Acute Effects of Percussive Massage Therapy on Thoracolumbar Fascia Thickness and Ultrasound Echo Intensity in Healthy Male Individuals: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Sample Size Calculation
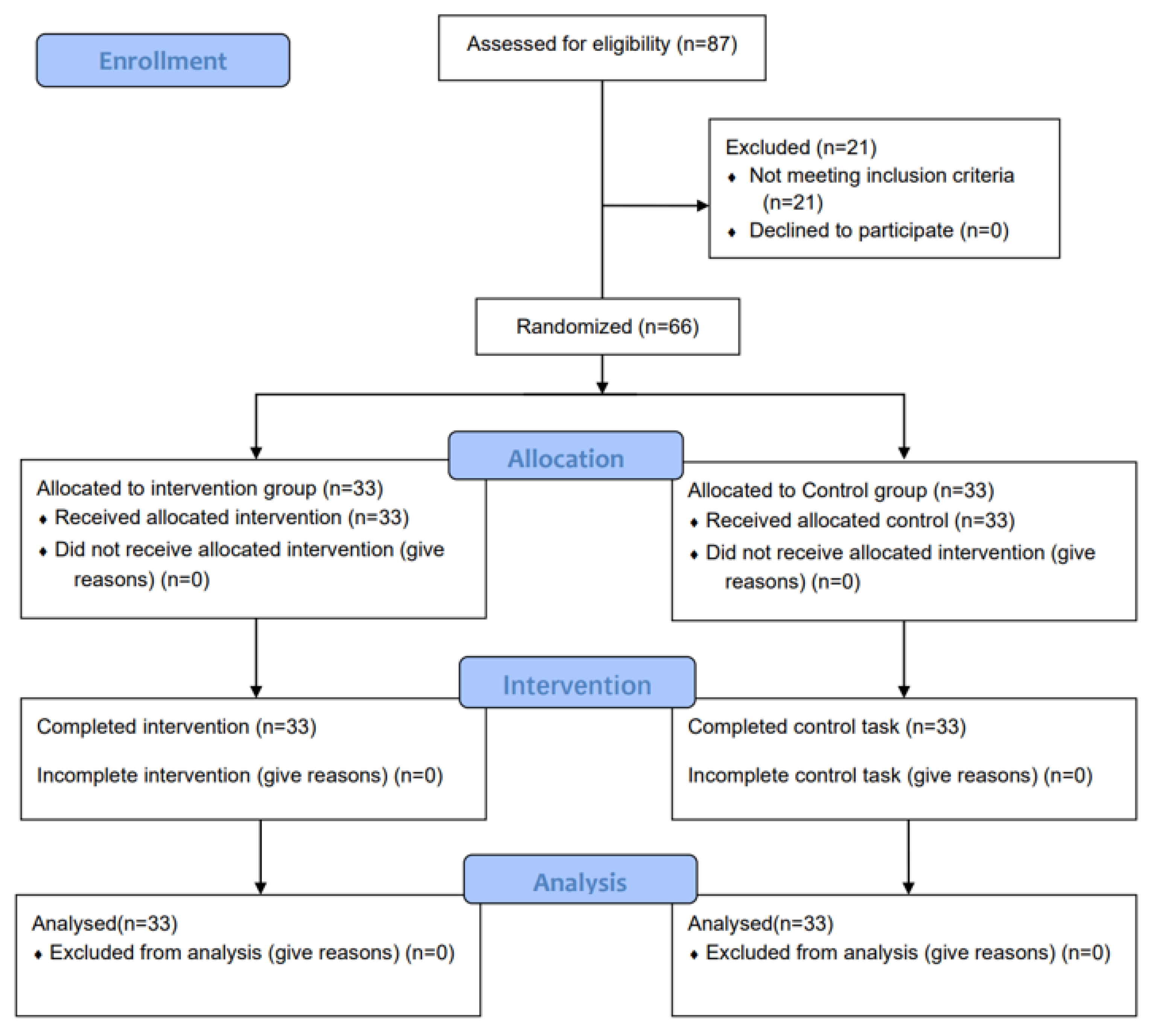
2.3. Participants
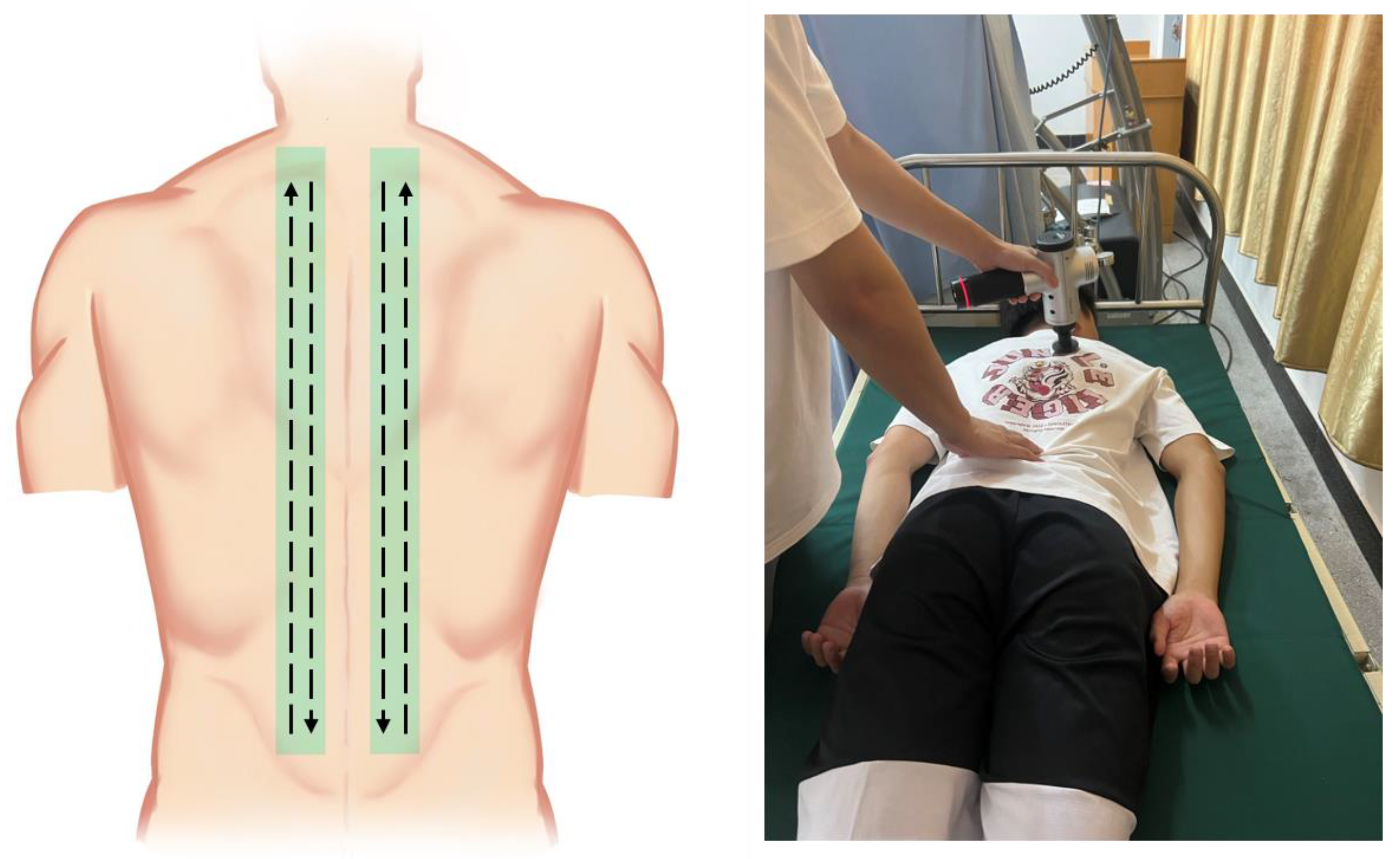
2.4. Intervention Protocol
2.5. Outcomes Measurement
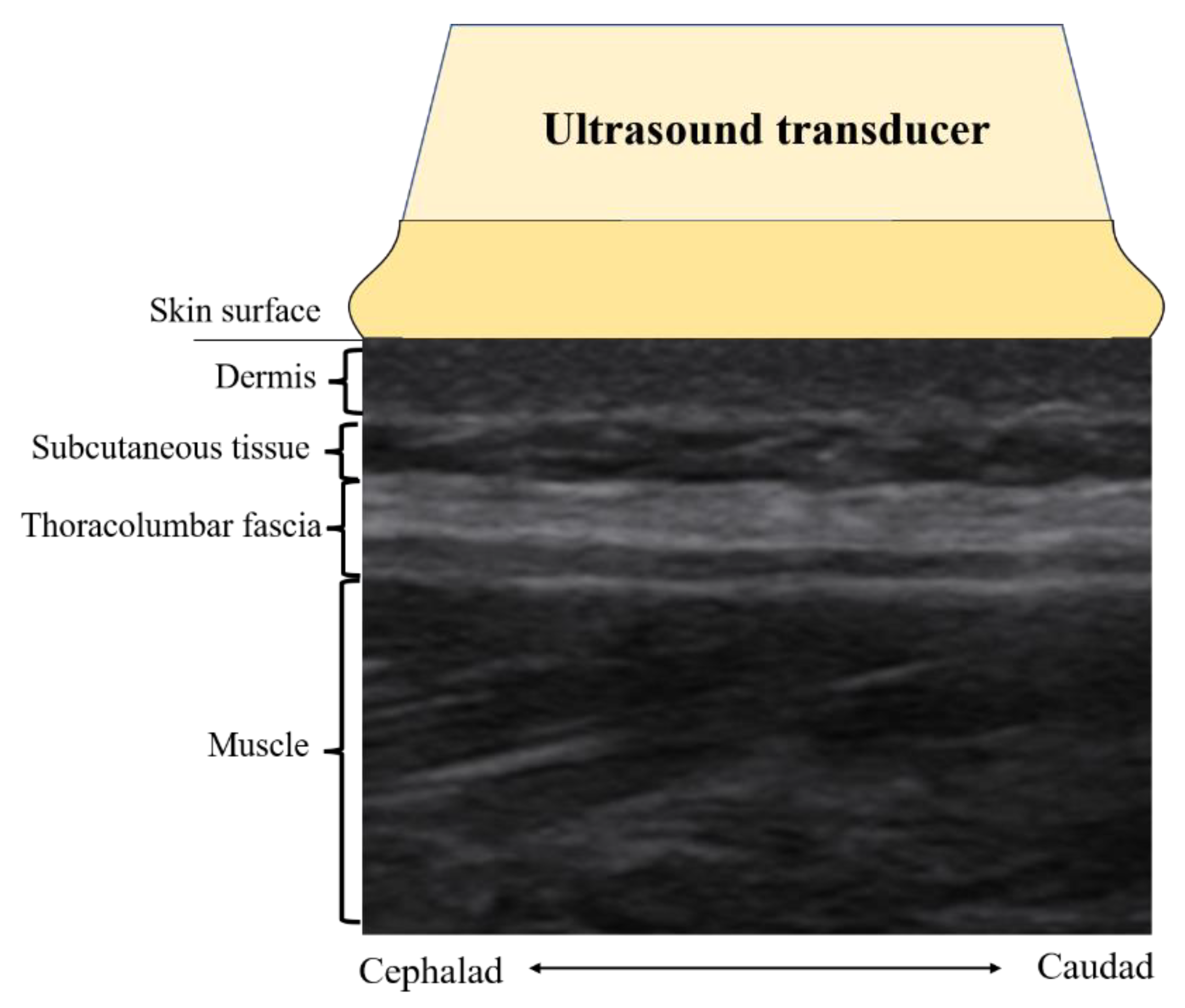
2.5.1. Ultrasound Imaging Assessment
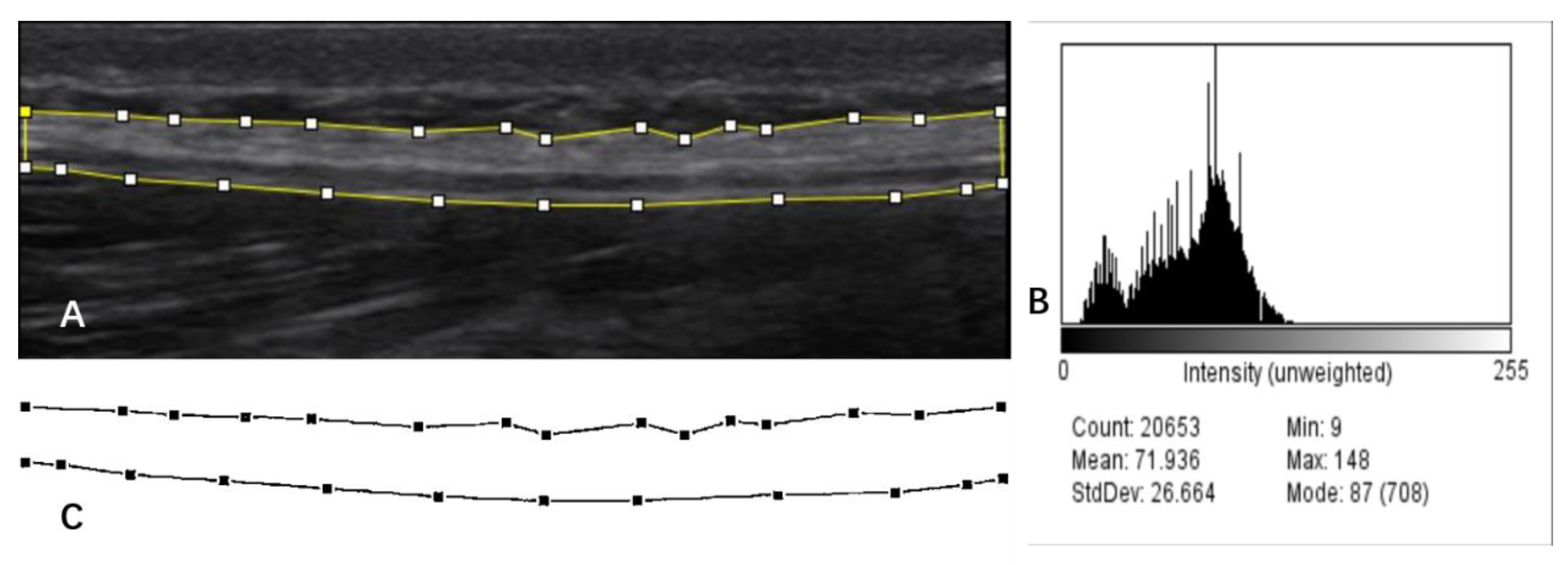
Fascial Echo Intensity Measures
Fascia Thickness Measures
2.5.2. Perceived Stiffness
2.5.3. Skin Temperature
2.5.4. Lumbar Flexibility
2.6. Statistics Analysis
3. Results
3.1. Echostructure Data
3.2. Secondary Outcomes
4. Discussion
5. Limitation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zügel, M.; Maganaris, C.N.; Wilke, J.; Jurkat-Rott, K.; Klingler, W.; Wearing, S.C.; Findley, T.; Barbe, M.F.; Steinacker, J.M.; Vleeming, A.; et al. Fascial Tissue Research in Sports Medicine: From Molecules to Tissue Adaptation, Injury and Diagnostics: Consensus Statement. Br. J. Sport. Med. 2018, 52, 1497. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M.; Keely, P.; Mao, J.; Hodge, L.M.; Schleip, R.; Deng, G.; Hinz, B.; Swartz, M.A.; De Valois, B.A.; Zick, S.; et al. Connecting (T)Issues: How Research in Fascia Biology Can Impact Integrative Oncology. Cancer Res. 2016, 76, 6159–6162. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, M. The Fascia of the Limbs and Back—A Review. J. Anat. 2009, 214, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Willard, F.H.; Vleeming, A.; Schuenke, M.D.; Danneels, L.; Schleip, R. The Thoracolumbar Fascia: Anatomy, Function and Clinical Considerations. J. Anat. 2012, 221, 507–536. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M.; Sherman, K.J. Pathophysiological Model for Chronic Low Back Pain Integrating Connective Tissue and Nervous System Mechanisms. Med. Hypotheses 2007, 68, 74–80. [Google Scholar] [CrossRef]
- Langevin, H.M.; Stevens-Tuttle, D.; Fox, J.R.; Badger, G.J.; Bouffard, N.A.; Krag, M.H.; Wu, J.; Henry, S.M. Ultrasound Evidence of Altered Lumbar Connective Tissue Structure in Human Subjects with Chronic Low Back Pain. BMC Musculoskelet. Disord. 2009, 10, 151. [Google Scholar] [CrossRef]
- Langevin, H.M.; Fox, J.R.; Koptiuch, C.; Badger, G.J.; Greenan-Naumann, A.C.; Bouffard, N.A.; Konofagou, E.E.; Lee, W.N.; Triano, J.J.; Henry, S.M. Reduced Thoracolumbar Fascia Shear Strain in Human Chronic Low Back Pain. BMC Musculoskelet. Disord. 2011, 12, 203. [Google Scholar] [CrossRef]
- Almaz, J.; Daniel, L.; Romero-morales, C.; Bravo-aguilar, M.; Calvo-lobo, C. Quantitative Ultrasound Imaging Differences in Multifidus and Thoracolumbar Fasciae between Athletes with and without Chronic Lumbopelvic Pain: A Case-Control Study. J. Clin. Med. 2020, 9, 2647. [Google Scholar]
- Schilder, A.; Hoheisel, U.; Magerl, W.; Benrath, J.; Klein, T.; Treede, R.D. Sensory Findings after Stimulation of the Thoracolumbar Fascia with Hypertonic Saline Suggest Its Contribution to Low Back Pain. Pain 2014, 155, 222–231. [Google Scholar] [CrossRef]
- Wilke, J.; Schleip, R.; Klingler, W.; Stecco, C. The Lumbodorsal Fascia as a Potential Source of Low Back Pain: A Narrative Review. BioMed Res. Int. 2017, 2017, 5349620. [Google Scholar] [CrossRef] [PubMed]
- Di Matteo, A.; Filippucci, E.; Cipolletta, E.; Martire, V.; Jesus, D.; Musca, A.; Corradini, D.; Isidori, M.; Salaffi, F.; Grassi, W. How Normal Is the Enthesis by Ultrasound in Healthy Subjects? Clin. Exp. Rheumatol. 2020, 38, 472–478. [Google Scholar] [PubMed]
- Pavan, P.G.; Stecco, A.; Stern, R.; Stecco, C. Painful Connections: Densification versus Fibrosis of Fascia. Curr. Pain Headache Rep. 2014, 18, 441. [Google Scholar] [CrossRef] [PubMed]
- Chase, P.B. The Rolf Method of Structural Integration on Fascial Tissue Stiffness, Elasticity, and Superficial Blood Perfusion in Healthy Individuals: The Prospective, Interventional Study. Front. Physiol. 2020, 11, 1062. [Google Scholar] [CrossRef]
- Cullen, M.F.L.; Casazza, G.A.; Davis, B.A. Passive Recovery Strategies after Exercise: A Narrative Literature Review of the Current Evidence. Curr. Sport. Med. Rep. 2021, 20, 351–358. [Google Scholar] [CrossRef]
- Stecco, A.; Meneghini, A.; Stern, R.; Stecco, C.; Imamura, M. Ultrasonography in Myofascial Neck Pain: Randomized Clinical trial for Diagnosis and Follow-Up. Surg. Radiol. Anat. 2014, 36, 243–253. [Google Scholar] [CrossRef]
- Stecco, A.; Stern, R.; Fantoni, I.; De Caro, R.; Stecco, C. Fascial Disorders: Implications for Treatment. PMR 2016, 8, 161–168. [Google Scholar] [CrossRef]
- Cheatham, S.W.; Baker, R.T.; Behm, D.G.; Stull, K.; Kolber, M.J. Mechanical Percussion Devices: A Survey of Practice Patterns among Healthcare Professionals. Int. J. Sport. Phys. Ther. 2021, 16, 766–777. [Google Scholar] [CrossRef]
- Lupowitz, L. Vibration Therapy—A Clinical Commentary. Int. J. Sport. Phys. Ther. 2022, 17, 984–987. [Google Scholar] [CrossRef]
- García-Sillero, M.; Jurado-Castro, J.M.; Benítez-Porres, J.; Vargas-Molina, S. Acute Effects of a Percussive Massage Treatment on Movement Velocity during Resistance Training. Int. J. Environ. Res. Public Health 2021, 18, 7726. [Google Scholar] [CrossRef]
- Konrad, A.; Glashüttner, C.; Reiner, M.M.; Bernsteiner, D.; Tilp, M. The Acute Effects of a Percussive Massage Treatment with a Hypervolt Device on Plantar Flexor Muscles’ Range of Motion and Performance. J. Sport. Sci. Med. 2020, 19, 690–694. [Google Scholar]
- García-Sillero, M.; Benítez-Porres, J.; García-Romero, J.; Bonilla, D.A.; Petro, J.L.; Vargas-Molina, S. Comparison of Interventional Strategies to Improve Recovery after Eccentric Exercise-Induced Muscle Fatigue. Int. J. Environ. Res. Public Health 2021, 18, 647. [Google Scholar] [CrossRef] [PubMed]
- Trainer, J.H.; Pascarella, M.; Paul, R.W.; Thomas, S.J. Acute Effects of Percussive Therapy on the Posterior Shoulder Muscles Differ Based on the Athlete’s Soreness Response. Int. J. Sport. Phys. Ther. 2022, 17, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Guang, H.; Ji, L.; Shi, Y. Focal Vibration Stretches Muscle Fibers by Producing Muscle Waves. IEEE Trans. Neural Syst. Rehabil. Eng. 2018, 26, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Eungpinichpong, W.; Sucharit, W.; Huang, X. Immediate Effects of Percussive Massage Treatment on Thoracolumbar Fascia Thickness: A Quasi-Experimental Design in Healthy Individuals. Arch. Allied Health Sci. 2022, 34, 22–32. [Google Scholar]
- Peloquin, K.; Barnhardt, M.; Behling, G.; Braun, S. The Immediate Effect of Percussion Myofascial Release Therapy on Hamstring Flexibility and Hip Flexion Range of Motion Among Active Young Adults. Int. J. Res. Phys. 2022, 17, 48–61. [Google Scholar]
- Oatyimprai, K.; Eungpinichpong, W.; Buranruk, O.; Konharn, K.; Tudpor, K. Effect of Traditional Thai Massage on Muscle Oxygen Saturation in Low Back Pain Patients: A Preliminary Study. Int. J. GEOMATE 2020, 19, 54–61. [Google Scholar] [CrossRef]
- Imamura, M.; Furlan, A.D.; Dryden, T.; Irvin, E. Evidence-Informed Management of Chronic Low Back Pain with Massage. Spine J. 2008, 8, 121–133. [Google Scholar] [CrossRef]
- De Coninck, K.; Hambly, K.; Dickinson, J.W.; Passfield, L. Measuring the Morphological Characteristics of Thoracolumbar Fascia in Ultrasound Images: An Inter-Rater Reliability Study. BMC Musculoskelet. Disord. 2018, 19, 180. [Google Scholar] [CrossRef]
- Wilke, J.; Macchi, V.; De Caro, R.; Stecco, C. Fascia Thickness, Aging and Flexibility: Is There an Association? J. Anat. 2019, 234, 43–49. [Google Scholar] [CrossRef]
- Pirri, C.; Stecco, C.; Fede, C.; Macchi, V.; Özçakar, L. Ultrasound Imaging of the Fascial Layers: You See (Only) What You Know. J. Ultrasound Med. 2020, 39, 827–828. [Google Scholar] [CrossRef]
- Caresio, C.; Molinari, F.; Emanuel, G.; Minetto, M.A. Muscle Echo Intensity: Reliability and Conditioning Factors. Clin. Physiol. Funct. Imaging 2015, 35, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Siegmund, L.A.; Barkley, J.E.; Knapp, D.; Peer, K.S. Acute Effects of Local Vibration with Biomechanical Muscle Stimulation on Low-Back Flexibility and Perceived Stiffness. Athl. Train. Sport. Health Care 2014, 6, 37–45. [Google Scholar] [CrossRef]
- Robinson, H.S.; Mengshoel, A.M. Assessments of Lumbar Flexion Range of Motion. Spine 2014, 39, E270–E275. [Google Scholar] [CrossRef] [PubMed]
- Tousignant, M.; Poulin, L.; Marchand, S.; Viau, A.; Place, C. The Modified-Modified Schober Test for Range of Motion Assessment of Lumbar Flexion in Patients with Low Back Pain: A Study of Criterion Validity, Intra-and Inter-Rater Reliability and Minimum Metrically Detectable Change. Disabil. Rehabil. 2005, 27, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: New York, NY, USA, 1988. [Google Scholar] [CrossRef]
- Park, H.S.; Martin, B.J. Contribution of the Tonic Vibration Reflex to Muscle Stress and Muscle Fatigue. Scand. J. Work. Environ. Health 1993, 19, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Bordoni, B.; Zanier, E. Understanding Fibroblasts in Order to Comprehend the Osteopathic Treatment of the Fascia. Evid.-Based Complement. Altern. Med. 2015, 2015, 860934. [Google Scholar] [CrossRef]
- Liang, H.W.; Wang, T.G.; Chen, W.S.; Hou, S.M. Thinner Plantar Fascia Predicts Decreased Pain after Extracorporeal Shock Wave Therapy. Clin. Orthop. Relat. Res. 2007, 460, 219–225. [Google Scholar] [CrossRef]
- Zullo, A.; Fleckenstein, J.; Schleip, R.; Hoppe, K.; Wearing, S.; Klingler, W. Structural and Functional Changes in the Coupling of Fascial Tissue, Skeletal Muscle, and Nerves During Aging. Front. Physiol. 2020, 11, 592. [Google Scholar] [CrossRef]
- Stock, M.S.; Thompson, B.J. Echo Intensity as an Indicator of Skeletal Muscle Quality: Applications, Methodology, and Future directions. Eur. J. Appl. Physiol. 2021, 121, 369–380. [Google Scholar] [CrossRef]
- Pillen, S.; Tak, R.O.; Zwarts, M.J.; Lammens, M.M.Y.; Verrijp, K.N.; Arts, I.M.P.; van der Laak, J.A.; Hoogerbrugge, P.M.; van Engelen, B.G.M.; Verrips, A. Skeletal Muscle Ultrasound: Correlation between Fibrous Tissue and Echo Intensity. Ultrasound Med. Amp Biol. 2009, 35, 443–446. [Google Scholar] [CrossRef]
- Stecco, A.; Cowman, M.; Pirri, N.; Raghavan, P.; Pirri, C. Densification: Hyaluronan Aggregation in Different Human Organs. Bioengineering 2022, 9, 159. [Google Scholar] [CrossRef] [PubMed]
- Krause, F.; Wilke, J.; Niederer, D.; Vogt, L.; Banzer, W. Acute Effects of Foam Rolling on Passive Stiffness, Stretch Sensation and Fascial Sliding: A Randomized Controlled Trial. Hum. Mov. Sci. 2019, 67, 102514. [Google Scholar] [CrossRef] [PubMed]
- Griefahn, A.; Knicker, A.; von Piekartz, H. Efficacy of Foam Rolling with Additional Vibration Stimulation on the Mobility of the Thoracolumbar Fascia. An Observational Study. J. Bodyw. Mov. Ther. 2021, 26, 84–93. [Google Scholar] [CrossRef]
- Okamoto, T.; Masuhara, M.; Ikuta, K. Acute Effects of Self-Myofascial Release Using a Foam Roller on Arterial Function. J. Strength Cond. Res. 2014, 28, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Stecco, C.; Stern, R.; Porzionato, A.; MacChi, V.; Masiero, S.; Stecco, A.; De Caro, R. Hyaluronan within Fascia in the Etiology of Myofascial Pain. Surg. Radiol. Anat. 2011, 33, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Roman, M.; Chaudhry, H.; Bukiet, B.; Stecco, A.; Findley, T.W. Mathematical Analysis of the Flow of Hyaluronic Acid around Fascia during Manual Therapy Motions. J. Am. Osteopath. Assoc. 2013, 113, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M. Fascia Mobility, Proprioception, and Myofascial Pain. Life 2021, 11, 668. [Google Scholar] [CrossRef] [PubMed]
- Tadmor, R.; Chen, N.; Israelachvili, J.N. Thin Film Rheology and Lubricity of Hyaluronic Acid Solutions at a Normal Physiological Concentration. J. Biomed. Mater. Res. 2002, 61, 514–523. [Google Scholar] [CrossRef]
- Behm, D.G.; Wilke, J. Do Self-Myofascial Release Devices Release Myofascia? Rolling Mechanisms: A Narrative Review. Sport. Med. 2019, 49, 1173–1181. [Google Scholar] [CrossRef]
- Schleip, R.; Gabbiani, G.; Wilke, J.; Naylor, I.; Hinz, B.; Zorn, A.; Jäger, H.; Breul, R.; Schreiner, S.; Klingler, W. Fascia Is Able to Actively Contract and May Thereby Influence Musculoskeletal Dynamics: A Histochemical and Mechanographic Investigation. Front. Physiol. 2019, 10, 336. [Google Scholar] [CrossRef]
- Schleip, R.; Klingler, W. Active Contractile Properties of Fascia. Clin. Anat. 2019, 32, 891–895. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | PT Group (n = 33) | Control Group (n = 33) |
|---|---|---|
| Age (years) | 21.6 ± 4.4 | 22.6 ± 3.7 |
| Height (cm) | 176.2 ± 5.8 | 174.9 ± 5.6 |
| Weight (kg) | 68.9 ± 7.9 | 70.5 ± 5.9 |
| BMI (kg/m2) | 22.2 ± 1.9 | 23 ± 1.5 |
| Outcomes | Group | Before | After | p-Value |
|---|---|---|---|---|
| Fascia thickness L (mm) | PT | 2.56 ± 0.64 | 2.57 ± 0.63 | 0.835 |
| Control | 2.56 ± 0.69 | 2.56 ± 0.66 | 1.000 | |
| Fascia thickness R (mm) | PT | 2.54 ± 0.6 | 2.47 ± 0.59 | 0.127 |
| Control | 2.69 ±0.67 | 2.67 ± 0.69 | 0.152 | |
| Echo intensity L (CSAEI) | PT | 71.9 ± 9.7 | 68.5 ± 9.5 | <0.001 |
| Control | 73.9 ± 8 | 73.6 ± 8.1 | 0.270 | |
| Echo intensity R (CSAEI) | PT | 73.7 ± 8.1 | 68.4 ± 9.2 | <0.001 |
| Control | 74.2 ± 8 | 73.4 ± 7.8 | 0.02 |
| Outcomes | Group | Baseline | After Intervention | After Intervention (Adjusted) | Difference (95% CI) | p-Value | Effect Size | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Mean | SE | |||||
| Fascia thickness L (mm) | PT | 2.56 | 0.111 | 2.57 | 0.109 | 2.57 | 0.031 | 0.009 (−0.079 to 0.097) | 0.837 | 0.03 |
| CG | 2.56 | 0.119 | 2.56 | 0.115 | 2.56 | 0.031 | ||||
| Fascia thickness R (mm) | PT | 2.54 | 0.105 | 2.47 | 0.102 | 2.55 | 0.035 | −0.047 (−0.15 to 0.05) | 0.349 | 0.12 |
| CG | 2.69 | 0.117 | 2.67 | 0.119 | 2.59 | 0.035 | ||||
| Echo intensity L (CSAEI) | PT | 71.9 | 1.69 | 68.5 | 1.65 | 69.4 | 0.62 | −3.36 (−5.1 to −1.6) | <0.001 | 0.48 |
| CG | 73.9 | 1.39 | 73.9 | 1.39 | 72.7 | 0.62 | ||||
| Echo intensity R (CSAEI) | PT | 73.7 | 1.4 | 68.4 | 1.61 | 68.7 | 0.6 | −4.39 (−6.1 to −2.7) | <0.001 | 0.64 |
| CG | 74.2 | 1.39 | 73.4 | 1.36 | 73.1 | 0.6 | ||||
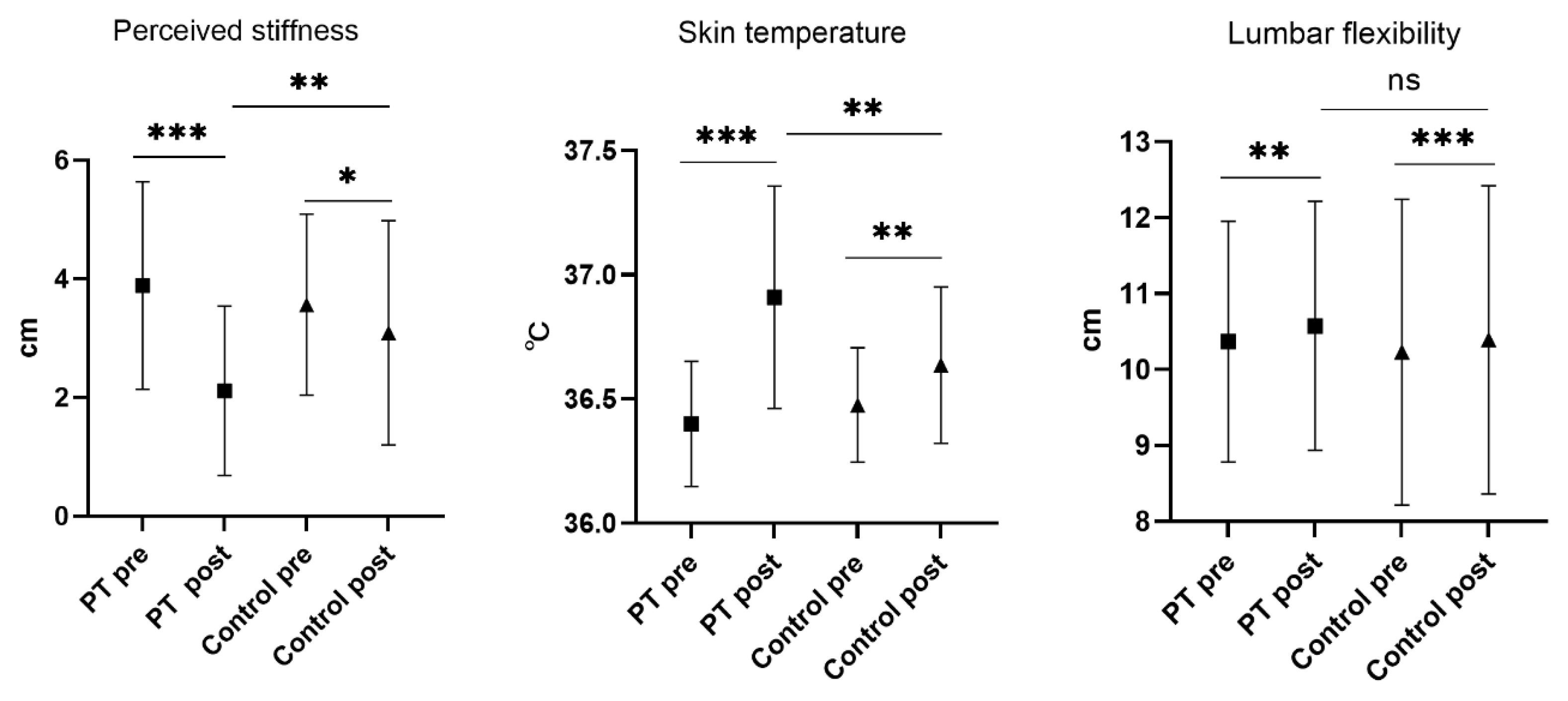
| Perceived stiffness (cm) | PT | 3.89 | 0.3 | 2.11 | 0.25 | 2.01 | 0.23 | −1.18 (−1.84 to −0.52) | <0.001 | 0.45 |
| CG | 3.56 | 0.27 | 3.09 | 0.33 | 3.19 | 0.23 | ||||
| Skin temperature (°C) | PT | 36.4 | 0.04 | 36.9 | 0.08 | 36.9 | 0.67 | 0.29 (0.11 to 0.48) | 0.002 | 0.40 |
| CG | 36.5 | 0.04 | 36.6 | 0.05 | 36.6 | 0.67 | ||||
| Lumbar flexibility (cm) | PT | 10.4 | 0.28 | 10.6 | 0.29 | 10.5 | 0.53 | 0.04 (−0.1 to 1.94) | 0.550 | 0.08 |
| CG | 10.2 | 0.35 | 10.4 | 0.35 | 10.4 | 0.53 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.; Huang, X.; Li, Y.; Sucharit, W.; Sirasaporn, P.; Eungpinichpong, W. Acute Effects of Percussive Massage Therapy on Thoracolumbar Fascia Thickness and Ultrasound Echo Intensity in Healthy Male Individuals: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2023, 20, 1073. https://doi.org/10.3390/ijerph20021073
Yang C, Huang X, Li Y, Sucharit W, Sirasaporn P, Eungpinichpong W. Acute Effects of Percussive Massage Therapy on Thoracolumbar Fascia Thickness and Ultrasound Echo Intensity in Healthy Male Individuals: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2023; 20(2):1073. https://doi.org/10.3390/ijerph20021073
Chicago/Turabian StyleYang, Chao, Xingyu Huang, Ying Li, Wiraphong Sucharit, Patpiya Sirasaporn, and Wichai Eungpinichpong. 2023. "Acute Effects of Percussive Massage Therapy on Thoracolumbar Fascia Thickness and Ultrasound Echo Intensity in Healthy Male Individuals: A Randomized Controlled Trial" International Journal of Environmental Research and Public Health 20, no. 2: 1073. https://doi.org/10.3390/ijerph20021073
APA StyleYang, C., Huang, X., Li, Y., Sucharit, W., Sirasaporn, P., & Eungpinichpong, W. (2023). Acute Effects of Percussive Massage Therapy on Thoracolumbar Fascia Thickness and Ultrasound Echo Intensity in Healthy Male Individuals: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 20(2), 1073. https://doi.org/10.3390/ijerph20021073







