OSA and Chronic Respiratory Disease: Mechanisms and Epidemiology
Abstract
:1. Introduction
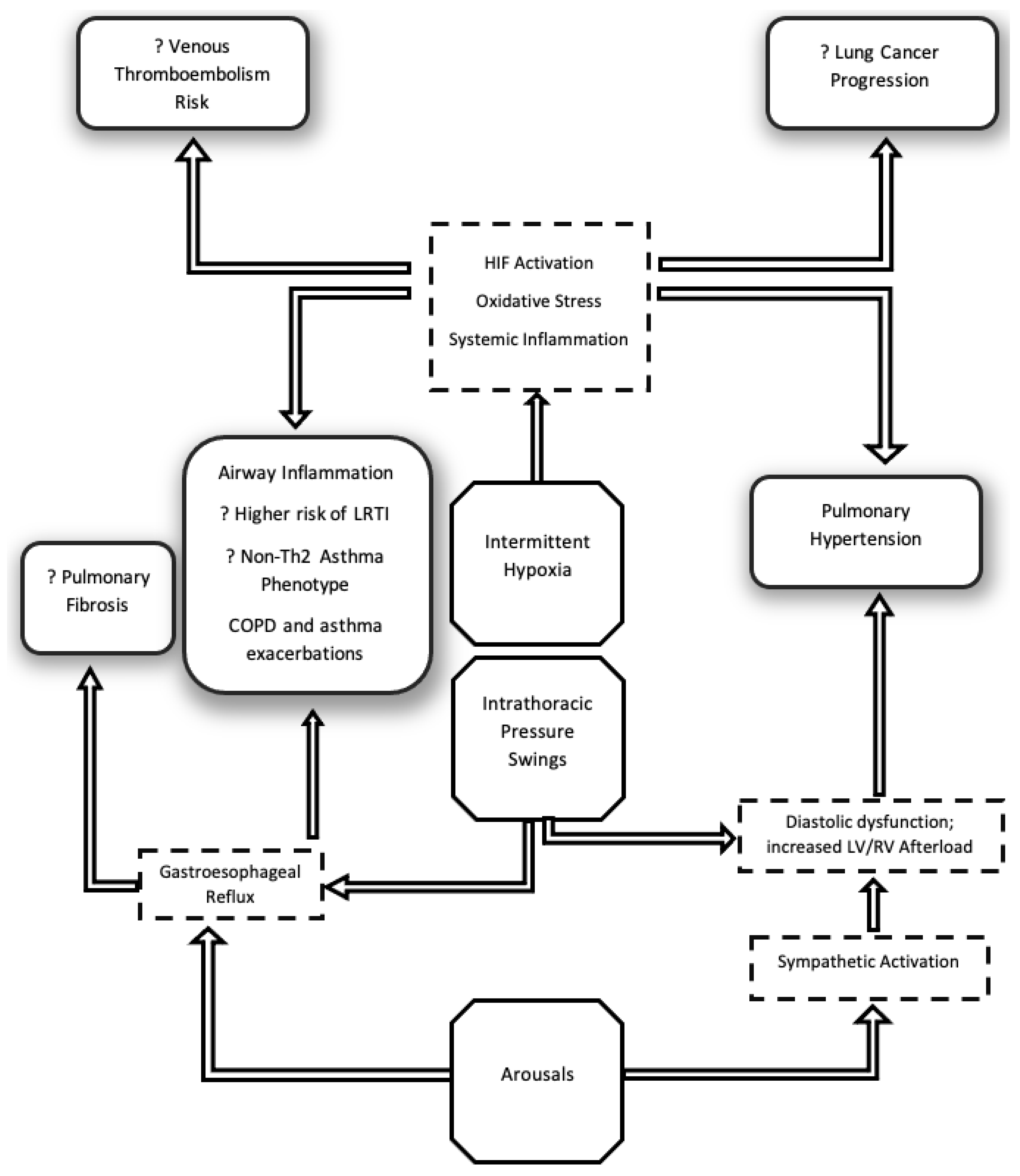
2. Pathophysiological Considerations
3. OSA and Asthma
4. OSA and Other Airway Disorders
5. OSA-COPD Overlap Syndrome
6. OSA and Hypoventilation Syndromes
7. OSA and Refractory Chronic Cough
8. OSA and Interstitial Lung Disease
9. OSA and Sarcoidosis
10. OSA and Pulmonary Vascular Disease and Venous Thromboembolism
11. OSA and Lung Cancer
12. Future Directions for Research
13. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lyons, M.M.; Bhatt, N.Y.; Pack, A.I.; Magalang, U.J. Global burden of sleep-disordered breathing and its implications. Respirology 2020, 25, 690–702. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Sundar, K.M. Evaluation and Management of Adults with Obstructive Sleep Apnea Syndrome. Lung 2021, 199, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Kendzerska, T.; Gershon, A.S.; Hawker, G.; Leung, R.S.; Tomlinson, G. Obstructive Sleep Apnea and Risk of Cardiovascular Events and All-Cause Mortality: A Decade-Long Historical Cohort Study. PLoS Med. 2014, 11, e1001599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomas, R.J.; Chen, S.; Eden, U.T.; Prerau, M.J. Quantifying statistical uncertainty in metrics of sleep disordered breathing. Sleep Med. 2020, 65, 161–169. [Google Scholar] [CrossRef]
- Punjabi, N.M.; Patil, S.; Crainiceanu, C.; Aurora, R.N. Variability and Misclassification of Sleep Apnea Severity Based on Multi-Night Testing. Chest 2020, 158, 365–373. [Google Scholar] [CrossRef]
- Lechat, B.; Naik, G.; Reynolds, A.; Aishah, A.; Scott, H.; Loffler, K.A.; Vakulin, A.; Escourrou, P.; McEvoy, R.D.; Adams, R.J.; et al. Multinight Prevalence, Variability, and Diagnostic Misclassification of Obstructive Sleep Apnea. Am. J. Respir. Crit. Care Med. 2022, 205, 563–569. [Google Scholar] [CrossRef]
- Bikov, A.; Losonczy, G.; Kunos, L. Role of lung volume and airway inflammation in obstructive sleep apnea. Respir. Investig. 2017, 55, 326–333. [Google Scholar] [CrossRef] [Green Version]
- Tuleta, I.; Stöckigt, F.; Juergens, U.R.; Pizarro, C.; Schrickel, J.W.; Kristiansen, G.; Nickenig, G.; Skowasch, D. Intermittent Hypoxia Contributes to the Lung Damage by Increased Oxidative Stress, Inflammation, and Disbalance in Protease/Antiprotease System. Lung 2016, 194, 1015–1020. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, K.; Liu, K.; Li, Z.; Yang, J.; Dong, Y.; Nie, M.; Chen, J.; Ruan, Y.; Kang, J. Obstructive sleep apnea exacerbates airway inflammation in patients with chronic obstructive pulmonary disease. Sleep Med. 2015, 16, 1123–1130. [Google Scholar] [CrossRef]
- Shukla, S.D.; Walters, E.H.; Simpson, J.L.; Keely, S.; Wark, P.A.; O’Toole, R.; Hansbro, P.M. Hypoxia-inducible factor and bacterial infections in chronic obstructive pulmonary disease. Respirology 2020, 25, 53–63. [Google Scholar] [CrossRef]
- Bin Kim, J.; Seo, B.S.; Kim, J.H. Effect of arousal on sympathetic overactivity in patients with obstructive sleep apnea. Sleep Med. 2019, 62, 86–91. [Google Scholar] [CrossRef]
- Adir, Y.; Humbert, M.; Chaouat, A. Sleep-related breathing disorders and pulmonary hypertension. Eur. Respir. J. 2021, 57, 2002258. [Google Scholar] [CrossRef] [PubMed]
- Zeidler, M.R.; Martin, J.L.; Kleerup, E.C.; Schneider, H.; Mitchell, M.N.; Hansel, N.N.; Sundar, K.; Schotland, H.; Basner, R.C.; Wells, J.M.; et al. Sleep disruption as a predictor of quality of life among patients in the subpopulations and intermediate outcome measures in COPD study (SPIROMICS). Sleep 2018, 41, zsy044. [Google Scholar] [CrossRef] [PubMed]
- Troy, L.K.; Corte, T.J. Sleep disordered breathing in interstitial lung disease: A review. World J. Clin. Cases 2014, 2, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Stuckless, T.J.R.; Vermeulen, T.D.; Brown, C.V.; Boulet, L.M.; Shafer, B.M.; Wakeham, D.J.; Steinback, C.D.; Ayas, N.T.; Floras, J.S.; Foster, G.E. Acute intermittent hypercapnic hypoxia and sympathetic neurovascular transduction in men. J. Physiol. 2020, 598, 473–487. [Google Scholar] [CrossRef]
- Díaz-García, E.; García-Tovar, S.; Alfaro, E.; Jaureguizar, A.; Casitas, R.; Sánchez-Sánchez, B.; Zamarrón, E.; Fernández-Lahera, J.; López-Collazo, E.; Cubillos-Zapata, C.; et al. Inflammasome Activation: A Keystone of Proinflammatory Response in Obstructive Sleep Apnea. Am. J. Respir. Crit. Care Med. 2022. [Google Scholar] [CrossRef]
- Tregub, P.P.; Malinovskaya, N.A.; Morgun, A.V.; Osipova, E.D.; Kulikov, V.P.; Kuzovkov, D.A.; Kovzelev, P.D. Hypercapnia potentiates HIF-1α activation in the brain of rats exposed to intermittent hypoxia. Respir. Physiol. Neurobiol. 2020, 278, 103442. [Google Scholar] [CrossRef]
- Leslie, K.O. Idiopathic Pulmonary Fibrosis May Be a Disease of Recurrent, Tractional Injury to the Periphery of the Aging Lung: A Unifying Hypothesis Regarding Etiology and Pathogenesis. Arch. Pathol. Lab. Med. 2012, 136, 591–600. [Google Scholar] [CrossRef] [Green Version]
- Reddel, H.K.; Bacharier, L.B.; Bateman, E.D.; Brightling, C.E.; Brusselle, G.G.; Buhl, R.; Cruz, A.A.; Duijts, L.; Drazen, J.M.; FitzGerald, J.M.; et al. Global Initiative for Asthma Strategy 2021: Executive Summary and Rationale for Key Changes. Am. J. Respir. Crit. Care Med. 2022, 205, 17–35. [Google Scholar] [CrossRef]
- Kuruvilla, M.E.; Lee, F.E.-H.; Lee, G.B. Understanding Asthma Phenotypes, Endotypes, and Mechanisms of Disease. Clin. Rev. Allergy Immunol. 2019, 56, 219–233. [Google Scholar] [CrossRef]
- Teodorescu, M.; Broytman, O.; Curran-Everett, D.; Sorkness, R.L.; Crisafi, G.M.; Bleecker, E.R.; Erzurum, S.C.; Gaston, B.M.; Wenzel, S.E.; Jarjour, N.N.; et al. Obstructive Sleep Apnea Risk, Asthma Burden, and Lower Airway Inflammation in Adults in the Severe Asthma Research Program (SARP) II. J. Allergy Clin. Immunol. Pract. 2015, 3, 566–575.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, B.; Nyenhuis, S.M.; Weaver, T.E. Obstructive sleep apnea and asthma: Associations and treatment implications. Sleep Med. Rev. 2014, 18, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.-L.; Qin, Z.; Shen, H.; Jin, H.-Y.; Wang, W.; Wang, Z.-F. Association of Obstructive Sleep Apnea with Asthma: A Meta-Analysis. Sci. Rep. 2017, 7, 4088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teodorescu, M.; Barnet, J.H.; Hagen, E.W.; Palta, M.; Young, T.B.; Peppard, P.E. Association Between Asthma and Risk of Developing Obstructive Sleep Apnea. JAMA J. Am. Med. Assoc. 2015, 313, 156–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, B.; Nyenhuis, S.M.; Imayama, I.; Siddiqi, A.; Teodorescu, M. Asthma and Obstructive Sleep Apnea Overlap: What Has the Evidence Taught Us? Am. J. Respir. Crit. Care Med. 2020, 201, 1345–1357. [Google Scholar] [CrossRef]
- Alkhalil, M.; Schulman, E.; Getsy, J. Obstructive sleep apnea syndrome and asthma: What are the links? J. Clin. Sleep Med. 2009, 5, 71–78. [Google Scholar] [CrossRef] [Green Version]
- Morrison, J.F.J.; Pearson, S.B.; Dean, H.G. Parasympathetic nervous system in nocturnal asthma. BMJ 1988, 296, 1427–1429. [Google Scholar] [CrossRef]
- DesJardin, J.A.; Sutarik, J.M.; Suh, B.Y.; Ballard, R.D. Influence of sleep on pulmonary capillary volume in normal and asthmatic subjects. Am. J. Respir. Crit. Care Med. 1995, 152, 193–198. [Google Scholar] [CrossRef]
- Dagg, K.D.; Thomson, L.J.; Clayton, R.A.; Ramsay, S.G.; Thomson, N. Effect of acute alterations in inspired oxygen tension on methacholine induced bronchoconstriction in patients with asthma. Thorax 1997, 52, 453–457. [Google Scholar] [CrossRef] [Green Version]
- Taillé, C.; Rouvel-Tallec, A.; Stoica, M.; Danel, C.; Dehoux, M.; Marin-Esteban, V.; Pretolani, M.; Aubier, M.; D’Ortho, M.-P. Obstructive Sleep Apnoea Modulates Airway Inflammation and Remodelling in Severe Asthma. PLoS ONE 2016, 11, e0150042. [Google Scholar] [CrossRef] [Green Version]
- Tay, T.R.; Hew, M. Comorbid “treatable traits” in difficult asthma: Current evidence and clinical evaluation. Allergy 2018, 73, 1369–1382. [Google Scholar] [CrossRef] [PubMed]
- Althoff, M.D.; Ghincea, A.; Wood, L.G.; Holguin, F.; Sharma, S. Asthma and Three Colinear Comorbidities: Obesity, OSA, and GERD. J. Allergy Clin. Immunol. Pract. 2021, 9, 3877–3884. [Google Scholar] [CrossRef] [PubMed]
- Dixon, A.E.; Holguin, F.; Sood, A.; Salome, C.M.; Pratley, R.E.; Beuther, D.A.; Celedon, J.C.; Shore, S.A. An Official American Thoracic Society Workshop Report: Obesity and Asthma. Proc. Am. Thorac. Soc. 2010, 7, 325–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brinke, A.T.; Sterk, P.J.; Masclee, A.A.M.; Spinhoven, P.; Schmidt, J.T.; Zwinderman, A.H.; Rabe, K.F.; Bel, E.H. Risk factors of frequent exacerbations in difficult-to-treat asthma. Eur. Respir. J. 2005, 26, 812–818. [Google Scholar] [CrossRef]
- Schan, C.A.; Harding, S.M.; Haile, J.M.; Bradley, L.A.; Richter, J.E. Gastroesophageal Reflux-induced Bronchoconstriction. An intraesophageal acid infusion study using state-of-the-art technology. Chest 1994, 106, 731–737. [Google Scholar] [CrossRef]
- Lim, K.G.; Morgenthaler, T.I.; Katzka, D.A. Sleep and Nocturnal Gastroesophageal Reflux: An Update. Chest 2018, 154, 963–971. [Google Scholar] [CrossRef]
- Ing, A.; Ngu, M.C.; Breslin, A.B. Obstructive sleep apnea and gastroesophageal reflux. Am. J. Med. 2000, 108 (Suppl. 1), 120–125. [Google Scholar] [CrossRef]
- Tawk, M.; Goodrich, S.; Kinasewitz, G.; Orr, W. The Effect of 1 Week of Continuous Positive Airway Pressure Treatment in Obstructive Sleep Apnea Patients With Concomitant Gastroesophageal Reflux. Chest 2006, 130, 1003–1008. [Google Scholar] [CrossRef]
- Bousquet, J.; Schünemann, H.; Samolinski, B.; Demoly, P.; Baena-Cagnani, C.; Bachert, C.; Bonini, S.; Boulet, L.; Brozek, J.; Canonica, G.; et al. Allergic Rhinitis and its Impact on Asthma (ARIA): Achievements in 10 years and future needs. J. Allergy Clin. Immunol. 2012, 130, 1049–1062. [Google Scholar] [CrossRef] [Green Version]
- Becerra, M.B.; Becerra, B.J.; Teodorescu, M. Healthcare burden of obstructive sleep apnea and obesity among asthma hospitalizations: Results from the U.S.-based Nationwide Inpatient Sample. Respir. Med. 2016, 117, 230–236. [Google Scholar] [CrossRef]
- Kim, M.-Y.; Jo, E.-J.; Kang, S.-Y.; Chang, Y.-S.; Yoon, I.-Y.; Cho, S.-H.; Min, K.-U.; Kim, S.-H. Obstructive sleep apnea is associated with reduced quality of life in adult patients with asthma. Ann. Allergy Asthma Immunol. 2013, 110, 253–257.e1. [Google Scholar] [CrossRef] [PubMed]
- Davies, S.E.; Bishopp, A.; Wharton, S.; Turner, A.M.; Mansur, A.H. Does Continuous Positive Airway Pressure (CPAP) treatment of obstructive sleep apnoea (OSA) improve asthma-related clinical outcomes in patients with co-existing conditions?- A systematic review. Respir. Med. 2018, 143, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, G.S. Does CPAP for obstructive sleep apnoea improve asthma control? Respirology 2018, 23, 972–973. [Google Scholar] [CrossRef] [PubMed]
- Ciftci, T.U.; Ciftci, B.; Guven, S.F.; Kokturk, O.; Turktas, H. Effect of nasal continuous positive airway pressure in uncontrolled nocturnal asthmatic patients with obstructive sleep apnea syndrome. Respir. Med. 2005, 99, 529–534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lafond, C.; Sériès, F.; Lemiere, C. Impact of CPAP on asthmatic patients with obstructive sleep apnoea. Eur. Respir. J. 2007, 29, 307–311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serrano-Pariente, J.; Plaza, V.; Soriano, J.B.; Mayos, M.; López-Viña, A.; Picado, C.; Vigil, L.; The CPASMA Trial Group. Asthma outcomes improve with continuous positive airway pressure for obstructive sleep apnea. Allergy 2017, 72, 802–812. [Google Scholar] [CrossRef] [Green Version]
- Ng, S.S.; Chan, T.-O.; To, K.-W.; Chan, K.K.; Ngai, J.; Yip, W.-H.; Lo, R.L.; Ko, F.W.; Hui, D.S. Continuous positive airway pressure for obstructive sleep apnoea does not improve asthma control. Respirology 2018, 23, 1055–1062. [Google Scholar] [CrossRef]
- Wang, J.; Cui, Z.; Liu, S.; Gao, X.; Gao, P.; Shi, Y.; Guo, S.; Li, P. Early use of noninvasive techniques for clearing respiratory secretions during noninvasive positive-pressure ventilation in patients with acute exacerbation of chronic obstructive pulmonary disease and hypercapnic encephalopathy: A prospective cohort study. Medicine 2017, 96, e6371. [Google Scholar] [CrossRef]
- Júnior, N.S.F.; Urbano, J.J.; Santos, I.R.; Silva, A.S.; Perez, E.A.; Souza, A.H.; Nascimento, O.; Jardim, J.R.; Insalaco, G.; Oliveira, L.V.F.; et al. Evaluation of obstructive sleep apnea in non-cystic fibrosis bronchiectasis: A cross-sectional study. PLoS ONE 2017, 12, e0185413. [Google Scholar] [CrossRef] [Green Version]
- Borekci, S.; Sekibag, Y.; Harbiyeli, D.O.; Musellim, B. The Frequency of Obstructive Sleep Apnea in Patients with Non-cystic Fibrosis Bronchiectasis. Turk. Thorac. J. 2021, 22, 333–338. [Google Scholar] [CrossRef]
- Cole, P.J. Inflammation: A two-edged sword—The model of bronchiectasis. Eur. J. Respir. Dis. Suppl. 1986, 147, 6–15. [Google Scholar] [PubMed]
- Peters, C.A.; Altose, M.D.; Coticchia, J.M. Tracheomalacia secondary to obstructive sleep apnea. Am. J. Otolaryngol. 2005, 26, 422–425. [Google Scholar] [CrossRef] [PubMed]
- Kolakowski, C.A.; Rollins, D.R.; Jennermann, T.; Stevens, A.D.; Good, J.T.; Denson, J.; Martin, R.J. Clarifying the link between sleep disordered breathing and tracheal collapse: A retrospective analysis. Sleep Sci. Pract. 2018, 2, 10. [Google Scholar] [CrossRef]
- Ehtisham, M.; Azhar Munir, R.; Klopper, E.; Hammond, K.; Musani, A.I. Correlation between tracheobronchomalacia/hyper dynamic airway collapse and obstructive sleep apnea. In C70 Sleep Disordered Breathing in Pediatric and Adult Medical Disorders; American Thoracic Society: Denver, CO, USA, 2015; p. A5037. [Google Scholar]
- Flenley, D. Sleep in Chronic Obstructive Lung Disease. Clin. Chest Med. 1985, 6, 651–661. [Google Scholar] [CrossRef]
- Weitzenblum, E.; Chaouat, A.; Kessler, R.; Canuet, M. Overlap Syndrome: Obstructive Sleep Apnea in Patients with Chronic Obstructive Pulmonary Disease. Proc. Am. Thorac. Soc. 2008, 5, 237–241. [Google Scholar] [CrossRef]
- Verbraecken, J.; McNicholas, W.T. Respiratory mechanics and ventilatory control in overlap syndrome and obesity hypoventilation. Respir. Res. 2013, 14, 132. [Google Scholar] [CrossRef] [Green Version]
- Malhotra, A.; Schwartz, A.R.; Schneider, H.; Owens, R.L.; Deyoung, P.; Han, M.K.; Wedzicha, J.A.; Hansel, N.N.; Zeidler, M.R.; Wilson, K.C.; et al. Research Priorities in Pathophysiology for Sleep-disordered Breathing in Patients with Chronic Obstructive Pulmonary Disease. An Official American Thoracic Society Research Statement. Am. J. Respir. Crit. Care Med. 2018, 197, 289–299. [Google Scholar] [CrossRef] [Green Version]
- Wellman, A.; Eckert, D.; Jordan, A.; Edwards, B.; Passaglia, C.; Jackson, A.C.; Gautam, S.; Owens, R.L.; Malhotra, A.; White, D.P. A method for measuring and modeling the physiological traits causing obstructive sleep apnea. J. Appl. Physiol. 2011, 110, 1627–1637. [Google Scholar] [CrossRef] [Green Version]
- Krachman, S.L.; Tiwari, R.; Vega, M.E.; Yu, D.; Soler, X.; Jaffe, F.; Kim, V.; Swift, I.; D’Alonzo, G.E.; Criner, G.J. Effect of Emphysema Severity on the Apnea–Hypopnea Index in Smokers with Obstructive Sleep Apnea. Ann. Am. Thorac. Soc. 2016, 13, 1129–1135. [Google Scholar] [CrossRef] [Green Version]
- Adler, D.; Bailly, S.; Benmerad, M.; Joyeux-Faure, M.; Jullian-Desayes, I.; Soccal, P.M.; Janssens, J.P.; Sapène, M.; Grillet, Y.; Stach, B.; et al. Clinical presentation and comorbidities of obstructive sleep apnea-COPD overlap syndrome. PLoS ONE 2020, 15, e0235331. [Google Scholar] [CrossRef]
- Steveling, E.H.; Clarenbach, C.F.; Miedinger, D.; Enz, C.; Dürr, S.; Maier, S.; Sievi, N.; Zogg, S.; Leuppi, J.D.; Kohler, M. Predictors of the Overlap Syndrome and Its Association with Comorbidities in Patients with Chronic Obstructive Pulmonary Disease. Respiration 2014, 88, 451–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orr, J.E.; Schmickl, C.N.; Edwards, B.A.; Deyoung, P.N.; Brena, R.; Sun, X.S.; Jain, S.; Malhotra, A.; Owens, R.L. Pathogenesis of obstructive sleep apnea in individuals with the COPD + OSA Overlap syndrome versus OSA alone. Physiol. Rep. 2020, 8, e14371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Donoghue, F.J.; Catcheside, P.; Ellis, E.E.; Grunstein, R.R.; Pierce, R.J.; Rowland, L.S.; Collins, E.R.; Rochford, S.E.; McEvoy, R.D. Sleep hypoventilation in hypercapnic chronic obstructive pulmonary disease: Prevalence and associated factors. Eur. Respir. J. 2003, 21, 977–984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwon, J.S.; Wolfe, L.F.; Lu, B.S.; Kalhan, R. Hyperinflation is Associated with Lower Sleep Efficiency in COPD with Co-existent Obstructive Sleep Apnea. COPD J. Chronic Obstr. Pulm. Dis. 2009, 6, 441–445. [Google Scholar] [CrossRef] [PubMed]
- He, B.-T.; Lu, G.; Xiao, S.-C.; Chen, R.; Steier, J.; Moxham, J.; Polkey, M.I.; Luo, Y.-M. Coexistence of OSA may compensate for sleep related reduction in neural respiratory drive in patients with COPD. Thorax 2017, 72, 256–262. [Google Scholar] [CrossRef] [Green Version]
- Soler, X.; Liao, S.-Y.; Marin, J.M.; Lorenzi-Filho, G.; Jen, R.; Deyoung, P.; Owens, R.L.; Ries, A.L.; Malhotra, A. Age, gender, neck circumference, and Epworth sleepiness scale do not predict obstructive sleep apnea (OSA) in moderate to severe chronic obstructive pulmonary disease (COPD): The challenge to predict OSA in advanced COPD. PLoS ONE 2017, 12, e0177289. [Google Scholar] [CrossRef]
- Adler, D.; Lozeron, E.D.; Janssens, J.P.; Soccal, P.M.; Lador, F.; Brochard, L.; Pépin, J.L. Obstructive sleep apnea in patients surviving acute hypercapnic respiratory failure is best predicted by static hyperinflation. PLoS ONE 2018, 13, e0205669. [Google Scholar] [CrossRef]
- Mermigkis, C.; Kopanakis, A.; Foldvary-Schaefer, N.; Golish, J.; Polychronopoulos, V.; Schiza, S.; Amfilochiou, A.; Siafakas, N.; Bouros, D. Health-related quality of life in patients with obstructive sleep apnoea and chronic obstructive pulmonary disease (overlap syndrome). Int. J. Clin. Pract. 2007, 61, 207–211. [Google Scholar] [CrossRef]
- Kendzerska, T.; Leung, R.S.; Aaron, S.D.; Ayas, N.; Sandoz, J.S.; Gershon, A.S. Cardiovascular Outcomes and All-Cause Mortality in Patients with Obstructive Sleep Apnea and Chronic Obstructive Pulmonary Disease (Overlap Syndrome). Ann. Am. Thorac. Soc. 2019, 16, 71–81. [Google Scholar] [CrossRef]
- Ganga, H.V.; Nair, S.U.; Puppala, V.K.; Miller, W.L. Risk of new-onset atrial fibrillation in elderly patients with the overlap syndrome: A retrospective cohort study. J. Geriatr. Cardiol. 2013, 10, 129–134. [Google Scholar] [CrossRef]
- Putcha, N.; Crainiceanu, C.; Norato, G.; Samet, J.; Quan, S.F.; Gottlieb, D.J.; Redline, S.; Punjabi, N.M. Influence of Lung Function and Sleep-disordered Breathing on All-Cause Mortality. A Community-based Study. Am. J. Respir. Crit. Care Med. 2016, 194, 1007–1014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryan, S.; Taylor, C.T.; McNicholas, W.T. Selective Activation of Inflammatory Pathways by Intermittent Hypoxia in Obstructive Sleep Apnea Syndrome. Circulation 2005, 112, 2660–2667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vernooy, J.H.; Küçükaycan, M.; Jacobs, J.A.; Chavannes, N.; Buurman, W.A.; Dentener, M.A.; Wouters, E.F. Local and Systemic Inflammation in Patients with Chronic Obstructive Pulmonary Disease: Soluble tumor necrosis factor receptors are increased in sputum. Am. J. Respir. Crit. Care Med. 2002, 166, 1218–1224. [Google Scholar] [CrossRef] [PubMed]
- Marin, J.M.; Soriano, J.B.; Carrizo, S.J.; Boldova, A.; Celli, B.R. Outcomes in Patients with Chronic Obstructive Pulmonary Disease and Obstructive Sleep Apnea: The overlap syndrome. Am. J. Respir. Crit. Care Med. 2010, 182, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Adler, D.; Dupuis-Lozeron, E.; Merlet-Violet, R.; Pépin, J.-L.; Espa-Cervena, K.; Muller, H.; Janssens, J.-P.; Brochard, L. Comorbidities and Subgroups of Patients Surviving Severe Acute Hypercapnic Respiratory Failure in the Intensive Care Unit. Am. J. Respir. Crit. Care Med. 2017, 196, 200–207. [Google Scholar] [CrossRef]
- Valko, L.; Baglyas, S.; Gyarmathy, V.A.; Gal, J.; Lorx, A. Home mechanical ventilation: Quality of life patterns after six months of treatment. BMC Pulm. Med. 2020, 20, 1–13. [Google Scholar] [CrossRef]
- Naranjo, M.; Willes, L.; Prillaman, B.A.; Quan, S.F.; Sharma, S. Undiagnosed OSA May Significantly Affect Outcomes in Adults Admitted for COPD in an Inner-City Hospital. Chest 2020, 158, 1198–1207. [Google Scholar] [CrossRef]
- Resta, O.; Barbaro, M.P.F.; Brindicci, C.; Nocerino, M.C.; Caratozzolo, G.; Carbonara, M. Hypercapnia in Overlap Syndrome: Possible Determinant Factors. Sleep Breath. 2002, 6, 11–17. [Google Scholar] [CrossRef]
- Sharma, B.; Neilan, T.G.; Kwong, R.Y.; Mandry, D.; Owens, R.L.; McSharry, D.; Bakker, J.P.; Malhotra, A. Evaluation of Right Ventricular Remodeling Using Cardiac Magnetic Resonance Imaging in Co-Existent Chronic Obstructive Pulmonary Disease and Obstructive Sleep Apnea. COPD J. Chronic Obstr. Pulm. Dis. 2013, 10, 4–10. [Google Scholar] [CrossRef] [Green Version]
- Shaya, F.T.; Lin, P.-J.; Aljawadi, M.H.; Scharf, S.M. Elevated economic burden in obstructive lung disease patients with concomitant sleep apnea syndrome. Sleep Breath. 2009, 13, 317–323. [Google Scholar] [CrossRef]
- Du, W.; Liu, J.; Zhou, J.; Ye, D.; OuYang, Y.; Deng, Q. Obstructive sleep apnea, COPD, the overlap syndrome, and mortality: Results from the 2005–2008 National Health and Nutrition Examination Survey. Int. J. Chronic Obstr. Pulm. Dis. 2018, 13, 665–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shawon, S.R.; Perret, J.L.; Senaratna, C.V.; Lodge, C.; Hamilton, G.S.; Dharmage, S.C. Current evidence on prevalence and clinical outcomes of co-morbid obstructive sleep apnea and chronic obstructive pulmonary disease: A systematic review. Sleep Med. Rev. 2016, 32, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.Y.; Yee, M.B.J.; Wong, M.K.; Grunstein, M.R.; Piper, B.A. A pilot randomized trial comparing CPAP vs bilevel PAP spontaneous mode in the treatment of hypoventilation disorder in patients with obesity and obstructive airway disease. J. Clin. Sleep Med. 2022, 18, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Toraldo, D.M.; De Nuccio, F.; Nicolardi, G. Fixed-pressure nCPAP in patients with obstructive sleep apnea (OSA) syndrome and chronic obstructive pulmonary disease (COPD): A 24-month follow-up study. Sleep Breath. 2010, 14, 115–123. [Google Scholar] [CrossRef]
- Stanchina, M.L.; Welicky, L.M.; Donat, W.; Lee, D.; Corrao, W.; Malhotra, A. Impact of CPAP Use and Age on Mortality in Patients with Combined COPD and Obstructive Sleep Apnea: The Overlap Syndrome. J. Clin. Sleep Med. 2013, 9, 767–772. [Google Scholar] [CrossRef] [Green Version]
- Platt, A.B.; Kuna, S.T.; Field, S.H.; Chen, Z.; Gupta, R.; Roche, D.F.; Christie, J.D.; Asch, D.A. Adherence to Sleep Apnea Therapy and Use of Lipid-Lowering Drugs: A Study of the Healthy-User Effect. Chest 2010, 137, 102–108. [Google Scholar] [CrossRef] [Green Version]
- Billings, M.E.; Auckley, D.; Benca, R.; Foldvary-Schaefer, N.; Iber, C.; Redline, S.; Rosen, C.L.; Zee, P.; Kapur, V. Race and Residential Socioeconomics as Predictors of CPAP Adherence. Sleep 2011, 34, 1653–1658. [Google Scholar] [CrossRef] [Green Version]
- Marin, J.M.; Carrizo, S.J.; Vicente, E.; Agusti, A.G. Long-term cardiovascular outcomes in men with obstructive sleep apnoea-hypopnoea with or without treatment with continuous positive airway pressure: An observational study. Lancet 2005, 365, 1046–1053. [Google Scholar] [CrossRef]
- Patil, S.P.; Ayappa, I.A.; Caples, S.M.; Kimoff, R.J.; Patel, S.; Harrod, C.G. Treatment of Adult Obstructive Sleep Apnea With Positive Airway Pressure: An American Academy of Sleep Medicine Systematic Review, Meta-Analysis, and GRADE Assessment. J. Clin. Sleep Med. 2019, 15, 301–334. [Google Scholar] [CrossRef]
- Machado, M.-C.L.; Vollmer, W.M.; Togeiro, S.M.; Bilderback, A.L.; Oliveira, M.-V.C.; Leitao, F.S.; Queiroga, F.; Lorenzi-Filho, G.; Krishnan, J.A. CPAP and survival in moderate-to-severe obstructive sleep apnoea syndrome and hypoxaemic COPD. Eur. Respir. J. 2010, 35, 132–137. [Google Scholar] [CrossRef] [Green Version]
- Masa, J.F.; Pépin, J.L.; Borel, J.C.; Mokhlesi, B.; Murphy, P.B.; Sánchez-Quiroga, M. Obesity hypoventilation syndrome. Eur. Respir. Rev. 2019, 28, 180097. [Google Scholar] [CrossRef] [PubMed]
- Medicine, A.A.o.S. International Classification of Sleep Disorders, 3rd ed.; American Academy of Sleep Medicine: Darien, IL, USA, 2014. [Google Scholar]
- Laaban, J.-P.; Chailleux, E. Daytime Hypercapnia in Adult Patients With Obstructive Sleep Apnea Syndrome in France, Before Initiating Nocturnal Nasal Continuous Positive Airway Pressure Therapy. Chest 2005, 127, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Masa, J.F.; Corral, J.; Alonso, M.L.; Ordax, E.; Troncoso, M.F.; González, M.; Lopez-Martínez, S.; Marin, J.M.; Marti, S.; Díaz-Cambriles, T.; et al. Efficacy of Different Treatment Alternatives for Obesity Hypoventilation Syndrome. Pickwick Study. Am. J. Respir. Crit. Care Med. 2015, 192, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Olson, A.L.; Zwillich, C. The obesity hypoventilation syndrome. Am. J. Med. 2005, 118, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.L.; Nzekwu, M.-M.U. The Effects of Body Mass Index on Lung Volumes. Chest 2006, 130, 827–833. [Google Scholar] [CrossRef]
- Steier, J.; Jolley, C.J.; Seymour, J.; Roughton, M.; Polkey, M.I.; Moxham, J. Neural respiratory drive in obesity. Thorax 2009, 64, 719–725. [Google Scholar] [CrossRef] [Green Version]
- Zwillich, C.W.; Sutton, F.D.; Pierson, D.J.; Creagh, E.M.; Weil, J.V. Decreased hypoxic ventilatory drive in the obesity-hypoventilation syndrome. Am. J. Med. 1975, 59, 343–348. [Google Scholar] [CrossRef]
- Randerath, W. More Than Obstruction: Rethinking Obesity Hypoventilation? Ann. Am. Thorac. Soc. 2020, 17, 282–283. [Google Scholar] [CrossRef]
- Javaheri, S.; Simbartl, L.A. Respiratory Determinants of Diurnal Hypercapnia in Obesity Hypoventilation Syndrome. What Does Weight Have to Do with It? Ann. Am. Thorac. Soc. 2014, 11, 945–950. [Google Scholar] [CrossRef]
- Lin, C.-K. Work of breathing and respiratory drive in obesity. Respirology 2012, 17, 402–411. [Google Scholar] [CrossRef]
- Berger, K.I.; Ayappa, I.; Sorkin, I.B.; Norman, R.G.; Rapoport, D.M.; Goldring, R.M. CO2 homeostasis during periodic breathing in obstructive sleep apnea. J. Appl. Physiol. 2000, 88, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Kittivoravitkul, P.; Aboussouan, L.S.; Kaw, R.; Hatipoğlu, U.; Wang, L. Determinants of Wake PCO2 and Increases in Wake PCO2 over Time in Patients with Obstructive Sleep Apnea. Ann. Am. Thorac. Soc. 2016, 13, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Randerath, W.; Verbraecken, J.; Andreas, S.; Arzt, M.; Bloch, K.E.; Brack, T.; Buyse, B.; De Backer, W.; Eckert, D.; Grote, L.; et al. Definition, discrimination, diagnosis and treatment of central breathing disturbances during sleep. Eur. Respir. J. 2017, 49, 1600959. [Google Scholar] [CrossRef] [PubMed]
- Manuel, A.R.G.; Hart, N.; Stradling, J.R. Is a Raised Bicarbonate, Without Hypercapnia, Part of the Physiologic Spectrum of Obesity-Related Hypoventilation? Chest 2015, 147, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Ouanes-Besbes, L.; Hammouda, Z.; Besbes, S.; Nouira, W.; Lahmar, M.; Ben Abdallah, S.; Ouanes, I.; Dachraoui, F.; Abroug, F. Diagnosis of Sleep Apnea Syndrome in the Intensive Care Unit: A Case Series of Survivors of Hypercapnic Respiratory Failure. Ann. Am. Thorac. Soc. 2021, 18, 727–729. [Google Scholar] [CrossRef] [PubMed]
- Thille, A.W.; Córdoba-Izquierdo, A.; Maitre, B.; Boyer, L.; Brochard, L.; Drouot, X. High prevalence of sleep apnea syndrome in patients admitted to ICU for acute hypercapnic respiratory failure: A preliminary study. Intensiv. Care Med. 2018, 44, 267–269. [Google Scholar] [CrossRef]
- Peppard, P.E.; Young, T.; Barnet, J.H.; Palta, M.; Hagen, E.W.; Hla, K.M. Increased Prevalence of Sleep-Disordered Breathing in Adults. Am. J. Epidemiol. 2013, 177, 1006–1014. [Google Scholar] [CrossRef] [Green Version]
- Benditt, J.O.; Boitano, L.J. Pulmonary Issues in Patients with Chronic Neuromuscular Disease. Am. J. Respir. Crit. Care Med. 2013, 187, 1046–1055. [Google Scholar] [CrossRef]
- Gupta, K.; Nagappa, M.; Prasad, A.; Abrahamyan, L.; Wong, J.; Weingarten, T.N.; Chung, F. Risk factors for opioid-induced respiratory depression in surgical patients: A systematic review and meta-analyses. BMJ Open 2018, 8, e024086. [Google Scholar] [CrossRef] [Green Version]
- Guilleminault, C.; Cao, M.; Yue, H.J.; Chawla, P. Obstructive Sleep Apnea and Chronic Opioid Use. Lung 2010, 188, 459–468. [Google Scholar] [CrossRef]
- Freire, C.; Sennes, L.U.; Polotsky, V.Y. Opioids and obstructive sleep apnea. J. Clin. Sleep Med. 2022, 18, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.; Garden, F.L.; Marks, G.B.; Vedam, H. Population Prevalence of Hypercapnic Respiratory Failure from Any Cause. Am. J. Respir. Crit. Care Med. 2022, 205, 966–967. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.B.; Geske, J.B.; Kathuria, P.; Cuttica, M.; Schimmel, D.; Courtney, D.M.; Waterer, G.W.; Wunderink, R. Analysis of National Trends in Admissions for Pulmonary Embolism. Chest 2016, 150, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Jennum, P.; Kjellberg, J. Health, social and economical consequences of sleep-disordered breathing: A controlled national study. Thorax 2011, 66, 560–566. [Google Scholar] [CrossRef] [Green Version]
- Meservey, A.J.; Burton, M.C.; Priest, J.; Teneback, C.C.; Dixon, A.E. Risk of Readmission and Mortality Following Hospitalization with Hypercapnic Respiratory Failure. Lung 2020, 198, 121–134. [Google Scholar] [CrossRef]
- Wilson, M.W.; Labaki, W.W.; Choi, P.J. Mortality and Healthcare Use of Patients with Compensated Hypercapnia. Ann. Am. Thorac. Soc. 2021, 18, 2027–2032. [Google Scholar] [CrossRef]
- Castro-Añón, O.; De Llano, L.A.P.; Sánchez, S.D.L.F.; Golpe, R.; Marote, L.M.; Castro-Castro, J.; Quintela, A.G. Obesity-Hypoventilation Syndrome: Increased Risk of Death over Sleep Apnea Syndrome. PLoS ONE 2015, 10, e0117808. [Google Scholar] [CrossRef] [Green Version]
- Marik, P.E.; Chen, C. The clinical characteristics and hospital and post-hospital survival of patients with the obesity hypoventilation syndrome: Analysis of a large cohort. Obes. Sci. Pract. 2016, 2, 40–47. [Google Scholar] [CrossRef] [Green Version]
- Berg, G.; Delaive, K.; Manfreda, J.; Walld, R.; Kryger, M.H. The Use of Health-Care Resources in Obesity-Hypoventilation Syndrome. Chest 2001, 120, 377–383. [Google Scholar] [CrossRef] [Green Version]
- Mokhlesi, B.; Masa, J.F.; Afshar, M.; Pacheco, V.A.; Berlowitz, D.J.; Borel, J.-C.; Budweiser, S.; Carrillo, A.; Castro-Añón, O.; Ferrer, M.; et al. The Effect of Hospital Discharge with Empiric Noninvasive Ventilation on Mortality in Hospitalized Patients with Obesity Hypoventilation Syndrome. An Individual Patient Data Meta-Analysis. Ann. Am. Thorac. Soc. 2020, 17, 627–637. [Google Scholar] [CrossRef]
- Mokhlesi, B.; Masa, J.F.; Brozek, J.L.; Gurubhagavatula, I.; Murphy, P.B.; Piper, A.J.; Tulaimat, A.; Afshar, M.; Balachandran, J.S.; Dweik, R.A.; et al. Evaluation and Management of Obesity Hypoventilation Syndrome. An Official American Thoracic Society Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2019, 200, e6–e24. [Google Scholar] [CrossRef] [Green Version]
- Mokhlesi, B.; Won, C.H.; Make, B.J.; Selim, B.J.; Sunwoo, B.Y.; Gay, P.C.; Owens, R.L.; Wolfe, L.F.; Benditt, J.O.; Aboussouan, L.S.; et al. Optimal Noninvasive Medicare Access Promotion: Patients With Hypoventilation Syndromes: A Technical Expert Panel Report From the American College of Chest Physicians, the American Association for Respiratory Care, the American Academy of Sleep Medicine, and the American Thoracic Society. Chest 2021, 160, e377–e387. [Google Scholar] [CrossRef] [PubMed]
- Morice, A.; Dicpinigaitis, P.; McGarvey, L.; Birring, S.S. Chronic cough: New insights and future prospects. Eur. Respir. Rev. 2021, 30, 210127. [Google Scholar] [CrossRef] [PubMed]
- Sundar, K.M.; Willis, A.M.; Smith, S.; Hu, N.; Kitt, J.P.; Birring, S.S. A Randomized, Controlled, Pilot Study of CPAP for Patients with Chronic Cough and Obstructive Sleep Apnea. Lung 2020, 198, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Sundar, K.M.; Daly, S.E.; Willis, A.M. A longitudinal study of CPAP therapy for patients with chronic cough and obstructive sleep apnoea. Cough 2013, 9, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sundar, K.M.; Daly, S.E.; Pearce, M.J.; Alward, W.T. Chronic cough and obstructive sleep apnea in a community-based pulmonary practice. Cough 2010, 6, 2–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sundar, K.M.; Stark, A.C.; Hu, N.; Barkmeier-Kraemer, J. Is laryngeal hypersensitivity the basis of unexplained or refractory chronic cough? ERJ Open Res. 2021, 7, 00793–2020. [Google Scholar] [CrossRef]
- Choi, J.; Lee, E.; Hong, S.; Chung, S.; Jung, Y.; Kim, H. Potential Therapeutic Effect of Continuous Positive Airway Pressure on Laryngopharyngeal Reflux in Obstructive Sleep Apnea Patients. J. Clin. Med. 2021, 10, 2861. [Google Scholar] [CrossRef]
- Poliacek, I.; Simera, M.; Veternik, M.; Kotmanova, Z.; Pitts, T.; Hanacek, J.; Plevkova, J.; Machac, P.; Visnovcova, N.; Misek, J.; et al. The course of lung inflation alters the central pattern of tracheobronchial cough in cat—The evidence for volume feedback during cough. Respir. Physiol. Neurobiol. 2016, 229, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Lancaster, L.H.; Mason, W.R.; Parnell, J.A.; Rice, T.; Loyd, J.; Milstone, A.P.; Collard, H.R.; Malow, B.A. Obstructive Sleep Apnea Is Common in Idiopathic Pulmonary Fibrosis. Chest 2009, 136, 772–778. [Google Scholar] [CrossRef] [Green Version]
- Mermigkis, C.; Stagaki, E.; Tryfon, S.; Schiza, S.; Amfilochiou, A.; Polychronopoulos, V.; Panagou, P.; Galanis, N.; Kallianos, A.; Mermigkis, D.; et al. How common is sleep-disordered breathing in patients with idiopathic pulmonary fibrosis? Sleep Breath. 2010, 14, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Gille, T.; Didier, M.; Boubaya, M.; Moya, L.; Sutton, A.; Carton, Z.; Baran-Marszak, F.; Sadoun-Danino, D.; Israël-Biet, D.; Cottin, V.; et al. Obstructive sleep apnoea and related comorbidities in incident idiopathic pulmonary fibrosis. Eur. Respir. J. 2017, 49, 1601934. [Google Scholar] [CrossRef] [PubMed]
- Papadogiannis, G.; Bouloukaki, I.; Mermigkis, C.; Michelakis, S.; Ermidou, C.; Mauroudi, E.; Moniaki, V.; Tzanakis, N.; Antoniou, K.M.; Schiza, S.E. Patients with idiopathic pulmonary fibrosis with and without obstructive sleep apnea: Differences in clinical characteristics, clinical outcomes, and the effect of PAP treatment. J. Clin. Sleep Med. 2021, 17, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.L.; Dai, H.P.; Zhang, H.; Gao, B.; Zhang, L.; Han, T.; Wang, C. Obstructive Sleep Apnea in Patients With Fibrotic Interstitial Lung Disease and COPD. J. Clin. Sleep Med. 2019, 15, 1807–1815. [Google Scholar] [CrossRef] [PubMed]
- Pihtili, A.; Bingol, Z.; Kiyan, E.; Cuhadaroglu, C.; Issever, H.; Gulbaran, Z. Obstructive sleep apnea is common in patients with interstitial lung disease. Sleep Breath. 2013, 17, 1281–1288. [Google Scholar] [CrossRef]
- Cardoso, A.V.; Pereira, N.; Neves, I.; Santos, V.; Jesus, J.M.; Melo, N.; Mota, P.C.; Morais, A.; Drummond, M. Obstructive sleep apnoea in patients with fibrotic diffuse parenchymal lung disease-characterization and treatment compliance assessment. Can. J. Respir. Ther. 2018, 54, 35–40. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, Y.; Dai, L. The prevalence of obstructive sleep apnea in interstitial lung disease: A systematic review and meta-analysis. Sleep Breath. 2021, 25, 1219–1228. [Google Scholar] [CrossRef]
- Mermigkis, C.; Chapman, J.; Golish, J.; Mermigkis, D.; Budur, K.; Kopanakis, A.; Polychronopoulos, V.; Burgess, R.; Foldvary-Schaefer, N. Sleep-Related Breathing Disorders in Patients with Idiopathic Pulmonary Fibrosis. Lung 2007, 185, 173–178. [Google Scholar] [CrossRef]
- Stanchina, M.L.; Malhotra, A.; Fogel, R.B.; Trinder, J.; Edwards, J.K.; Schory, K.; White, D.P. The Influence of Lung Volume on Pharyngeal Mechanics, Collapsibility, and Genioglossus Muscle Activation during Sleep. Sleep 2003, 26, 851–856. [Google Scholar] [CrossRef]
- Aronson, R.M.; Carley, D.W.; Önal, E.; Wilborn, J.; Lopata, M. Upper airway muscle activity and the thoracic volume dependence of upper airway resistance. J. Appl. Physiol. 1991, 70, 430–438. [Google Scholar] [CrossRef]
- Bosi, M.; Milioli, G.; Parrino, L.; Fanfulla, F.; Tomassetti, S.; Melpignano, A.; Trippi, I.; Vaudano, A.E.; Ravaglia, C.; Mascetti, S.; et al. Quality of life in idiopathic pulmonary fibrosis: The impact of sleep disordered breathing. Respir. Med. 2019, 147, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Bosi, M.; Milioli, G.; Fanfulla, F.; Tomassetti, S.; Ryu, J.; Parrino, L.; Riccardi, S.; Melpignano, A.; Vaudano, A.E.; Ravaglia, C.; et al. OSA and Prolonged Oxygen Desaturation During Sleep are Strong Predictors of Poor Outcome in IPF. Lung 2017, 195, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.W.; Lee, T.Y.; Johannson, K.A.; Assayag, D.; Morisset, J.; Fell, C.D.; Fisher, J.H.; Shapera, S.; Gershon, A.S.; Cox, G.; et al. A cluster-based analysis evaluating the impact of comorbidities in fibrotic interstitial lung disease. Respir. Res. 2020, 21, 1–9. [Google Scholar] [CrossRef]
- Vozoris, N.T.; Wilton, A.S.; Austin, P.C.; Kendzerska, T.; Ryan, C.M.; Gershon, A.S. Morbidity and mortality reduction associated with polysomnography testing in idiopathic pulmonary fibrosis: A population-based cohort study. BMC Pulm. Med. 2021, 21, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Adegunsoye, A.; Neborak, J.M.; Zhu, D.; Cantrill, B.; Garcia, N.; Oldham, J.M.; Noth, I.; Vij, R.; Kuzniar, T.J.; Bellam, S.K.; et al. CPAP Adherence, Mortality, and Progression-Free Survival in Interstitial Lung Disease and OSA. Chest 2020, 158, 1701–1712. [Google Scholar] [CrossRef]
- Mermigkis, C.; Bouloukaki, I.; Antoniou, K.; Papadogiannis, G.; Giannarakis, I.; Varouchakis, G.; Siafakas, N.; Schiza, S.E. Obstructive sleep apnea should be treated in patients with idiopathic pulmonary fibrosis. Sleep Breath. 2015, 19, 385–391. [Google Scholar] [CrossRef]
- Mermigkis, C.; Bouloukaki, I.; Antoniou, K.M.; Mermigkis, D.; Psathakis, K.; Giannarakis, I.; Varouchakis, G.; Siafakas, N.; Schiza, S.E. CPAP therapy in patients with idiopathic pulmonary fibrosis and obstructive sleep apnea: Does it offer a better quality of life and sleep? Sleep Breath. 2013, 17, 1137–1143. [Google Scholar] [CrossRef]
- Baughman, R.P.; Lower, E.E.; Tami, T. Upper airway.4: Sarcoidosis of the upper respiratory tract (SURT). Thorax 2010, 65, 181–186. [Google Scholar] [CrossRef] [Green Version]
- Lal, C.; Medarov, B.I.; Judson, M.A. Interrelationship Between Sleep-Disordered Breathing and Sarcoidosis. Chest 2015, 148, 1105–1114. [Google Scholar] [CrossRef]
- Mari, P.-V.; Pasciuto, G.; Siciliano, M.; Simonetti, J.; Ballacci, F.; Macagno, F.; Iovene, B.; Martone, F.; Corbo, G.M.; Richeldi, L. Obstructive sleep apnea in sarcoidosis and impact of CPAP treatment on fatigue. Sarcoidosis Vasc. Diffus. Lung Dis. 2020, 37, 169–178. [Google Scholar] [CrossRef]
- Benn, B.S.; Lehman, Z.; Kidd, S.A.; Miaskowski, C.; Sunwoo, B.Y.; Ho, M.; Sun, S.; Ramstein, J.; Gelfand, J.M.; Koth, L.L. Sleep disturbance and symptom burden in sarcoidosis. Respir. Med. 2018, 144, S35–S40. [Google Scholar] [CrossRef] [PubMed]
- Bosse-Henck, A.; Koch, R.; Wirtz, H.; Hinz, A. Fatigue and Excessive Daytime Sleepiness in Sarcoidosis: Prevalence, Predictors, and Relationships between the Two Symptoms. Respiration 2017, 94, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Drent, M.; Lower, E.E.; De Vries, J. Sarcoidosis-associated fatigue. Eur. Respir. J. 2012, 40, 255–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chervin, R.D. Sleepiness, Fatigue, Tiredness, and Lack of Energy in Obstructive Sleep Apnea. Chest 2000, 118, 372–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marrone, O.; Bonsignore, M.R. Pulmonary haemodynamics in obstructive sleep apnoea. Sleep Med. Rev. 2002, 6, 175–193. [Google Scholar] [CrossRef]
- Nathan, S.D.; Barbera, J.A.; Gaine, S.P.; Harari, S.; Martinez, F.J.; Olschewski, H.; Olsson, K.M.; Peacock, A.J.; Pepke-Zaba, J.; Provencher, S.; et al. Pulmonary hypertension in chronic lung disease and hypoxia. Eur. Respir. J. 2019, 53, 1801914. [Google Scholar] [CrossRef] [Green Version]
- Kholdani, C.; Fares, W.H.; Mohsenin, V. Pulmonary Hypertension in Obstructive Sleep Apnea: Is it Clinically Significant? A Critical Analysis of the Association and Pathophysiology. Pulm. Circ. 2015, 5, 220–227. [Google Scholar] [CrossRef] [Green Version]
- Masa, J.F.; Benítez, M.I.D.; Javaheri, S.; Mogollon, M.V.; Sánchez-Quiroga, M.; de Terreros, F.J.G.; Corral, J.; Gallego, R.; Romero, A.; Caballero-Eraso, C.; et al. Risk factors associated with pulmonary hypertension in obesity hypoventilation syndrome. J. Clin. Sleep Med. 2022, 18, 983–992. [Google Scholar] [CrossRef]
- García-Ortega, A.; Mañas, E.; López-Reyes, R.; Selma, M.J.; García-Sánchez, A.; Oscullo, G.; Jiménez, D.; Martínez-García, M. Obstructive sleep apnoea and venous thromboembolism: Pathophysiological links and clinical implications. Eur. Respir. J. 2019, 53, 1800893. [Google Scholar] [CrossRef]
- Genuardi, M.V.; Rathore, A.; Ogilvie, R.P.; DeSensi, R.S.; Borker, P.V.; Magnani, J.W.; Patel, S.R. Incidence of VTE in Patients With OSA: A Cohort Study. Chest 2021, 161, 1073–1082. [Google Scholar] [CrossRef]
- Alonso-Fernández, A.; Toledo-Pons, N.; Garcia-Rio, F. Obstructive sleep apnea and venous thromboembolism: Overview of an emerging relationship. Sleep Med. Rev. 2020, 50, 101233. [Google Scholar] [CrossRef] [PubMed]
- Nieto, F.; Peppard, P.; Young, T.; Finn, L.; Hla, K.; Farré, R. Sleep-disordered breathing and cancer mortality: Results from the Wisconsin Sleep Cohort Study. Am. J. Respir. Crit. Care Med. 2012, 186, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Campos-Rodriguez, F.; Martinez-Garcia, M.; Martinez, M.; Duran-Cantolla, J.; Peña, M.L.; Masdeu, M.; Gonzalez, M.; Campo, F.; Gallego, I.; Marin, J.; et al. Association between Obstructive Sleep Apnea and Cancer Incidence in a Large Multicenter Spanish Cohort. Am. J. Respir. Crit. Care Med. 2013, 187, 99–105. [Google Scholar] [CrossRef] [Green Version]
- Hunyor, I.; Cook, K.M. Models of intermittent hypoxia and obstructive sleep apnea: Molecular pathways and their contribution to cancer. Am. J. Physiol. Integr. Comp. Physiol. 2018, 315, R669–R687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.-J.; Ouyang, C.; Yu, B.; Chen, C.; Xu, X.-F.; Ye, X.-Q. Role of hypoxia-inducible factor-2α in lung cancer. Oncol. Rep. 2021, 45, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheong, A.J.Y.; Tan, B.K.J.; Teo, Y.H.; Tan, N.K.W.; Yap, D.W.T.; Sia, C.-H.; Ong, T.H.; Leow, L.C.; See, A.; Toh, S.T. Obstructive Sleep Apnea and Lung Cancer: A Systematic Review and Meta-Analysis. Ann. Am. Thorac. Soc. 2022, 19, 469–475. [Google Scholar] [CrossRef]
- Gozal, D.; Ham, S.; Mokhlesi, B. Sleep Apnea and Cancer: Analysis of a Nationwide Population Sample. Sleep 2016, 39, 1493–1500. [Google Scholar] [CrossRef]
- Schiza, S.E.; Bouloukaki, I.; Bolaki, M.; Antoniou, K.M. Obstructive sleep apnea in pulmonary fibrosis. Curr. Opin. Pulm. Med. 2020, 26, 443–448. [Google Scholar] [CrossRef]
- Cade, B.E.; Dashti, H.S.; Hassan, S.M.; Redline, S.; Karlson, E.W. Sleep Apnea and COVID-19 Mortality and Hospitalization. Am. J. Respir. Crit. Care Med. 2020, 202, 1462–1464. [Google Scholar] [CrossRef]
- Maas, M.B.; Kim, M.; Malkani, R.G.; Abbott, S.M.; Zee, P.C. Obstructive Sleep Apnea and Risk of COVID-19 Infection, Hospitalization and Respiratory Failure. Sleep Breath. 2021, 25, 1155–1157. [Google Scholar] [CrossRef] [PubMed]
- Strausz, S.; Kiiskinen, T.; Broberg, M.; Ruotsalainen, S.; Koskela, J.; Bachour, A.; Palotie, A.; Palotie, T.; Ripatti, S.; Ollila, H.M.; et al. Sleep apnoea is a risk factor for severe COVID-19. BMJ Open Respir. Res. 2021, 8, e000845. [Google Scholar] [CrossRef] [PubMed]
- Guillien, A.; Cadiou, S.; Slama, R.; Siroux, V. The Exposome Approach to Decipher the Role of Multiple Environmental and Lifestyle Determinants in Asthma. Int. J. Environ. Res. Public Health 2021, 18, 1138. [Google Scholar] [CrossRef] [PubMed]
| Study | Design | Patients | Key Findings | Limitations |
|---|---|---|---|---|
| Ng et al. (2018) [47] | RCT, CPAP vs. no CPAP. 122 patients tested, 37 randomized. Hong Kong. ACT | Adults with asthma who snore and have nocturnal symptoms. Randomized if PSG showed AHI > 10 events/h | 33.6% had AHI over 10 events/hr. No difference in ACT score change (CPAP + 3.2, Control + 2.4, p = 0.57) | Small sample size. |
| Davies et al. 2018 [42] | Metanalysis of 8 observational studies. Mean duration of CPAP use 19.5 weeks. | Adults with Asthma and OSA treated with CPAP | ACQ scores improved (0.59, 2 studies). No difference in FEV1 (4 studies) | High risk of bias, significant heterogeneity. |
| Serrano-Pariente et al. (2017) [46] | Before–after; 6-month follow-up; 99 patients | Asthma + new diagnosis of OSA starting CPAP | ACQ improved from 1.39 to 1.0 | No control group, unclear if the effect in addition to regression to mean. Mean change of 0.39 less than MCID (0.5) |
| Study | Design | Patients | Key Findings | Limitations |
|---|---|---|---|---|
| Marin et al. (2010) [75] | Prospective cohort study, n = 651, 9.4 years mean follow-up, Sleep clinics in Spain. | Patients with COPD referred for sleep evaluation. Compared COPD, OVS and declined CPAP, OVS and tried CPAP | COPD and OVS with CPAP had similar mortality, but RR for AECOPD (1.7) and death (1.79) in OVS without CPAP were elevated. | Acceptance of CPAP recommendations is likely a marker for a broad range of health behaviors that influence mortality. |
| Machado et al. (2010) [91] | Prospective cohort study, n = 95. Pulmonary clinics in Brazil. Median follow-up 41 months. Cox proportional hazard modeling. | Patients with COPD on LTOT for 6+ months with mod–severe OSA on PSG. Compared patients who started CPAP to those who didn’t. | HR for death was 0.19 in the CPAP-treated group. | Acceptance and insurance coverage of CPAP reflects health behaviors and socioeconomic status. |
| Stanchina et al. (2013) [86] | Retrospective cohort. n = 227 patients with overlap | Diagnosis by ICD code and confirmed by survey. Associated CPAP use in first 3 months to mortality. | Each hour of nightly adherence is associated with an HR of 0.71 for mortality. Age is also independently associated (HR 1.14), but FEV1, smoking, and O2 are not. | Retrospective, no control group. CPAP benefit is associated with strong confounders for mortality [87] |
| Toraldo et al. (2010) [85] | Prospective case series, n = 12. Italy. Follow-up at 3, 12, and 24 months | Patients with BMI 30+, moderate obstruction, severe OSA. Starting nasal CPAP | FEV1, FRC, mPAP, PaCO2, and PaO2 improved by 3 months, then stable. ESS improved at 3 and further at 12 months. | No control group. Severe disease (majority hypercapnic at the start) |
| Study | Design | Patients | Key Findings | Limitations |
|---|---|---|---|---|
| Mermigkis et al. (2013) [149] | Prospective single-center cohort study n = 23 (Greece) | Patients with incident IPF underwent a Type I PSG; 12 patients were found to have moderate to severe OSA * and were placed on CPAP. | With CPAP, there was a significant improvement in sleep-related QOL, as measured by the FOSQ at 1,3, and 6 months. There was no significant change in other QOL measures, including ESS. | Single-center, small population. Compliance was poor, with 2/12 patients not able to tolerate PAP therapy. |
| Mermigkis et al. (2015) [148] | Prospective multicenter cohort study n = 92 (Greece) | Patients with incident IPF underwent a Type 1 PSG; 60 patients had moderate to severe OSA *; of these patients, 55 agreed to CPAP. | Good compliance group ** (n = 37) had significant improvements in all QOL measures at one year. At two-year follow-up, significant mortality benefit with good compliance, 3 deaths vs. 0 deaths. | Poor compliance. |
| Adegunsoye et al. (2020) [147] | Retrospective observational multicenter cohort study (United States) n = 160 | Patients with ILD who had undergone a Type 1 PSG; 94 patients with moderate to severe OSA *; of these patients, 51 with untreated/poor compliance, and 43 with good compliance ** | No difference in all-cause mortality, progression-free survival, or lung transplantation with moderate/severe OSA treatment. Among sub-population requiring supplemental oxygen, CPAP compliance was associated with improved progression-free survival. | Retrospective study. Inclusion of various forms of ILD, including IPF. Unclear length of ILD diagnosis. Low overall event rate led to the study being underpowered. |
| Papadogiannis et al. (2021) [135] | Prospective single-center cohort study n = 45 (Greece) | Patients with incident IPF underwent Type 1 PSG; 29 patients with moderate to severe OSA were started on CPAP. | Of the 29 patients on CPAP, 11 (38%) had good compliance **. Compared to poor compliance, good compliance group saw improvements in QOL measures before and after CPAP. No survival benefit was seen, except in a sub-group averaging ≥ 6 h usage, 70% of nights. | Poor compliance. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Locke, B.W.; Lee, J.J.; Sundar, K.M. OSA and Chronic Respiratory Disease: Mechanisms and Epidemiology. Int. J. Environ. Res. Public Health 2022, 19, 5473. https://doi.org/10.3390/ijerph19095473
Locke BW, Lee JJ, Sundar KM. OSA and Chronic Respiratory Disease: Mechanisms and Epidemiology. International Journal of Environmental Research and Public Health. 2022; 19(9):5473. https://doi.org/10.3390/ijerph19095473
Chicago/Turabian StyleLocke, Brian W., Janet J. Lee, and Krishna M. Sundar. 2022. "OSA and Chronic Respiratory Disease: Mechanisms and Epidemiology" International Journal of Environmental Research and Public Health 19, no. 9: 5473. https://doi.org/10.3390/ijerph19095473
APA StyleLocke, B. W., Lee, J. J., & Sundar, K. M. (2022). OSA and Chronic Respiratory Disease: Mechanisms and Epidemiology. International Journal of Environmental Research and Public Health, 19(9), 5473. https://doi.org/10.3390/ijerph19095473






