Identification of Alzheimer’s Disease by Imaging: A Comprehensive Review
Abstract
1. Introduction
1.1. Image Processing and Its Applications in Disease Detection
1.2. Evidence of Alzheimer’s Disease Influencing Central Nervous System
1.3. Evidence of Alzheimer’s Disease Influencing the Visual System at the Molecular Stage
1.4. Neurochemistry Deficiency in the AD Retina
2. Non-Invasive Imaging of the Retina
2.1. Image Reconstruction-Recognizing Objects over Background Luminance
2.2. Image Filtering-Biomarkers for AD Diagnosis Based on Structural Changes in the Retina, Optic Nerves, and Lenses
2.3. Image Segmentation-Optic Nerve Fibre Imaging and AD
2.4. Image Registration
3. Discussion
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Doody, R.S.; Thomas, R.G.; Farlow, M.; Iwatsubo, T.; Vellas, B.; Joffe, S.; Kieburtz, K.; Raman, R.; Sun, X.; Aisen, P.S.; et al. Phase 3 Trials of Solanezumab for Mild-to-Moderate Alzheimer’s Disease. N. Engl. J. Med. 2014, 370, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Connolly, A.; Gaehl, E.; Martin, H.; Morris, J.; Purandare, N. Underdiagnosis of Dementia in Primary Care: Variations in the Observed Prevalence and Comparisons to the Expected Prevalence. Aging Ment. Health 2011, 15, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, L.; Anthony Crowther, R.; Martin, K.R.; Berg, N.; Coleman, M.; Goedert, M.; Spillantini, M.G. Tau Inclusions in Retinal Ganglion Cells of Human P301S Tau Transgenic Mice: Effects on Axonal Viability. Neurobiol. Aging 2011, 32, 419–433. [Google Scholar] [CrossRef] [PubMed]
- Koronyo-Hamaoui, M.; Koronyo, Y.; Ljubimov, A.V.; Miller, C.A.; Ko, M.K.; Black, K.L.; Schwartz, M.; Farkas, D.L. Identification of Amyloid Plaques in Retinas from Alzheimer’s Patients and Noninvasive in Vivo Optical Imaging of Retinal Plaques in a Mouse Model. Neuroimage 2011, 54, S204–S217. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Rasool, S.; Yang, Z.; Glabe, C.G.; Schreiber, S.S.; Ge, J.; Tan, Z. Amyloid-Peptide Vaccinations Reduce β-Amyloid Plaques but Exacerbate Vascular Deposition and Inflammation in the Retina of Alzheimer’s Transgenic Mice. Am. J. Pathol. 2009, 175, 2099–2110. [Google Scholar] [CrossRef]
- Zhang-Nunes, S.X.; Maat-Schieman, M.L.C.; Duinen, S.G.; Roos, R.A.C.; Frosch, M.P.; Greenberg, S.M. The Cerebral β-Amyloid Angiopathies: Hereditary and Sporadic. Brain Pathol. 2006, 16, 30–39. [Google Scholar] [CrossRef]
- Ardiles, Á.O.; Tapia-Rojas, C.C.; Mandal, M.; Alexandre, F.; Kirkwood, A.; Inestrosa, N.C.; Palacios, A.G. Postsynaptic Dysfunction Is Associated with Spatial and Object Recognition Memory Loss in a Natural Model of Alzheimer’s Disease. Proc. Natl. Acad. Sci. USA 2012, 109, 13835–13840. [Google Scholar] [CrossRef] [PubMed]
- Ardiles, A.O.; Ewer, J.; Acosta, M.L.; Kirkwood, A.; Martinez, A.D.; Ebensperger, L.A.; Bozinovic, F.; Lee, T.M.; Palacios, A.G. Octodon Degus (Molina 1782): A Model in Comparative Biology and Biomedicine. Cold Spring Harb. Protoc. 2013, 2013, pdb.emo071357. [Google Scholar] [CrossRef]
- Bruban, J.; Dinet, V.; Mascarelli, F. The Role of Amyloid-β in Retinal Degeneration. In Advances in Experimental Medicine and Biology; Springer: Boston, MA, USA, 2012; pp. 67–74. [Google Scholar]
- Bull, N.D.; Guidi, A.; Goedert, M.; Martin, K.R.; Spillantini, M.G. Reduced Axonal Transport and Increased Excitotoxic Retinal Ganglion Cell Degeneration in Mice Transgenic for Human Mutant P301S Tau. PLoS ONE 2012, 7, e34724. [Google Scholar] [CrossRef]
- Francis, P.T.; Palmer, A.M.; Snape, M.; Wilcock, G.K. The Cholinergic Hypothesis of Alzheimer’s Disease: A Review of Progress. J. Neurol. Neurosurg. Psychiatry 1999, 66, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Masland, R.H.; Tauchi, M. The Cholinergic Amacrine Cell. Trends Neurosci. 1986, 9, 218–223. [Google Scholar] [CrossRef]
- Schmidt, M.; Humphrey, M.F.; Wassle, H. Action and Localization of Acetylcholine in the Cat Retina. J. Neurophysiol. 1987, 58, 997–1015. [Google Scholar] [CrossRef] [PubMed]
- Strenn, K.; Dal-Bianco, P.; Weghaupt, H.; Koch, G.; Vass, C.; Gottlob, I. Pattern Electroretinogram and Luminance Electroretinogram in Alzheimer’s Disease. In Age-Associated Neurological Diseases; Springer: Vienna, Austria, 1991; pp. 73–80. [Google Scholar]
- Bulut, M.; Kurtuluş, F.; Gözkaya, O.; Erol, M.K.; Cengiz, A.; Akıdan, M.; Yaman, A. Evaluation of Optical Coherence Tomography Angiographic Findings in Alzheimer’s Type Dementia. Br. J. Ophthalmol. 2018, 102, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Casaletto, K.B.; Ward, M.E.; Baker, N.S.; Bettcher, B.M.; Gelfand, J.M.; Li, Y.; Chen, R.; Dutt, S.; Miller, B.; Kramer, J.H.; et al. Retinal Thinning Is Uniquely Associated with Medial Temporal Lobe Atrophy in Neurologically Normal Older Adults. Neurobiol. Aging 2017, 51, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Fischer, H. Three Main Layers of the Eye. Available online: http://open.umich.edu/education/med/resources/second-look-series/materials (accessed on 25 April 2022).
- Ko, F.; Muthy, Z.A.; Gallacher, J.; Sudlow, C.; Rees, G.; Yang, Q.; Keane, P.A.; Petzold, A.; Khaw, P.T.; Reisman, C.; et al. Association of Retinal Nerve Fiber Layer Thinning with Current and Future Cognitive Decline. JAMA Neurol. 2018, 75, 1198. [Google Scholar] [CrossRef]
- Yan, Y.; Wu, X.; Wang, X.; Geng, Z.; Wang, L.; Xiao, G.; Wu, Y.; Zhou, S.; Liao, R.; Wei, L.; et al. The Retinal Vessel Density Can Reflect Cognitive Function in Patients with Alzheimer’s Disease: Evidence from Optical Coherence Tomography Angiography. J. Alzheimer’s Dis. 2021, 79, 1307–1316. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.; Yang, M.; Zhu, L.; Zhou, Q.; Li, X.; Chen, Z.; Zou, C. Retinal Levels of Amyloid Beta Correlate with Cerebral Levels of Amyloid Beta in Young APPswe/PS1dE9 Transgenic Mice before Onset of Alzheimer’s Disease. Behav. Neurol. 2020, 2020, 1574816. [Google Scholar] [CrossRef] [PubMed]
- Chua, J.; Hu, Q.; Ke, M.; Tan, B.; Hong, J.; Yao, X.; Hilal, S.; Venketasubramanian, N.; Garhöfer, G.; Cheung, C.Y.; et al. Retinal Microvasculature Dysfunction Is Associated with Alzheimer’s Disease and Mild Cognitive Impairment. Alzheimer’s Res. Ther. 2020, 12, 161. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, X.; Azhati, G.; Li, T.; Xu, G.; Liu, F. Retinal Microvascular Attenuation in Mental Cognitive Impairment and Alzheimer’s Disease by Optical Coherence Tomography Angiography. Acta Ophthalmol. 2020, 98, e781–e787. [Google Scholar] [CrossRef]
- Kim, J.-I.; Kang, B.-H. Decreased Retinal Thickness in Patients with Alzheimer’s Disease Is Correlated with Disease Severity. PLoS ONE 2019, 14, e0224180. [Google Scholar] [CrossRef]
- Zabel, P.; Kałużny, J.J.; Wiłkość-Dębczyńska, M.; Gębska-Tołoczko, M.; Suwała, K.; Kucharski, R.; Araszkiewicz, A. Peripapillary Retinal Nerve Fiber Layer Thickness in Patients with Alzheimer’s Disease: A Comparison of Eyes of Patients with Alzheimer’s Disease, Primary Open-Angle Glaucoma, and Preperimetric Glaucoma and Healthy Controls. Med. Sci. Monit. 2019, 25, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, D.; Castilla-Marti, M.; Rodríguez-Gómez, O.; Valero, S.; Piferrer, A.; Martínez, G.; Martínez, J.; Serra, J.; Moreno-Grau, S.; Hernández-Olasagarre, B.; et al. Usefulness of Peripapillary Nerve Fiber Layer Thickness Assessed by Optical Coherence Tomography as a Biomarker for Alzheimer’s Disease. Sci. Rep. 2018, 8, 16345. [Google Scholar] [CrossRef]
- Koronyo, Y.; Biggs, D.; Barron, E.; Boyer, D.S.; Pearlman, J.A.; Au, W.J.; Kile, S.J.; Blanco, A.; Fuchs, D.-T.; Ashfaq, A.; et al. Retinal Amyloid Pathology and Proof-of-Concept Imaging Trial in Alzheimer’s Disease. JCI Insight 2017, 2, e93621. [Google Scholar] [CrossRef] [PubMed]
- Haan, J.; Verbraak, F.D.; Visser, P.J.; Bouwman, F.H. Retinal Thickness in Alzheimer’s Disease: A Systematic Review and Meta-analysis. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2017, 6, 162–170. [Google Scholar] [CrossRef]
- Mutlu, U.; Bonnemaijer, P.W.M.; Ikram, M.A.; Colijn, J.M.; Cremers, L.G.M.; Buitendijk, G.H.S.; Vingerling, J.R.; Niessen, W.J.; Vernooij, M.W.; Klaver, C.C.W.; et al. Retinal Neurodegeneration and Brain MRI Markers: The Rotterdam Study. Neurobiol. Aging 2017, 60, 183–191. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, H.; Li, W.; Cao, X.; Li, C. Amyloid Plaques in Retina for Diagnosis in Alzheimer’s Patients: A Meta-Analysis. Front. Aging Neurosci. 2016, 8, 267. [Google Scholar] [CrossRef] [PubMed]
- Pillai, J.A.; Bermel, R.; Bonner-Jackson, A.; Rae-Grant, A.; Fernandez, H.; Bena, J.; Jones, S.E.; Ehlers, J.P.; Leverenz, J.B. Retinal Nerve Fiber Layer Thinning in Alzheimer’s Disease. Am. J. Alzheimer’s Dis. Other Dement.® 2016, 31, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-S. Digital Image Enhancement and Noise Filtering by Use of Local Statistics. IEEE Trans. Pattern Anal. Mach. Intell. 1980, PAMI-2, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Kuan, D.T.; Sawchuk, A.A.; Strand, T.C.; Chavel, P. Adaptive Noise Smoothing Filter for Images with Signal-Dependent Noise. IEEE Trans. Pattern Anal. Mach. Intell. 1985, PAMI-7, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Frost, V.S.; Stiles, J.A.; Shanmugan, K.S.; Holtzman, J.C. A Model for Radar Images and Its Application to Adaptive Digital Filtering of Multiplicative Noise. IEEE Trans. Pattern Anal. Mach. Intell. 1982, PAMI-4, 157–166. [Google Scholar] [CrossRef]
- Daneshmand, P.G.; Mehridehnavi, A.; Rabbani, H. Reconstruction of Optical Coherence Tomography Images Using Mixed Low Rank Approximation and Second Order Tensor Based Total Variation Method. IEEE Trans. Med. Imaging 2021, 40, 865–878. [Google Scholar] [CrossRef] [PubMed]
- Cronin-Golomb, A.; Gilmore, G.C.; Neargarder, S.; Morrison, S.R.; Laudate, T.M. Enhanced Stimulus Strength Improves Visual Cognition in Aging and Alzheimer’s Disease. Cortex 2007, 43, 952–966. [Google Scholar] [CrossRef]
- Neargarder, S.A.; Stone, E.R.; Cronin-Golomb, A.; Oross, S. The Impact of Acuity on Performance of Four Clinical Measures of Contrast Sensitivity in Alzheimer’s Disease. J. Gerontol. B Psychol. Sci. Soc. Sci. 2003, 58, P54–P62. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, G.C.; Morrison, S.R.; Groth, K.E. Magnocellular Deficit Hypothesis in Alzheimer’s Disease. In Vision in Alzheimer’s Disease; Karger: Basel, Switzerland, 2004; pp. 173–198. [Google Scholar]
- Scialfa, C.T.; Kline, D.W.; Wood, P.K. Structural Modeling of Contrast Sensitivity in Adulthood. J. Opt. Soc. Am. A 2002, 19, 158. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, G.C.; Groth, K.E.; Thomas, C.W. Stimulus Contrast and Word Reading Speed in Alzheimer’s Disease. Exp. Aging Res. 2005, 31, 15–33. [Google Scholar] [CrossRef]
- Hinton, D.R.; Sadun, A.A.; Blanks, J.C.; Miller, C.A. Optic-Nerve Degeneration in Alzheimer’s Disease. N. Engl. J. Med. 1986, 315, 485–487. [Google Scholar] [CrossRef]
- Danesh-Meyer, H.V.; Birch, H.; Ku, J.Y.-F.; Carroll, S.; Gamble, G. Reduction of Optic Nerve Fibers in Patients with Alzheimer Disease Identified by Laser Imaging. Neurology 2006, 67, 1852–1854. [Google Scholar] [CrossRef] [PubMed]
- Kergoat, H.; Kergoat, M.-J.; Justino, L.; Chertkow, H.; Robillard, A.; Bergman, H. An Evaluation of the Retinal Nerve Fiber Layer Thickness by Scanning Laser Polarimetry in Individuals with Dementia of the Alzheimer Type. Acta Ophthalmol. Scand. 2001, 79, 187–191. [Google Scholar] [CrossRef]
- Parisi, V.; Restuccia, R.; Fattapposta, F.; Mina, C.; Bucci, M.G.; Pierelli, F. Morphological and Functional Retinal Impairment in Alzheimer’s Disease Patients. Clin. Neurophysiol. 2001, 112, 1860–1867. [Google Scholar] [CrossRef]
- Iseri, P.K.; Altinaş, Ö.; Tokay, T.; Yüksel, N. Relationship between Cognitive Impairment and Retinal Morphological and Visual Functional Abnormalities in Alzheimer Disease. J. Neuro-Ophthalmol. 2006, 26, 18–24. [Google Scholar] [CrossRef]
- Valenti, D.A. Neuroimaging of Retinal Nerve Fiber Layer in AD Using Optical Coherence Tomography. Neurology 2007, 69, 1060. [Google Scholar] [CrossRef] [PubMed]
- Kromer, R.; Serbecic, N.; Hausner, L.; Froelich, L.; Aboul-Enein, F.; Beutelspacher, S.C. Detection of Retinal Nerve Fiber Layer Defects in Alzheimer’s Disease Using SD-OCT. Front. Psychiatry 2014, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Mentis, M.; Grady, C.; Maisog, J.M.; Vanmeter, J.; Horwitz, B.; Pietrini, P.; Schapiro, M.; Rapoport, S. Visual Cortical Dysfunction in Alzheimer’s Disease Evaluated with a Temporally Graded “Stress Test” during PET. Am. J. Psychiatry 1996, 153, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Valenti, D. The Anterior Visual System and Circadian Function with Reference to Alzheimer’s Disease. In Vision in Alzheimer’s Disease; Karger: Basel, Switzerland, 2004; pp. 1–29. [Google Scholar]
- Ho, C.-Y.; Troncoso, J.C.; Knox, D.; Stark, W.; Eberhart, C.G. Beta-Amyloid, Phospho-Tau and Alpha-Synuclein Deposits Similar to Those in the Brain Are Not Identified in the Eyes of Alzheimer’s and Parkinson’s Disease Patients. Brain Pathol. 2014, 24, 25–32. [Google Scholar] [CrossRef]
- Isas, J.M.; Luibl, V.; Johnson, L.V.; Kayed, R.; Wetzel, R.; Glabe, C.G.; Langen, R.; Chen, J. Soluble and Mature Amyloid Fibrils in Drusen Deposits. Investig. Ophthalmol. Vis. Sci. 2010, 51, 1304. [Google Scholar] [CrossRef]
- Salobrar-García, E.; de Hoz, R.; Ramírez, A.I.; López-Cuenca, I.; Rojas, P.; Vazirani, R.; Amarante, C.; Yubero, R.; Gil, P.; Pinazo-Durán, M.D.; et al. Changes in Visual Function and Retinal Structure in the Progression of Alzheimer’s Disease. PLoS ONE 2019, 14, e0220535. [Google Scholar] [CrossRef]
- Zabel, P.; Kaluzny, J.J.; Zabel, K.; Kaluzna, M.; Lamkowski, A.; Jaworski, D.; Makowski, J.; Gebska-Toloczko, M.; Kucharski, R. Quantitative Assessment of Retinal Thickness and Vessel Density Using Optical Coherence Tomography Angiography in Patients with Alzheimer’s Disease and Glaucoma. PLoS ONE 2021, 16, e0248284. [Google Scholar] [CrossRef] [PubMed]
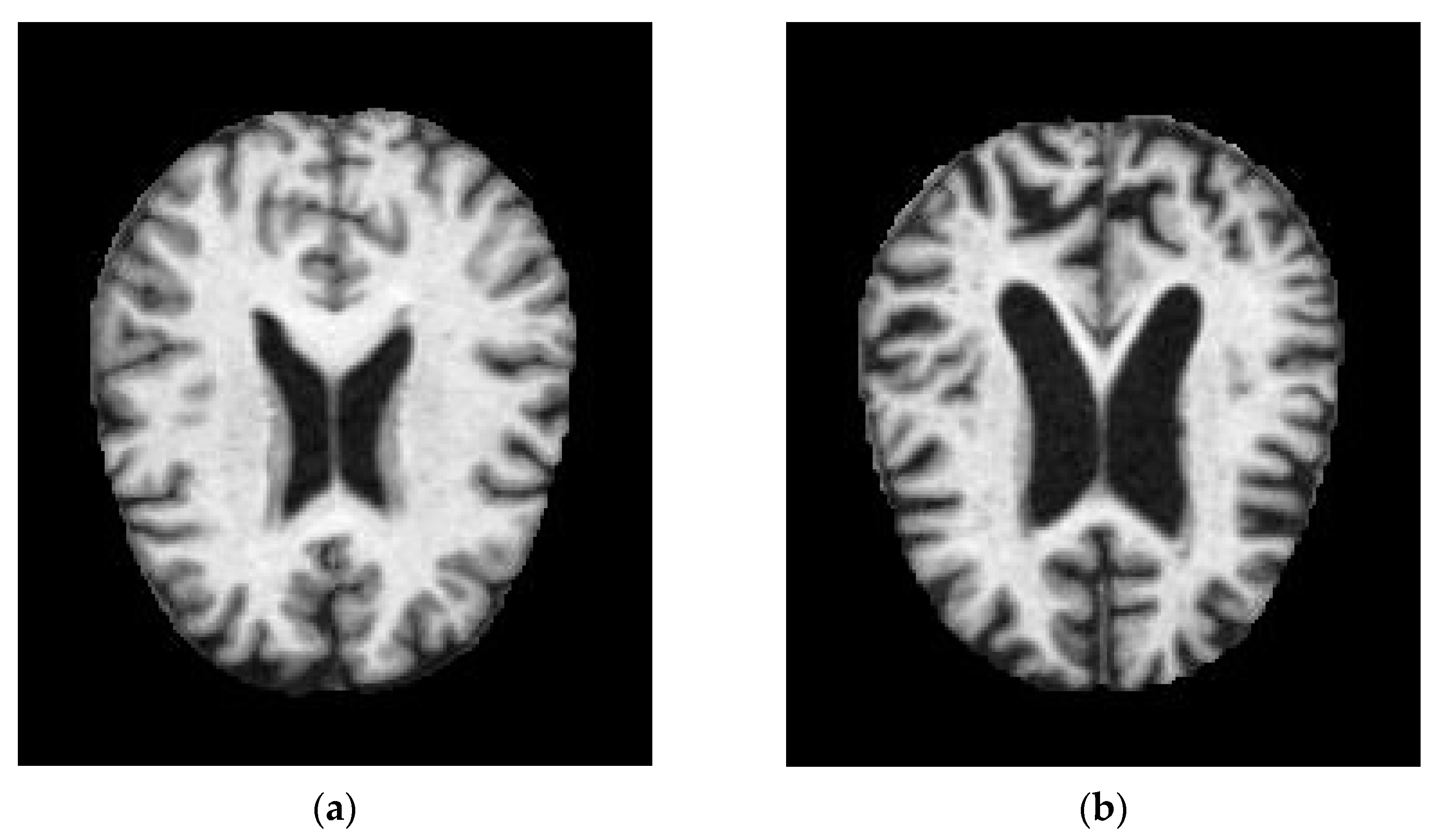
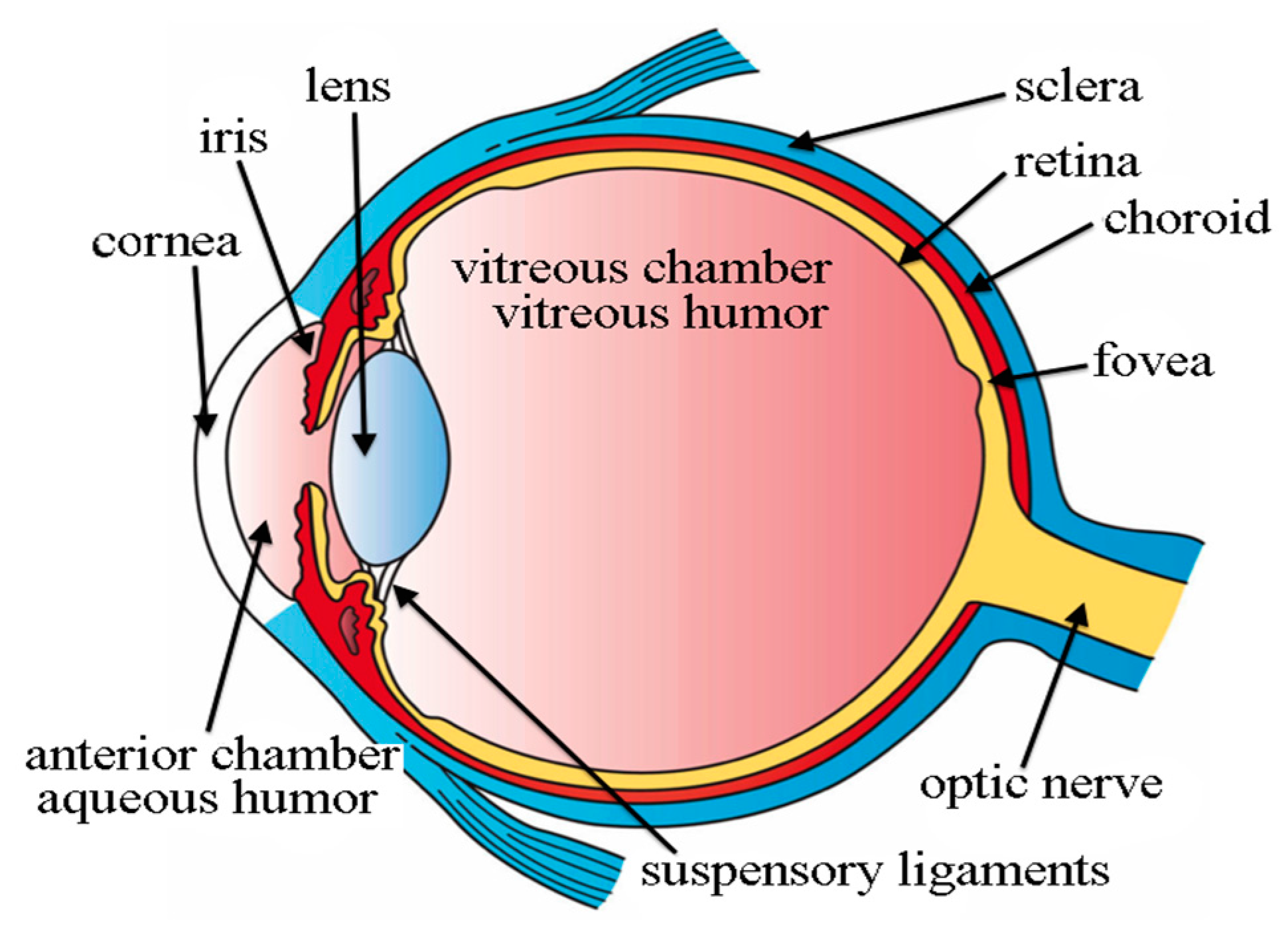
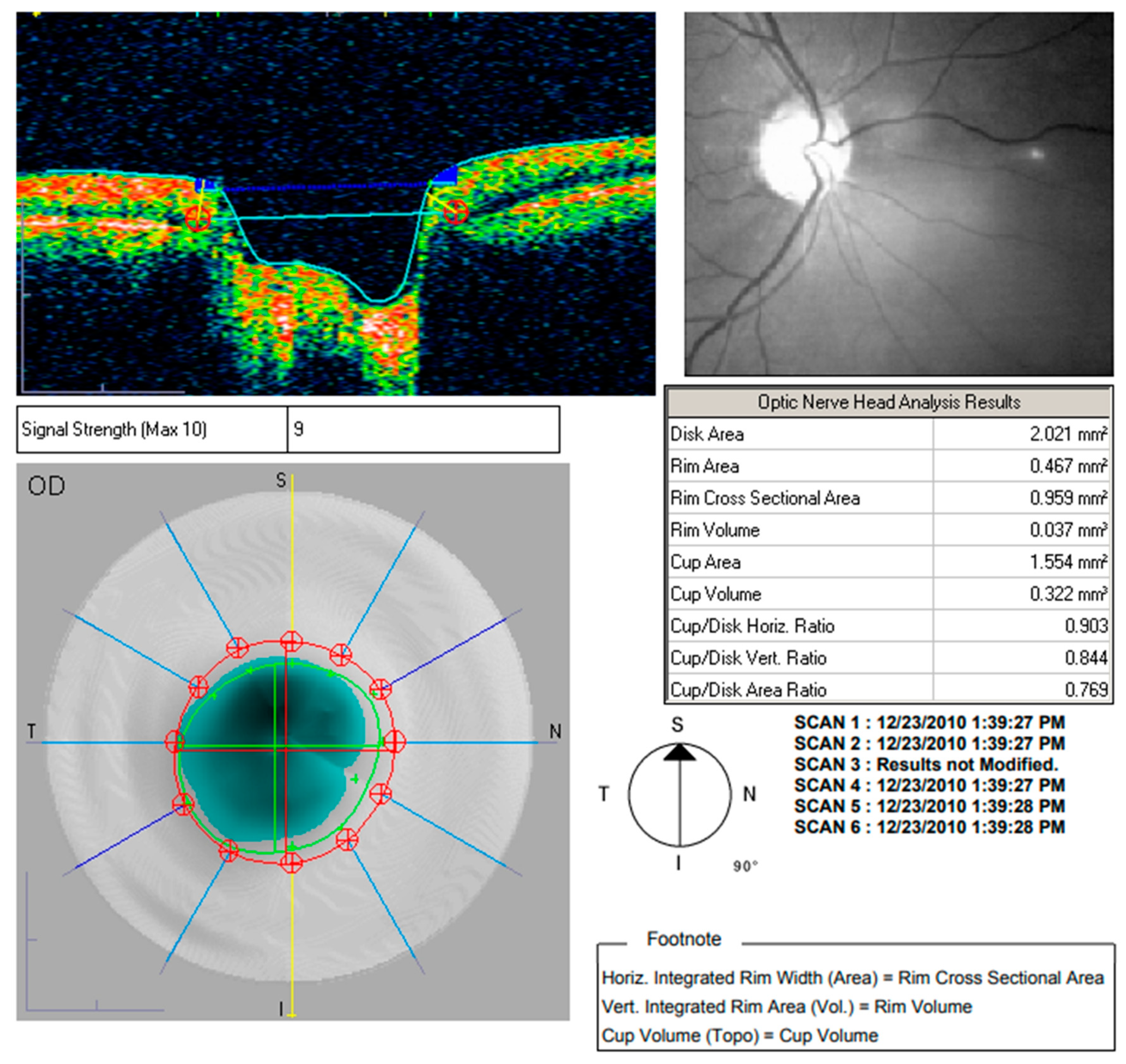
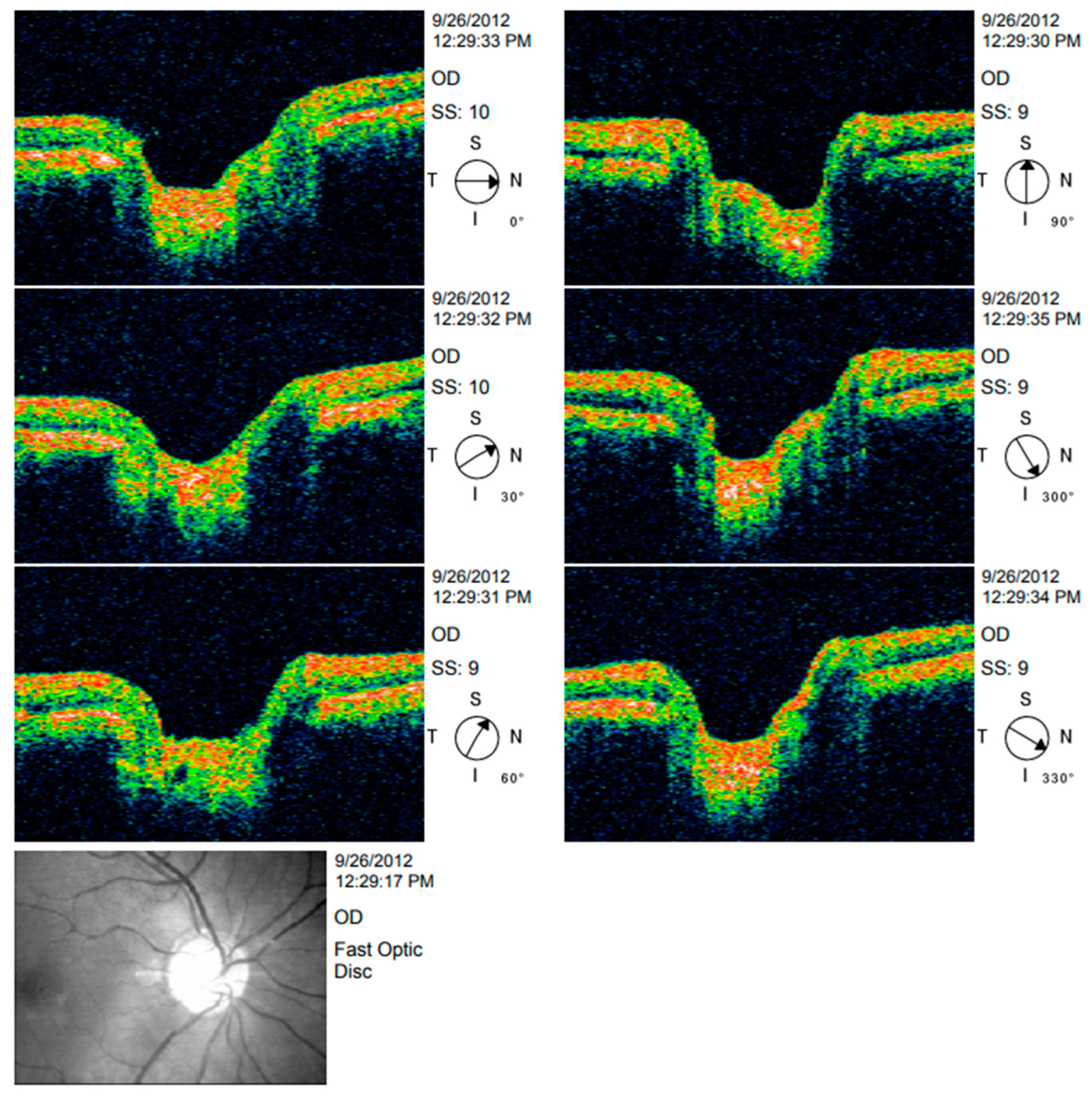
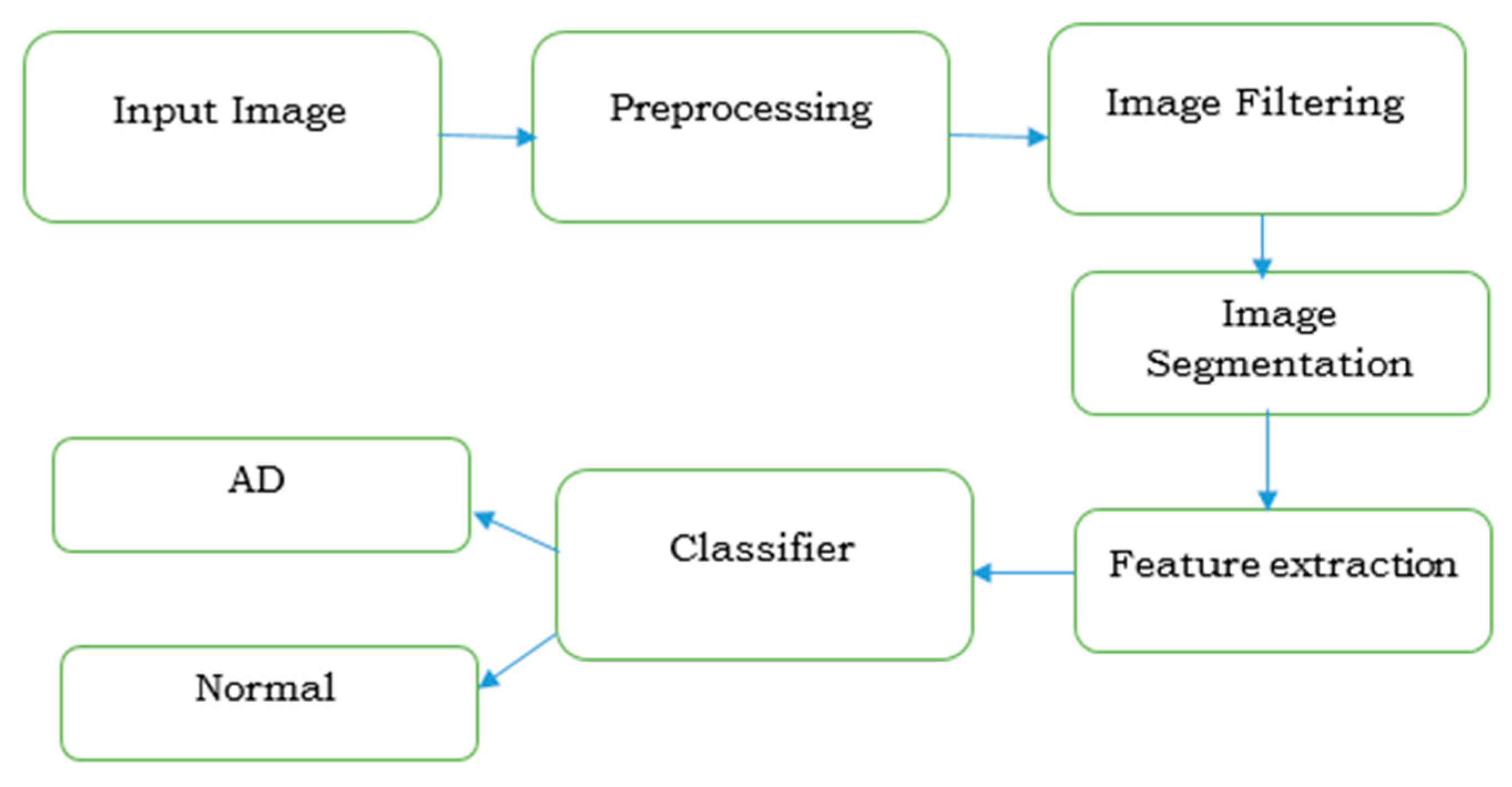
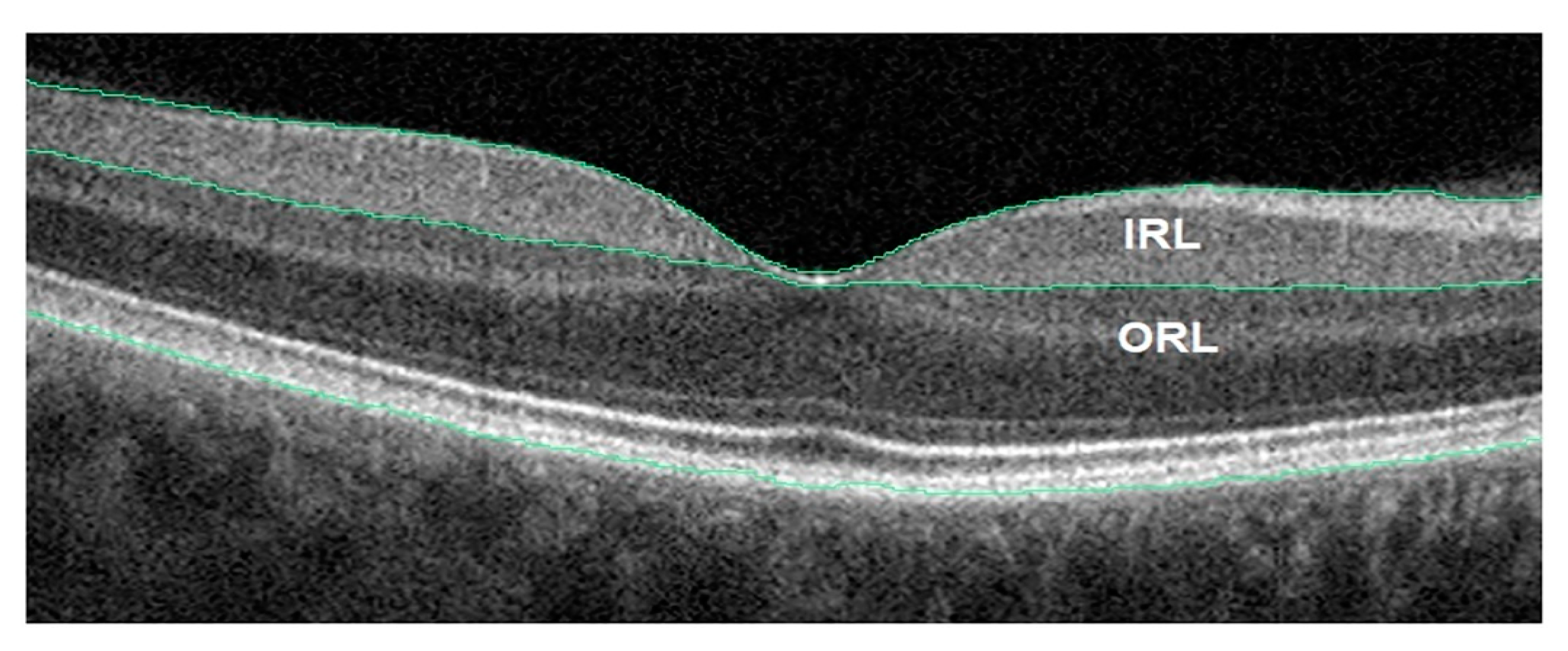
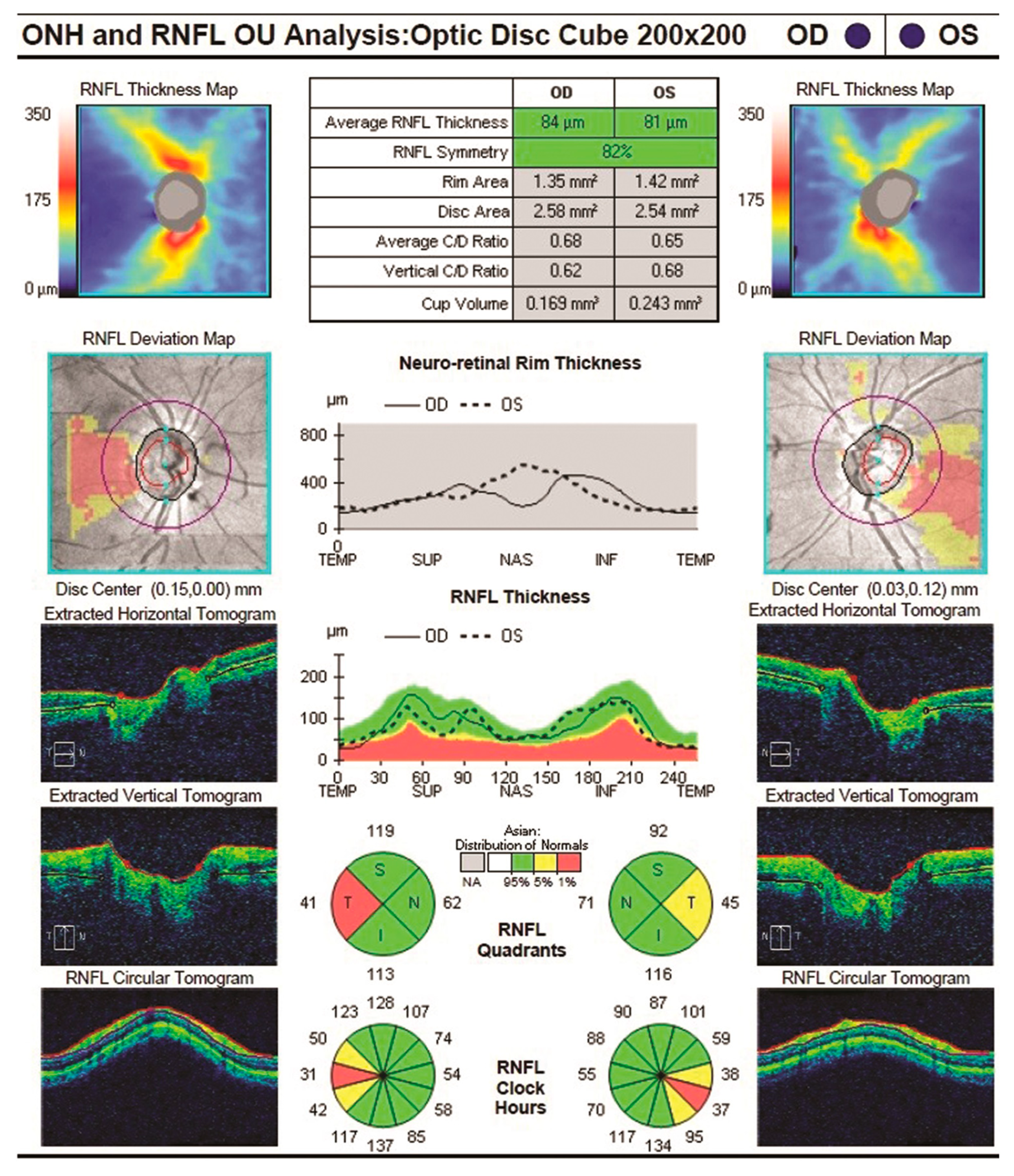
| Year, Authors [Ref] | Significant Finding |
|---|---|
| Yan et al., 2021 [19] | rNFL and retinal vascular density were considerably lower in persons with Alzheimer’s disease than in healthy controls. Some patients’ cognitive dysfunction has been linked to a decrease in retinal vessel density. |
| Mei et al., 2020 [20] | Investigation of the link between the retinal and cerebral Aβ content in APP mice. In the retina, curcumin can stain Aβ, but it has been found to suppress the level of this staining compound. |
| Chua et al., 2020 [21] | AD patients’ superficial and deep capillary plexus showed considerably decreased vascular density compared with healthy controls. |
| Wu et al., 2020 [22] | OCT-A on AD patients revealed significantly lower deep retinal capillary plexus micro vascular densities than on matched controls. |
| Kim et al., 2019 [23] | Both individuals with serious Alzheimer’s disease and those with mild to medium Alzheimer’s disease had thinning of the rNFL assessed by OCT. |
| Zabel et al., 2019 [24] | Comparing patients with AD to healthy controls, their rNFL was significantly thinner, but this was not statistically significant. SD-OCT observed changes that were not specific. |
| Ko et al., 2018 [18] | Researchers found that patients without the neurodegenerative disease had lower cognitive function and the possibility of cognitive reduction as a result of the presence of thinning rNFL. |
| Sanchez’ et al., 2018 [25] | For both cognitively healthy and Alzheimer’s disease persons, the thickness of the peripapillary rNFL is the same. |
| Koronyo et al., 2017 [26] | Curcumin fluorochrome was used to detect a deposit at the retinal level. Imagery of the retina A was captured using solid lipid curcumin and an adapted scanning laser eye scope. |
| Den Haan et al., 2017 [27] | Mild cognitive impairment (MCI) and Alzheimer’s disease (AD) patients exhibit reduced retinal nerve fibre zone and macular thickness compared with healthy controls, respectively. |
| Mutlu et al., 2017 [28] | Gray and white matter volumes were found to be less when the RNFL and GCL were smaller as measured by OCT. |
| Casaletto et al., 2017 [16] | On OCT, overall macular and macular ganglion cell volumes were shown to decrease. |
| Jiang et al., 2016 [29] | A meta-analysis of five studies with few sample sizes was unable to make any conclusions about pathological retinal degeneration. |
| Pillai et al., 2016 [30] | There is no variation in rNFL, GCL, or macular volume on OCT in Alzheimer’s disease patients compared with healthy control subjects. |
| Koronyo Hamaoui et al., 2011 [4] | It was discovered that systemic administration of curcumin caused the development of retinal postmortem eyes of Alzheimer’s patients to show a buildup of plaques. These plaques can be seen and accumulated at a primitive phase of illness. |
| Patient | Mean | NS | N | NI | TI | T | TS |
|---|---|---|---|---|---|---|---|
| 1 | 90.0 ± 8.5 | 75.0 ± 1.4 | 46.0 ± 9.9 | 111.5 ± 21.9 | 143.5 ± 9.2 | 73.0 ± 1.4 | 153.0 ± 17.0 |
| 2 | 102.0 ± 8.5 | 105.5 ± 3.5 | 60.5 ± 6.4 | 112.0 ± 7.1 | 166.5 ± 13.4 | 77.5 ± 17.7 | 157.5 ± 0.7 |
| 3 | 100.0 ± 0.0 | 99.0 ± 4.2 | 99.5 ± 0.7 | 127.5 ± 2.1 | 130.5 ± 2.1 | 61.5 ± 0.7 | 122.5 ± 0.7 |
| 4 | 99.0 | 124.0 | 92.0 | 127.0 | 134.0 | 58.0 | 108.0 |
| 5 | 110.0 ± 5.7 | 116.5 ± 4.9 | 81.0 ± 5.7 | 129.0 ± 4.2 | 148.5 ± 2.1 | 86.5 ± 13.4 | 151.0 ± 5.7 |
| 6 | 95.5 ± 4.9 | 110.5 ± 16.3 | 66.0 ± 2.8 | 91.5 ± 21.9 | 143.5 ± 23.3 | 68.0 ± 1.4 | 148.5 ± 14.8 |
| 7 | 108.0 ± 1.4 | 111.5 ± 16.3 | 87.0 ± 0.0 | 125.5 ± 4.9 | 147.5 ± 3.5 | 77.5 ± 4.9 | 139.0 ± 0.0 |
| 8 | 85.0 ± 1.4 | 85.5 ± 4.9 | 55.0 ± 1.4 | 73.0 ± 5.7 | 129.0 ± 9.9 | 81.5 ± 20.5 | 118.5 ± 7.8 |
| 9 | 90.5 ± 2.1 | 91.5 ± 7.8 | 67.0 ± 2.8 | 80.5 ± 2.1 | 147.0 ± 4.2 | 66.0 ± 0.0 | 139.0 ± 11.3 |
| 10 | 93.0 ± 0.0 | 110.0 ± 0.0 | 66.5 ± 0.7 | 124..0 ± 5.7 | 141.5 ± 7.8 | 59.5 ± 0.7 | 114.5 ± 0.7 |
| Control | Mild AD | Moderate AD | Mild AD vs. Control | Moderate AD vs. Control | Mild AD vs. Moderate AD | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| (n = 40) | (n = 39) | (n = 18) | % Difference | p-Value | % Difference | p-Value | % Difference | p-Value | ||
| Visual Acuity (dec) | 1.00 ± 0.10 | 0.90 ± 0.20 | 0.90 ± 0.30 | −10.00 | <0.001 ** | −10.00 | 0.003 ** | 0.00 | 0.921 | |
| Constrast Sensitivity (cpd) | 3 | 1.63 ± 0.29 | 1.49 ± 0.46 | 1.49 ± 0.29 | −8.59 | <0.001 ** | −8.59 | 0.009 ** | 0.00 | 0.422 |
| 6 | 1.84 ± 0.44 | 1.70 ± 0.33 | 1.55 ± 0.28 | −7.61 | <0.001 ** | −15.76 | <0.001 ** | −8.82 | 0.373 | |
| 12 | 1.54 ± 0.29 | 1.25 ± 0.32 | 1.16 ± 0.30 | −18.83 | <0.001 ** | −24.68 | <0.001 ** | −7.20 | 0.599 | |
| 18 | 1.10 ± 0.29 | 0.64 ± 0.56 | 0.64 ± 0.57 | −41.82 | <0.001 ** | −41.82 | <0.001 ** | 0.00 | 0.781 | |
| Number of Errors | ||||||||||
| Total | 5.0 ± 5.0 | 7.0 ± 4.0 | 12.00 ± 8.0 | 40.00 | 0.034 * | 140.00 | <0.001 ** | 71.43 | 0.001 ** | |
| Tritan | 2.0 ± 2.0 | 3.0 ± 3.0 | 6.0 ± 5.0 | 50.00 | 0.002 ** | 200.00 | <0.001 ** | 100.00 | 0.017 * | |
| Deutan | 1.0 ± 3.0 | 3.0 ± 3.0 | 5.0 ± 3.0 | 200.00 | 0.003 ** | 400.00 | <0.001 ** | 66.67 | 0.002 ** | |
| PDT | 14.00 ± 2.00 | 12.00 ± 4.00 | 11.50 ± 3.0 | −14.29 | <0.001 ** | −17.86 | <0.001 ** | −4.17 | 0.650 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
T., P.; V., S. Identification of Alzheimer’s Disease by Imaging: A Comprehensive Review. Int. J. Environ. Res. Public Health 2023, 20, 1273. https://doi.org/10.3390/ijerph20021273
T. P, V. S. Identification of Alzheimer’s Disease by Imaging: A Comprehensive Review. International Journal of Environmental Research and Public Health. 2023; 20(2):1273. https://doi.org/10.3390/ijerph20021273
Chicago/Turabian StyleT., Prasath, and Sumathi V. 2023. "Identification of Alzheimer’s Disease by Imaging: A Comprehensive Review" International Journal of Environmental Research and Public Health 20, no. 2: 1273. https://doi.org/10.3390/ijerph20021273
APA StyleT., P., & V., S. (2023). Identification of Alzheimer’s Disease by Imaging: A Comprehensive Review. International Journal of Environmental Research and Public Health, 20(2), 1273. https://doi.org/10.3390/ijerph20021273






