Self-Reported Cannabis Use and HIV Viral Control among Patients with HIV Engaged in Care: Results from a National Cohort Study
Abstract
1. Introduction
2. Methods
2.1. Study Design and Data Sources
2.2. Study Population
2.3. Study Measures
2.4. Statistical Analyses
3. Results
3.1. Eligible Participants
3.2. Baseline Participant Characteristics
3.3. Factors Associated with Cannabis Use: Bivariable Analyses
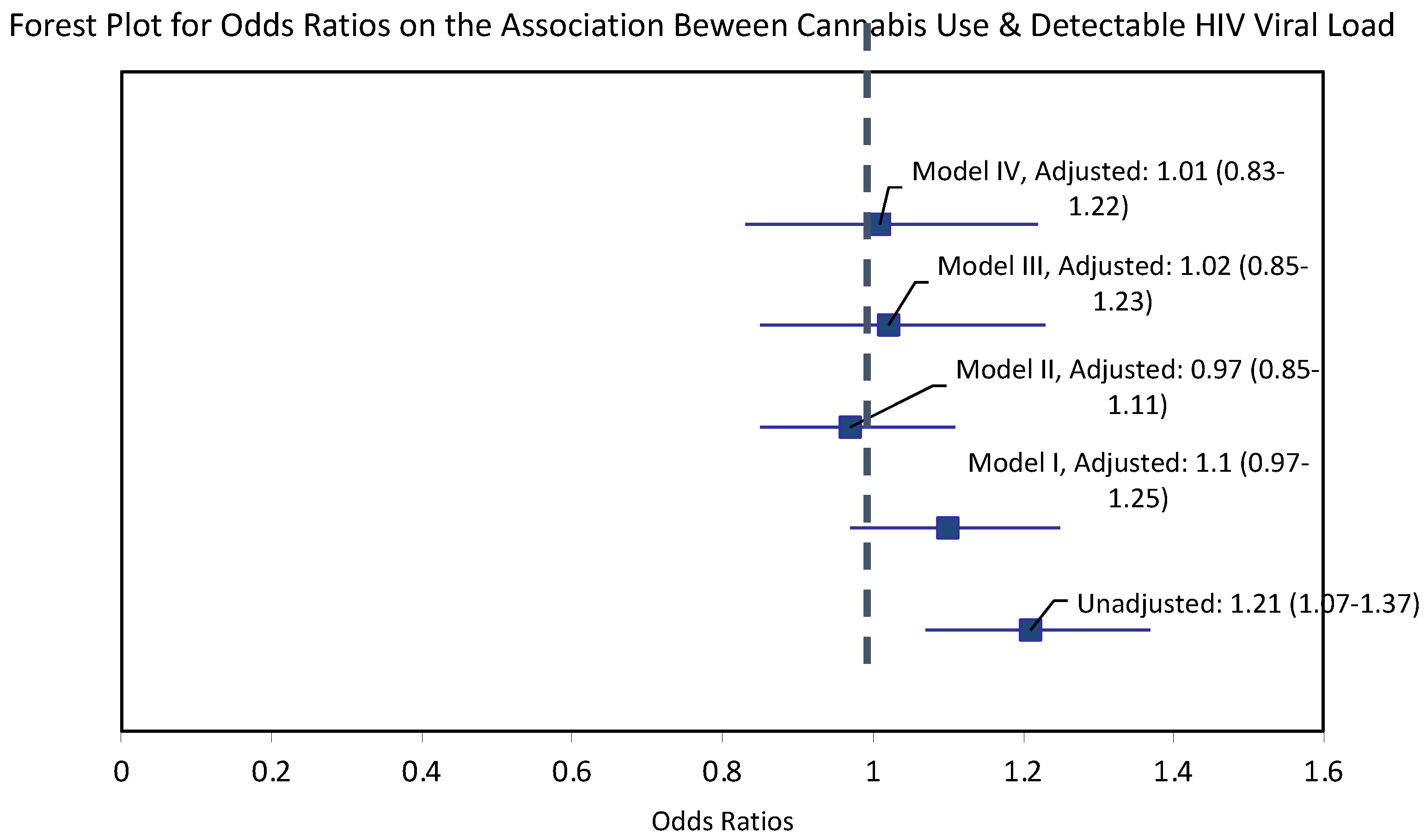
3.4. Association between Cannabis Use and HIV Viral Load
4. Discussion
4.1. Limitations
4.2. Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shiau, S.; Arpadi, S.M.; Yin, M.T.; Martins, S.S. Patterns of Drug Use and HIV Infection among Adults in a Nationally Representative Sample. Addict. Behav. 2017, 68, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Woolridge, E.; Barton, S.; Samuel, J.; Osorio, J.; Dougherty, A.; Holdcroft, A. Cannabis Use in HIV for Pain and Other Medical Symptoms. J. Pain Symptom Manag. 2005, 29, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Harris, G.E.; Dupuis, L.; Mugford, G.J.; Johnston, L.; Haase, D.; Page, G.; Haldane, H.; Harris, N.; Midodzi, W.K.; Dow, G. Patterns and Correlates of Cannabis Use among Individuals with HIV/AIDS in Maritime Canada. Can. J. Infect. Dis. Med. Microbiol. 2014, 25, e1–e7. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Torben, W.; Kenway, C.S.; Schiro, F.R.; Mohan, M. Longitudinal Examination of the Intestinal Lamina Propria Cellular Compartment of Simian Immunodeficiency Virus-Infected Rhesus Macaques Provides Broader and Deeper Insights into the Link between Aberrant MicroRNA Expression and Persistent Immune Activation. J. Virol. 2016, 90, 5003–5019. [Google Scholar] [CrossRef]
- Adams, J.W.; Bryant, K.J.; Edelman, E.J.; Fiellin, D.A.; Gaither, J.R.; Gordon, A.J.; Gordon, K.S.; Kraemer, K.L.; Mimiaga, M.J.; Operario, D.; et al. Association of Cannabis, Stimulant, and Alcohol Use with Mortality Prognosis Among HIV-Infected Men. AIDS Behav. 2018, 22, 1341–1351. [Google Scholar] [CrossRef]
- Keen, L.; Abbate, A.; Blanden, G.; Priddie, C.; Moeller, F.G.; Rathore, M. Confirmed Marijuana Use and Lymphocyte Count in Black People Living with HIV. Drug Alcohol Depend. 2019, 198, 112–115. [Google Scholar] [CrossRef]
- D’souza, G.; Matson, P.A.; Grady, C.D.; Nahvi, S.; Merenstein, D.; Weber, K.M.; Greenblatt, R.; Burian, P.; Wilson, T.E. Medicinal and Recreational Marijuana Use among HIV-Infected Women in the Women’s Interagency HIV Study (WIHS) Cohort, 1994–2010. J. Acquir. Immune Defic. Syndr. 1999 2012, 61, 618–626. [Google Scholar] [CrossRef]
- Kipp, A.M.; Rebeiro, P.F.; Shepherd, B.E.; Brinkley-Rubinstein, L.; Turner, M.; Bebawy, S.; Sterling, T.R.; Hulgan, T. Daily Marijuana Use Is Associated with Missed Clinic Appointments Among HIV-Infected Persons Engaged in HIV Care. AIDS Behav. 2017, 21, 1996–2004. [Google Scholar] [CrossRef]
- Montgomery, L.; Bagot, K.; Brown, J.L.; Haeny, A.M. The Association Between Marijuana Use and HIV Continuum of Care Outcomes: A Systematic Review. Curr. HIV/AIDS Rep. 2019, 16, 17–28. [Google Scholar] [CrossRef]
- Slawson, G.; Milloy, M.-J.; Balneaves, L.; Simo, A.; Guillemi, S.; Hogg, R.; Montaner, J.; Wood, E.; Kerr, T. High-Intensity Cannabis Use and Adherence to Antiretroviral Therapy Among People Who Use Illicit Drugs in a Canadian Setting. AIDS Behav. 2015, 19, 120–127. [Google Scholar] [CrossRef]
- Vidot, D.C.; Lerner, B.; Gonzalez, R. Cannabis Use, Medication Management and Adherence Among Persons Living with HIV. AIDS Behav. 2017, 21, 2005–2013. [Google Scholar] [CrossRef] [PubMed]
- Kuhns, L.M.; Hotton, A.L.; Garofalo, R.; Muldoon, A.L.; Jaffe, K.; Bouris, A.; Voisin, D.; Schneider, J. An Index of Multiple Psychosocial, Syndemic Conditions Is Associated with Antiretroviral Medication Adherence Among HIV-Positive Youth. AIDS Patient Care STDs 2016, 30, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Okafor, C.N.; Zhou, Z.; Burrell, L.E.; Kelso, N.E.; Whitehead, N.E.; Harman, J.S.; Cook, C.L.; Cook, R.L. Marijuana Use and Viral Suppression in Persons Receiving Medical Care for HIV-Infection. Am. J. Drug Alcohol Abuse 2017, 43, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Lake, S.; Kerr, T.; Capler, R.; Shoveller, J.; Montaner, J.; Milloy, M.-J. High-Intensity Cannabis Use and HIV Clinical Outcomes among HIV-Positive People Who Use Illicit Drugs in Vancouver, Canada. Int. J. Drug Policy 2017, 42, 63–70. [Google Scholar] [CrossRef]
- Lorenz, D.R.; Dutta, A.; Mukerji, S.S.; Holman, A.; Uno, H.; Gabuzda, D. Marijuana Use Impacts Midlife Cardiovascular Events in HIV-Infected Men. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2017, 65, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Bonn-Miller, M.O.; Oser, M.L.; Bucossi, M.M.; Trafton, J.A. Cannabis Use and HIV Antiretroviral Therapy Adherence and HIV-Related Symptoms. J. Behav. Med. 2014, 37, 1–10. [Google Scholar] [CrossRef]
- Lee, J.T.; Saag, L.A.; Kipp, A.M.; Logan, J.; Shepherd, B.E.; Koethe, J.R.; Turner, M.; Bebawy, S.; Sterling, T.R.; Hulgan, T. Self-Reported Cannabis Use and Changes in Body Mass Index, CD4 T-Cell Counts, and HIV-1 RNA Suppression in Treated Persons with HIV. AIDS Behav. 2020, 24, 1275–1280. [Google Scholar] [CrossRef]
- Gross, I.M.; Hosek, S.; Richards, M.H.; Fernandez, M.I. Predictors and Profiles of Antiretroviral Therapy Adherence Among African American Adolescents and Young Adult Males Living with HIV. AIDS Patient Care STDs 2016, 30, 324–338. [Google Scholar] [CrossRef]
- Zhang, Y.; Wilson, T.E.; Adedimeji, A.; Merenstein, D.; Milam, J.; Cohen, J.; Cohen, M.; Golub, E.T. The Impact of Substance Use on Adherence to Antiretroviral Therapy Among HIV-Infected Women in the United States. AIDS Behav. 2018, 22, 896–908. [Google Scholar] [CrossRef]
- Newville, H.; Berg, K.M.; Gonzalez, J.S. The Interaction of Active Substance Use, Depression, and Antiretroviral Adherence in Methadone Maintenance. Int. J. Behav. Med. 2015, 22, 214–222. [Google Scholar] [CrossRef]
- Newville, H.; Roley, J.; Sorensen, J.L. Prescription Medication Misuse among HIV-Infected Individuals Taking Antiretroviral Therapy. J. Subst. Abuse Treat. 2015, 48, 56–61. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hightow-Weidman, L.; LeGrand, S.; Choi, S.K.; Egger, J.; Hurt, C.B.; Muessig, K.E. Exploring the HIV Continuum of Care among Young Black MSM. PLoS ONE 2017, 12, e0179688. [Google Scholar] [CrossRef] [PubMed]
- Tucker, J.S.; Burnam, M.A.; Sherbourne, C.D.; Kung, F.-Y.; Gifford, A.L. Substance Use and Mental Health Correlates of Nonadherence to Antiretroviral Medications in a Sample of Patients with Human Immunodeficiency Virus Infection. Am. J. Med. 2003, 114, 573–580. [Google Scholar] [CrossRef]
- Wilson, K.J.; Doxanakis, A.; Fairley, C.K. Predictors for Non-Adherence to Antiretroviral Therapy. Sex. Health 2004, 1, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Pacek, L.R.; Towe, S.L.; Hobkirk, A.L.; Nash, D.; Goodwin, R.D. Frequency of Cannabis Use and Medical Cannabis Use Among Persons Living with HIV in the United States: Findings from a Nationally Representative Sample. AIDS Educ. Prev. 2018, 30, 169–181. [Google Scholar] [CrossRef]
- Slawek, D.E.; Arnsten, J.; Sohler, N.; Zhang, C.; Grossberg, R.; Stein, M.; Cunningham, C.O. Daily and Near-Daily Cannabis Use Is Associated with HIV Viral Load Suppression in People Living with HIV Who Use Cocaine. AIDS Care 2020, 33, 1350–1357. [Google Scholar] [CrossRef]
- Ellis, R.J.; Wilson, N.; Peterson, S. Cannabis and Inflammation in HIV: A Review of Human and Animal Studies. Viruses 2021, 13, 1521. [Google Scholar] [CrossRef]
- Molina, P.E.; Winsauer, P.; Zhang, P.; Walker, E.; Birke, L.; Amedee, A.; Stouwe, C.V.; Troxclair, D.; McGoey, R.; Varner, K.; et al. Cannabinoid Administration Attenuates the Progression of Simian Immunodeficiency Virus. AIDS Res. Hum. Retrovir. 2011, 27, 585–592. [Google Scholar] [CrossRef]
- Justice, A.C.; Dombrowski, E.; Conigliaro, J.; Fultz, S.L.; Gibson, D.; Madenwald, T.; Goulet, J.; Simberkoff, M.; Butt, A.A.; Rimland, D.; et al. Veterans Aging Cohort Study (VACS): Overview and Description. Med. Care 2006, 44, S13–S24. [Google Scholar] [CrossRef]
- Conigliaro, J.; Madenwald, T.; Bryant, K.; Braithwaite, S.; Gordon, A.; Fultz, S.L.; Maisto, S.; Samet, J.; Kraemer, K.; Cook, R.; et al. The Veterans Aging Cohort Study: Observational Studies of Alcohol Use, Abuse, and Outcomes among Human Immunodeficiency Virus-Infected Veterans. Alcohol. Clin. Exp. Res. 2004, 28, 313–321. [Google Scholar] [CrossRef]
- Tate, J.P.; Sterne, J.A.C.; Justice, A.C. Veterans Aging Cohort Study (VACS) and the Antiretroviral Therapy Cohort Collaboration (ART-CC) Albumin, White Blood Cell Count, and Body Mass Index Improve Discrimination of Mortality in HIV-Positive Individuals. AIDS 2019, 33, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Edelman, E.J.; Li, Y.; Barry, D.; Brennan Braden, J.; Crystal, S.; Kerns, R.D.; Gaither, J.R.; Gordon, K.S.; Manhapra, A.; Merlin, J.S.; et al. Trajectories of Self-Reported Opioid Use Among Patients with HIV Engaged in Care: Results from a National Cohort Study. J. Acquir. Immune Defic. Syndr. 1999 2020, 84, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Sohler, N.L.; Starrels, J.L.; Khalid, L.; Bachhuber, M.A.; Arnsten, J.H.; Nahvi, S.; Jost, J.; Cunningham, C.O. Cannabis Use Is Associated with Lower Odds of Prescription Opioid Analgesic Use Among HIV-Infected Individuals with Chronic Pain. Subst. Use Misuse 2018, 53, 1602–1607. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.; Degenhardt, L. Prevalence and Correlates of Cannabis Use in Developed and Developing Countries. Curr. Opin. Psychiatry 2007, 20, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Hart, L.G.; Larson, E.H.; Lishner, D.M. Rural Definitions for Health Policy and Research. Am. J. Public Health 2005, 95, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Greysen, S.R.; Horwitz, L.I.; Covinsky, K.E.; Gordon, K.; Ohl, M.E.; Justice, A.C. Does Social Isolation Predict Hospitalization and Mortality among HIV+ and Uninfected Older Veterans? J. Am. Geriatr. Soc. 2013, 61, 1456–1463. [Google Scholar] [CrossRef]
- Williams, E.C.; McGinnis, K.A.; Tate, J.P.; Matson, T.E.; Rubinsky, A.D.; Bobb, J.F.; Lapham, G.T.; Edelman, E.J.; Catz, S.L.; Satre, D.D.; et al. HIV Disease Severity Is Sensitive to Temporal Changes in Alcohol Use: A National Study of VA Patients with HIV. J. Acquir. Immune Defic. Syndr. 1999 2019, 81, 448–455. [Google Scholar] [CrossRef]
- Viswanathan, S.; Justice, A.C.; Alexander, G.C.; Brown, T.T.; Gandhi, N.R.; McNicholl, I.R.; Rimland, D.; Rodriguez-Barradas, M.C.; Jacobson, L.P. Adherence and HIV RNA Suppression in the Current Era of Highly Active Antiretroviral Therapy. J. Acquir. Immune Defic. Syndr. 1999 2015, 69, 493–498. [Google Scholar] [CrossRef]
- Williams, E.C.; McGinnis, K.A.; Rubinsky, A.D.; Matson, T.E.; Bobb, J.F.; Lapham, G.T.; Edelman, E.J.; Satre, D.D.; Catz, S.L.; Richards, J.E.; et al. Alcohol Use and Antiretroviral Adherence Among Patients Living with HIV: Is Change in Alcohol Use Associated with Change in Adherence? AIDS Behav. 2021, 25, 203–214. [Google Scholar] [CrossRef]
- Viswanathan, S.; Detels, R.; Mehta, S.H.; Macatangay, B.J.C.; Kirk, G.D.; Jacobson, L.P. Level of Adherence and HIV RNA Suppression in the Current Era of Highly Active Antiretroviral Therapy (HAART). AIDS Behav. 2015, 19, 601–611. [Google Scholar] [CrossRef]
- Park, L.S.; Tate, J.P.; Sigel, K.; Brown, S.T.; Crothers, K.; Gibert, C.; Goetz, M.B.; Rimland, D.; Rodriguez-Barradas, M.C.; Bedimo, R.J.; et al. Association of Viral Suppression with Lower AIDS-Defining and Non-AIDS-Defining Cancer Incidence in HIV-Infected Veterans: A Prospective Cohort Study. Ann. Intern. Med. 2018, 169, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Justice, A.C.; Holmes, W.; Gifford, A.L.; Rabeneck, L.; Zackin, R.; Sinclair, G.; Weissman, S.; Neidig, J.; Marcus, C.; Chesney, M.; et al. Development and Validation of a Self-Completed HIV Symptom Index. J. Clin. Epidemiol. 2001, 54, S77–S90. [Google Scholar] [CrossRef]
- Edelman, E.J.; Gordon, K.; Rodriguez-Barradas, M.C.; Justice, A.C. Vacs Project Team Patient-Reported Symptoms on the Antiretroviral Regimen Efavirenz/Emtricitabine/Tenofovir. AIDS Patient Care STDs 2012, 26, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B. The PHQ-9: Validity of a Brief Depression Severity Measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.W.; Patient Health Questionnaire Primary Care Study Group. Validation and Utility of a Self-Report Version of PRIME-MD: The PHQ Primary Care Study. JAMA 1999, 282, 1737–1744. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.; Kosinski, M.; Keller, S.D. A 12-Item Short-Form Health Survey: Construction of Scales and Preliminary Tests of Reliability and Validity. Med. Care 1996, 34, 220–233. [Google Scholar] [CrossRef]
- Bush, K.; Kivlahan, D.R.; McDonell, M.B.; Fihn, S.D.; Bradley, K.A. The AUDIT Alcohol Consumption Questions (AUDIT-C): An Effective Brief Screening Test for Problem Drinking. Ambulatory Care Quality Improvement Project (ACQUIP). Alcohol Use Disorders Identification Test. Arch. Intern. Med. 1998, 158, 1789–1795. [Google Scholar] [CrossRef]
- Barry, D.T.; Marshall, B.D.L.; Becker, W.C.; Gordon, A.J.; Crystal, S.; Kerns, R.D.; Gaither, J.R.; Gordon, K.S.; Justice, A.C.; Fiellin, D.A.; et al. Duration of Opioid Prescriptions Predicts Incident Nonmedical Use of Prescription Opioids among U.S. Veterans Receiving Medical Care. Drug Alcohol Depend. 2018, 191, 348–354. [Google Scholar] [CrossRef]
- Edelman, E.J.; Gordon, K.S.; Crothers, K.; Akgün, K.; Bryant, K.J.; Becker, W.C.; Gaither, J.R.; Gibert, C.L.; Gordon, A.J.; Marshall, B.D.L.; et al. Association of Prescribed Opioids with Increased Risk of Community-Acquired Pneumonia Among Patients with and without HIV. JAMA Intern. Med. 2019, 179, 297–304. [Google Scholar] [CrossRef]
- Dowell, D.; Haegerich, T.M.; Chou, R. CDC Guideline for Prescribing Opioids for Chronic Pain—United States, 2016. MMWR Morb. Mortal. Wkly. Rep. Recomm. Rep. 2016, 65, 1–49. [Google Scholar] [CrossRef]
- Finnerup, N.B. Nonnarcotic Methods of Pain Management. N. Engl. J. Med. 2019, 380, 2440–2448. [Google Scholar] [CrossRef]
- Jones, J.D.; Mogali, S.; Comer, S.D. Polydrug Abuse: A Review of Opioid and Benzodiazepine Combination Use. Drug Alcohol Depend. 2012, 125, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Evoy, K.E.; Morrison, M.D.; Saklad, S.R. Abuse and Misuse of Pregabalin and Gabapentin. Drugs 2017, 77, 403–426. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, M.; Freitas, D.; Bangsberg, D.R.; Miaskowski, C.; Kushel, M.B. Non-Medical Use of Non-Opioid Psychotherapeutic Medications in a Community-Based Cohort of HIV-Infected Indigent Adults. Drug Alcohol Depend. 2014, 143, 263–267. [Google Scholar] [CrossRef]
- Parthipan, A.; Banerjee, I.; Humphreys, K.; Asch, S.M.; Curtin, C.; Carroll, I.; Hernandez-Boussard, T. Predicting Inadequate Postoperative Pain Management in Depressed Patients: A Machine Learning Approach. PLoS ONE 2019, 14, e0210575. [Google Scholar] [CrossRef] [PubMed]
- Mason, B.J.; Crean, R.; Goodell, V.; Light, J.M.; Quello, S.; Shadan, F.; Buffkins, K.; Kyle, M.; Adusumalli, M.; Begovic, A.; et al. A Proof-of-Concept Randomized Controlled Study of Gabapentin: Effects on Cannabis Use, withdrawal and Executive Function Deficits in Cannabis-Dependent Adults. Neuropsychopharmacology 2012, 37, 1689–1698. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.-Y.; Zeger, S.L. Longitudinal Data Analysis Using Generalized Linear Models. Biometrika 1986, 73, 13–22. [Google Scholar] [CrossRef]
- McGowan, C.C.; Weinstein, D.D.; Samenow, C.P.; Stinnette, S.E.; Barkanic, G.; Rebeiro, P.F.; Sterling, T.R.; Moore, R.D.; Hulgan, T. Drug Use and Receipt of Highly Active Antiretroviral Therapy among HIV-Infected Persons in Two U.S. Clinic Cohorts. PLoS ONE 2011, 6, e18462. [Google Scholar] [CrossRef]
- Cescon, A.M.; Cooper, C.; Chan, K.; Palmer, A.K.; Klein, M.B.; Machouf, N.; Loutfy, M.R.; Raboud, J.; Rachlis, A.; Ding, E.; et al. Factors Associated with Virological Suppression among HIV-Positive Individuals on Highly Active Antiretroviral Therapy in a Multi-Site Canadian Cohort. HIV Med. 2011, 12, 352–360. [Google Scholar] [CrossRef]
- Obel, N.; Omland, L.H.; Kronborg, G.; Larsen, C.S.; Pedersen, C.; Pedersen, G.; Sørensen, H.T.; Gerstoft, J. Impact of Non-HIV and HIV Risk Factors on Survival in HIV-Infected Patients on HAART: A Population-Based Nationwide Cohort Study. PLoS ONE 2011, 6, e22698. [Google Scholar] [CrossRef]
- Milloy, M.-J.; Marshall, B.; Kerr, T.; Richardson, L.; Hogg, R.; Guillemi, S.; Montaner, J.S.G.; Wood, E. High-Intensity Cannabis Use Associated with Lower Plasma Human Immunodeficiency Virus-1 RNA Viral Load among Recently Infected People Who Use Injection Drugs. Drug Alcohol Rev. 2015, 34, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Chou, F.Y.; Armstrong, H.L.; Wang, L.; Bacani, N.; Lachowsky, N.J.; Patterson, T.L.; Walsh, Z.; Olarewaju, G.; Card, K.G.; Roth, E.A.; et al. A Longitudinal Analysis of Cannabis Use and Mental Health Symptoms among Gay, Bisexual, and Other Men Who Have Sex with Men in Vancouver, Canada. J. Affect. Disord. 2019, 247, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Parent, N.; Ferlatte, O.; Milloy, M.-J.; Fast, D.; Knight, R. The Sexualised Use of Cannabis among Young Sexual Minority Men: “I’m Actually Enjoying This for the First Time”. Cult. Health Sex. 2021, 23, 883–898. [Google Scholar] [CrossRef] [PubMed]
- Braithwaite, R.S.; Bryant, K.J. Influence of Alcohol Consumption on Adherence to and Toxicity of Antiretroviral Therapy and Survival. Alcohol Res. Health 2010, 33, 280–287. [Google Scholar]
- Winsauer, P.J.; Molina, P.E.; Amedee, A.M.; Filipeanu, C.M.; McGoey, R.R.; Troxclair, D.A.; Walker, E.M.; Birke, L.L.; Stouwe, C.V.; Howard, J.M.; et al. Tolerance to Chronic Delta-9-Tetrahydrocannabinol (Δ9-THC) in Rhesus Macaques Infected with Simian Immunodeficiency Virus. Exp. Clin. Psychopharmacol. 2011, 19, 154–172. [Google Scholar] [CrossRef] [PubMed]
- Rock, R.B.; Gekker, G.; Hu, S.; Sheng, W.S.; Cabral, G.A.; Martin, B.R.; Peterson, P.K. WIN55,212-2-Mediated Inhibition of HIV-1 Expression in Microglial Cells: Involvement of Cannabinoid Receptors. J. Neuroimmune Pharmacol. 2007, 2, 178–183. [Google Scholar] [CrossRef]
- Peterson, P.K.; Gekker, G.; Hu, S.; Cabral, G.; Lokensgard, J.R. Cannabinoids and Morphine Differentially Affect HIV-1 Expression in CD4(+) Lymphocyte and Microglial Cell Cultures. J. Neuroimmunol. 2004, 147, 123–126. [Google Scholar] [CrossRef]
- Darke, S. Self-Report among Injecting Drug Users: A Review. Drug Alcohol Depend. 1998, 51, 253–263; discussion 267–268. [Google Scholar] [CrossRef]
- Johnson, M.; Fisher, D.; Montoya, I.; Booth, R.; Rhodes, F.; Andersen, M.; Zhuo, Z.; Williams, M. Reliability and Validity of Not-in-Treatment Drug Users’ Follow-Up Self-Reports. AIDS Behav. 2000, 4, 373–380. [Google Scholar] [CrossRef]
- Smith, P.C.; Schmidt, S.M.; Allensworth-Davies, D.; Saitz, R. A Single-Question Screening Test for Drug Use in Primary Care. Arch. Intern. Med. 2010, 170, 1155–1160. [Google Scholar] [CrossRef]
- Volkow, N.D.; Weiss, S.R.B. Importance of a Standard Unit Dose for Cannabis Research. Addiction 2020, 115, 1219–1221. [Google Scholar] [CrossRef] [PubMed]
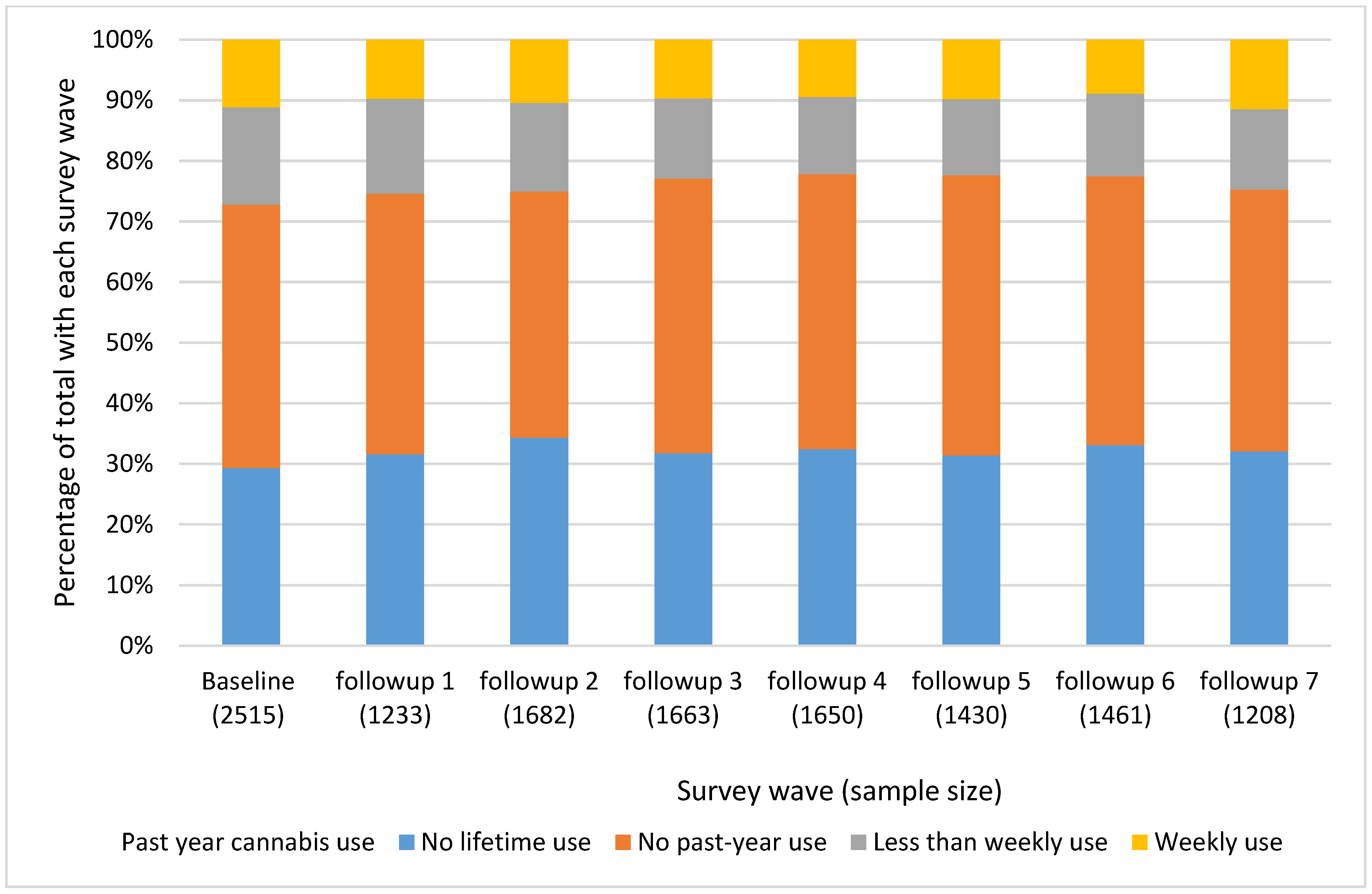
| Characteristic | Overall (n = 2515) | No Past-Year Use (n = 1830, 72.8%) | Past-Year Use (n = 685, 27.2%) | Global p-Value |
|---|---|---|---|---|
| Demographics | ||||
| Age, mean (SD) | 50.1 (8.8) | 50.6 (9.0) | 48.7 (8.2) | <0.0001 |
| Gender, n (%) | 0.494 | |||
| Male | 2447 (97.3) | 1783 (97.4) | 664 (96.9) | |
| Female | 68 (2.7) | 47 (2.6) | 21 (3.1) | |
| Race/ethnicity, n (%) | 0.160 | |||
| Non-Hispanic White | 524 (20.8) | 363 (19.8) | 161 (23.5) | |
| Non-Hispanic Black | 1646 (65.5) | 1214 (66.3) | 432 (63.1) | |
| Hispanic (any race) | 247 (9.8) | 185 (10.1) | 62 (9.1) | |
| Other (multiple race or unknown) | 98 (3.9) | 68 (3.7) | 30 (4.4) | |
| Education, n (%) | 0.077 | |||
| High school or less | 987 (39.7) | 736 (40.8) | 251 (36.9) | |
| Some college or more | 1500 (60.3) | 1070 (59.3) | 430 (63.1) | |
| Marital status, n (%) | 0.159 | |||
| Never married | 610 (24.6) | 440 (24.4) | 170 (25.2) | |
| Married/living with a partner | 981 (39.6) | 734 (40.7) | 247 (36.7) | |
| Divorced/widowed | 885 (35.7) | 628 (34.9) | 257 (38.1) | |
| Housing instability ever, n (%) | 982 (39.3) | 680 (37.4) | 302 (44.3) | 0.002 |
| Location of residence, n (%) | 0.512 | |||
| Urban | 2368 (95.0) | 1726 (95.2) | 642 (94.3) | |
| Suburban | 70 (2.8) | 50 (2.8) | 20 (2.9) | |
| Rural | 56 (2.3) | 37 (2.0) | 19 (2.8) | |
| Annual income, n (%) | 0.013 | |||
| <$11,999 | 1213 (49.8) | 859 (48.6) | 354 (53.2) | |
| $12,000–$49,999 | 1038 (42.6) | 761 (43.0) | 277 (41.6) | |
| ≥$50,000 | 184 (7.6) | 149 (8.4) | 35 (5.3) | |
| Social Isolation Score | 0.032 | |||
| <4 | 680 (27.0) | 516 (28.2) | 164 (23.9) | |
| ≥4 | 1835 (73.0) | 1314 (71.8) | 521 (76.1) | |
| HIV-related factors | ||||
| CD4 cell count, cells/mm3, median (IQR) | 374 (228, 568) | 377 (231, 578) | 365 (219, 549) | 0.295 |
| HIV viral load <500 copies/mL, n (%) | 1690 (67.2) | 1259 (68.8) | 431 (62.9) | 0.005 |
| VACS Index 2.0 score, median (IQR) | 56 (46, 66) | 56 (46, 66) | 56 (46, 67) | 0.109 |
| ART adherent, n (%) | 1551 (61.7) | 1164 (63.6) | 387 (56.5) | 0.001 |
| Other health conditions and status, n (%) | ||||
| HCV co-infection | 929 (36.9) | 664 (36.3) | 265 (38.7) | 0.267 |
| Any cancer | 524 (20.8) | 375 (20.5) | 149 (21.8) | 0.489 |
| Anxiety symptoms | 905 (37.1) | 619 (34.9) | 286 (42.9) | 0.0003 |
| Depressive symptoms | 534 (21.5) | 347 (19.2) | 187 (27.5) | <0.0001 |
| Pain interference | 830 (33.3) | 582 (32.1) | 248 (36.4) | 0.043 |
| Other substance use, n (%) | ||||
| Smokes cigarettes | 1915 (76.1) | 1337 (73.1) | 578 (84.4) | <0.0001 |
| Unhealthy alcohol use | 876 (34.8) | 599 (32.7) | 277 (40.4) | 0.0003 |
| Past-year stimulants or cocaine | 538 (21.4) | 252 (13.8) | 286 (41.8) | <0.0001 |
| Prescribed opioid receipt, n (%) | 0.001 | |||
| No opioid receipt | 1802 (71.7) | 1346 (73.6) | 456 (66.6) | |
| Short-term + low dose | 449 (17.9) | 308 (16.8) | 141 (20.6) | |
| Short-term + high dose | 40 (1.6) | 33 (1.8) | 7 (1.0) | |
| Long-term + low dose | 159 (6.3) | 100 (5.5) | 59 (8.6) | |
| Long-term + high dose | 65 (2.6) | 43 (2.4) | 22 (3.2) | |
| Prescribed benzodiazepine | 0.336 | |||
| None | 2147 (85.4) | 1573 (86.0) | 574 (83.8) | |
| Low dose | 269 (10.7) | 190 (10.4) | 79 (11.5) | |
| High dose | 99 (3.9) | 67 (3.7) | 32 (4.7) | |
| Prescribed gabapentin | 0.320 | |||
| None | 2240 (89.1) | 1639 (89.6) | 601 (87.7) | |
| Low dose | 127 (5.1) | 91 (5.0) | 36 (5.3) | |
| High dose | 148 (5.9) | 100 (5.5) | 48 (7.0) | |
| Prescribed antidepressant | 0.017 | |||
| None | 1557 (61.9) | 1161 (63.4) | 396 (57.8) | |
| Short-term | 314 (12.5) | 227 (12.4) | 87 (12.7) | |
| Long-term | 644 (25.6) | 442 (24.2) | 202 (29.5) | |
| Site | 0.193 | |||
| Atlanta | 403 (16.0) | 299 (16.3) | 104 (15.2) | |
| Bronx | 258 (10.3) | 190 (10.4) | 68 (9.9) | |
| Houston | 335 (13.3) | 242 (13.2) | 93 (13.6) | |
| Los Angeles | 334 (13.3) | 225 (12.3) | 109 (15.9) | |
| New York | 399 (15.9) | 295 (16.1) | 104 (15.2) | |
| Baltimore | 282 (11.2) | 214 (11.7) | 68 (9.9) | |
| Washington DC | 408 (16.2) | 302 (16.5) | 106 (15.5) | |
| Pittsburgh | 96 (3.8) | 63 (3.4) | 33 (4.8) | |
| Calendar year | 0.084 | |||
| 2002–2006 | 781 (31.5) | 583 (32.3) | 198 (29.3) | |
| 2007–2011 | 976 (39.3) | 717 (39.7) | 259 (38.3) | |
| 2012–2017 | 724 (29.2) | 505 (28.0) | 219 (32.4) | |
| Average follow-up years, mean (SD) | 7.0 (3.7) | 6.9 (3.7) | 7.3 (3.7) | 0.030 |
| Died during study | 904 (35.9) | 666 (36.4) | 238 (34.7) | 0.443 |
| Unadjusted Odds Ratios [95% CI] | Model I, Adjusted Odds Ratios [95% CI] | Model II, Adjusted Odds Ratios [95% CI] | Model III, Adjusted Odds Ratios [95% CI] | Model IV, Adjusted Odds Ratios [95% CI] | |
|---|---|---|---|---|---|
| Past-year cannabis | 1.21 (1.07–1.37) | 1.10 (0.97–1.25) | 0.97 (0.85–1.11) | 1.02 (0.85–1.23) | 1.01 (0.83–1.22) |
| Age | 0.95 (0.94, 0.95) | 0.95 (0.94–0.95) | 0.96 (0.95–0.97) | 0.96 (0.95–0.97) | |
| Race/ethnicity | |||||
| Non-Hispanic White | Ref | Ref | Ref | Ref | |
| Non-Hispanic Black | 1.21 (1.02–1.43) | 1.15 (0.97–1.37) | 1.21 (0.97–1.50) | 1.12 (0.90–1.40) | |
| Hispanic (any race) | 0.85 (0.65–1.10) | 0.82 (0.64–1.07) | 0.93 (0.67–1.29) | 0.93 (0.67–1.30) | |
| Other (multiple race or unknown) | 1.00 (0.69–1.44) | 0.96 (0.66–1.40) | 1.19 (0.76–1.86) | 1.14 (0.72–1.79) | |
| Sex | |||||
| Male | Ref | Ref | Ref | Ref | |
| Female | 0.62 (0.40–0.94) | 0.62 (0.41–0.95) | 0.64 (0.36–1.14) | 0.56 (0.32–0.97) | |
| Unhealthy alcohol use | |||||
| Yes | 1.14 (1.01–1.29) | 0.83 (0.70–0.98) | 0.78 (0.66–0.93) | ||
| No | Ref | Ref | Ref | ||
| Past-year stimulant/cocaine use | |||||
| Yes | 1.56 (1.35–1.79) | 1.45 (1.18–1.79) | 1.37 (1.11–1.69) | ||
| No | Ref | Ref | Ref | ||
| Opioid Use | |||||
| No opioid receipt | Ref | Ref | Ref | ||
| Short-term + low dose | 1.02 (0.90–1.15) | 0.98 (0.81–1.20) | 0.97 (0.80–1.19) | ||
| Short-term + high dose | 1.04 (0.67–1.60) | 1.86 (0.99–3.49) | 1.72 (0.92–3.24) | ||
| Long-term + low dose | 1.21 (0.99–1.49) | 1.27 (0.93–1.73) | 1.25 (0.90–1.72) | ||
| Long-term + high dose | 0.97 (0.70–1.35) | 0.82 (0.50–1.35) | 0.85 (0.52–1.40) | ||
| Marital Status | |||||
| Never married | Ref | Ref | |||
| Married/living with a partner | 1.12 (0.88–1.43) | 1.10 (0.86–1.42) | |||
| Divorced/widowed | 1.03 (0.80–1.33) | 1.01 (0.78–1.30) | |||
| Homeless | |||||
| Yes | 1.01 (0.84–1.22) | 0.98 (0.81–1.18) | |||
| No | Ref | Ref | |||
| Income | |||||
| <$11,999 | Ref | Ref | |||
| $12,000–$49,999 | 1.08 (0.90–1.30) | 1.07 (0.89–1.29) | |||
| ≥$50,000 | 1.04 (0.74–1.47) | 1.05 (0.75–1.49) | |||
| Social score | |||||
| <4 | Ref | Ref | |||
| ≥4 | 1.01 (0.81–1.26) | 1.03 (0.82–1.29) | |||
| HCV | |||||
| Yes | 0.98 (0.82–1.17) | 0.93 (0.78–1.12) | |||
| No | Ref | Ref | |||
| Anxiety | |||||
| Yes | 1.04 (0.86–1.27) | 1.02 (0.84–1.24) | |||
| No | Ref | Ref | |||
| Depression | |||||
| Yes | 1.13 (0.92–1.40) | 1.07 (0.86–1.32) | |||
| No | Ref | Ref | |||
| Smoking | |||||
| Yes | 0.81 (0.66–0.99) | 0.83 (0.67–1.01) | |||
| No | Ref | Ref | |||
| Antidepressant | |||||
| None | Ref | Ref | |||
| Short-term | 1.24 (0.97–1.57) | 1.23 (0.96–1.58) | |||
| Long-term | 1.13 (0.92–1.38) | 1.27 (1.03–1.55) | |||
| ART adherence | |||||
| Yes | 0.41 (0.35–0.48) | ||||
| No | Ref |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahji, A.; Li, Y.; Vickers-Smith, R.; Crystal, S.; Kerns, R.D.; Gordon, K.S.; Macmadu, A.; Skanderson, M.; So-Armah, K.; Sung, M.L.; et al. Self-Reported Cannabis Use and HIV Viral Control among Patients with HIV Engaged in Care: Results from a National Cohort Study. Int. J. Environ. Res. Public Health 2022, 19, 5649. https://doi.org/10.3390/ijerph19095649
Bahji A, Li Y, Vickers-Smith R, Crystal S, Kerns RD, Gordon KS, Macmadu A, Skanderson M, So-Armah K, Sung ML, et al. Self-Reported Cannabis Use and HIV Viral Control among Patients with HIV Engaged in Care: Results from a National Cohort Study. International Journal of Environmental Research and Public Health. 2022; 19(9):5649. https://doi.org/10.3390/ijerph19095649
Chicago/Turabian StyleBahji, Anees, Yu Li, Rachel Vickers-Smith, Stephen Crystal, Robert D. Kerns, Kirsha S. Gordon, Alexandria Macmadu, Melissa Skanderson, Kaku So-Armah, Minhee L. Sung, and et al. 2022. "Self-Reported Cannabis Use and HIV Viral Control among Patients with HIV Engaged in Care: Results from a National Cohort Study" International Journal of Environmental Research and Public Health 19, no. 9: 5649. https://doi.org/10.3390/ijerph19095649
APA StyleBahji, A., Li, Y., Vickers-Smith, R., Crystal, S., Kerns, R. D., Gordon, K. S., Macmadu, A., Skanderson, M., So-Armah, K., Sung, M. L., Bhondoekhan, F., Marshall, B. D. L., & Edelman, E. J. (2022). Self-Reported Cannabis Use and HIV Viral Control among Patients with HIV Engaged in Care: Results from a National Cohort Study. International Journal of Environmental Research and Public Health, 19(9), 5649. https://doi.org/10.3390/ijerph19095649






