The Impact of Podiatric Intervention on the Quality of Life and Pain in Children and Adolescents with Hypermobility
Abstract
:1. Introduction
2. Materials and Methods
2.1. Research Participants
2.2. Procedure
2.3. Adverse Events
2.4. Primary Outcome Measures
2.5. Secondary Outcome Measures
2.6. Statistical Analysis
3. Results
4. Discussion
4.1. Summary of Findings
4.2. Clinical Implications
4.3. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malfait, F.; Francomano, C.; Byers, P.; Belmont, J.; Berglund, B.; Black, J.; Bloom, L.; Bowen, J.M.; Brady, A.F.; Burrows, N.P.; et al. The 2017 international classification of the Ehlers–Danlos syndromes. Am. J. Med. Genet. Part C Semin. Med. Genet. 2017, 175, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Kirk, J.A.; Ansell, B.M.; Bywaters, E.G. The hypermobility syndrome. Musculoskeletal complaints associated with generalized joint hypermobility. Ann. Rheum. Dis. 1967, 26, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Sobhani-Eraghi, A.; Motalebi, M.; Sarreshtehdari, S.; Molazem-Sanandaji, B.; Hasanlu, Z. Prevalence of joint hypermobility in children and adolescents: A systematic review and meta-analysis. J. Res. Med. Sci. 2020, 25, 104. [Google Scholar]
- Sperotto, F.; Balzarin, M.; Parolin, M.; Monteforte, N.; Vittadello, F.; Zulian, F. Joint hypermobility, growing pain and obesity are mutually exclusive as causes of musculoskeletal pain in schoolchildren. Clin. Exp. Rheumatol. 2014, 32, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Remvig, L.; Kümmel, C.; Kristensen, J.H.; Boas, G.; Juul-Kristensen, B. Prevalence of generalized joint hypermobility, arthralgia and motor competence in 10-year-old school children. Int. Musculoskelet. Med. 2011, 33, 137–145. [Google Scholar] [CrossRef]
- Hasija, R.P.; Khubchandani, R.P.; Shenoi, S. Joint hypermobility in Indian children. Clin. Exp. Rheumatol. 2008, 26, 146–150. [Google Scholar]
- Hass, J.; Hass, R. Arthrochalasis multiplex congenita: Congenital flaccidity of the joints. J. Bone Jt. Surg. Am. 1958, 40, 663–674. [Google Scholar] [CrossRef]
- Malfait, F.; Hakim, A.; De Paepe, A.; Grahame, R. The genetic basis of the joint hypermobility syndromes. Rheumatology 2006, 45, 502–507. [Google Scholar] [CrossRef]
- Castori, M.; Tinkle, B.; Levy, H.; Grahame, R.; Malfait, F.; Hakim, A. A framework for the classification of joint hypermobility and related conditions. Am. J. Med. Genet. Part C Semin. Med. Genet. 2017, 175, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Hanewinkel-van Kleef, Y.B.; Helders, P.J.; Takken, T.; Engelbert, R.H. Motor performance in children with generalized hypermobility: The influence of muscle strength and exercise capacity. Pediatr. Phys. Ther. 2009, 21, 194–200. [Google Scholar] [CrossRef]
- Pacey, V.; Tofts, L.; Adams, R.; Nicholson, L. Factors affecting change in children with joint hypermobility syndrome: Results of a prospective longitudinal study. Intern. Med. J. 2015, 45, 9. [Google Scholar]
- Grahame, R. Hypermobility: An important but often neglected area within rheumatology. Nat. Clin. Pract. Rheumatol. 2008, 4, 522–524. [Google Scholar] [CrossRef]
- Uziel, Y.; Chapnick, G.; Jaber, L.; Nemet, D.; Hashkes, P.J. Five-year outcome of children with “growing pains”: Correlations with pain threshold. J. Pediatr. 2010, 156, 838–840. [Google Scholar] [CrossRef]
- Sperotto, F.; Brachi, S.; Vittadello, F.; Zulian, F. Musculoskeletal pain in schoolchildren across puberty: A 3-year follow-up study. Pediatr. Rheumatol. 2015, 13, 16. [Google Scholar] [CrossRef]
- Engelbert, R.H.H.; Kooijmans, F.T.C.; van Riet, A.M.H.; Feitsma, T.M.; Uiterwaal, C.S.P.M.; Helders, P.J.M. The relationship between generalized joint hypermobility and motor development. Pediatr. Phys. Ther. 2005, 17, 258–263. [Google Scholar] [CrossRef]
- Fatoye, F.; Palmer, S.; Macmillan, F.; Rowe, P.; van der Linden, M. Proprioception and muscle torque deficits in children with hypermobility syndrome. Rheumatology 2009, 48, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Wesley, A.; Bray, P.; Munns, C.F.; Pacey, V. Impact of heritable disorders of connective tissue on daily life of children: Parent perspectives. J. Paediatr. Child Health 2021, 57, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Fatoye, F.; Palmer, S.; Macmillan, F.; Rowe, P.; van der Linden, M. Pain intensity and quality of life perception in children with hypermobility syndrome. Rheumatol. Int. 2012, 32, 1277–1284. [Google Scholar] [CrossRef]
- Mu, W.; Muriello, M.; Clemens, J.L.; Wang, Y.; Smith, C.H.; Tran, P.T.; Rowe, P.C.; Francomano, C.A.; Kline, A.D.; Bodurtha, J. Factors affecting quality of life in children and adolescents with hypermobile Ehlers-Danlos syndrome/hypermobility spectrum disorders. Am. J. Med. Genet. Part A 2019, 179, 561–569. [Google Scholar] [CrossRef]
- Schubert-Hjalmarsson, E.; Öhman, A.; Kyllerman, M.; Beckung, E. Pain, balance, activity, and participation in children with hypermobility syndrome. Pediatr. Phys. Ther. 2012, 24, 339–344. [Google Scholar] [CrossRef]
- Pacey, V.; Tofts, L.; Adams, R.D.; Munns, C.F.; Nicholson, L.L. Exercise in children with joint hypermobility syndrome and knee pain: A randomised controlled trial comparing exercise into hypermobile versus neutral knee extension. Pediatr. Rheumatol. 2013, 11, 30. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.O.; Bacon, H.; Jerman, E.; Easton, V.; Armon, K.; Poland, F.; Macgregor, A.J. Physiotherapy and occupational therapy interventions for people with benign joint hypermobility syndrome: A systematic review of clinical trials. Disabil. Rehabil. 2014, 36, 797–803. [Google Scholar] [CrossRef]
- Peterson, B.; Coda, A.; Pacey, V.; Hawke, F. Physical and mechanical therapies for lower limb symptoms in children with Hypermobility Spectrum Disorder and Hypermobile Ehlers-Danlos Syndrome: A systematic review. J. Foot Ankle Res. 2018, 11, 59. [Google Scholar] [CrossRef]
- Palmer, S.; Davey, I.; Oliver, L.; Preece, A.; Sowerby, L.; House, S. The effectiveness of conservative interventions for the management of syndromic hypermobility: A systematic literature review. Clin. Rheumatol. 2021, 40, 1113–1129. [Google Scholar] [CrossRef]
- McDermott, P.; Wolfe, E.; Lowry, C.; Robinson, K.; French, H.P. Evaluating the immediate effects of wearing foot orthotics in children with Joint Hypermobility Syndrome (JHS) by analysis of temperospatial parameters of gait and dynamic balance: A preliminary study. Gait Posture 2018, 60, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, R.L.; Peng, H.L.; Lee, W.C. Short-term effects of customized arch support insoles on symptomatic flexible flatfoot in children: A randomized controlled trial. Medicine 2018, 97, e10655. [Google Scholar] [CrossRef]
- Juul-Kristensen, B.; Schmedling, K.; Rombaut, L.; Lund, H.; Engelbert, R.H. Measurement properties of clinical assessment methods for classifying generalized joint hypermobility—A systematic review. Am. J. Med. Genet. Part C Semin. Med. Genet. 2017, 175, 116–147. [Google Scholar] [CrossRef]
- Reina-Bueno, M.; Vázquez-Bautista, C.; Palomo-Toucedo, I.C.; Domínguez-Maldonado, G.; Castillo-López, J.M.; Munuera-Martínez, P.V. Custom-Made Foot Orthoses Reduce Pain and Fatigue in Patients with Ehlers-Danlos Syndrome. A Pilot Study. Int. J. Environ. Res. Public Health 2020, 17, 1359. [Google Scholar] [CrossRef] [PubMed]
- Smits-Engelsman, B.; Klerks, M.; Kirby, A. Beighton score: A valid measure for generalized hypermobility in children. J. Pediatr. 2011, 158, 119–123.e4. [Google Scholar] [CrossRef]
- Redmond, A.C.; Crosbie, J.; Ouvrier, R.A. Development and validation of a novel rating system for scoring standing foot posture: The Foot Posture Index. Clin. Biomech. 2006, 21, 89–98. [Google Scholar] [CrossRef]
- Dars, S.; Uden, H.; Kumar, S.; Banwell, H.A. When, why and how foot orthoses (FOs) should be prescribed for children with flexible pes planus: A Delphi survey of podiatrists. PeerJ 2018, 6, e4667. [Google Scholar] [CrossRef] [PubMed]
- Boonstra, A.M.; Preuper, H.R.S.; Balk, G.A.; Stewart, R.E. Cut-off points for mild, moderate, and severe pain on the visual analogue scale for pain in patients with chronic musculoskeletal pain. Pain 2014, 155, 2545–2550. [Google Scholar] [CrossRef]
- Maarj, M.; Pacey, V.; Tofts, L.; Clapham, M.; Garcia, X.G.; Coda, A. Validation of an electronic visual analog scale app for pain evaluation in children and adolescents with symptomatic hypermobility: Cross-sectional study. JMIR Pediatr. Parent. 2022, 5, e41930. [Google Scholar] [CrossRef] [PubMed]
- Varni, J.W.; Seid, M.; Smith Knight, T.; Burwinkle, T.; Brown, J.; Szer, I.S. The PedsQL in pediatric rheumatology: Reliability, validity, and responsiveness of the Pediatric Quality of Life Inventory Generic Core Scales and Rheumatology Module. Arthritis Rheum. 2002, 46, 714–725. [Google Scholar] [CrossRef]
- Varni, J.W.; Seid, M.; Kurtin, P.S. PedsQL 4.0: Reliability and validity of the Pediatric Quality of Life Inventory version 4.0 generic core scales in healthy and patient populations. Med. Care 2001, 39, 800–812. [Google Scholar] [CrossRef] [PubMed]
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: Guidelines for the six-minute walk test. Am. J. Respir. Crit. Care Med. 2002, 166, 111–117. [Google Scholar] [CrossRef]
- Holland, A.E.; Spruit, M.A.; Troosters, T.; Puhan, M.A.; Pepin, V.; Saey, D.; McCormack, M.C.; Carlin, B.W.; Sciurba, F.C.; Pitta, F.; et al. An official European Respiratory Society/American Thoracic Society technical standard: Field walking tests in chronic respiratory disease. Eur. Respir. J. 2014, 44, 1428–1446. [Google Scholar] [CrossRef]
- Lammers, A.E.; Hislop, A.A.; Flynn, Y.; Haworth, S.G. The 6-minute walk test: Normal values for children of 4–11 years of age. Arch. Dis. Child. 2008, 93, 464–468. [Google Scholar] [CrossRef]
- Varni, J.W.; Burwinkle, T.M.; Szer, I.S. The PedsQL Multidimensional Fatigue Scale in pediatric rheumatology: Reliability and validity. J. Rheumatol. 2004, 31, 2494–2500. [Google Scholar]
- Scheper, M.C.; Nicholson, L.L.; Adams, R.D.; Tofts, L.; Pacey, V. The natural history of children with joint hypermobility syndrome and Ehlers-Danlos hypermobility type: A longitudinal cohort study. Rheumatology 2017, 56, 2073–2083. [Google Scholar] [CrossRef]
- Ferguson, L.; Scheman, J. Patient global impression of change scores within the context of a chronic pain rehabilitation program. J. Pain 2009, 10, S73. [Google Scholar] [CrossRef]
- Dhanani, S.; Quenneville, J.; Perron, M.; Abdolell, M.; Feldman, B.M. Minimal difference in pain associated with change in quality of life in children with rheumatic disease. Arthritis Care Res. 2002, 47, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, G.-N.; Alfonso, M.-N.; Pilar, A.-G.; Jesus, M.-A.; Angela Margaret, E. International normative data for paediatric foot posture assessment: A cross-sectional investigation. BMJ Open 2019, 9, e023341. [Google Scholar]
- Tsigaras, G.; Goulios, V.; Exadaktylou, S.; Besios, T.; Milioudi, M.; Chandolias, K.; Laspa, V.; Xristara, A. Efficacy of customized foot orthoses in children aged 6–7 years old with symptomatic flexible flat foot (SFFF). Open J. Prev. Med. 2020, 10, 217–244. [Google Scholar] [CrossRef]
- Pacey, V.; Tofts, L.; Adams, R.D.; Munns, C.F.; Nicholson, L.L. Quality of life prediction in children with joint hypermobility syndrome. J. Paediatr. Child Health 2015, 51, 689–695. [Google Scholar] [CrossRef]
- Janisse, D.J. Prescription footwear for arthritis of the foot and ankle. Clin. Orthop. Relat. Res. 1998, 349, 100–107. [Google Scholar] [CrossRef]
- Ibrahim, M.; El Hilaly, R.; Taher, M.; Morsy, A. A pilot study to assess the effectiveness of orthotic insoles on the reduction of plantar soft tissue strain. Clin. Biomech. 2013, 28, 68–72. [Google Scholar] [CrossRef]
- Fatoye, F.A.; Palmer, S.; Van der Linden, M.L.; Rowe, P.J.; Macmillan, F. Gait kinematics and passive knee joint range of motion in children with hypermobility syndrome. Gait Posture 2011, 33, 447–451. [Google Scholar] [CrossRef]
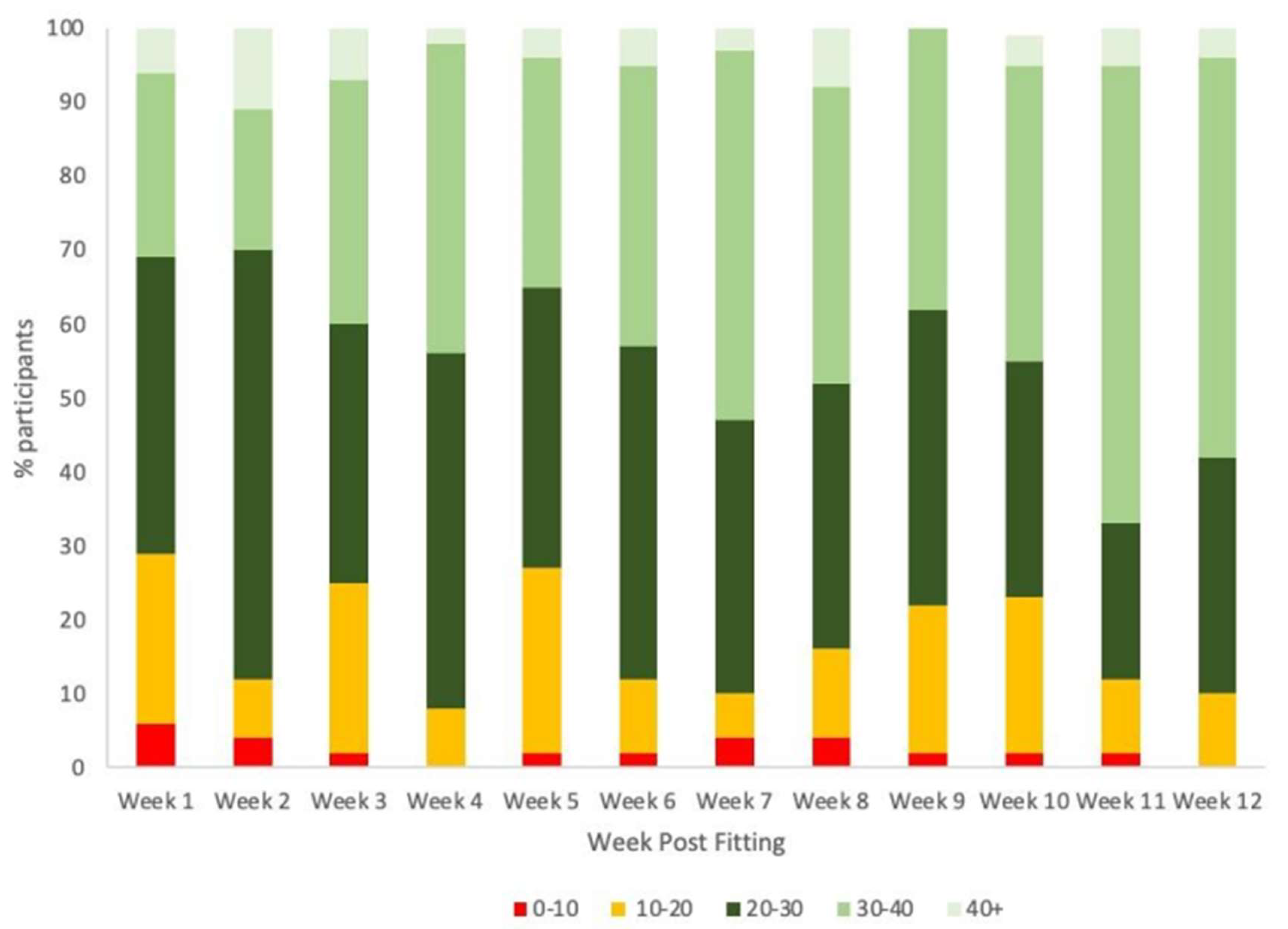
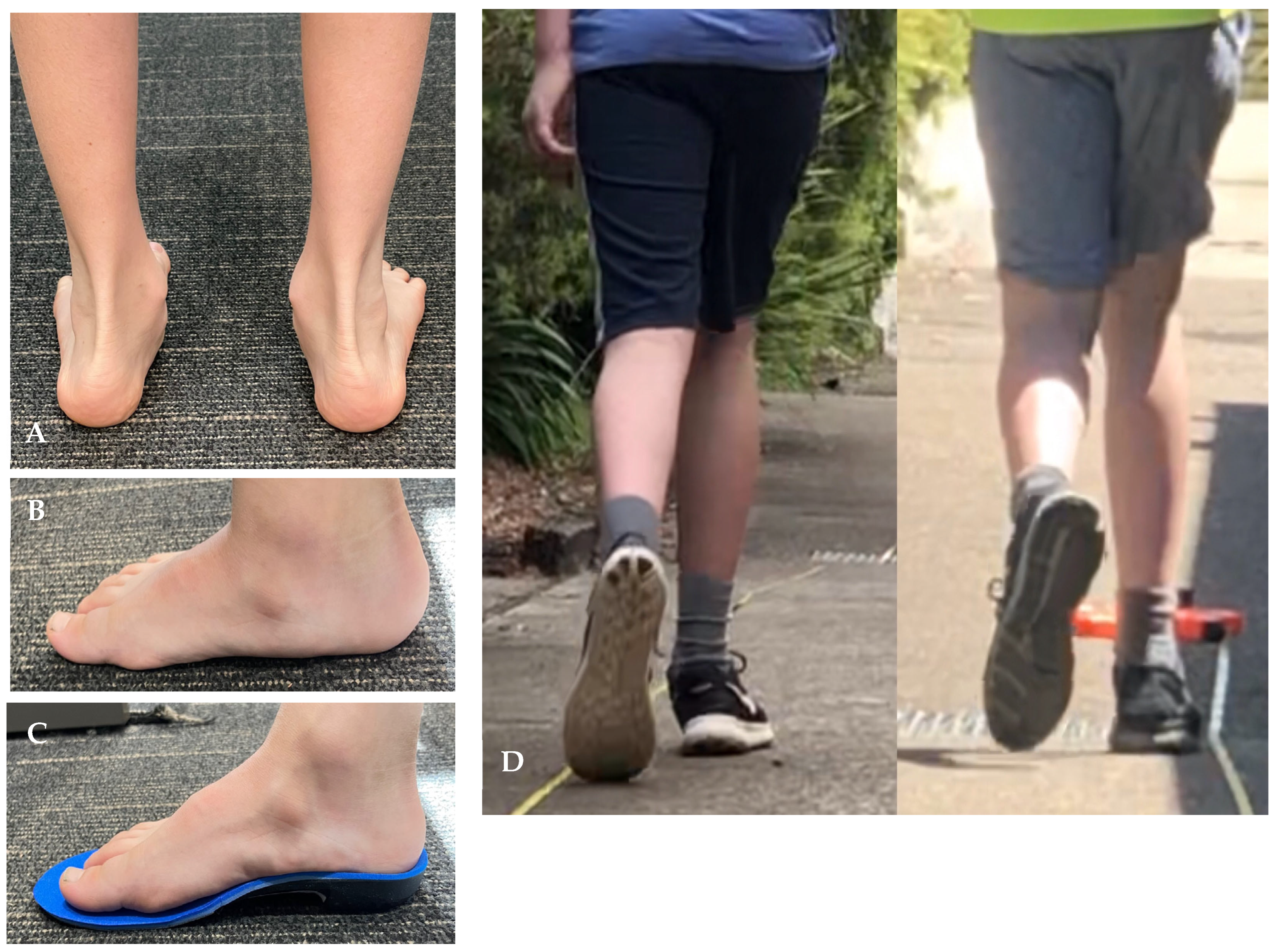
| Characteristic | Study Participants N = 53 |
|---|---|
| Age, years | |
| Mean (SD) | 10.6 (3.7) |
| Gender, n (%) | |
| Female | 35 (66%) |
| Male | 18 (34%) |
| Beighton score/9 | |
| Mean (SD) | 7 (1.24) |
| Height, m | |
| Mean (SD) | 1.43 (0.18) |
| Weight, kg | |
| Median (Range) | 33 (20, 82) |
| Body mass index, kg/m2 | |
| Median (Range) | 17.2 (12.0, 29.8) |
| Foot Posture Index (FPI/12 *) | |
| Left foot | |
| Mean (SD) | 9 (2.25) |
| Right foot | |
| Mean (SD) | 10 (2.26) |
| Outcome | Mean (SD) | 1-Month vs. Baseline | 3-Months vs. Baseline | 3-Months vs. 1-Month | |||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | 1 m | 3 m | Mean Diff (95%CI) | p-Value | Mean Diff (95%CI) | p-Value | Mean Diff (95%CI) | p-Value | |
| Primary outcomes | |||||||||
| Pain | |||||||||
| Visual analogue scale (VAS)/100 mm | 55 (17) | 29 (21) | 22 (18) | −27 (−33 to −21) | <0.001 | −33 (−39 to 27) | <0.001 | −6 (−11.9 to −0.4) | 0.036 |
| HRQoL | |||||||||
| Self-reported PedsQL Generic Core Scale Total/100 | 57 (17) | 68 (14) | 71 (12) | 11 (7.0 to 15) | <0.001 | 13 (9.4 to 17) | <0.001 | 2 (−1.6 to 6.3) | 0.242 |
| Physical Functioning Domain Score/100 | 49 (18) | 69 (17) | 67 (16) | 20 (15 to 25) | <0.001 | 18 (13 to 23) | <0.001 | 2 (−6.9 to 2.5) | 0.352 |
| Emotional Functioning Domain Score/100 | 60 (23) | 68 (21) | 71 (19) | 8 (3.0 to 14) | 0.003 | 12 (6.2 to 17) | <0.001 | 3 (−2.2 to 8.7) | 0.247 |
| Social Functioning Domain Score/100 | 68 (19) | 73 (20) | 75 (17) | 4 (−0.9 to 9.7) | 0.10 | 7 (2.0 to 13) | 0.007 | 3 (−2.4 to 8.2) | 0.282 |
| School Functioning Domain Score/100 | 59 (24) | 62 (19) | 67 (19) | 3 (−2.2 to 8.2) | 0.25 | 8 (2.5 to 13) | 0.004 | 5 (−0.5 to 9.9) | 0.078 |
| Parent reported PedsQL, Generic Core Scale Total/100 | 52 (14) | 66 (14) | 68 (15) | 14 (10 to 17) | <0.001 | 16 (12 to 20) | <0.001 | 2.3 (−1.4 to 5.9) | 0.214 |
| Physical Functioning Domain Score/100 | 46 (17) | 63 (17) | 67 (17) | 17 (12 to 23) | <0.001 | 22 (16 to 27) | <0.001 | 4 (−1.2 to 9.3) | 0.127 |
| Emotional Functioning Domain Score/100 | 50 (19) | 62 (19) | 68 (19) | 12 (6.4 to 18) | <0.001 | 18 (13 to 24) | <0.001 | 6 (0.6 to 11.9) | 0.030 |
| Social Functioning Domain Score/100 | 59 (21) | 70 (19) | 75 (20) | 11 (5.8 to 16) | <0.001 | 16 (11 to 21) | <0.001 | 5 (0.4 10.5) | 0.034 |
| School Functioning Domain Score/100 | 59 (20) | 67 (20) | 68 (19) | 8.7 (3.7 to 14) | <0.001 | 9.2 (4.2 to 14) | <0.001 | 0.5 (−4.4 to 5.4) | 0.842 |
| Secondary outcomes | |||||||||
| Functional Endurance | |||||||||
| Six-Minute walk test (6MWT) meters | 477 (68) | 503 (57) | 510 (59) | 27 (18 to 36) | <0.001 | 33 (24 to 42) | <0.001 | 6.8 (−2.2 to 15.8) | 0.140 |
| Fatigue | |||||||||
| Self-reported PedsQL MFS Total Score/100 | 52 (19) | 65 (16) | 67 (15) | 13 (8.7 to 17) | <0.001 | 15 (10 to 19) | <0.001 | 1.8 (−2.2 to 5.8) | 0.370 |
| General Domain Score/100 | 51 (21) | 64 (17) | 66 (17) | 13 (8.2 to 19) | <0.001 | 15 (9.6 to 20) | <0.001 | 1.5 (−3.7 to 6.6) | 0.581 |
| Sleep/Rest Domain Score/100 | 51 (21) | 64 (18) | 67 (20) | 13 (7.5 to 18) | <0.001 | 16 (11 to 22) | <0.001 | 3.7 (−1.7 to 8.9) | 0.177 |
| Cognitive Domain Score/100 | 56 (23) | 64 (18) | 67 (18) | 8 (3.3 to 13) | 0.001 | 12 (6.7 to 17) | <0.001 | 3.4 (−1.6 to 8.4) | 0.179 |
| Parent reported Peds QL MFS Total Score/100 | 53 (16) | 66 (16) | 68 (16) | 14 (9.7 to 18) | <0.001 | 16 (12 to 20) | <0.001 | 2.2 (−1.8 to 6.2) | 0.279 |
| General Domain Score/100 | 48 (18) | 63 (17) | 65 (17) | 15 (11 to 20) | <0.001 | 17 (12 to 22) | <0.001 | 1.5 (−3.2 to 6.2) | 0.523 |
| Sleep/Rest Domain Score/100 | 57 (19) | 70 (19) | 73 (19) | 12 (7.8 to 17) | <0.001 | 16 (12 to 21) | <0.001 | 3.9 (−0.7 to 8.4) | 0.095 |
| Cognitive Domain Score/100 | 52 (22) | 64 (22) | 65 (25) | 12 (7.0 to 18) | <0.001 | 14 (8.2 to 19) | <0.001 | 1.2 (−4.1 to 6.5) | 0.568 |
| PGIC * | Participant n/N (n%) | |
|---|---|---|
| 1-Month | 3-Months | |
| No change | 2/53 (3.8%) | - |
| Slight Improvement (SI) | 8/53 (15%) | 10/53 (18.8%) |
| Much Improved (MI) | 29/53 (54.7%) | 22/53 (41.5%) |
| Very Much Improved (VMI) | 13/53 (24.5%) | 20/53 (37.7%) |
| Unknown | 1/53 (2%) | 1/53 (2%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maarj, M.; Pacey, V.; Tofts, L.; Clapham, M.; Coda, A. The Impact of Podiatric Intervention on the Quality of Life and Pain in Children and Adolescents with Hypermobility. Int. J. Environ. Res. Public Health 2023, 20, 6623. https://doi.org/10.3390/ijerph20176623
Maarj M, Pacey V, Tofts L, Clapham M, Coda A. The Impact of Podiatric Intervention on the Quality of Life and Pain in Children and Adolescents with Hypermobility. International Journal of Environmental Research and Public Health. 2023; 20(17):6623. https://doi.org/10.3390/ijerph20176623
Chicago/Turabian StyleMaarj, Muhammad, Verity Pacey, Louise Tofts, Matthew Clapham, and Andrea Coda. 2023. "The Impact of Podiatric Intervention on the Quality of Life and Pain in Children and Adolescents with Hypermobility" International Journal of Environmental Research and Public Health 20, no. 17: 6623. https://doi.org/10.3390/ijerph20176623
APA StyleMaarj, M., Pacey, V., Tofts, L., Clapham, M., & Coda, A. (2023). The Impact of Podiatric Intervention on the Quality of Life and Pain in Children and Adolescents with Hypermobility. International Journal of Environmental Research and Public Health, 20(17), 6623. https://doi.org/10.3390/ijerph20176623







