia and chronic kidney disease: A review. Nephro-Urol. Mon. 2015, 7, e27233. [Google Scholar]
- Krishnan, E. Reduced Glomerular Function and Prevalence of Gout: NHANES 2009–10. PLoS ONE 2012, 7, e50046. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, W.; Qian, T.; Sun, H.; Xu, Q.; Hou, X.; Hu, W.; Zhang, G.; Drummond, G.R.; Sobey, C.G. Reduced renal function may explain the higher prevalence of hyperuricemia in older people. Sci. Rep. 2021, 11, 1302. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Liu, X.; Sun, X.; Wang, Y. The impact of serum uric acid on the natural history of glomerular filtration rate: A retrospective study in the general population. PeerJ 2016, 4, e1859. [Google Scholar] [CrossRef]
- Koratala, A.; Singhania, G.; Alquadan, K.F.; Shimada, M.; Johnson, R.J.; Ejaz, A.A. Serum uric acid exhibits inverse relationship with estimated glomerular filtration rate. Nephron 2016, 134, 231–237. [Google Scholar] [CrossRef]
- Selig, J.P.; Little, T.D. Autoregressive and cross-lagged panel analysis for longitudinal data. In Handbook of Developmental Research Methods; Guilford Press: New York, NY, USA, 2012. [Google Scholar]
- Bai, Y.; Yang, A.; Pu, H.; Dai, M.; Cheng, N.; Ding, J.; Li, J.; Hu, X.; Ren, X. Cohort profile: The China metal-exposed workers cohort study (Jinchang cohort). Int. J. Epidemiol. 2017, 46, 1095–1096e. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.; Feldman, M.I.; Kusek, J.W.; Eggers, P.; van Lente, F.; Greene, T.; Coresh, J. A New Equation to Estimate Glomerular Filtration Rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Dong, X.; Zhang, H.; Wang, F.; Liu, X.; Yang, K.; Tu, R. Epidemiology and prevalence of hyperuricemia among men and women in Chinese rural population: The Henan Rural Cohort Study. Mod. Rheumatol. 2020, 30, 910–920. [Google Scholar] [CrossRef]
- Lipkowitz, M.S. Regulation of uric acid excretion by the kidney. Curr. Rheumatol. Rep. 2012, 14, 179–188. [Google Scholar] [CrossRef]
- Russo, E.; Viazzi, F.; Pontremoli, R.; Barbagallo, C.M.; Bombelli, M.; Casiglia, E.; Cicero, A.F.G.; Cirillo, M.; Cirillo, P.; Desideri, G. Association of uric acid with kidney function and albuminuria: The Uric Acid Right for heArt Health (URRAH) Project. J. Nephrol. 2022, 35, 211–221. [Google Scholar] [CrossRef]
- Reboldi, G.; Verdecchia, P.; Fiorucci, G.; Beilin, L.G.; Eguchi, K.; Imai, Y.; Kario, K.; Ohkubo, T.; Pierdomenico, S.D.; Schwartz, J.E. Glomerular hyperfiltration is a predictor of adverse cardiovascular outcomes. Kidney Int. 2018, 93, 195–203. [Google Scholar] [CrossRef]
- Kuwabara, M.; Niwa, K.; Hisatome, I. Hyperfiltration could be a risk factor for development of hypertension: A five-year cohort study. Eur. Heart J. 2019, 40, 3346. [Google Scholar] [CrossRef]
- Har, R.; Scholey, J.W.; Daneman, D.; Mahmud, F.H.; Dekker, R.; Lai, V. The effect of renal hyperfiltration on urinary inflammatory cytokines/chemokines in patients with uncomplicated type 1 diabetes mellitus. Diabetologia 2013, 56, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.J.; Li, B.L.; Wang, S.B.; Chen, M.L.; Deng, R.T.; Ye, C.Q.; Liu, L.; Fang, A.-J.; Xiong, S.-L.; Wen, S. Changes of the tubular markers in type 2 diabetes mellitus with glomerular hyperfiltration. Diabetes Res. Clin. Pract. 2012, 95, 105–109. [Google Scholar] [CrossRef]
- Cortinovis, M.; Perico, N.; Ruggenenti, P.; Remuzzi, A.; Remuzzi, G. Glomerular hyperfiltration. Nat. Rev. Nephrol. 2022, 18, 435–451. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Lu, A.; Lu, X.; Zhang, L.; Fang, H.; Zhou, L.; Yang, T. Activation of renal (pro) renin receptor contributes to high fructose-induced salt sensitivity. Hypertension 2017, 69, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Mulay, S.R.; Shi, C.; Ma, X.; Anders, H.J. Novel insights into crystal-induced kidney injury. Kidney Dis. 2018, 4, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, R.; Ninomiya, D.; Akase, T.; Kikuchi, A.; Kumagi, T. Interactive association of baseline and changes in serum uric acid on renal dysfunction among community-dwelling persons. J. Clin. Lab. Anal. 2020, 34, e23166. [Google Scholar] [CrossRef]
- Kritmetapak, K.; Charoensri, S.; Thaopanya, R.; Pongchaiyakul, C. Elevated Serum Uric Acid is Associated with Rapid Decline in Kidney Function: A 10-Year Follow-Up Study. Int. J. Gen. Med. 2020, 13, 945–953. [Google Scholar] [CrossRef]
- Tada, K.; Maeda, T.; Takahashi, K.; Ito, K.; Yasuno, T.; Funakoshi, S.; Satoh, A.; Kawazoe, M.; Yoshimura, C. Association between serum uric acid and new onset and progression of chronic kidney disease in a Japanese general population: Iki epidemiological study of atherosclerosis and chronic kidney disease. Clin. Exp. Nephrol. 2021, 25, 751–759. [Google Scholar] [CrossRef]
- Wu, N.; Xia, J.; Chen, S.; Yu, C.; Xu, Y.; Xu, C.; Pan, X.F. Serum uric acid and risk of incident chronic kidney disease: A national cohort study and updated meta-analysis. Nutr. Metab. 2021, 18, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chou, Y.-C.; Kuan, J.-C.; Yang, T.; Chou, W.Y.; Hsieh, P.C.; Bai, C.H.; Sun, C.A. Elevated uric acid level as a significant predictor of chronic kidney disease: A cohort study with repeated measurements. J. Nephrol. 2015, 28, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Akasaka, H.; Yoshida, H.; Takizawa, H.; Hanawa, N.; Tobisawa, T.; Tanaka, M.; Miura, T. The impact of elevation of serum uric acid level on the natural history of glomerular filtration rate (GFR) and its sex difference. Nephrol. Dial. Transplant. 2014, 29, 1932–1939. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-H.; Tsai, C.-C.; Liu, Y.-H.; Wu, P.Y.; Huang, J.C.; Chung, T.L.; Chen, S.C. Sex Difference in the Associations among Hyperuricemia with New-Onset Chronic Kidney Disease in a Large Taiwanese Population Follow-Up Study. Nutrients 2022, 14, 3832. [Google Scholar] [CrossRef]
- Chertow, G.M.; Beddhu, S. Modification of eGFR-based CKD definitions: Perfect, or enemy of the good? J. Am. Soc. Nephrol. 2019, 30, 1807–1809. [Google Scholar] [CrossRef]
- Hirst, J.A.; Hill, N.; O’callaghan, C.A.; Lasserson, D.; McManus, R.J.; Ogburn, E.; Hobbs, F.R. Prevalence of chronic kidney disease in the community using data from OxRen: A UK population-based cohort study. Br. J. Gen. Pract. 2020, 70, e285–e293. [Google Scholar] [CrossRef]


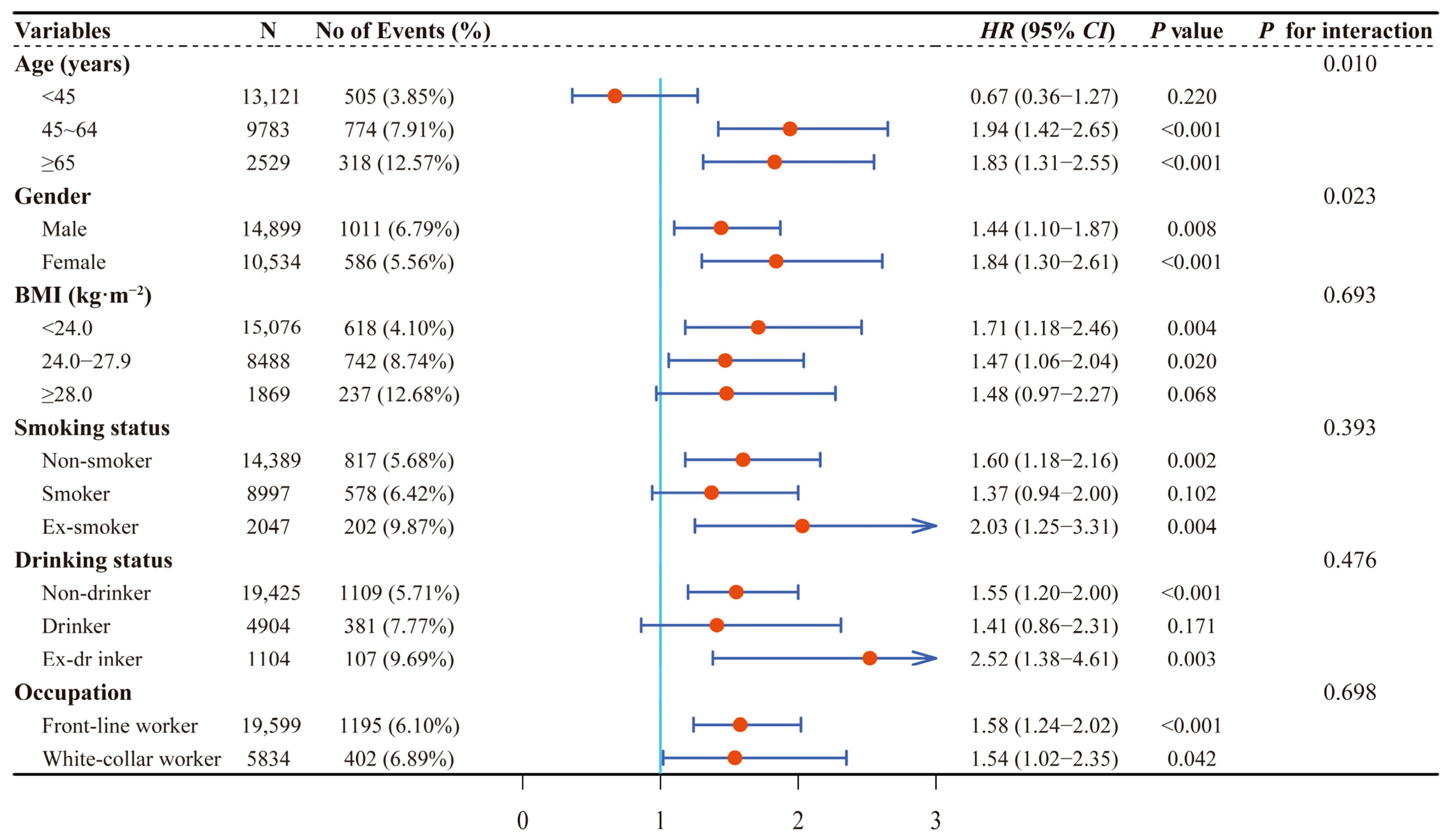
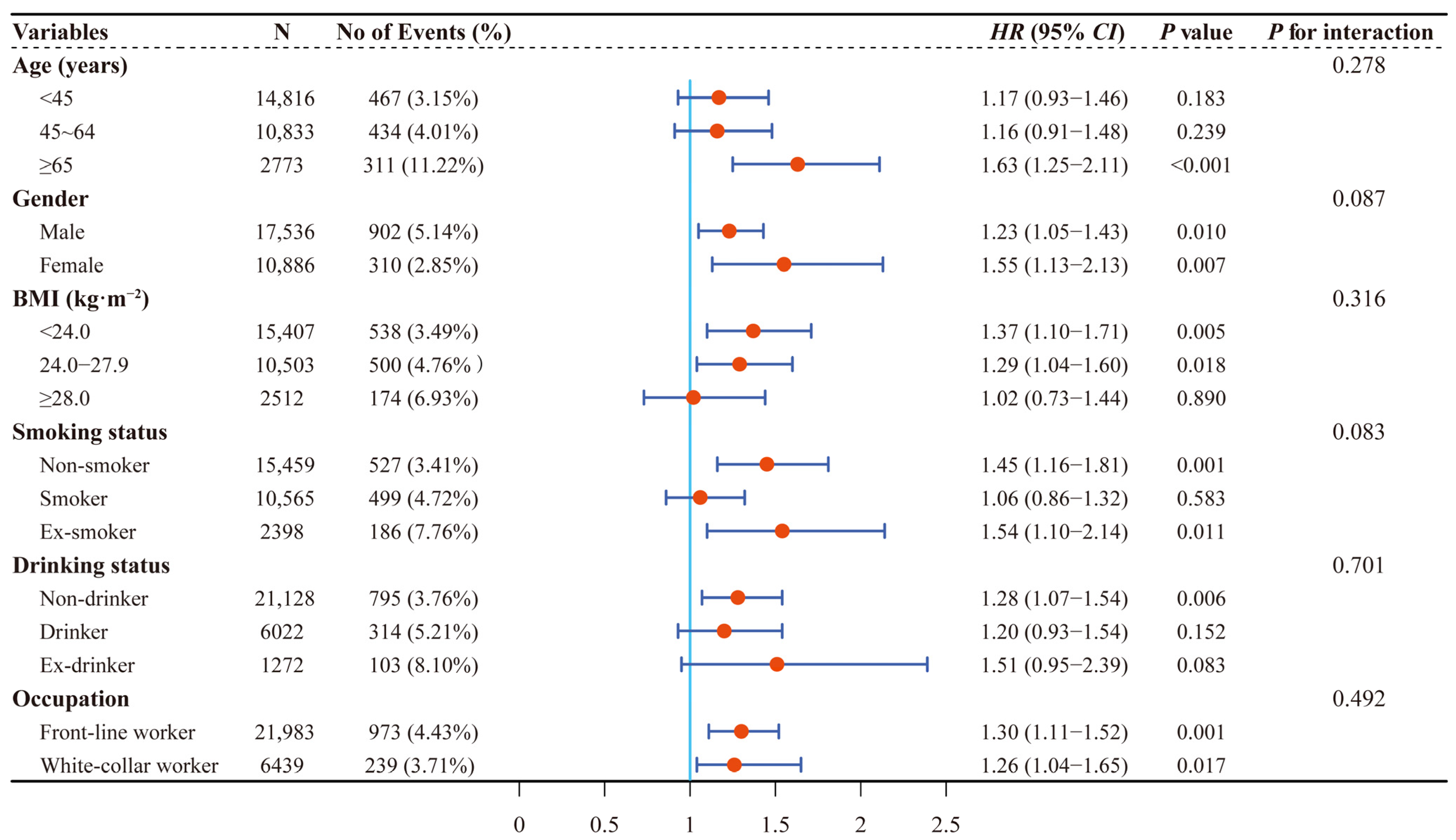
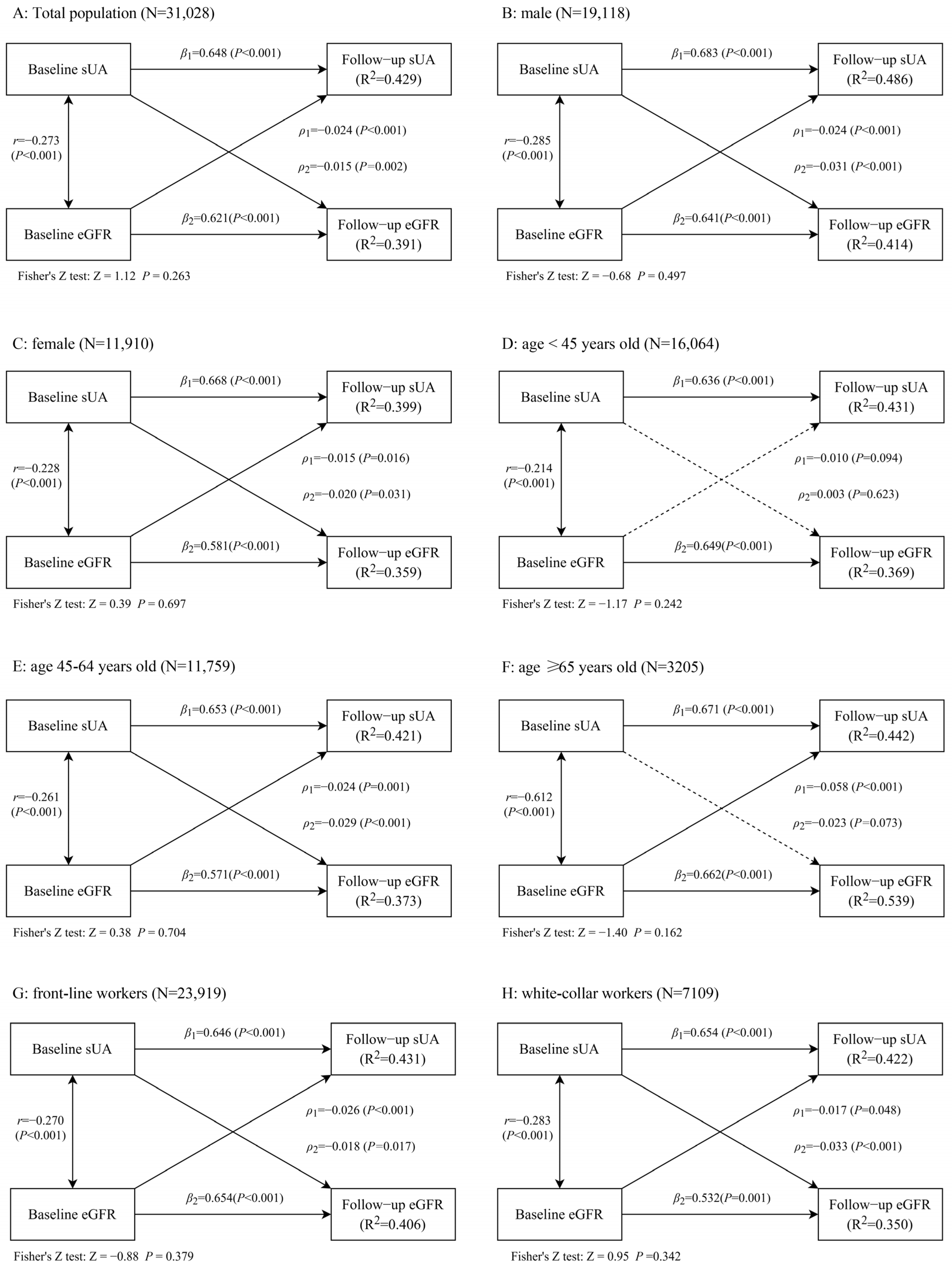
| N | No of Events (%) | Model 1 | Model 2 | |||
|---|---|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | |||
| CKD at Baseline | ||||||
| Non-CKD | 24,684 | 1502 (6.08%) | 1.00 | 1.00 | ||
| CKD | 749 | 95 (12.68%) | 1.94 (1.58–2.39) | <0.001 | 1.58 (1.28–1.95) | <0.001 |
| Groups of eGFR | ||||||
| (mL·min−1·1.73 m−2) | ||||||
| Group1 (<60) | 135 | 35 (25.93%) | 3.88 (2.77–5.43) | <0.001 | 3.56 (2.50–5.05) | <0.001 |
| Group2 (60~<90) | 3689 | 438 (11.87%) | 1.72 (1.53–1.92) | <0.001 | 1.61 (1.42–1.83) | <0.001 |
| Group3 (90~<120) | 19,191 | 999 (5.21%) | 1.00 | 1.00 | ||
| Group4 (≥120) | 2418 | 125 (5.17%) | 1.74 (1.44–2.10) | <0.001 | 1.74 (1.42–2.14) | <0.001 |
| N | No of Events (%) | Model 1 | Model 2 | |||
|---|---|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | |||
| HUA at Baseline | ||||||
| Non-HUA | 24,553 | 933 (3.80%) | 1.00 | 1.00 | ||
| HUA | 3869 | 279 (7.21%) | 1.70 (1.48–1.93) | <0.001 | 1.28 (1.12–1.47) | <0.001 |
| sUA (μmol/L) | ||||||
| Q1 (≤285) | 7024 | 192 (2.73%) | 1.00 | 1.00 | ||
| Q2 (266–318) | 7087 | 260 (3.67%) | 1.22 (1.01–1.47) | 0.036 | 1.03 (0.85–1.25) | 0.738 |
| Q3 (319–374) | 7091 | 309 (4.36%) | 1.46 (1.22–1.74) | <0.001 | 1.04 (0.85–1.27) | 0.705 |
| Q4 (≥375) | 7220 | 451 (6.25%) | 2.02 (1.70–2.39) | <0.001 | 1.24 (1.01–1.51) | 0.038 |
| p trend | <0.001 | <0.001 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Z.; Wang, X.; Zhang, J.; Yang, C.; Du, H.; Dou, F.; Li, J.; Zhao, Y.; Quan, P.; Hu, X. The Bidirectional Relationship between Chronic Kidney Disease and Hyperuricemia: Evidence from a Population-Based Prospective Cohort Study. Int. J. Environ. Res. Public Health 2023, 20, 1728. https://doi.org/10.3390/ijerph20031728
Ma Z, Wang X, Zhang J, Yang C, Du H, Dou F, Li J, Zhao Y, Quan P, Hu X. The Bidirectional Relationship between Chronic Kidney Disease and Hyperuricemia: Evidence from a Population-Based Prospective Cohort Study. International Journal of Environmental Research and Public Health. 2023; 20(3):1728. https://doi.org/10.3390/ijerph20031728
Chicago/Turabian StyleMa, Zhibin, Xiao Wang, Jia Zhang, Chao Yang, Hongmei Du, Feng Dou, Jianjian Li, Yini Zhao, Peiqin Quan, and Xiaobin Hu. 2023. "The Bidirectional Relationship between Chronic Kidney Disease and Hyperuricemia: Evidence from a Population-Based Prospective Cohort Study" International Journal of Environmental Research and Public Health 20, no. 3: 1728. https://doi.org/10.3390/ijerph20031728
APA StyleMa, Z., Wang, X., Zhang, J., Yang, C., Du, H., Dou, F., Li, J., Zhao, Y., Quan, P., & Hu, X. (2023). The Bidirectional Relationship between Chronic Kidney Disease and Hyperuricemia: Evidence from a Population-Based Prospective Cohort Study. International Journal of Environmental Research and Public Health, 20(3), 1728. https://doi.org/10.3390/ijerph20031728






