How Antimicrobial Resistance Is Linked to Climate Change: An Overview of Two Intertwined Global Challenges
Abstract
1. Introduction
2. Needs for a Global Solution against AMR
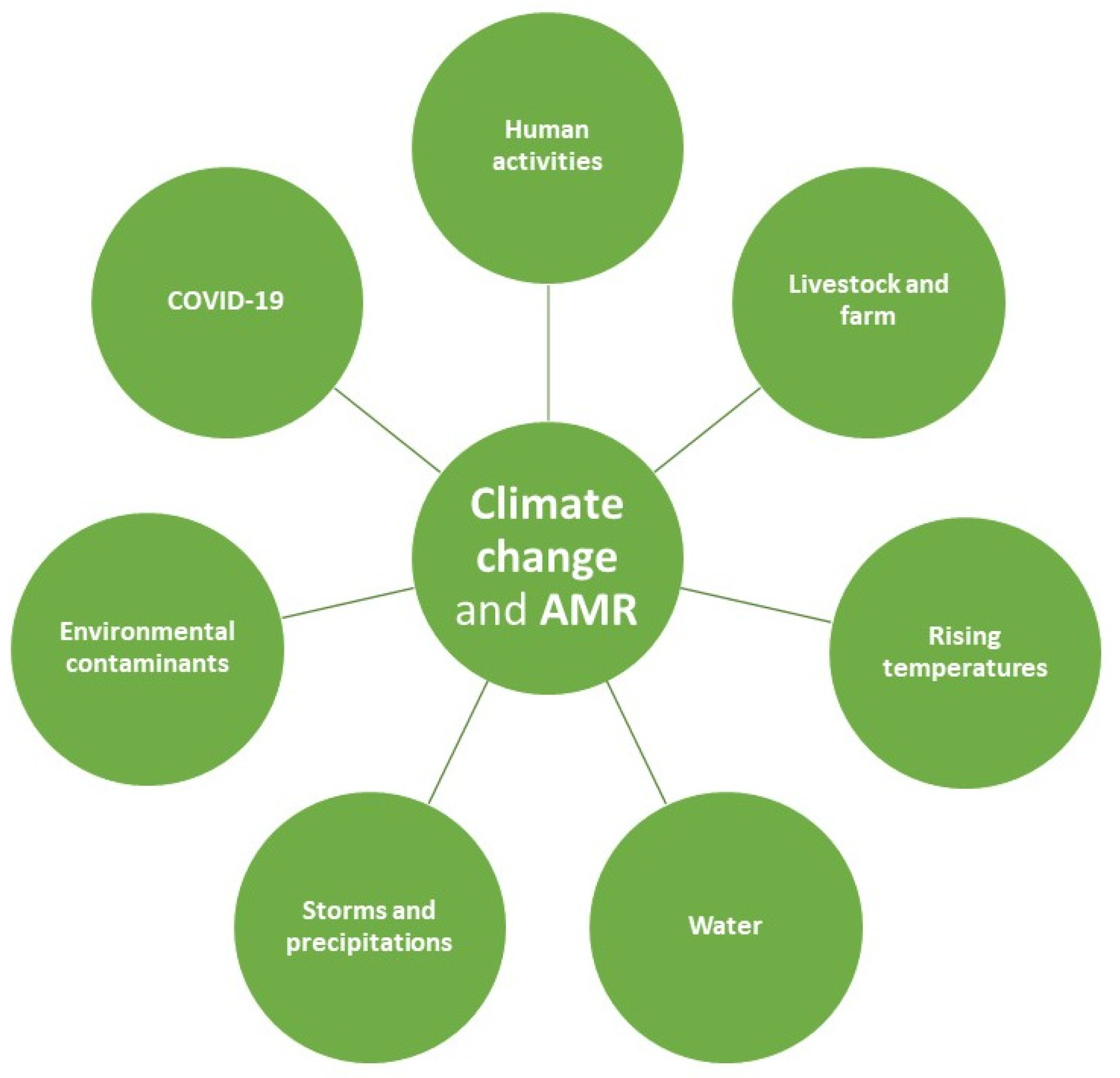
3. Antimicrobial Resistance and Climate Change: The One Health Approach
4. Antimicrobial Resistance and Climate Change: Two Intertwined Global Challenges
4.1. The Relationship between Climate Change and Antimicrobial Resistance at Human, Animal and Environmental Levels
4.2. Current Strategies and Future Interdisciplinary Research on the Relationship between Antimicrobial Resistance and Climate Change
4.3. The Economic and Political Interventions to Counteract Antimicrobial Resistance and Climate Change
4.4. The Impact of COVID-19 Pandemic on Antimicrobial Resistance and Climate Change
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cassini, A.; Högberg, L.D.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect. Dis. 2019, 19, 56–66. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Antimicrobial Resistance Surveillance in Europe 2022—2020 Data. 2022. Available online: https://www.ecdc.europa.eu/en/publications-data/antimicrobial-resistance-surveillance-europe-2022-2020-data (accessed on 1 November 2022).
- Watts, N.; Amann, M.; Ayeb-Karlsson, S.; Belesova, K.; Bouley, T.; Boykoff, M.; Byass, P.; Cai, W.; Campbell-Lendrum, D.; Chambers, J.; et al. The Lancet Countdown on health and climate change: From 25 years of inaction to a global transformation for public health. Lancet 2018, 391, 581–630. [Google Scholar] [CrossRef] [PubMed]
- Romanello, M.; Di Napoli, C.; Drummond, P.; Green, C.; Kennard, H.; Lampard, P.; Scamman, D.; Arnell, N.; Ayeb-Karlsson, S.; Ford, L.B.; et al. The 2022 report of the Lancet Countdown on health and climate change: Health at the mercy of fossil fuels. Lancet 2022, 400, 1619–1654. [Google Scholar] [CrossRef]
- Harper, S.L.; Cunsolo, A.; Babujee, A.; Coggins, S.; Aguilar, M.D.; Wright, C.J. Climate change and health in North America: Literature review protocol. Syst. Rev. 2021, 10, 3. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.; Remenyi, T.A.; White, C.J.; Johnston, F.H. Heatwave and health impact research: A global review. Health Place 2018, 53, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, M.; Arbuthnott, K.; Kovats, S.; Hajat, S.; Falloon, P. The use of climate information to estimate future mortality from high ambient temperature: A systematic literature review. PLoS ONE 2017, 12, e0180369. [Google Scholar] [CrossRef] [PubMed]
- Rataj, E.; Kunzweiler, K.; Garthus-Niegel, S. Extreme weather events in developing countries and related injuries and mental health disorders—A systematic review. BMC Public Health 2016, 16, 1020. [Google Scholar] [CrossRef]
- Saulnier, D.D.; Brolin Ribacke, K.; von Schreeb, J. No Calm After the Storm: A Systematic Review of Human Health Following Flood and Storm Disasters. Prehosp. Disaster Med. 2017, 32, 568–579. [Google Scholar] [CrossRef]
- Levy, K.; Woster, A.P.; Goldstein, R.S.; Carlton, E.J. Untangling the Impacts of Climate Change on Waterborne Diseases: A Systematic Review of Relationships between Diarrheal Diseases and Temperature, Rainfall, Flooding, and Drought. Environ. Sci. Technol. 2016, 50, 4905–4922. [Google Scholar] [CrossRef]
- Semenza, J.C.; Herbst, S.; Rechenburg, A.; Suk, J.E.; Höser, C.; Schreiber, C.; Kistemann, T. Climate Change Impact Assessment of Food- and Waterborne Diseases. Crit. Rev. Environ. Sci. Technol. 2012, 42, 857–890. [Google Scholar] [CrossRef]
- Cann, K.F.; Thomas, D.R.; Salmon, R.L.; Wyn-Jones, A.P.; Kay, D. Extreme water-related weather events and waterborne disease. Epidemiol. Infect. 2013, 141, 671–686. [Google Scholar] [CrossRef]
- Andrade, L.; O’Dwyer, J.; O’Neill, E.; Hynds, P. Surface water flooding, groundwater contamination, and enteric disease in developed countries: A scoping review of connections and consequences. Environ. Pollut. 2018, 236, 540–549. [Google Scholar] [CrossRef]
- Lake, I.R.; Hooper, L.; Abdelhamid, A.; Bentham, G.; Boxall, A.B.; Draper, A.; Fairweather-Tait, S.; Hulme, M.; Hunter, P.R.; Nichols, G.; et al. Climate change and food security: Health impacts in developed countries. Environ. Health Perspect. 2012, 120, 1520–1526. [Google Scholar] [CrossRef]
- Springmann, M.; Mason-D’Croz, D.; Robinson, S.; Garnett, T.; Godfray, H.C.; Gollin, D.; Rayner, M.; Ballon, P.; Scarborough, P. Global and regional health effects of future food production under climate change: A modelling study. Lancet 2016, 387, 1937–1946. [Google Scholar] [CrossRef] [PubMed]
- Park, M.S.; Park, K.H.; Bahk, G.J. Interrelationships between Multiple Climatic Factors and Incidence of Foodborne Diseases. Int. J. Environ. Res. Public Health 2018, 15, 2482. [Google Scholar] [CrossRef] [PubMed]
- Lake, I.R.; Gillespie, I.A.; Bentham, G.; Nichols, G.L.; Lane, C.; Adak, G.K.; Threlfall, E.J. A re-evaluation of the impact of temperature and climate change on foodborne illness. Epidemiol. Infect. 2009, 137, 1538–1547. [Google Scholar] [CrossRef]
- Lake, I.R.; Barker, G.C. Climate Change, Foodborne Pathogens and Illness in Higher-Income Countries. Curr. Environ. Health Rep. 2018, 5, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Campbell-Lendrum, D.; Manga, L.; Bagayoko, M.; Sommerfeld, J. Climate change and vector-borne diseases: What are the implications for public health research and policy? Philos. Trans. R Soc. Lond. B Biol. Sci. 2015, 370, 20130552. [Google Scholar] [CrossRef]
- Sweileh, W.M. Bibliometric analysis of peer-reviewed literature on climate change and human health with an emphasis on infectious diseases. Glob. Health 2020, 16, 44. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Climate Change and Health. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/climate-change-and-health (accessed on 1 November 2022).
- Fox, N.J.; Marion, G.; Davidson, R.S.; White, P.C.; Hutchings, M.R. Climate-driven tipping-points could lead to sudden, high-intensity parasite outbreaks. R. Soc. Open Sci. 2015, 2, 140296. [Google Scholar] [CrossRef]
- Maugeri, A.; Barchitta, M.; Basile, G.; Agodi, A. How COVID-19 Has Influenced Public Interest in Antimicrobials, Antimicrobial Resistance and Related Preventive Measures: A Google Trends Analysis of Italian Data. Antibiotics 2022, 11, 379. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Maugeri, A.; La Rosa, M.C.; La Mastra, C.; Murolo, G.; Basile, G.; Agodi, A. Carbapenem Consumption and Rate of carbapenemresistant gram-negative bacteria: Results from the Sicilian Surveillance System. Ann. Ig. 2021, 33, 289–296. [Google Scholar] [CrossRef]
- Barchitta, M.; Maugeri, A.; La Rosa, M.C.; La Mastra, C.; Murolo, G.; Corrao, G.; Agodi, A. Burden of Healthcare-Associated Infections in Sicily, Italy: Estimates from the Regional Point Prevalence Surveys 2016–2018. Antibiotics 2021, 10, 1360. [Google Scholar] [CrossRef]
- Barchitta, M.; Maugeri, A.; La Rosa, M.C.; La Mastra, C.; Murolo, G.; Agodi, A. Three-Year Trends of Healthcare-Associated Infections and Antibiotic Use in Acute Care Hospitals: Findings from 2016–2018 Point Prevalence Surveys in Sicily, Italy. Antibiotics 2020, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Quattrocchi, A.; Maugeri, A.; Rosa, M.C.; Mastra, C.; Basile, G.; Giuffrida, G.; Rinaldi, F.M.; Murolo, G.; Agodi, A. The “Obiettivo Antibiotico” Campaign on Prudent Use of Antibiotics in Sicily, Italy: The Pilot Phase. Int. J. Environ. Res. Public Health 2020, 17, 3077. [Google Scholar] [CrossRef]
- Valencia, C.; Hammami, N.; Agodi, A.; Lepape, A.; Herrejon, E.P.; Blot, S.; Vincent, J.L.; Lambert, M.L. Poor adherence to guidelines for preventing central line-associated bloodstream infections (CLABSI): Results of a worldwide survey. Antimicrob. Resist. Infect. Control 2016, 5, 49. [Google Scholar] [CrossRef]
- Agodi, A.; Auxilia, F.; Barchitta, M.; Brusaferro, S.; D’Errico, M.M.; Montagna, M.T.; Pasquarella, C.; Tardivo, S.; Mura, I.; SPIN-UTI Network of the GISIOWorking Group of the Italian Society of Hygiene. Antibiotic consumption and resistance: Results of the SPIN-UTI project of the GISIO-SItI. Epidemiol. Prev. 2015, 39, 94–98. [Google Scholar] [PubMed]
- Agodi, A.; Barchitta, M.; Quattrocchi, A.; Maugeri, A.; Aldisio, E.; Marchese, A.E.; Mattaliano, A.R.; Tsakris, A. Antibiotic trends of Klebsiella pneumoniae and Acinetobacter baumannii resistance indicators in an intensive care unit of Southern Italy, 2008–2013. Antimicrob. Resist. Infect. Control 2015, 4, 43. [Google Scholar] [CrossRef]
- Gudipati, S.; Zervos, M.; Herc, E. Can the One Health Approach Save Us from the Emergence and Reemergence of Infectious Pathogens in the Era of Climate Change: Implications for Antimicrobial Resistance? Antibiotics 2020, 9, 599. [Google Scholar] [CrossRef]
- Jones, G.; Rebelo, H. Responses of bats to climate change: Learning from the past and predicting the future. In Bat Evolution, Ecology, and Conservation; Springer: New York, NY, USA, 2013. [Google Scholar]
- World Health Organization (WHO). Coronavirus Disease 2019 (COVID-19) Situation Report—57. 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200317-sitrep-57-covid-19.pdf (accessed on 1 November 2022).
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.; Wertheim, H.F.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H.; et al. Antibiotic resistance-the need for global solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Ukuhor, H.O. The interrelationships between antimicrobial resistance, COVID-19, past, and future pandemics. J. Infect. Public Health 2021, 14, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Pitchforth, E.; Smith, E.; Taylor, J.; Davies, S.; Ali, G.C.; d’Angelo, C. Global action on antimicrobial resistance: Lessons from the history of climate change and tobacco control policy. BMJ Glob. Health 2022, 7, e009283. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Global Database for the Tripartite Antimicrobial Resistance Country Self-Assessment Survey. 2019. Available online: https://www.who.int/publications/m/item/tripartite-amr-country-self-assessment-survey-(tracss)-2019-2020 (accessed on 1 November 2022).
- Ford, J.D. Indigenous health and climate change. Am. J. Public Health 2012, 102, 1260–1266. [Google Scholar] [CrossRef] [PubMed]
- Laxminarayan, R.; Van Boeckel, T.; Frost, I.; Kariuki, S.; Khan, E.A.; Limmathurotsakul, D.; Larsson, D.G.J.; Levy-Hara, G.; Mendelson, M.; Outterson, K.; et al. The Lancet Infectious Diseases Commission on antimicrobial resistance: 6 years later. Lancet Infect. Dis. 2020, 20, e51–e60. [Google Scholar] [CrossRef] [PubMed]
- Collaborators, G.A.R. Global mortality associated with 33 bacterial pathogens in 2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2022, 400, 2221–2248. [Google Scholar] [CrossRef]
- Davies, J.; Davies, D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef]
- Waters, D.; Jawad, I.; Ahmad, A.; Lukšić, I.; Nair, H.; Zgaga, L.; Theodoratou, E.; Rudan, I.; Zaidi, A.K.; Campbell, H. Aetiology of community-acquired neonatal sepsis in low and middle income countries. J. Glob. Health 2011, 1, 154–170. [Google Scholar]
- Holmberg, S.D.; Solomon, S.L.; Blake, P.A. Health and economic impacts of antimicrobial resistance. Rev. Infect. Dis. 1987, 9, 1065–1078. [Google Scholar] [CrossRef]
- Dadgostar, P. Antimicrobial Resistance: Implications and Costs. Infect. Drug Resist. 2019, 12, 3903–3910. [Google Scholar] [CrossRef]
- Smith, R.; Coast, J. The true cost of antimicrobial resistance. BMJ 2013, 346, f1493. [Google Scholar] [CrossRef]
- Morgan, D.J.; Okeke, I.N.; Laxminarayan, R.; Perencevich, E.N.; Weisenberg, S. Non-prescription antimicrobial use worldwide: A systematic review. Lancet Infect. Dis. 2011, 11, 692–701. [Google Scholar] [CrossRef]
- Food and Drug Administration, Summary Report on Antimicrobials Sold or Distributed for Use in Food-Producing Animals. 2021. Available online: https://www.fda.gov/media/154820/download (accessed on 1 November 2022).
- Barchitta, M.; Sabbatucci, M.; Furiozzi, F.; Iannazzo, S.; Maugeri, A.; Maraglino, F.; Prato, R.; Agodi, A.; Pantosti, A. Knowledge, attitudes and behaviors on antibiotic use and resistance among healthcare workers in Italy, 2019: Investigation by a clustering method. Antimicrob. Resist. Infect. Control 2021, 10, 134. [Google Scholar] [CrossRef]
- Mishra, R.P.; Oviedo-Orta, E.; Prachi, P.; Rappuoli, R.; Bagnoli, F. Vaccines and antibiotic resistance. Curr. Opin. Microbiol. 2012, 15, 596–602. [Google Scholar] [CrossRef] [PubMed]
- Omenn, G.S. Evolution in health and medicine Sackler colloquium: Evolution and public health. Proc. Natl. Acad. Sci. USA 2010, 107 (Suppl. S1), 1702–1709. [Google Scholar] [CrossRef]
- Barchitta, M.; Maugeri, A.; Vinci, R.; Agodi, A. The Inverse Relationship between Influenza Vaccination and Antimicrobial Resistance: An Ecological Analysis of Italian Data. Vaccines 2022, 10, 554. [Google Scholar] [CrossRef] [PubMed]
- Maugeri, A.; Barchitta, M.; Agodi, A. Vaccination coverage in Italian children and antimicrobial resistance: An ecological analysis. Antimicrob. Resist. Infect. Control 2022, 11, 136. [Google Scholar] [CrossRef]
- O’Neill, C. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations; HM Government: London, UK, 2016. Available online: https://amrreview.org/sites/default/files/160518_Final%20paper_with%20cover.pdf (accessed on 1 November 2022).
- World Health Organization (WHO). Strategic Priorities on Antimicrobial Resistance. Available online: https://apps.who.int/iris/bitstream/handle/10665/351719/9789240041387-eng.pdf (accessed on 1 November 2022).
- Harrison, E.M.; Paterson, G.K.; Holden, M.T.; Larsen, J.; Stegger, M.; Larsen, A.R.; Petersen, A.; Skov, R.L.; Christensen, J.M.; Bak Zeuthen, A.; et al. Whole genome sequencing identifies zoonotic transmission of MRSA isolates with the novel mecA homologue mecC. EMBO Mol. Med. 2013, 5, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Patz, J.A.; Epstein, P.R.; Burke, T.A.; Balbus, J.M. Global climate change and emerging infectious diseases. JAMA 1996, 275, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.P.; Gonçalves, Y.M.; Novo, M.T.; Sousa, C.A.; Melim, M.; Grácio, A.J. Vector monitoring of Aedes aegypti in the Autonomous Region of Madeira, Portugal. Euro Surveill. 2007, 12, E071115.071116. [Google Scholar] [CrossRef]
- Omazic, A.; Bylund, H.; Boqvist, S.; Högberg, A.; Björkman, C.; Tryland, M.; Evengård, B.; Koch, A.; Berggren, C.; Malogolovkin, A.; et al. Identifying climate-sensitive infectious diseases in animals and humans in Northern regions. Acta Vet. Scand. 2019, 61, 53. [Google Scholar] [CrossRef] [PubMed]
- Semenza, J.C.; Menne, B. Climate change and infectious diseases in Europe. Lancet Infect. Dis. 2009, 9, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Jobling, M.G. Trust but Verify: Uncorroborated Assemblies of Plasmid Genomes from Next-Generation Sequencing Data Are Likely Spurious. Microb. Drug Resist. 2019, 25, 1521–1524. [Google Scholar] [CrossRef]
- Lipp, E.K.; Huq, A.; Colwell, R.R. Effects of global climate on infectious disease: The cholera model. Clin. Microbiol. Rev. 2002, 15, 757–770. [Google Scholar] [CrossRef] [PubMed]
- Constantin de Magny, G.; Colwell, R.R. Cholera and climate: A demonstrated relationship. Trans. Am. Clin. Climatol. Assoc. 2009, 120, 119–128. [Google Scholar]
- Zinsstag, J.; Crump, L.; Schelling, E.; Hattendorf, J.; Maidane, Y.O.; Ali, K.O.; Muhummed, A.; Umer, A.A.; Aliyi, F.; Nooh, F.; et al. Climate change and One Health. FEMS Microbiol. Lett. 2018, 365, fny085. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M.; Ward, M.; van Bunnik, B.; Farrar, J. Antimicrobial resistance in humans, livestock and the wider environment. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140083. [Google Scholar] [CrossRef] [PubMed]
- Cleaveland, S.; Sharp, J.; Abela-Ridder, B.; Allan, K.J.; Buza, J.; Crump, J.A.; Davis, A.; Del Rio Vilas, V.J.; de Glanville, W.A.; Kazwala, R.R.; et al. One Health contributions towards more effective and equitable approaches to health in low- and middle-income countries. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2017, 372, 20160168. [Google Scholar] [CrossRef]
- De Jongh, E.J.; Harper, S.L.; Yamamoto, S.S.; Wright, C.J.; Wilkinson, C.W.; Ghosh, S.; Otto, S.J.G. One Health, One Hive: A scoping review of honey bees, climate change, pollutants, and antimicrobial resistance. PLoS ONE 2022, 17, e0242393. [Google Scholar] [CrossRef]
- Regueira, M.S.; Tintino, S.R.; da Silva, A.R.P.; Costa, M.D.S.; Boligon, A.A.; Matias, E.F.F.; de Queiroz Balbino, V.; Menezes, I.R.A.; Melo Coutinho, H.D. Seasonal variation of Brazilian red propolis: Antibacterial activity, synergistic effect and phytochemical screening. Food Chem. Toxicol. 2017, 107, 572–580. [Google Scholar] [CrossRef]
- Prodělalová, J.; Malenovská, H.; Moutelíková, R.; Titěra, D. Virucides in apiculture: Persistence of surrogate enterovirus under simulated field conditions. Pest Manag. Sci. 2017, 73, 2544–2549. [Google Scholar] [CrossRef]
- Runckel, C.; Flenniken, M.L.; Engel, J.C.; Ruby, J.G.; Ganem, D.; Andino, R.; DeRisi, J.L. Temporal analysis of the honey bee microbiome reveals four novel viruses and seasonal prevalence of known viruses, Nosema, and Crithidia. PLoS ONE 2011, 6, e20656. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Protecting Health from Climate Change: Global Research Priorities. Available online: https://www.who.int/publications/i/item/9789241598187 (accessed on 1 November 2022).
- Cars, O.; Högberg, L.D.; Murray, M.; Nordberg, O.; Sivaraman, S.; Lundborg, C.S.; So, A.D.; Tomson, G. Meeting the challenge of antibiotic resistance. BMJ 2008, 337, a1438. [Google Scholar] [CrossRef] [PubMed]
- McGough, S.F.; MacFadden, D.R.; Hattab, M.W.; Mølbak, K.; Santillana, M. Rates of increase of antibiotic resistance and ambient temperature in Europe: A cross-national analysis of 28 countries between 2000 and 2016. Euro Surveill. 2020, 25, 1900414. [Google Scholar] [CrossRef] [PubMed]
- MacFadden, D.R.; McGough, S.F.; Fisman, D.; Santillana, M.; Brownstein, J.S. Antibiotic Resistance Increases with Local Temperature. Nat. Clim. Chang. 2018, 8, 510–514. [Google Scholar] [CrossRef]
- Pietikäinen, J.; Pettersson, M.; Bååth, E. Comparison of temperature effects on soil respiration and bacterial and fungal growth rates. FEMS Microbiol. Ecol. 2005, 52, 49–58. [Google Scholar] [CrossRef]
- Philipsborn, R.; Ahmed, S.M.; Brosi, B.J.; Levy, K. Climatic Drivers of Diarrheagenic Escherichia coli Incidence: A Systematic Review and Meta-analysis. J. Infect. Dis. 2016, 214, 6–15. [Google Scholar] [CrossRef]
- Casadevall, A.; Kontoyiannis, D.P.; Robert, V. On the Emergence of Candida auris: Climate Change, Azoles, Swamps, and Birds. mBio 2019, 10, e01397-19. [Google Scholar] [CrossRef]
- Arora, P.; Singh, P.; Wang, Y.; Yadav, A.; Pawar, K.; Singh, A.; Padmavati, G.; Xu, J.; Chowdhary, A. Environmental Isolation of Candida auris from the Coastal Wetlands of Andaman Islands, India. mBio 2021, 12, e03181-20. [Google Scholar] [CrossRef]
- Chaves, L.F.; Koenraadt, C.J. Climate change and highland malaria: Fresh air for a hot debate. Q. Rev. Biol. 2010, 85, 27–55. [Google Scholar] [CrossRef]
- Ryan, S.J.; Carlson, C.J.; Mordecai, E.A.; Johnson, L.R. Global expansion and redistribution of Aedes-borne virus transmission risk with climate change. PLoS Negl. Trop. Dis. 2019, 13, e0007213. [Google Scholar] [CrossRef]
- Institute of Medicine, (US) Committee for the Study on Malaria Prevention and Control. Malaria: Obstacles and Opportunities; Oaks, S.C., Jr., Mitchell, V.S., Pearson, G.W., Carpenter, C.C.J., Eds.; The National Academies Press: Washington, DC, USA, 1991. [CrossRef]
- Alonso, P.L.; Smith, T.; Schellenberg, J.R.; Masanja, H.; Mwankusye, S.; Urassa, H.; Bastos de Azevedo, I.; Chongela, J.; Kobero, S.; Menendez, C. Randomised trial of efficacy of SPf66 vaccine against Plasmodium falciparum malaria in children in southern Tanzania. Lancet 1994, 344, 1175–1181. [Google Scholar] [CrossRef] [PubMed]
- Aik, J.; Heywood, A.E.; Newall, A.T.; Ng, L.C.; Kirk, M.D.; Turner, R. Climate variability and salmonellosis in Singapore—A time series analysis. Sci. Total Environ. 2018, 639, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Shuman, E.K. Global climate change and infectious diseases. N. Engl. J. Med. 2010, 362, 1061–1063. [Google Scholar] [CrossRef] [PubMed]
- McDonald, L.C.; Banerjee, S.N.; Jarvis, W.R. Seasonal variation of Acinetobacter infections: 1987–1996. Nosocomial Infections Surveillance System. Clin. Infect. Dis. 1999, 29, 1133–1137. [Google Scholar] [CrossRef]
- Perencevich, E.N.; McGregor, J.C.; Shardell, M.; Furuno, J.P.; Harris, A.D.; Morris, J.G.; Fisman, D.N.; Johnson, J.A. Summer Peaks in the Incidences of Gram-Negative Bacterial Infection Among Hospitalized Patients. Infect. Control Hosp. Epidemiol. 2008, 29, 1124–1131. [Google Scholar] [CrossRef]
- Gradel, K.O.; Nielsen, S.L.; Pedersen, C.; Knudsen, J.D.; Østergaard, C.; Arpi, M.; Jensen, T.G.; Kolmos, H.J.; Søgaard, M.; Lassen, A.T.; et al. Seasonal Variation of Escherichia coli, Staphylococcus aureus, and Streptococcus pneumoniae Bacteremia According to Acquisition and Patient Characteristics: A Population-Based Study. Infect. Control Hosp. Epidemiol. 2016, 37, 946–953. [Google Scholar] [CrossRef]
- Al-Hasan, M.N.; Lahr, B.D.; Eckel-Passow, J.E.; Baddour, L.M. Seasonal variation in Escherichia coli bloodstream infection: A population-based study. Clin. Microbiol. Infect. 2009, 15, 947–950. [Google Scholar] [CrossRef]
- Al-Hasan, M.N.; Lahr, B.D.; Eckel-Passow, J.E.; Baddour, L.M. Epidemiology and outcome of Klebsiella species bloodstream infection: A population-based study. Mayo Clin. Proc. 2010, 85, 139–144. [Google Scholar] [CrossRef]
- Al-Hasan, M.N.; Lahr, B.D.; Eckel-Passow, J.E.; Baddour, L.M. Temporal trends in Enterobacter species bloodstream infection: A population-based study from 1998-2007. Clin. Microbiol. Infect. 2011, 17, 539–545. [Google Scholar] [CrossRef]
- Anderson, D.J.; Richet, H.; Chen, L.F.; Spelman, D.W.; Hung, Y.J.; Huang, A.T.; Sexton, D.J.; Raoult, D. Seasonal variation in Klebsiella pneumoniae bloodstream infection on 4 continents. J. Infect. Dis. 2008, 197, 752–756. [Google Scholar] [CrossRef]
- Chazan, B.; Colodner, R.; Edelstein, H.; Raz, R. Seasonal variation in Escherichia coli bloodstream infections in northern Israel. Clin. Microbiol. Infect. 2011, 17, 851–854. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Matsumura, Y.; Yamamoto, M.; Nagao, M.; Ito, Y.; Takakura, S.; Ichiyama, S. Erratum to: Seasonal trend and clinical presentation of Bacillus cereus bloodstream infection: Association with summer and indwelling catheter. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 875–883. [Google Scholar] [CrossRef] [PubMed]
- Alcorn, K.; Gerrard, J.; Macbeth, D.; Steele, M. Seasonal variation in health care-associated bloodstream infection: Increase in the incidence of gram-negative bacteremia in nonhospitalized patients during summer. Am. J. Infect. Control 2013, 41, 1205–1208. [Google Scholar] [CrossRef] [PubMed]
- Eber, M.R.; Shardell, M.; Schweizer, M.L.; Laxminarayan, R.; Perencevich, E.N. Seasonal and temperature-associated increases in gram-negative bacterial bloodstream infections among hospitalized patients. PLoS ONE 2011, 6, e25298. [Google Scholar] [CrossRef]
- Fortaleza, C.M.; Caldeira, S.M.; Moreira, R.G.; Akazawa, R.T.; Corrente, J.E.; de Souza, L.o.R.; da Cunha, A.R. Tropical healthcare epidemiology: Weather determinants of the etiology of bloodstream infections in a Brazilian hospital. Infect. Control Hosp. Epidemiol. 2014, 35, 85–88. [Google Scholar] [CrossRef]
- Zangbar, B.; Rhee, P.; Pandit, V.; Hsu, C.H.; Khalil, M.; Okeefe, T.; Neumayer, L.; Joseph, B. Seasonal Variation in Emergency General Surgery. Ann. Surg. 2016, 263, 76–81. [Google Scholar] [CrossRef]
- Durkin, M.J.; Dicks, K.V.; Baker, A.W.; Lewis, S.S.; Moehring, R.W.; Chen, L.F.; Sexton, D.J.; Anderson, D.J. Seasonal Variation of Common Surgical Site Infections: Does Season Matter? Infect. Control Hosp. Epidemiol. 2015, 36, 1011–1016. [Google Scholar] [CrossRef]
- Koljonen, V.; Tukiainen, E.; Pipping, D.; Kolho, E. ‘Dog days’ surgical site infections in a Finnish trauma hospital during 2002–2005. J. Hosp. Infect. 2009, 71, 290–291. [Google Scholar] [CrossRef]
- Freeman, J.T.; Anderson, D.J.; Sexton, D.J. Seasonal peaks in Escherichia coli infections: Possible explanations and implications. Clin. Microbiol. Infect. 2009, 15, 951–953. [Google Scholar] [CrossRef]
- Richet, H. Seasonality in Gram-negative and healthcare-associated infections. Clin. Microbiol. Infect. 2012, 18, 934–940. [Google Scholar] [CrossRef]
- Kaba, H.E.J.; Kuhlmann, E.; Scheithauer, S. Thinking outside the box: Association of antimicrobial resistance with climate warming in Europe—A 30 country observational study. Int. J. Hyg. Environ. Health 2020, 223, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Collignon, P.; Athukorala, P.C.; Senanayake, S.; Khan, F. Antimicrobial resistance: The major contribution of poor governance and corruption to this growing problem. PLoS ONE 2015, 10, e0116746. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; McIlroy, S.E.; Archana, A.; Baker, D.M.; Panagiotou, G. A pollution gradient contributes to the taxonomic, functional, and resistome diversity of microbial communities in marine sediments. Microbiome 2019, 7, 104. [Google Scholar] [CrossRef] [PubMed]
- Coates-Marnane, J.; Olley, J.; Burton, J.; Grinham, A. The impact of a high magnitude flood on metal pollution in a shallow subtropical estuarine embayment. Sci. Total Environ. 2016, 569–570, 716–731. [Google Scholar] [CrossRef] [PubMed]
- Knapp, C.W.; McCluskey, S.M.; Singh, B.K.; Campbell, C.D.; Hudson, G.; Graham, D.W. Antibiotic resistance gene abundances correlate with metal and geochemical conditions in archived Scottish soils. PLoS ONE 2011, 6, e27300. [Google Scholar] [CrossRef]
- Seiler, C.; Berendonk, T.U. Heavy metal driven co-selection of antibiotic resistance in soil and water bodies impacted by agriculture and aquaculture. Front. Microbiol. 2012, 3, 399. [Google Scholar] [CrossRef]
- Singer, A.C.; Shaw, H.; Rhodes, V.; Hart, A. Review of Antimicrobial Resistance in the Environment and Its Relevance to Environmental Regulators. Front. Microbiol. 2016, 7, 1728. [Google Scholar] [CrossRef]
- Kusi, J.; Ojewole, C.O.; Ojewole, A.E.; Nwi-Mozu, I. Antimicrobial Resistance Development Pathways in Surface Waters and Public Health Implications. Antibiotics 2022, 11, 821. [Google Scholar] [CrossRef]
- Burnham, J.P. Climate change and antibiotic resistance: A deadly combination. Ther. Adv. Infect. Dis. 2021, 8, 2049936121991374. [Google Scholar] [CrossRef]
- Vieira, M.T.; Marlow, M.A.; Aguiar-Alves, F.; Pinheiro, M.G.; Freitas Alves, M.e.F.; Santos Cruz, M.L.; Saavedra Gaspar, M.C.; Rocha, R.; Velarde, L.G.; Araújo Cardoso, C.A. Living Conditions as a Driving Factor in Persistent Methicillin-resistant Staphylococcus aureus Colonization Among HIV-infected Youth. Pediatr. Infect. Dis. J. 2016, 35, 1126–1131. [Google Scholar] [CrossRef]
- Immergluck, L.C.; Leong, T.; Malhotra, K.; Parker, T.C.; Ali, F.; Jerris, R.C.; Rust, G.S. Correction to: Geographic surveillance of community associated MRSA infections in children using electronic health record data. BMC Infect. Dis. 2019, 19, 396. [Google Scholar] [CrossRef] [PubMed]
- Blakiston, M.R.; Freeman, J.T. Population-level exposures associated with MRSA and ESBL-E. coli infection across district health boards in Aotearoa New Zealand: An ecological study. N. Z. Med. J. 2020, 133, 62–69. [Google Scholar] [PubMed]
- Cardoso, M.R.; Cousens, S.N.; de Góes Siqueira, L.F.; Alves, F.M.; D’Angelo, L.A. Crowding: Risk factor or protective factor for lower respiratory disease in young children? BMC Public Health 2004, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Gowrisankar, G.; Chelliah, R.; Ramakrishnan, S.R.; Elumalai, V.; Dhanamadhavan, S.; Brindha, K.; Antony, U.; Elango, L. Chemical, microbial and antibiotic susceptibility analyses of groundwater after a major flood event in Chennai. Sci. Data 2017, 4, 170135. [Google Scholar] [CrossRef]
- Pingfeng, Y. Elevated Levels of Pathogenic Indicator Bacteria and Antibiotic Resistance Genes After Hurricane Harvey’s Flooding in Houston. Environ. Sci. Technol. Lett. 2018, 5, 481–486. [Google Scholar]
- Von Seidlein, L.; Deen, J.L. Preventing cholera outbreaks through early targeted interventions. PLoS Med. 2018, 15, e1002510. [Google Scholar] [CrossRef]
- Nelson, E.J.; Andrews, J.R.; Maples, S.; Barry, M.; Clemens, J.D. Is a Cholera Outbreak Preventable in Post-earthquake Nepal? PLoS Negl. Trop. Dis. 2015, 9, e0003961. [Google Scholar] [CrossRef]
- Brander, R.L.; Walson, J.L.; John-Stewart, G.C.; Naulikha, J.M.; Ndonye, J.; Kipkemoi, N.; Rwigi, D.; Singa, B.O.; Pavlinac, P.B. Correlates of multi-drug non-susceptibility in enteric bacteria isolated from Kenyan children with acute diarrhea. PLoS Negl. Trop. Dis. 2017, 11, e0005974. [Google Scholar] [CrossRef]
- Prüss, A. Review of epidemiological studies on health effects from exposure to recreational water. Int. J. Epidemiol. 1998, 27, 1–9. [Google Scholar] [CrossRef]
- Holcomb, D.; Palli, L.; Setty, K.; Uprety, S. Water and health seminar and special issue highlight ideas that will change the field. Int. J. Hyg. Environ. Health 2020, 226, 113529. [Google Scholar] [CrossRef]
- Chyerochana, N.; Kongprajug, A.; Somnark, P.; Leelapanang Kamphaengthong, P.; Mongkolsuk, S.; Sirikanchana, K. Distributions of enterococci and human-specific bacteriophages of enterococci in a tropical watershed. Int. J. Hyg. Environ. Health 2020, 226, 113482. [Google Scholar] [CrossRef] [PubMed]
- Kay, D.; Fleisher, J.M.; Salmon, R.L.; Jones, F.; Wyer, M.D.; Godfree, A.F.; Zelenauch-Jacquotte, Z.; Shore, R. Predicting likelihood of gastroenteritis from sea bathing: Results from randomised exposure. Lancet 1994, 344, 905–909. [Google Scholar] [CrossRef] [PubMed]
- Van Asperen, I.A.; Medema, G.; Borgdorff, M.W.; Sprenger, M.J.; Havelaar, A.H. Risk of gastroenteritis among triathletes in relation to faecal pollution of fresh waters. Int. J. Epidemiol. 1998, 27, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, K.J.; Patel, S.; Gibson, M.K.; Lauber, C.L.; Knight, R.; Fierer, N.; Dantas, G. Bacterial phylogeny structures soil resistomes across habitats. Nature 2014, 509, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Karkman, A.; Do, T.T.; Walsh, F.; Virta, M.P.J. Antibiotic-Resistance Genes in Waste Water. Trends Microbiol. 2018, 26, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, G.; Ye, C.; Liu, W. Bacterial community and climate change implication affected the diversity and abundance of antibiotic resistance genes in wetlands on the Qinghai-Tibetan Plateau. J. Hazard. Mater. 2019, 361, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.X.; Zhang, T.; Fang, H.H. Antibiotic resistance genes in water environment. Appl. Microbiol. Biotechnol. 2009, 82, 397–414. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, L.; Manaia, C.; Merlin, C.; Schwartz, T.; Dagot, C.; Ploy, M.C.; Michael, I.; Fatta-Kassinos, D. Urban wastewater treatment plants as hotspots for antibiotic resistant bacteria and genes spread into the environment: A review. Sci. Total Environ. 2013, 447, 345–360. [Google Scholar] [CrossRef]
- Gandra, S.; Alvarez-Uria, G.; Turner, P.; Joshi, J.; Limmathurotsakul, D.; van Doorn, H.R. Antimicrobial Resistance Surveillance in Low- and Middle-Income Countries: Progress and Challenges in Eight South Asian and Southeast Asian Countries. Clin. Microbiol. Rev. 2020, 33, e00048-19. [Google Scholar] [CrossRef]
- George, A. Antimicrobial Resistance (AMR) in the Food Chain: Trade, One Health and Codex. Trop. Med. Infect. Dis. 2019, 4, 54. [Google Scholar] [CrossRef]
- Hendriksen, R.S.; Bortolaia, V.; Tate, H.; Tyson, G.H.; Aarestrup, F.M.; McDermott, P.F. Using Genomics to Track Global Antimicrobial Resistance. Front. Public Health 2019, 7, 242. [Google Scholar] [CrossRef] [PubMed]
- Munk, P.; Knudsen, B.E.; Lukjancenko, O.; Duarte, A.S.R.; Van Gompel, L.; Luiken, R.E.C.; Smit, L.A.M.; Schmitt, H.; Garcia, A.D.; Hansen, R.B.; et al. Author Correction: Abundance and diversity of the faecal resistome in slaughter pigs and broilers in nine European countries. Nat. Microbiol. 2018, 3, 1186. [Google Scholar] [CrossRef]
- Waseem, H.; Jameel, S.; Ali, J.; Saleem Ur Rehman, H.; Tauseef, I.; Farooq, U.; Jamal, A.; Ali, M.I. Contributions and Challenges of High Throughput qPCR for Determining Antimicrobial Resistance in the Environment: A Critical Review. Molecules 2019, 24, 163. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Peng, Y.; Chan, C.L.; On, H.; Wai, H.K.; Shekhawat, S.S.; Gupta, A.B.; Varshney, A.K.; Chuanchuen, R.; Zhou, X.; et al. Metagenomic Survey Reveals More Diverse and Abundant Antibiotic Resistance Genes in Municipal Wastewater Than Hospital Wastewater. Front. Microbiol. 2021, 12, 712843. [Google Scholar] [CrossRef] [PubMed]
- Pazda, M.; Kumirska, J.; Stepnowski, P.; Mulkiewicz, E. Antibiotic resistance genes identified in wastewater treatment plant systems—A review. Sci. Total Environ. 2019, 697, 134023. [Google Scholar] [CrossRef]
- Arias-Andres, M.; Klümper, U.; Rojas-Jimenez, K.; Grossart, H.P. Microplastic pollution increases gene exchange in aquatic ecosystems. Environ. Pollut. 2018, 237, 253–261. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, W.; Yang, X.; Wang, J.; Lin, H.; Yang, Y. Microplastics are a hotspot for antibiotic resistance genes: Progress and perspective. Sci. Total Environ. 2021, 773, 145643. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, G.; Song, W.; Ye, C.; Lin, H.; Li, Z.; Liu, W. Plastics in the marine environment are reservoirs for antibiotic and metal resistance genes. Environ. Int. 2019, 123, 79–86. [Google Scholar] [CrossRef]
- Dong, H.; Chen, Y.; Wang, J.; Zhang, Y.; Zhang, P.; Li, X.; Zou, J.; Zhou, A. Interactions of microplastics and antibiotic resistance genes and their effects on the aquaculture environments. J. Hazard. Mater. 2021, 403, 123961. [Google Scholar] [CrossRef]
- Song, R.; Sun, Y.; Li, X.; Ding, C.; Huang, Y.; Du, X.; Wang, J. Biodegradable microplastics induced the dissemination of antibiotic resistance genes and virulence factors in soil: A metagenomic perspective. Sci. Total Environ. 2022, 828, 154596. [Google Scholar] [CrossRef]
- Nyoni, N.M.B.; Grab, S.; Archer, E.R.M. Heat stress and chickens: Climate risk effects on rural poultry farming in low-income countries. Clim. Dev. 2019, 11, 83–90. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Chaudhury, R.R. Antibiotic Resistance in India: Drivers and Opportunities for Action. PLoS Med. 2016, 13, e1001974. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.P.; Bu, D.P.; Carrique-Mas, J.; Fèvre, E.M.; Gilbert, M.; Grace, D.; Hay, S.I.; Jiwakanon, J.; Kakkar, M.; Kariuki, S.; et al. Antibiotic resistance: Mitigation opportunities in livestock sector development. Animal 2017, 11, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, J.F.; Grace, D. The consequences of human actions on risks for infectious diseases: A review. Infect. Ecol. Epidemiol. 2015, 5, 30048. [Google Scholar] [CrossRef] [PubMed]
- Silbergeld, E.K.; Graham, J.; Price, L.B. Industrial food animal production, antimicrobial resistance, and human health. Annu. Rev. Public Health 2008, 29, 151–169. [Google Scholar] [CrossRef]
- Rigos, G.; Troisi, G.M. Antibacterial Agents in Mediterranean Finfish Farming: A Synopsis of Drug Pharmacoki-netics in Important Euryhaline Fish Species and Possible Environmental Implications. Fish Biol. Fish. 2005, 15, 53–73. [Google Scholar] [CrossRef]
- Watts, J.E.M.; Schreier, H.J.; Lanska, L.; Hale, M.S. The Rising Tide of Antimicrobial Resistance in Aquaculture: Sources, Sinks and Solutions. Mar. Drugs 2017, 15, 158. [Google Scholar] [CrossRef]
- Zago, V.; Veschetti, L.; Patuzzo, C.; Malerba, G.; Lleo, M.M. Shewanella algae and Vibrio spp. strains isolated in Italian aquaculture farms are reservoirs of antibiotic resistant genes that might constitute a risk for human health. Mar. Pollut. Bull. 2020, 154, 111057. [Google Scholar] [CrossRef]
- Anastasiou, T.I.; Mandalakis, M.; Krigas, N.; Vézignol, T.; Lazari, D.; Katharios, P.; Dailianis, T.; Antonopoulou, E. Comparative Evaluation of Essential Oils from Medicinal-Aromatic Plants of Greece: Chemical Composition, Antioxidant Capacity and Antimicrobial Activity against Bacterial Fish Pathogens. Molecules 2019, 25, 148. [Google Scholar] [CrossRef]
- Pepi, M.; Focardi, S. Antibiotic-Resistant Bacteria in Aquaculture and Climate Change: A Challenge for Health in the Mediterranean Area. Int. J. Environ. Res. Public Health 2021, 18, 5723. [Google Scholar] [CrossRef]
- Reverter, M.; Sarter, S.; Caruso, D.; Avarre, J.C.; Combe, M.; Pepey, E.; Pouyaud, L.; Vega-Heredía, S.; de Verdal, H.; Gozlan, R.E. Aquaculture at the crossroads of global warming and antimicrobial resistance. Nat. Commun. 2020, 11, 1870. [Google Scholar] [CrossRef] [PubMed]
- The Lancet Respiratory Medicine. Antimicrobial resistance-what can we learn from climate change? Lancet Respir. Med. 2016, 4, 845. [Google Scholar] [CrossRef] [PubMed]
- Hay, A.D.; Redmond, N.M.; Turnbull, S.; Christensen, H.; Thornton, H.; Little, P.; Thompson, M.; Delaney, B.; Lovering, A.M.; Muir, P.; et al. Development and internal validation of a clinical rule to improve antibiotic use in children presenting to primary care with acute respiratory tract infection and cough: A prognostic cohort study. Lancet Respir. Med. 2016, 4, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Global Leaders Group on Antimicrobial Resistance; World Health Organization (WHO). Antimicrobial Resistance and the Climate Crisis. 2021. Available online: https://www.amrleaders.org/resources/m/item/antimicrobial-resistance-and-the-climate-crisis (accessed on 1 November 2022).
- Rodríguez-González, A.; Zanin, M.; Menasalvas-Ruiz, E. Public Health and Epidemiology Informatics: Can Artificial Intelligence Help Future Global Challenges? An Overview of Antimicrobial Resistance and Impact of Climate Change in Disease Epidemiology. Yearb. Med. Inform. 2019, 28, 224–231. [Google Scholar] [CrossRef]
- González, C.; Wang, O.; Strutz, S.E.; González-Salazar, C.; Sánchez-Cordero, V.; Sarkar, S. Climate change and risk of leishmaniasis in north america: Predictions from ecological niche models of vector and reservoir species. PLoS Negl. Trop. Dis. 2010, 4, e585. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.; Gu, J.; Zhou, Z.; Wang, Z. Artificial neural networks for infectious diarrhea prediction using meteorological factors in Shanghai (China). Appl. Soft Comput. 2013, 35, 280–290. [Google Scholar] [CrossRef]
- Gunasekaran Manogaran, D.L. Disease Surveillance System for Big Climate Data Processing and Dengue Transmission. Int. J. Ambient. Comput. Intell. 2018, 8, 88–105. [Google Scholar] [CrossRef]
- Woolhouse, M.; Farrar, J. Policy: An intergovernmental panel on antimicrobial resistance. Nature 2014, 509, 555–557. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Library of AMR National Action Plans. 2021. Available online: https://www.who.int/teams/surveillance-prevention-control-AMR/national-action-plan-monitoring-evaluation/library-of-national-action-plans (accessed on 1 November 2022).
- World Health Organization (WHO). Global Action Plan on Antimicrobial Resistance. 2015. Available online: https://www.who.int/publications/i/item/9789241509763 (accessed on 1 November 2022).
- World Health Organization (WHO). Health and Climate Change Survey Report: Tracking Global Progress. 2018. Available online: https://apps.who.int/iris/handle/10665/329972 (accessed on 1 November 2022).
- Hallegatte, S. Lifelines: The Resilient Infrastructure Opportunity. Sustainable Infrastructure; World Bank Group: Washington, DC, USA, 2019. [Google Scholar]
- Weldon, I.; Rogers Van Katwyk, S.; Burci, G.L.; Giur, D.; de Campos, T.C.; Eccleston-Turner, M.; Fryer, H.R.; Giubilini, A.; Hale, T.; Harrison, M.; et al. Governing Global Antimicrobial Resistance: 6 Key Lessons from the Paris Climate Agreement. Am. J. Public Health 2022, 112, 553–557. [Google Scholar] [CrossRef]
- Rogers Van Katwyk, S.; Grimshaw, J.M.; Mendelson, M.; Taljaard, M.; Hoffman, S.J. Government policy interventions to reduce human antimicrobial use: Protocol for a systematic review and meta-analysis. Syst. Rev. 2017, 6, 256. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Antimicrobial Resistance and Use Surveillance System (GLASS) Report: Early Implementation 2020. 2021. Available online: https://www.who.int/publications/i/item/9789240005587 (accessed on 1 November 2022).
- Van Katwyk, S.R.; Balasegaram, M.; Boriello, P.; Farrar, J.; Giubilini, A.; Harrison, M.; Kieny, M.P.; Kirchhelle, C.; Liu, J.; Outterson, K.; et al. A roadmap for sustainably governing the global antimicrobial commons. Lancet 2019, 394, 1788–1789. [Google Scholar] [CrossRef] [PubMed]
- Kirchhelle, C.; Atkinson, P.; Broom, A.; Chuengsatiansup, K.; Ferreira, J.P.; Fortané, N.; Frost, I.; Gradmann, C.; Hinchliffe, S.; Hoffman, S.J.; et al. Setting the standard: Multidisciplinary hallmarks for structural, equitable and tracked antibiotic policy. BMJ Glob. Health 2020, 5, e003091. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, S.J.; Røttingen, J.A.; Frenk, J. International Law Has a Role to Play in Addressing Antibiotic Resistance. J. Law Med. Ethics 2015, 43 (Suppl. 3), 65–67. [Google Scholar] [CrossRef] [PubMed]
- Livermore, D.M. The 2018 Garrod Lecture: Preparing for the Black Swans of resistance. J. Antimicrob. Chemother. 2018, 73, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Roope, L.S.J.; Smith, R.D.; Pouwels, K.B.; Buchanan, J.; Abel, L.; Eibich, P.; Butler, C.C.; Tan, P.S.; Walker, A.S.; Robotham, J.V.; et al. The challenge of antimicrobial resistance: What economics can contribute. Science 2019, 364, aau4679. [Google Scholar] [CrossRef]
- Lambraki, I.A.; Cousins, M.; Graells, T.; Léger, A.; Abdelrahman, S.; Desbois, A.P.; Gallagher, R.; Staaf Larsson, B.; Mattson, B.; Henriksson, P.; et al. Governing Antimicrobial Resistance (AMR) in a Changing Climate: A Participatory Scenario Planning Approach Applied to Sweden in 2050. Front. Public Health 2022, 10, 831097. [Google Scholar] [CrossRef]
- Khan, S.; Akbar, S.M.F.; Yahiro, T.; Mahtab, M.A.; Kimitsuki, K.; Hashimoto, T.; Nishizono, A. Dengue Infections during COVID-19 Period: Reflection of Reality or Elusive Data Due to Effect of Pandemic. Int. J. Environ. Res. Public Health 2022, 19, 768. [Google Scholar] [CrossRef]
- Rahman, M.; Islam, M.; Shimanto, M.H.; Ferdous, J.; Rahman, A.A.S.; Sagor, P.S.; Chowdhury, T. A global analysis on the effect of temperature, socio-economic and environmental factors on the spread and mortality rate of the COVID-19 pandemic. Environ. Dev. Sustain. 2021, 23, 9352–9366. [Google Scholar] [CrossRef]
- Jamshidi, S.; Baniasad, M.; Niyogi, D. Global to USA County Scale Analysis of Weather, Urban Density, Mobility, Homestay, and Mask Use on COVID-19. Int. J. Environ. Res. Public Health 2020, 17, 7847. [Google Scholar] [CrossRef]
- Islam, A.R.M.T.; Hasanuzzaman, M.; Azad, M.A.K.; Salam, R.; Toshi, F.Z.; Khan, M.S.I.; Alam, G.M.M.; Ibrahim, S.M. Effect of meteorological factors on COVID-19 cases in Bangladesh. Environ. Dev. Sustain. 2021, 23, 9139–9162. [Google Scholar] [CrossRef]
- Meo, S.A.; Abukhalaf, A.A.; Alomar, A.A.; Sumaya, O.Y.; Sami, W.; Shafi, K.M.; Meo, A.S.; Usmani, A.M.; Akram, J. Effect of heat and humidity on the incidence and mortality due to COVID-19 pandemic in European countries. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 9216–9225. [Google Scholar] [CrossRef] [PubMed]
- McMullen, K.M.; Smith, B.A.; Rebmann, T. Impact of SARS-CoV-2 on hospital acquired infection rates in the United States: Predictions and early results. Am. J. Infect. Control 2020, 48, 1409–1411. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, S.G.; Ahammad, S.Z. COVID-19 and antimicrobial resistance: A cross-study. Sci. Total Environ. 2022, 807, 150873. [Google Scholar] [CrossRef]
- Petrie, B.; Barden, R.; Kasprzyk-Hordern, B. A review on emerging contaminants in wastewaters and the environment: Current knowledge, understudied areas and recommendations for future monitoring. Water Res. 2015, 72, 3–27. [Google Scholar] [CrossRef]
- Appa, R.; Mhaisalkar, V.A.; Bafana, A.; Saravana Devi, S.; Krishnamurthi, K.; Chakrabarti, T.; Naoghare, P.K. Simultaneous quantitative monitoring of four indicator contaminants of emerging concern (CEC) in different water sources of Central India using SPE/LC-(ESI)MS-MS. Environ. Monit. Assess. 2018, 190, 489. [Google Scholar] [CrossRef]
- Barbosa, M.O.; Ribeiro, A.R.; Ratola, N.; Hain, E.; Homem, V.; Pereira, M.F.R.; Blaney, L.; Silva, A.M.T. Spatial and seasonal occurrence of micropollutants in four Portuguese rivers and a case study for fluorescence excitation-emission matrices. Sci. Total Environ. 2018, 644, 1128–1140. [Google Scholar] [CrossRef]
- Dindarloo, K.; Aghamolaei, T.; Ghanbarnejad, A.; Turki, H.; Hoseinvandtabar, S.; Pasalari, H.; Ghaffari, H.R. Pattern of disinfectants use and their adverse effects on the consumers after COVID-19 outbreak. J. Environ. Health Sci. Eng. 2020, 18, 1301–1310. [Google Scholar] [CrossRef]
- Buffet-Bataillon, S.; Branger, B.; Cormier, M.; Bonnaure-Mallet, M.; Jolivet-Gougeon, A. Effect of higher minimum inhibitory concentrations of quaternary ammonium compounds in clinical E. coli isolates on antibiotic susceptibilities and clinical outcomes. J. Hosp. Infect. 2011, 79, 141–146. [Google Scholar] [CrossRef]
- Jutkina, J.; Marathe, N.P.; Flach, C.F.; Larsson, D.G.J. Antibiotics and common antibacterial biocides stimulate horizontal transfer of resistance at low concentrations. Sci. Total Environ. 2018, 616–617, 172–178. [Google Scholar] [CrossRef]
- Guo, H.; Xue, S.; Nasir, M.; Gu, J.; Lv, J. Impacts of cadmium addition on the alteration of microbial community and transport of antibiotic resistance genes in oxytetracycline contaminated soil. J. Environ. Sci. 2021, 99, 51–58. [Google Scholar] [CrossRef]
- Zhao, Y.; Gao, J.; Wang, Z.; Dai, H.; Wang, Y. Responses of bacterial communities and resistance genes on microplastics to antibiotics and heavy metals in sewage environment. J. Hazard. Mater. 2021, 402, 123550. [Google Scholar] [CrossRef] [PubMed]
- Stern, N. Economic development, climate and values: Making policy. Proc. Biol. Sci. 2015, 282, 20150820. [Google Scholar] [CrossRef] [PubMed]
- Coast, J.; Smith, R.D. Distributional Considerations in Economic Responses to Antimicrobial Resistance. Public Health Ethics 2015, 8, 225–237. [Google Scholar] [CrossRef]
- Bruinsma, N.; Hutchinson, J.M.; van den Bogaard, A.E.; Giamarellou, H.; Degener, J.; Stobberingh, E.E. Influence of population density on antibiotic resistance. J. Antimicrob. Chemother. 2003, 51, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Beović, B.; Doušak, M.; Ferreira-Coimbra, J.; Nadrah, K.; Rubulotta, F.; Belliato, M.; Berger-Estilita, J.; Ayoade, F.; Rello, J.; Erdem, H. Antibiotic use in patients with COVID-19: A ’snapshot’ Infectious Diseases International Research Initiative (ID-IRI) survey. J. Antimicrob. Chemother. 2020, 75, 3386–3390. [Google Scholar] [CrossRef]
- Huttner, B.; Goossens, H.; Verheij, T.; Harbarth, S. on behalf of the CHAMP Consortium. Characteristics and outcomes of public campaigns aimed at improving the use of antibiotics in outpatients in high-income countries. Lancet Infect. Dis. 2010, 10, 17–31. [Google Scholar] [CrossRef]
- Roope, L.S.J.; Tonkin-Crine, S.; Butler, C.C.; Crook, D.; Peto, T.; Peters, M.; Walker, A.S.; Wordsworth, S. Reducing demand for antibiotic prescriptions: Evidence from an online survey of the general public on the interaction between preferences, beliefs and information, United Kingdom, 2015. Euro Surveill. 2018, 23, 1700424. [Google Scholar] [CrossRef]
- The Lancet Planetary Health. Welcome to The Lancet Planetary Health. Lancet Planet. Health 2017, 1, e1. [Google Scholar] [CrossRef]
| Microorganisms | Role of Climate Change | Disease |
|---|---|---|
| Campylobacter spp. and Salmonella spp. | Rising temperatures in water system contributes to better survival of these microorganisms [31,58,59,82] | Waterborne and foodborne diseases |
| Vibrio cholerae | Rising temperatures led to natural disasters, determining better conditions for the microorganism survival [59,116] | Waterborne diseases (Cholera) |
| Candida auris | Gained thermotolerance and salinity tolerance on the wetland ecosystem [31,76,77] | Fungal infection (Candidiasis) |
| Plasmodium falciparum | Rising temperatures and humidity contributes to increased transmissibility [56,78,80] | Vector-borne disease (Malaria) |
| Zika, Chikungunya and Dengue viruses, Tripanosoma cruzi | Warmer temperatures led to rising spread of vectors, even in winter months [83] | Vector-borne diseases (Zika, Chikungunya, Dengue and Chagas diseases) |
| Pseudomonas aeruginosa, Klebsiella pneumoniae, Escherichia coli, and Staphylococcus aureus | Warm-season changes in temperature contributes to their optimal growth conditions at 32–36 °C [101] | Gram negative infections (especially in healthcare settings) |
| SARS-CoV-2 | Increased aridity and prolonged droughts led to bats migration and increased viral transmission [32,33] | COVID-19 disease |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magnano San Lio, R.; Favara, G.; Maugeri, A.; Barchitta, M.; Agodi, A. How Antimicrobial Resistance Is Linked to Climate Change: An Overview of Two Intertwined Global Challenges. Int. J. Environ. Res. Public Health 2023, 20, 1681. https://doi.org/10.3390/ijerph20031681
Magnano San Lio R, Favara G, Maugeri A, Barchitta M, Agodi A. How Antimicrobial Resistance Is Linked to Climate Change: An Overview of Two Intertwined Global Challenges. International Journal of Environmental Research and Public Health. 2023; 20(3):1681. https://doi.org/10.3390/ijerph20031681
Chicago/Turabian StyleMagnano San Lio, Roberta, Giuliana Favara, Andrea Maugeri, Martina Barchitta, and Antonella Agodi. 2023. "How Antimicrobial Resistance Is Linked to Climate Change: An Overview of Two Intertwined Global Challenges" International Journal of Environmental Research and Public Health 20, no. 3: 1681. https://doi.org/10.3390/ijerph20031681
APA StyleMagnano San Lio, R., Favara, G., Maugeri, A., Barchitta, M., & Agodi, A. (2023). How Antimicrobial Resistance Is Linked to Climate Change: An Overview of Two Intertwined Global Challenges. International Journal of Environmental Research and Public Health, 20(3), 1681. https://doi.org/10.3390/ijerph20031681










