Vascular Effects, Potential Pathways and Mediators of Fetal Exposure to Alcohol and Cigarette Smoking during Pregnancy: A Narrative Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Eligibility Criteria
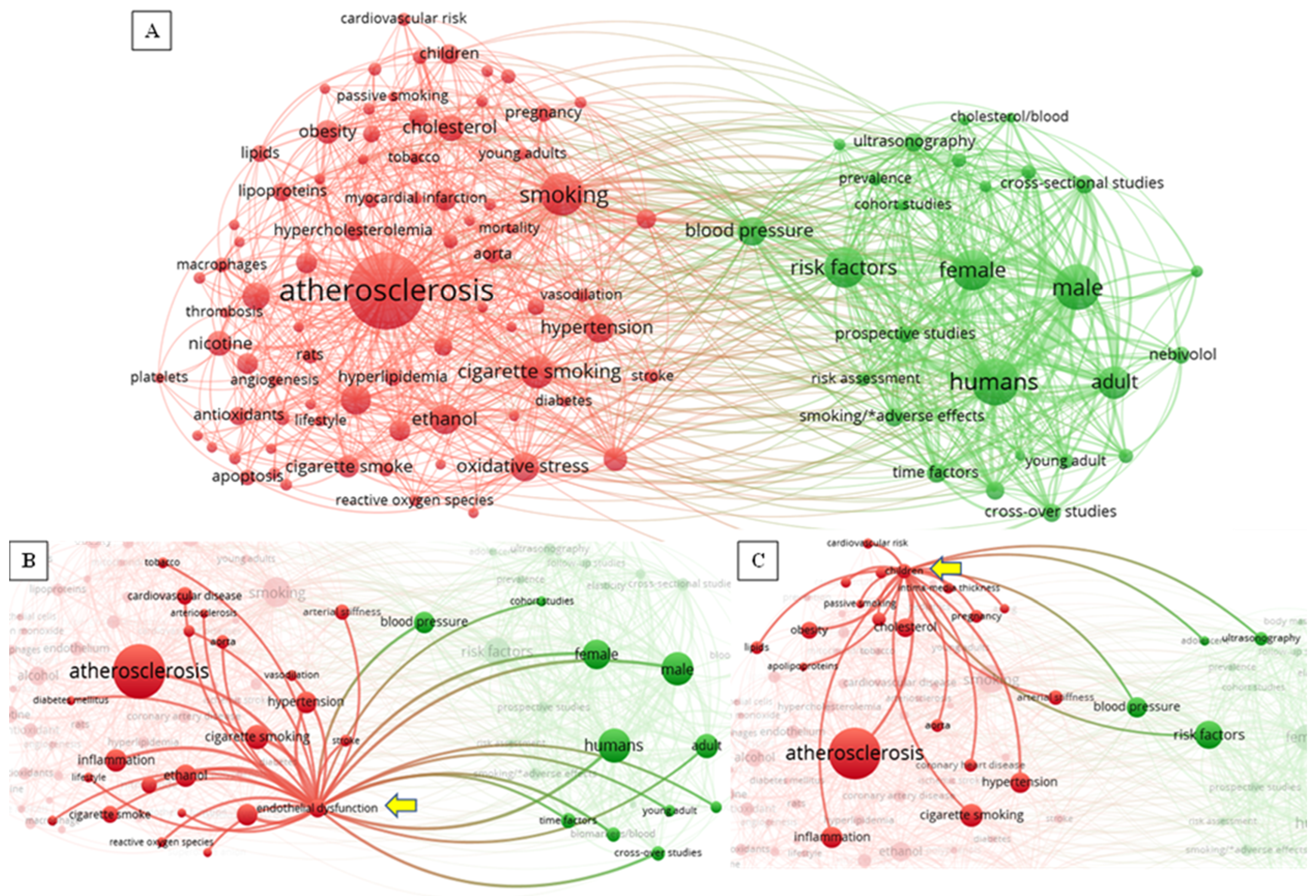
2.3. Graphical Bibliographical Analysis of Literature Search
3. Results
3.1. Teratogen Exposure during Pregnancy and Vascular Dysfunction
3.2. Teratogen Exposure during Pregnancy and Altered Lipid Metabolism
| Reference | Year, Country | Sample | Study Design | Analysis Methods | Outcome |
|---|---|---|---|---|---|
| Anderson, Walker and Stender. [34] | 2004, Copenhagen | 74 pregnant women and infants (Smoking = 30, Control = 44) | Prospective pregnancy/birth cohort study | Endothelial cells of fetuses’ umbilical veins were isolated after birth, and eNOS activity was studied by an eNOS immunoassay. | The eNOS activity in the umbilical veins of fetuses exposed to maternal smoking was significantly reduced (p = 0.006) by 40%, with a 32% lower concentration of eNOS, suggesting endothelial dysfunction and reduced fetal growth. |
| Gunes et al. [14] | 2007, Kayseri, Türkiye | 28 term neonates with smoking mothers and 28 term neonates with non-smoking mothers | Prospective pregnancy/birth cohort study | Aortic artery IMT was measured in newborns using high-resolution ultrasound. | The mean aIMT and weight-adjusted aIMT were significantly greater in neonates born to smoking mothers during pregnancy compared to neonates in the control group. Maternal smoking did not significantly decrease insulin-like growth factor I (IGF-I) and IGFBP-3 levels in their infants. |
| Geelhoed et al. [35] | 2011, Rotterdam, The Netherlands | 1120 mothers and their two-year-old children (≥5 cigarettes per day = 72), (<5 cigarettes per day = 68) | A prospective birth cohort study | Pulsed Doppler ultrasound assessed fetal and placental arterial resistance characteristics during the third trimester. At two years old, cardiac measurements were evaluated using M-mode and Doppler echocardiograms. | Continued smoking during pregnancy (≥5 cigarettes per day) was associated with increased fetal arterial resistance in the umbilical (p < 0.01), uterine (p < 0.01) and middle cerebral arteries (p < 0.05), and with a decrease in blood flow (p < 0.05) and diameter (p < 0.01) in the ascending aorta in the third trimester. Increased fetal umbilical artery resistance was also associated with a reduced aortic root diameter at two years old. |
| Iwashima et al. [15] | 2012, Hamamatsu, Japan | 96 mothers and newborns | Prospective pregnancy/birth cohort study | Abdominal aortic artery IMT in newborns using high-resolution ultrasound. | No associations were investigated between maternal smoking and newborn aIMT due to limitations. However, there were no significant differences in newborn aIMT when comparing newborns exposed to smoking mothers vs. non-smoking mothers. |
| Geerts et al. [17] | 2012, Utrecht, Netherlands | 259 five-year-old children and their parents (maternal smoking = 15, non-smoking = 244) | A prospective birth cohort study | cIMT and arterial wall distensibility were measured using ultrasound. | Children born to maternal smoking during pregnancy had significantly thicker cIMT (18.8 μm) at five years old (p = 0.04), with 15% significantly lower arterial wall distensibility. Maternal smoking was not associated with cIMT after adjusting for the child’s growth patterns. |
| Taal et al. [36] | 2013, The Netherlands | 5565 mothers and six-year-old children (non-smoking n = 4159, continued smoking n = 912, stopped smoking n = 494) | A prospective birth cohort study | Blood pressure, carotid–femoral pulse-wave velocity and left cardiac structures and function were assessed. | No association was observed between maternal cigarette smoking during pregnancy with systolic blood pressure (SBP), carotid–femoral PWV and left cardiac structures. Maternal smoking of 10 or more cigarettes per day was associated with a higher fractional shortening in childhood. A dose–response association was observed between the number of cigarettes smoked during the third trimester and diastolic blood pressure (DBP) in six-year-old children. |
| Dugmonits et al. [37] | 2019, Hungary | 113 mothers and neonates (non-smoking n = 62, smoking n = 51) | Ex vivo study | Red blood cells were isolated from fetal umbilical cord artery samples for morphological and molecular studies. | Circulating red blood cells in fetuses exposed to maternal smoking presented with impaired activation of nitric oxide synthase 3 (IGFBP-3), regardless of the phenotypic appearance, as well as an upregulation of the Arginase 1 (ARG1) enzyme, a molecule in the NOS3-NO pathway. The association between decreased NOS3 and maternal smoking was not investigated. |
| De Smidt et al. [7] | 2021, South Africa | 500 five-year-old children (control n = 146, smoking n = 167, alcohol n = 33, dual-exposed n = 154) | Prospective pregnancy/birth cohort | Ultrasound measurements of the aorta and carotid arteries IMT were performed at the age of five years | cIMT was significantly higher in children exposed to both alcohol and cigarette smoking during pregnancy compared to those not exposed (p = 0.008). Dual exposure was associated with higher right cIMT (p < 0.01). |
| Turan et al. [16] | 2021, Turkey | 74 mothers and term neonates aged 0–30 days (smoking n = 40, control n = 34) | Prospective pregnancy/birth cohort | Carotid IMT was measured by standard ultrasound examination, and fasting blood cholesterol was determined by an automatic chemistry analyzer. | There were no differences in carotid IMT and low-density lipoprotein (LDL)-cholesterol, triglycerides and total cholesterol between neonates exposed to maternal smoking (n = 40) and neonates in the control group (n = 34). However, neonates exposed to maternal smoking had significantly lower HDL levels (39.1 ± 17.8 mg/dL; p = 0.021). |
| Monasso et al. [38] | 2022, The Netherlands | 4639 mothers and 10-year-old children (control n = 3180, smoking n = 627, quit smoking n = 359) | Prospective pregnancy/birth cohort | Common carotid artery intima–media thickness and distensibility were assessed using standard ultrasound protocols. | Continued maternal tobacco smoking was not associated with arterial health markers such as cIMT and distensibility at the age of 10 years old. |
| Reference | Year, Country | Sample Size | Analysis Methods | Outcome |
|---|---|---|---|---|
| Turcotte et al. [27] | 2002, North Dakota | 31 female pregnant Sprague Dawley rats and offspring (control = 18, ethanol exposed = 13) | Vascular function was studied in fetal alcohol syndrome rats by evaluating force-generating capacity of thoracic aorta segments isolated from adult rat offspring. | Ethanol treatment resulted in altered vascular contractile function in aortic segments, whether the responses were endothelium-dependent or endothelium-independent. |
| Xiao et al. [22] | 2007, California, United States | 17 pregnant Sprague Dawley Rats (control = 9, nicotine-treated = 8) | Isolated aortic from adult male and female offspring at three months old. | Prenatal nicotine exposure decreased aortic endothelium-dependent vasodilation induced by acetylcholine (Ach) in male rat offspring. However, it increased endothelium-dependent relaxation in female offspring. Nicotine treatment resulted in vascular alterations differently in each gender. |
| Xiao et al. [21] | 2011, United States | 25 pregnant Sprague Dawley rats and male rat offspring | Aortas were isolated, contraction studies were performed and Western blot analysis was used to determine protein expression of NADPH oxidase (Nox2 and Nox4). Oxidative damage was determined by measuring malondialdehyde levels, superoxide production and superoxide dismutase. | Aortic relaxations induced by Ach were impaired in rat adult offspring exposed to nicotine treatment in pregnant rats. Nicotine exposure resulted in increased oxidative stress and heightened Nox2-dependent mediated hypertensive reactivity. Therefore, prenatal nicotine exposure in a rat model supports the concept of fetal programming of vascular dysfunction through heightened oxidative stress. |
| Gunes et al. [18] | 2011, Kayseri, Türkiye | 25 female adult white Sprague Dawley and offspring (control n = 5, nicotine group A n = 10, nicotine group B (n = 10) | Using histopathology, the abdomen aortic IMT in rat pups was studied at 45 days of age. | The aIMT was significantly higher in rat pups in both nicotine groups exposed to the nicotine treatment compared to the controls (p < 0.001). No significant difference in aIMT when comparing the two nicotine groups that received different concentrations of nicotine (nicotine group A: 6 mg/kg/day and nicotine group B: 3 mg/kg/day). |
| Parkington et al. [28] | 2014, Australia | Twelve female sheep (ewes) and their fetuses, six exposed and six non-exposed fetuses | Arteries were isolated on day 134 of pregnancy for vascular function tests using a wire myograph. Arterial stiffness was assessed using a pressure myograph. | Daily ethanol exposure during late pregnancy reduced coronary arteries’ endothelium-dependent vasodilatation sensitivity by 10-fold without fetal growth restriction. Ethanol exposure was also significantly associated with reduced endothelial NO synthase mRNA. Arterial stiffness was increased in all isolated arteries (coronary, renal, mesenteric, psoas muscle and cerebral). |
| Subramanian et al. [26] | 2015, United States | 12 Sprague Dawley rats and fetuses on gestational day 18 | Vascular studies were performed using wire myography to investigate uterine artery function to vasodilators and vasoconstrictors in a pregnant rat model. | Moderate alcohol exposure resulted in impaired acetylcholine-mediated and endothelium-dependent vasodilation in uterine arteries of pregnant mothers in the absence of growth deficits. |
| Naik et al. [25] | 2016, United States | 16 Sprague Dawley rats and fetuses (control = 8, alcohol exposed = 8) | The uterine artery was isolated on gestational day 20, and functional tests were performed using dual-chamber arteriography. | Daily binge alcohol exposure resulted in impaired endothelial vasodilation in the primary uterine artery by impeding the eNOS pathway. |
| Tobiasz et al. [24] | 2018, United States | 18 pregnant baboons (control = 9, alcohol exposed = 9) | Pregnant baboons were treated with alcohol three times in the 2nd semester. Fetal cardiovascular parameters were assessed using Doppler ultrasound. | Fetal cerebral blood flow (peak systolic velocity) decreased during alcohol exposure, but no significant differences were found in fetal cardiovascular indices at term. Therefore, alcohol affected vascular function, but no persistent changes were observed. |
| Alfourti et al. [20] | 2019, Libya | Pregnant Wister rats (sample size unknown) | Pregnant rats were treated with nicotine (1 mg/kg/day in 1 mL). The abdominal aorta was isolated for histological studies. | The three tunica layers of the abdominal aorta showed irregular alignment in rats exposed to nicotine compared to regular alignment in the control group. |
| Soraya et al. [19] | 2022, Iran | 20 Wister rats and their male rat offspring | Systemic hemodynamic measurements were analyzed in offspring on postnatal day 90, utilizing waveform contour analysis. Histology of the aortic wall and level of inflammatory factors of the aorta were assessed at postnatal days 21 and 90. | Aortic wall thickness significantly increased in the ethanol-exposed group at postnatal days 21 and 90. Inflammatory factors such as tumor necrosis factor (TNF)-α, endothelin-1 and nuclear factor (NF)-κ, and intercellular adhesion molecule (ICAM)-1 significantly increased in the ethanol-exposed group (p < 0.001), predisposing the aorta to atherosclerosis. SBP, DBP, mean arterial pressure (MAP) and dicrotic pressure were significantly higher in rat offspring with ethanol exposure. |
4. Discussion
4.1. Potential Mediators in Vascular Dysfunction
4.2. Additional Influences during Childhood
4.3. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zahorán, S.; Szántó, P.R.; Bódi, N.; Bagyánszki, M.; Maléth, J.; Hegyi, P.; Sári, T.; Hermesz, E. Sustained Maternal Smoking Triggers Endothelial-Mediated Oxidative Stress in the Umbilical Cord Vessels, Resulting in Vascular Dysfunction. Antioxidants 2021, 10, 583. [Google Scholar] [CrossRef] [PubMed]
- Bakker, H.; Jaddoe, V.W.V. Cardiovascular and Metabolic Influences of Fetal Smoke Exposure. Eur. J. Epidemiol. 2011, 26, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Balistreri, C.R. Fetal Programming as the Cause of All the Evils in Adult Humans: Atherosclerosis and Coronary Heart Disease Included. Cardiovasc. Med. 2020, 23, 1–6. [Google Scholar] [CrossRef]
- Benedict, L.M.; Sarma, D.; Moulick, A.; Stevens, R.; Mahan, V. Identifying and Preventing Cardiac Risk Factors from Fetal Life. J. Pediatr. Pediatr. Med. 2018, 2, 7–13. [Google Scholar]
- Toda, N.; Ayajiki, K. Vascular Actions of Nitric Oxide as Affected by Exposure to Alcohol. Alcohol Alcohol. 2010, 45, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Rodríguez, P.; Ramiro-Cortijo, D.; Reyes-Hernández, C.G.; López de Pablo, A.L.; Carmen González, M.; Arribas, S.M. Implication of Oxidative Stress in Fetal Programming of Cardiovascular Disease. Front. Physiol. 2018, 9, 602. [Google Scholar] [CrossRef]
- De Smidt, J.; Odendaal, H.J.; Nel, D.G.; Nolan, H.; Du Plessis, C.; Brink, L.T.; Oelofse, A. In Utero Teratogen Exposure and Cardiometabolic Risk in 5-Year-Old Children: A Prospective Pediatric Study. J. Matern. Neonatal Med. 2019, 34, 3740–3749. [Google Scholar] [CrossRef]
- Morley, R.; Dwyer, T.; Hynes, K.L.; Cochrane, J.; Ponsonby, A.L.; Parkington, H.C.; Carlin, J.B. Maternal Alcohol Intake and Offspring Pulse Wave Velocity. Neonatology 2010, 97, 204–211. [Google Scholar] [CrossRef]
- Kable, J.A.; Mehta, P.K.; Rashid, F.; Coles, C.D. Path Analysis of the Impact of Prenatal Alcohol on Adult Vascular Function. Alcohol. Clin. Exp. Res. 2023, 47, 116–126. [Google Scholar] [CrossRef]
- Skilton, M.R.; Celermajer, D.S.; Cosmi, E.; Crispi, F.; Gidding, S.S.; Raitakari, O.T.; Urbina, E.M. Natural History of Atherosclerosis and Abdominal Aortic Intima-Media Thickness: Rationale, Evidence, and Best Practice for Detection of Atherosclerosis in the Young. J. Clin. Med. 2019, 8, 1201. [Google Scholar] [CrossRef]
- Chen, Y.N.Y. Distributions of Diffuse Intimal Thickening in Human Arteries: Preferential Expression in Atherosclerosis-Prone Arteries from an Early Age. Virchows Arch. 2002, 441, 279–288. [Google Scholar] [CrossRef]
- Hayes, N.; Reid, N.; Akison, L.K.; Moritz, K.M. The Effect of Heavy Prenatal Alcohol Exposure on Adolescent Body Mass Index and Waist-to-Height Ratio at 12–13 Years. Int. J. Obes. 2021, 45, 2118–2125. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, P.; Datta, S.; Mishra, A.; Sinukumar, S.; Garg, A.; Joshi, P. Harmful Effects of Nicotine. Indian J. Med. Paediatr. Oncol. 2015, 36, 24. [Google Scholar] [CrossRef]
- Gunes, T.; Koklu, E.; Yikilmaz, A.; Ozturk, M.A.; Akcakus, M.; Kurtoglu, S.; Coskun, A.; Koklu, S. Influence of Maternal Smoking on Neonatal Aortic Intima-Media Thickness, Serum IGF-I and IGFBP-3 Levels. Eur. J. Pediatr. 2007, 166, 1039–1044. [Google Scholar] [CrossRef] [PubMed]
- Iwashima, S.; Ishikawa, T.; Akira, O.; Itou, H. Association of Abdominal Aortic Wall Thickness in the Newborn with Maternal Factors. Am. J. Perinatol. 2012, 29, 441–448. [Google Scholar] [CrossRef]
- Turan, H.; Meşe, T.; Doksoz, O.; Turan, V. Assessment of Carotid Artery Intima-Media Thickness and Lipid Profile in Term Neonates Born to Smoking Mothers. Iran. J. Pediatr. 2021, 31, 1–6. [Google Scholar] [CrossRef]
- Geerts, C.C.; Bots, M.L.; Van Der Ent, C.K.; Grobbee, D.E.; Uiterwaal, C.S.P.M. Parental Smoking and Vascular Damage in Their 5-Year-Old Children. Pediatrics 2012, 129, 45–54. [Google Scholar] [CrossRef]
- Gunes, T.; Akin, M.A.; Canoz, O.; Coban, D.; Ozcan, B.; Kose, M.; Ozturk, M.A.; Kurtoglu, S. Aortic Intima-Media Thickness in Nicotine-Exposed Rat Pups during Gestation and Lactation Period. Eur. J. Pediatr. 2011, 170, 1257–1262. [Google Scholar] [CrossRef]
- Soraya, H.; Sheikholeslami, S.; Shirpoor, A.; Majd, F.N.; Naderi, R.; Rasmi, Y. Influence of Maternal Ethanol Exposure on Systemic Hemodynamic Variables and Histopathological Changes in the Aorta Wall of Male Rat Offspring: A Three-Month Follow-Up. Iran. J. Med. Sci. 2022, 47, 468–476. [Google Scholar] [CrossRef]
- Alfourti, A.M.A.B.; Azzwali, A.A.A.; Azab, A.E. Effects of Maternal Nicotine Exposure during Pregnancy and Lactation on Blood Pressure of the Offspring and Blood Vessel Structure and Attenuation by Vitamin C. East Afr. Sch. J. Med. Sci. 2019, 2, 240–246. [Google Scholar]
- Xiao, D.; Huang, X.; Yang, S.; Zhang, L. Antenatal Nicotine Induces Heightened Oxidative Stress and Vascular Dysfunction in Rat Offspring. Br. J. Pharmacol. 2011, 164, 1400–1409. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.L.; Huang, X.; Lawrence, J.; Yang, S.; Zhang, L. Fetal and Neonatal Nicotine Exposure Differentially Regulates Vascular Contractility in Adult Male and Female Offspring. J. Pharmacol. Exp. Ther. 2007, 320, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Ramadoss, J.; Magness, R.R. Vascular Effects of Maternal Alcohol Consumption. Am. J. Physiol. -Heart Circ. Physiol. 2012, 303, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Tobiasz, A.M.; Duncan, J.R.; Bursac, Z.; Sullivan, R.D.; Tate, D.L.; Dopico, A.M.; Bukiya, A.N.; Mari, G. The Effect of Prenatal Alcohol Exposure on Fetal Growth and Cardiovascular Parameters in a Baboon Model of Pregnancy. Reprod. Sci. 2018, 25, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Naik, V.D.; Davis-Anderson, K.; Subramanian, K.; Lunde-Young, R.; Nemec, M.J.; Ramadoss, J. Mechanisms Underlying Chronic Binge Alcohol Exposure-Induced Uterine Artery Dysfunction in Pregnant Rat. Alcohol. Clin. Exp. Res. 2018, 42, 682–690. [Google Scholar] [CrossRef]
- Subramanian, K.; Naik, V.D.; Sathishkumar, K.; Yallampalli, C.; Saade, G.R.; Hankins, G.D.; Ramadoss, J. Chronic Binge Alcohol Exposure during Pregnancy Impairs Rat Maternal Uterine Vascular Function. Alcohol. Clin. Exp. Res. 2014, 38, 1832–1838. [Google Scholar] [CrossRef]
- Turcotte, L.A.; Aberle, N.S.; Norby, F.L.; Wang, G.J.; Ren, J. Influence of Prenatal Ethanol Exposure on Vascular Contractile Response in Rat Thoracic Aorta. Alcohol 2002, 26, 75–81. [Google Scholar] [CrossRef]
- Parkington, H.C.; Kenna, K.R.; Sozo, F.; Coleman, H.A.; Bocking, A.; Brien, J.F.; Harding, R.; Walker, D.W.; Morley, R.; Tare, M. Maternal Alcohol Consumption in Pregnancy Enhances Arterial Stiffness and Alters Vasodilator Function That Varies between Vascular Beds in Fetal Sheep. J. Physiol. 2014, 592, 2591–2603. [Google Scholar] [CrossRef]
- Dejong, K.; Olyaei, A.; Lo, J.O. Alcohol Use in Pregnancy. Clin. Obstet. Gynecol. 2019, 62, 142–155. [Google Scholar] [CrossRef]
- Parkington, H.C.; Coleman, H.A.; Wintour, E.M.; Tare, M. Prenatal Alcohol Exposure: Implications for Cardiovascular Function in the Fetus and Beyond. Clin. Exp. Pharmacol. Physiol. 2010, 37, 91–98. [Google Scholar] [CrossRef]
- Cajachagua-Torres, K.N.; El Marroun, H.; Reiss, I.K.M.; Santos, S.; Jaddoe, V.W.V. Foetal Tobacco and Cannabis Exposure, Body Fat and Cardio-Metabolic Health in Childhood. Pediatr. Obes. 2021, 17, e12863. [Google Scholar] [CrossRef] [PubMed]
- Akison, L.K.; Reid, N.; Wyllie, M.; Moritz, K.M. Adverse Health Outcomes in Offspring Associated with Fetal Alcohol Exposure: A Systematic Review of Clinical and Preclinical Studies with a Focus on Metabolic and Body Composition Outcomes. Alcohol. Clin. Exp. Res. 2019, 43, 1324–1343. [Google Scholar] [CrossRef] [PubMed]
- Weeks, O.; Fazeli, P.K.; Goessling, W.; Weeks, O.; Bossé, G.D.; Oderberg, I.M.; Akle, S.; Houvras, Y.; Wrighton, P.J.; Labella, K.; et al. Fetal Alcohol Spectrum Disorder Predisposes to Metabolic Abnormalities in Adulthood Fetal Alcohol Spectrum Disorder Predisposes to Metabolic Abnormalities in Adulthood. J. Clin. Investig. 2020, 130, 2252–2269. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.R.; Walker, L.R.; Stender, S. Reduced Endothelial Nitric Oxide Synthase Activity and Concentration in Fetal Umbilical Veins from Maternal Cigarette Smokers. Am. J. Obstet. Gynecol. 2004, 191, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Geelhoed, J.J.M.; El Marroun, H.; Verburg, B.O.; Van Osch-Gevers, L.; Hofman, A.; Huizink, A.C.; Moll, H.A.; Verhulst, F.C.; Helbing, W.A.; Steegers, E.A.P.; et al. Maternal Smoking during Pregnancy, Fetal Arterial Resistance Adaptations and Cardiovascular Function in Childhood. BJOG Int. J. Obstet. Gynaecol. 2011, 118, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Taal, H.R.; de Jonge, L.L.; van Osch-Gevers, L.; Steegers, E.A.P.; Hofman, A.; Helbing, W.A.; van der Heijden, A.J.; Jaddoe, V.W.V. Parental Smoking during Pregnancy and Cardiovascular Structures and Function in Childhood: The Generation R Study. Int. J. Epidemiol. 2013, 42, 1371–1380. [Google Scholar] [CrossRef]
- Dugmonits, K.N.; Chakraborty, P.; Hollandi, R.; Zahorán, S.; Pankotai-Bodó, G.; Horváth, P.; Orvos, H.; Hermesz, E. Maternal Smoking Highly Affects the Function, Membrane Integrity, and Rheological Properties in Fetal Red Blood Cells. Oxid. Med. Cell. Longev. 2019, 2019, 1–10. [Google Scholar] [CrossRef]
- Monasso, G.S.; Felix, J.F.; Gaillard, R.; Jaddoe, V.W. V Fetal and Childhood Exposure to Parental Tobacco Smoking and Arterial Health at Age 10 Years. Am. J. Hypertens. 2022, 35, 867–874. [Google Scholar] [CrossRef]
- Hernández, J.A.; López-Sánchez, R.C.; Rendón-Ramírez, A. Lipids and Oxidative Stress Associated with Ethanol-Induced Neurological Damage. Oxid. Med. Cell. Longev. 2016, 2016, 1–15. [Google Scholar] [CrossRef]
- Le Daré, B.; Lagente, V.; Gicquel, T. Ethanol and Its Metabolites: Update on Toxicity, Benefits, and Focus on Immunomodulatory Effects. Drug Metab. Rev. 2019, 51, 545–561. [Google Scholar] [CrossRef]
- Marianian, A.; Atalyan, A.; Bohora, S.; Darenskaya, M.; Grebenkina, L.; Kolesnikova, L.; Kolesnikov, S.; Mikhaylevich, I.; Protopopova, N.; Stockett, M.; et al. The Effect of Low Alcohol Consumption during Pregnancy on the Lipid Peroxidation-Antioxidant Defense System of Women, Their Alcohol-Exposed Infants, and Growth, Health, and Developmental Outcomes. Birth Defects Res. 2020, 112, 40–53. [Google Scholar] [CrossRef] [PubMed]
- Bosco, C.; Diaz, E. Pharmacology and Cell Metabolism Placental Hypoxia and Foetal Development Versus Alcohol Exposure in Pregnancy. Alcohol Alcohol. 2012, 47, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Matjuda, E.N.; Engwa, G.A.; Nkeh, S.; Anye, C.; Nkeh-chungag, B.N.; Goswami, N. Cardiovascular Risk Factors and Their Relationship with Vascular Dysfunction in South African Children of African Ancestry. J. Clin. Med. 2021, 10, 354. [Google Scholar] [CrossRef] [PubMed]
- Cyr, A.R.; Huckaby, L.V.; Shiva, S.S.; Zuckerbraun, B.S. Nitric Oxide and Endothelial Dysfunction. Crit. Care Clin. 2020, 36, 307–321. [Google Scholar] [CrossRef]
- Vaiserman, A.M. Long-Term Health Consequences of Early-Life Exposure to Substance Abuse: An Epigenetic Perspective. J. Dev. Orig. Health Dis. 2020, 4, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Ivorra, C.; Fraga, M.F.; Bayón, G.F.; Fernández, A.F.; Garcia-Vicent, C.; Chaves, F.J.; Redon, J.; Lurbe, E. DNA Methylation Patterns in Newborns Exposed to Tobacco in Utero. J. Transl. Med. 2015, 13, 1–9. [Google Scholar] [CrossRef]
- Everson, T.M.; Vives-Usano, M.; Seyve, E.; Cardenas, A.; Lacasaña, M.; Craig, J.M.; Lesseur, C.; Baker, E.R.; Fernandez-Jimenez, N.; Heude, B.; et al. Placental DNA Methylation Signatures of Maternal Smoking during Pregnancy and Potential Impacts on Fetal Growth. Nat. Commun. 2021, 12, 5095. [Google Scholar] [CrossRef]
- Cleal, J.; Poore, K.; Lewis, R. The Placental Exposome, Placental Epigenetic Adaptations and Lifelong Cardio-Metabolic Health. Mol. Asp. Med. 2022, 87, 101095. [Google Scholar] [CrossRef]
- Rauschert, S.; Melton, P.E.; Burdge, G.; Craig, J.M.; Godfrey, K.M.; Holbrook, J.D.; Lillycrop, K.; Mori, T.A.; Beilin, L.J.; Oddy, W.H.; et al. Maternal Smoking During Pregnancy Induces Persistent Epigenetic Changes Into Adolescence, Independent of Postnatal Smoke Exposure and Is Associated with Cardiometabolic Risk. Front. Genet. 2019, 10, 770. [Google Scholar] [CrossRef]
- Costantino, S.; Mohammed, S.A.; Ambrosini, S.; Paneni, F. Epigenetic Processing in Cardiometabolic Disease. Atherosclerosis 2018, 281, 150–158. [Google Scholar] [CrossRef]
- Epure, A.M.; Rios-Leyvraz, M.; Anker, D.; Di Bernardo, S.; da Costa, B.R.; Chiolero, A.; Sekarski, N. Risk Factors during First 1000 Days of Life for Carotid Intima-Media Thickness in Infants, Children, and Adolescents: A Systematic Review with Meta-Analyses. PLoS Med. 2020, 17, e1003414. [Google Scholar] [CrossRef] [PubMed]
- Monasso, G.S.; Silva, C.C.V.; Santos, S.; Goncalvez, R.; Gaillard, R.; Felix, J.F.; Jaddoe, V.W.V. Infant Weight Growth Patterns, Childhood BMI, and Arterial Health at Age 10 Years. Obesity 2022, 30, 770–778. [Google Scholar] [CrossRef] [PubMed]
- White, D.A.; Anand, G.M.; Qayum, O.; Ibezim, C.F.; Sherman, A.K.; Raghuveer, G. Modifiable Clinical Correlates of Vascular Health in Children and Adolescents with Dyslipidemia. Pediatr. Cardiol. 2019, 40, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Gambineri, A.; Conforti, A.; Di Nisio, A.; Laudisio, D.; Muscogiuri, G.; Barrea, L.; Savastano, S.; Colao, A. Maternal Obesity: Focus on Offspring Cardiometabolic Outcomes. Int. J. Obes. Suppl. 2020, 10, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Mourino, N.; Varela-Lema, L.; Ahluwalia, J.S.; Rey-Brandariz, J.; Candal-Pedreira, C.; Ruano-Ravina, A.; Vila-Farinas, A.; Torres, A.; Pérez-Rios, M. Maternal Smoking in Pregnancy and Blood Pressure during Childhood and Adolescence: A Meta-Analysis. Eur. J. Pediatr. 2023, 182, 2119–2132. [Google Scholar] [CrossRef]
- van Dijk, A.E.; van Eijsden, M.; Stronks, K.; Gemke, R.J.B.J.; Vrijkotte, T.G.M. The Association between Prenatal Psychosocial Stress and Blood Pressure in the Child at Age 5–7 Years. PLoS ONE 2012, 7, e43548. [Google Scholar] [CrossRef]
- Eick, S.M.; Meeker, J.D.; Brown, P.; Swartzendruber, A.; Rios-McConnell, R.; Shen, Y.; Milne, G.L.; Vélez Vega, C.; Rosario, Z.; Alshawabkeh, A.; et al. Associations between Socioeconomic Status, Psychosocial Stress, and Urinary Levels of 8-Iso-Prostaglandin-F2α during Pregnancy in Puerto Rico. Free Radic. Biol. Med. 2019, 143, 95–100. [Google Scholar] [CrossRef]
- Viikari, J.S.A.; Gall, S.; Venn, A.; Dwyer, T.; Magnussen, C.G.; Huynh, Q.L.; Raitakari, O.T.; Kähönen, M.; Juonala, M. Exposure to Parental Smoking in Childhood or Adolescence Is Associated with Increased Carotid Intima-Media Thickness in Young Adults: Evidence from the Cardiovascular Risk in Young Finns Study and the Childhood Determinants of Adult Health Study. Eur. Heart J. 2014, 35, 2484–2491. [Google Scholar] [CrossRef]
- Dong, G.H.; Wang, J.; Zeng, X.W.; Chen, L.; Qin, X.D.; Zhou, Y.; Li, M.; Yang, M.; Zhao, Y.; Ren, W.H.; et al. Interactions between Air Pollution and Obesity on Blood Pressure and Hypertension in Chinese Children. Epidemiology 2015, 26, 740–747. [Google Scholar] [CrossRef]
- Oldewage, T.; Egal, A.; Grobler, C. Lipid Profile, Hyperglycaemia, Systemic Inflammation and Anthropometry as Cardiovascular Risk Factors and Their Association with Dietary Intakes in Children from Rural Cofimvaba, Eastern Cape, South Africa. J. Consum. Sci. 2017, 2, 1–15. [Google Scholar]
- Psaltopoulou, T.; Hatzis, G. ScienceDirect Socioeconomic Status and Risk Factors for Cardiovascular Disease: Impact of Dietary Mediators. Hell. J. Cardiol. 2017, 58, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.; Williams, J.; Townsend, N.; Mikkelsen, B.; Roberts, N.; Foster, C.; Wickramasinghe, K. Socioeconomic Status and Non-Communicable Disease Behavioural Risk Factors in Low-Income and Lower-Middle-Income Countries: A Systematic Review. Lancet Glob. Health 2017, 5, e277–e289. [Google Scholar] [CrossRef] [PubMed]
- Kivimäki, M.; Vahtera, J.; Halonen, J.I.; Vineis, P.; Pentti, J.; Pahkala, K.; Rovio, S.; Viikari, J.; Juonala, M.; Ferrie, J.E.; et al. Neighbourhood Socioeconomic Disadvantage, Risk Factors, and Diabetes from Childhood to Middle Age in the Young Finns Study: A Cohort Study. Lancet Public Health 2018, 3, 365–373. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services, The Health Consequences of Smoking: 50 Years of Progress. A Report of the Surgeon General. Centers for Disease Control and Prevention Website. January 2014. Available online: https://www.cdc.gov/tobacco/sgr/50th-anniversary/index.htm (accessed on 30 May 2022).
- Hahad, O.; Prochaska, J.H.; Daiber, A.; Münzel, T. Review Article Environmental Noise-Induced Effects on Stress Hormones, Oxidative Stress, and Vascular Dysfunction: Key Factors in the Relationship between Cerebrocardiovascular and Psychological Disorders. Oxidative Med. Cell. Longev. 2019, 2019, 13. [Google Scholar] [CrossRef]
- Tibuakuu, M.; Michos, E.D.; Navas-Acien, A.; Jones, M.R. Air Pollution and Cardiovascular Disease: A Focus on Vulnerable Populations Worldwide. Curr. Epidemiol. Rep. 2018, 5, 370–378. [Google Scholar] [CrossRef]
- Hartel, T.C.; Turawa, E.B.; Oelofse, A.; De Smidt, J.J.A. Effect of Maternal Cigarette Smoking and Alcohol Consumption during Pregnancy on Birth Weight and Cardiometabolic Risk Factors in Infants, Children and Adolescents: A Systematic Review Protocol. BMJ Open 2022, 12, e061811. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hartel, T.C.; Oelofse, A.; De Smidt, J.J.A. Vascular Effects, Potential Pathways and Mediators of Fetal Exposure to Alcohol and Cigarette Smoking during Pregnancy: A Narrative Review. Int. J. Environ. Res. Public Health 2023, 20, 6398. https://doi.org/10.3390/ijerph20146398
Hartel TC, Oelofse A, De Smidt JJA. Vascular Effects, Potential Pathways and Mediators of Fetal Exposure to Alcohol and Cigarette Smoking during Pregnancy: A Narrative Review. International Journal of Environmental Research and Public Health. 2023; 20(14):6398. https://doi.org/10.3390/ijerph20146398
Chicago/Turabian StyleHartel, Tammy C., André Oelofse, and Juléy J. A. De Smidt. 2023. "Vascular Effects, Potential Pathways and Mediators of Fetal Exposure to Alcohol and Cigarette Smoking during Pregnancy: A Narrative Review" International Journal of Environmental Research and Public Health 20, no. 14: 6398. https://doi.org/10.3390/ijerph20146398
APA StyleHartel, T. C., Oelofse, A., & De Smidt, J. J. A. (2023). Vascular Effects, Potential Pathways and Mediators of Fetal Exposure to Alcohol and Cigarette Smoking during Pregnancy: A Narrative Review. International Journal of Environmental Research and Public Health, 20(14), 6398. https://doi.org/10.3390/ijerph20146398





