Prolonged Disability following Re-Exposure after Complete Recovery from Aerotoxic Syndrome: A Case Report
Abstract
1. Introduction
2. Detailed Case Description
2.1. Mechanism of Injury and Initial Presentation
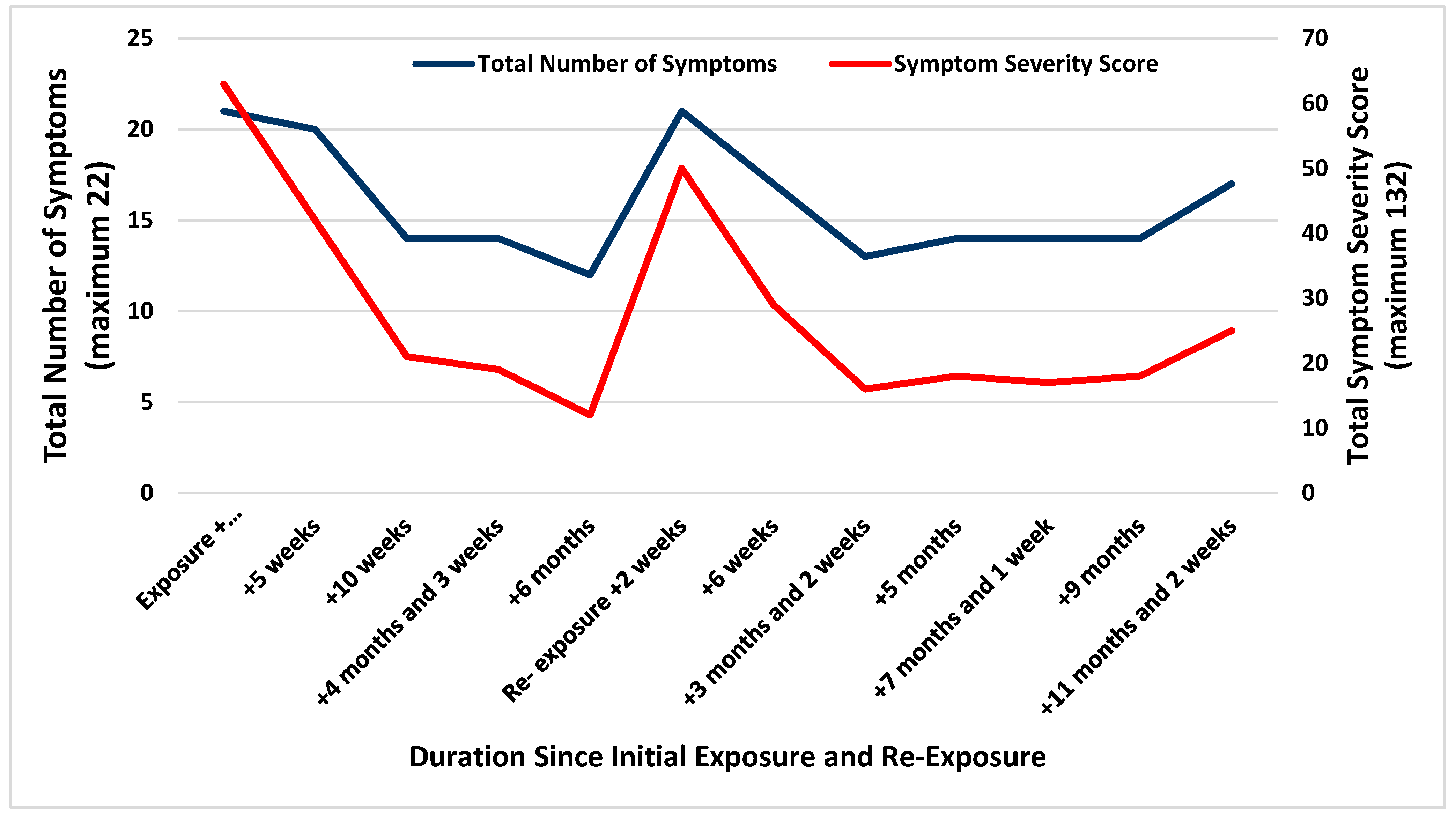
2.2. Evaluation and Management
2.3. Outcome and Disability
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mawdsley, S.E. Burden of Proof: The Debate Surrounding Aerotoxic Syndrome. J. Contemp. Hist. 2022, 57, 959–974. [Google Scholar] [CrossRef] [PubMed]
- Hageman, G.; Pal, T.M.; Nihom, J.; Mackenzie Ross, S.J.; Berg, M.V.D. Aerotoxic syndrome, discussion of possible diagnostic criteria. Clin. Toxicol. 2020, 58, 414–416. [Google Scholar] [CrossRef] [PubMed]
- Environmental Health Criteria 101—Tricresyl Phosphate. World Health Organization. 1990. Available online: https://apps.who.int/iris/bitstream/handle/10665/39784/9241571101-eng.pdf (accessed on 6 January 2023).
- Abou-Donia, M.B.; Abou-Donia, M.M.; ElMasry, E.M.; Monro, J.A.; Mulder, M.F. Autoantibodies to nervous system-specific proteins are elevated in sera of flight crew members: Biomarkers for nervous system injury. J. Toxicol. Environ. Health. Part A 2013, 76, 363–380. [Google Scholar] [CrossRef] [PubMed]
- Abou Donia, M.; Goot, F.R.; Mulder, M.F. Autoantibody markers of neural degeneration are associated with post-mortem histopathological alterations of a neurologically injured pilot. J. Biol. Phys. Chem. 2014, 14, 34–53. [Google Scholar] [CrossRef]
- Schopfer, L.M.; Masson, P.; Lamourette, P.; Simon, S.; Lockridge, O. Detection of cresyl phosphate-modified butyrylcholinesterase in human plasma for chemical exposure associated with aerotoxic syndrome. Anal. Biochem. 2014, 461, 17–26. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Budnik, L.T.; Baur, X.; Hageman, G.; Roig, J.; Coxon, L.; Furlong, C.E.; Gee, D.; Loraine, T.; Terry, A.V., Jr.; Midavaine, J.; et al. Health consequences of exposure to aircraft contaminated air and fume events: A narrative review and medical protocol for the investigation of exposed aircrew and passengers. Environ. Health A Glob. Access Sci. Source 2023, 22, 43. [Google Scholar] [CrossRef]
- SCAT3. Br. J. Sports Med. 2013, 47, 259.
- Michaelis, S.; Burdon, J.; Howard, C. Vyvyan & World Health Organization. Regional Office for Europe. Aerotoxic syndrome: A new occupational disease? Public Health Panor. 2017, 3, 198–211. [Google Scholar]
- National Center for Biotechnology Information (2023). PubChem Compound Summary for CID 6529, Tricresyl Phosphate. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Tricresyl-phosphate (accessed on 4 January 2023).
- Air Safety and Cabin Air Quality in the BAE 146 Aircraft. Parliament of the Commonwealth of Australia. (2000, October). Available online: https://www.aph.gov.au/binaries/senate/committee/rrat_ctte/completed_inquiries/1999-02/bae/report/report.pdf (accessed on 5 January 2023).
- Susser, M.; Stein, Z. An outbreak of tri-ortho-cresyl phosphate (T.O.C.P.) poisoning in Durban. Br. J. Ind. Med. 1957, 14, 111–120. [Google Scholar] [CrossRef][Green Version]
- Naughton, S.X.; Terry, A.V., Jr. Neurotoxicity in acute and repeated organophosphate exposure. Toxicology 2018, 408, 101–112. [Google Scholar] [CrossRef]
- Raad, M.; Nohra, E.; Chams, N.; Itani, M.; Talih, F.; Mondello, S.; Kobeissy, F. Autoantibodies in traumatic brain injury and central nervous system trauma. Neuroscience 2014, 281, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Kawata, K.; Liu, C.Y.; Merkel, S.F.; Ramirez, S.H.; Tierney, R.T.; Langford, D. Blood biomarkers for brain injury: What are we measuring? Neurosci. Biobehav. Rev. 2016, 68, 460–473. [Google Scholar] [CrossRef] [PubMed]
- Liyasova, M.; Li, B.; Schopfer, L.M.; Nachon, F.; Masson, P.; Furlong, C.E.; Lockridge, O. Exposure to tri-ocresyl phosphate detected in jet airplane passengers. Toxicol. Appl. Pharmacol. 2011, 256, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Reneman, L.; Schagen, S.B.; Mulder, M.; Mutsaerts, H.J.; Hageman, G.; de Ruiter, M.B. Cognitive impairment and associated loss in brain white microstructure in aircrew members exposed to engine oil fumes. Brain Imaging Behav. 2016, 10, 437–444. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Harrison, R.; Murawski, J.; Mcneely, E.; Guerriero, J.; Milton, D. Exposure to Aircraft Bleed Air Contaminants Among Airline Workers—A Guide for Health Care Providors. San Francisco, CA, USA: Occupational Health Research Consortium in Aviation (OHRCA). 2009. Available online: http://www.ohrca.org/medical-protocols-for-crews-exposed-to-engine-oil-fumes-on-aircraft/ (accessed on 30 October 2023).
- Kontos, A.; Collins, M. Concussion: A Clinical Profile Approach to Assessment and Treatment; American Psychological Association: Washington, DC, USA, 2018. [Google Scholar] [CrossRef]
- Tator, C.H.; Davis, H.S.; Dufort, P.A.; Tartaglia, M.C.; Davis, K.D.; Ebraheem, A.; Hiploylee, C. Postconcussion syndrome: Demographics and predictors in 221 patients. J. Neurosurg. 2016, 125, 1206–1216. [Google Scholar] [CrossRef] [PubMed]
- Hiploylee, C.; Dufort, P.A.; Davis, H.S.; Wennberg, R.A.; Tartaglia, M.C.; Mikulis, D.; Hazrati, L.N.; Tator, C.H. Longitudinal Study of Postconcussion Syndrome: Not Everyone Recovers. J. Neurotrauma 2017, 34, 1511–1523. [Google Scholar] [CrossRef] [PubMed]
- Tapia, R.N.; Eapen, B.C. Rehabilitation of Persistent Symptoms After Concussion. Phys. Med. Rehabil. Clin. N. Am. 2017, 28, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Kenzie, E.S.; Parks, E.L.; Bigler, E.D.; Lim, M.M.; Chesnutt, J.C.; Wakeland, W. Concussion as a Multi-Scale Complex System: An Interdisciplinary Synthesis of Current Knowledge. Front. Neurol. 2017, 8, 513. [Google Scholar] [CrossRef] [PubMed]
- Pervez, M.; Kitagawa, R.S.; Chang, T.R. Definition of Traumatic Brain Injury, Neurosurgery, Trauma Orthopedics, Neuroimaging, Psychology, and Psychiatry in Mild Traumatic Brain Injury. Neuroimaging Clin. N. Am. 2018, 28, 1–13. [Google Scholar] [CrossRef]
- Zanier, E.R.; Zoerle, T.; Di Lernia, D.; Riva, G. Virtual Reality for Traumatic Brain Injury. Front. Neurol. 2018, 9, 345. [Google Scholar] [CrossRef]
- Azizi, E.; Fielding, J.; Abel, L.A. Video game training in traumatic brain injury patients: An exploratory case report study using eye tracking. J. Eye Mov. Res. 2022, 15, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Christopher, E.; Alsaffarini, K.W.; Jamjoom, A.A. Mobile Health for Traumatic Brain Injury: A Systematic Review of the Literature and Mobile Application Market. Cureus 2019, 11, e5120. [Google Scholar] [CrossRef] [PubMed]
- Leddy, J.; Hinds, A.; Sirica, D.; Willer, B. The Role of Controlled Exercise in Concussion Management. PM R J. Inj. Funct. Rehabil. 2016, 8 (Suppl. 3), S91–S100. [Google Scholar] [CrossRef] [PubMed]
- Leddy, J.J.; Baker, J.G.; Willer, B. Active Rehabilitation of Concussion and Post-concussion Syndrome. Phys. Med. Rehabil. Clin. N. Am. 2016, 27, 437–454. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Creeden, R.; Blonien, N.; Schultz, J.K.; Wheeler, J.; Haltson, E.L.; McKinney, Z.J. Prolonged Disability following Re-Exposure after Complete Recovery from Aerotoxic Syndrome: A Case Report. Int. J. Environ. Res. Public Health 2023, 20, 7156. https://doi.org/10.3390/ijerph20247156
Creeden R, Blonien N, Schultz JK, Wheeler J, Haltson EL, McKinney ZJ. Prolonged Disability following Re-Exposure after Complete Recovery from Aerotoxic Syndrome: A Case Report. International Journal of Environmental Research and Public Health. 2023; 20(24):7156. https://doi.org/10.3390/ijerph20247156
Chicago/Turabian StyleCreeden, Race, Nick Blonien, Jill K. Schultz, Jessika Wheeler, Erik L. Haltson, and Zeke J. McKinney. 2023. "Prolonged Disability following Re-Exposure after Complete Recovery from Aerotoxic Syndrome: A Case Report" International Journal of Environmental Research and Public Health 20, no. 24: 7156. https://doi.org/10.3390/ijerph20247156
APA StyleCreeden, R., Blonien, N., Schultz, J. K., Wheeler, J., Haltson, E. L., & McKinney, Z. J. (2023). Prolonged Disability following Re-Exposure after Complete Recovery from Aerotoxic Syndrome: A Case Report. International Journal of Environmental Research and Public Health, 20(24), 7156. https://doi.org/10.3390/ijerph20247156





