Ecotoxicological Estimation of 4-Cumylphenol, 4-t-Octylphenol, Nonylphenol, and Volatile Leachate Phenol Degradation by the Microscopic Fungus Umbelopsis isabellina Using a Battery of Biotests
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Landfill Leachate Preparation
2.3. Microorganism and Growth Conditions
2.4. Biomass Estimation
2.5. Ecotoxicological Analysis
2.5.1. Microorganism-Based Bioassays
2.5.2. Plant Bioassays
2.5.3. Invertebrate-Based Bioassays
2.5.4. Endocrine Activity Assay
2.6. Statistical Analyses
2.7. Spatial Analyses
3. Results and Discussion
3.1. Ecotoxicological Assessment of Biodegradation Processes
3.1.1. Producers
3.1.2. Consumers
3.1.3. Decomposers
3.1.4. Yeast Screen Assay—Endocrine Activity
3.2. Growth Ability of U. isabellina in Leachate Presence
3.3. VP Utilization by U. isabellina
3.4. Evaluation of Sample Toxicity from U. isabellina Cultures Supplemented with Leachate
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Corvini, P.F.X.; Schäffer, A.; Schlosser, D. Microbial degradation of nonylphenol and other alkylphenols—Our evolving view. Appl. Microbiol. Biotechnol. 2006, 72, 223–243. [Google Scholar] [CrossRef] [PubMed]
- Michałowicz, J.; Duda, W. Phenols—Sources and toxicity. Pol. J. Environ. Stud. 2007, 16, 347–362. [Google Scholar]
- Ramos, R.L.; Moreira, V.R.; Lebron, Y.A.R.; Santos, A.V.; Santos, L.V.S.; Amaral, M.C.S. Phenolic compounds seasonal occurrence and risk assessment in surface and treated waters in Minas Gerais-Brazil. Environ. Pollut. 2021, 268, 115782. [Google Scholar] [CrossRef] [PubMed]
- Soares, A.; Guieysse, B.; Jefferson, B.; Cartmell, E.; Lester, J.N. Nonylphenol in the environment: A critical review on occurrence, fate, toxicity and treatment in wastewaters. Environ. Int. 2008, 34, 1033–1049. [Google Scholar] [CrossRef] [PubMed]
- Oluwasanu, A.A. Fate and Toxicity of Chlorinated Phenols of Environmental Implications: A Review. Med. Anal. Chem. Int. J. 2018, 2, 000126. [Google Scholar] [CrossRef]
- Wang, B.; Dong, F.; Chen, S.; Chen, M.; Bai, Y.; Tan, J.; Li, F.; Wang, Q. Phenolic endocrine disrupting chemicals in an urban receiving river (Panlong river) of Yunnan-Guizhou plateau: Occurrence, bioaccumulation and sources. Ecotoxicol. Environ. Saf. 2016, 128, 133–142. [Google Scholar] [CrossRef]
- Yu, F.; Pan, X.; Wang, B. Determination of four phenolic endocrine disrupting chemicals in Dianchi Lake, China. Int. J. Environ. Anal. Chem. 2012, 92, 1532–1545. [Google Scholar] [CrossRef]
- Im, J.K.; Yu, S.J.; Kim, S.; Kim, S.H.; Noh, H.R.; Kim, M.K. Occurrence, potential sources, and risk assessment of volatile organic compounds in the Han River Basin, South Korea. Int. J. Environ. Res. Public Health 2021, 18, 3727. [Google Scholar] [CrossRef]
- Zhao, J.L.; Huang, Z.; Zhang, Q.Q.; Ying-He, L.; Wang, T.T.; Yang, Y.Y.; Ying, G.G. Distribution and mass loads of xenoestrogens bisphenol a, 4-nonylphenol, and 4-tert-octylphenol in rainfall runoff from highly urbanized regions: A comparison with point sources of wastewater. J. Hazard. Mater. 2021, 401, 123747. [Google Scholar] [CrossRef]
- Xie, W.; Zhao, J.; Zhang, Q.; Ye, C.; Zheng, G.; Shan, Q.; Li, L.; Shao, X. Occurrence, distribution and bioaccumulation of alkylphenols in the Pearl River networks, South China. Ecol. Indic. 2020, 110, 105847. [Google Scholar] [CrossRef]
- Oberoi, A.S.; Philip, L. Variation in toxicity during the biodegradation of various heterocyclic and homocyclic aromatic hydrocarbons in single and multi-substrate systems. Ecotoxicol. Environ. Saf. 2017, 135, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Costa, E.; Piazza, V.; Gambardella, C.; Moresco, R.; Prato, E.; Biandolino, F.; Cassin, D.; Botter, M.; Maurizio, D.; D’Adamo, R.; et al. Ecotoxicological effects of sediments from Mar Piccolo, South Italy: Toxicity testing with organisms from different trophic levels. Environ. Sci. Pollut. Res. 2016, 23, 12755–12769. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Sanderson, H.; Roy, K.; Benfenati, E.; Leszczynski, J. Ecotoxicological assessment of pharmaceuticals and personal care products using predictive toxicology approaches. Green Chem. 2020, 22, 1458–1516. [Google Scholar] [CrossRef]
- Janicki, T.; Długoński, J.; Krupiński, M. Detoxification and simultaneous removal of phenolic xenobiotics and heavy metals with endocrine-disrupting activity by the non-ligninolytic fungus Umbelopsis isabellina. J. Hazard. Mater. 2018, 360, 661–669. [Google Scholar] [CrossRef]
- Szklarek, S.; Kiedrzyńska, E.; Kiedrzyński, M.; Mankiewicz-Boczek, J.; Mitsch, W.J.; Zalewski, M. Comparing ecotoxicological and physicochemical indicators of municipal wastewater effluent and river water quality in a Baltic Sea catchment in Poland. Ecol. Indic. 2021, 126, 107611. [Google Scholar] [CrossRef]
- Louati, I.; Dammak, M.; Nasri, R.; Belbahri, L.; Nasri, M.; Abdelkafi, S.; Mechichi, T. Biodegradation and detoxification of bisphenol A by bacteria isolated from desert soils. 3 Biotech 2019, 9, 228. [Google Scholar] [CrossRef] [PubMed]
- Mtibaà, R.; Ezzanad, A.; Aranda, E.; Pozo, C.; Ghariani, B.; Moraga, J.; Nasri, M.; Manuel Cantoral, J.; Garrido, C.; Mechichi, T. Biodegradation and toxicity reduction of nonylphenol, 4-tert-octylphenol and 2,4-dichlorophenol by the ascomycetous fungus Thielavia sp HJ22: Identification of fungal metabolites and proposal of a putative pathway. Sci. Total Environ. 2020, 708, 135129. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kwon, J.H. Enhancement of toxic efficacy of alkylated polycyclic aromatic hydrocarbons transformed by sphingobium quisquiliarum. Int. J. Environ. Res. Public Health 2020, 17, 6416. [Google Scholar] [CrossRef] [PubMed]
- Janicki, T.; Krupiński, M.; Długoński, J. Degradation and toxicity reduction of the endocrine disruptors nonylphenol, 4-tert-octylphenol and 4-cumylphenol by the non-ligninolytic fungus Umbelopsis isabellina. Bioresour. Technol. 2016, 200, 223–229. [Google Scholar] [CrossRef]
- Martinez-Haro, M.; Beiras, R.; Bellas, J.; Capela, R.; Coelho, J.P.; Lopes, I.; Moreira-Santos, M.; Reis-Henriques, A.M.; Ribeiro, R.; Santos, M.M.; et al. A review on the ecological quality status assessment in aquatic systems using community based indicators and ecotoxicological tools: What might be the added value of their combination? Ecol. Indic. 2015, 48, 8–16. [Google Scholar] [CrossRef]
- Duan, W.; Meng, F.; Lin, Y.; Wang, G. Toxicological effects of phenol on four marine microalgae. Environ. Toxicol. Pharmacol. 2017, 52, 170–176. [Google Scholar] [CrossRef]
- Suda, T.; Hata, T.; Kawai, S.; Okamura, H.; Nishida, T. Treatment of tetracycline antibiotics by laccase in the presence of 1-hydroxybenzotriazole. Bioresour. Technol. 2012, 103, 498–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yousefi-Ahmadipour, A.; Bozorgi-Koshalshahi, M.; Mogharabi, M.; Amini, M.; Ghazi-Khansari, M.; Faramarzi, M.A. Laccase-catalyzed treatment of ketoconazole, identification of biotransformed metabolites, determination of kinetic parameters, and evaluation of micro-toxicity. J. Mol. Catal. B Enzym. 2016, 133, 77–84. [Google Scholar] [CrossRef]
- Inoue, S.; Igarashi, Y.; Yoneda, Y.; Kawai, S.; Okamura, H.; Nishida, T. Elimination and detoxification of fungicide miconazole and antidepressant sertraline by manganese peroxidase-dependent lipid peroxidation system. Int. Biodeterior. Biodegrad. 2015, 100, 79–84. [Google Scholar] [CrossRef]
- Długoński, A. Recreational development of old landfill: The case study of górka rogowska landfill in Łódź City, Poland. Detritus 2018, 2, 155–162. [Google Scholar] [CrossRef]
- Wei, X.; Lyu, S.; Yu, Y.; Wang, Z.; Liu, H.; Pan, D.; Chen, J. Phylloremediation of Air Pollutants: Exploiting the Potential of Plant Leaves and Leaf-Associated Microbes. Front. Plant Sci. 2017, 8, 1318. [Google Scholar] [CrossRef] [PubMed]
- Wieczerzak, M.; Namieśnik, J.; Kudłak, B. Bioassays as one of the Green Chemistry tools for assessing environmental quality: A review. Environ. Int. 2016, 94, 341–361. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, P.; Thakur, I.S.; Kaushik, A. Bioassays for toxicological risk assessment of landfill leachate: A review. Ecotoxicol. Environ. Saf. 2017, 141, 259–270. [Google Scholar] [CrossRef]
- Długoński, A. Revitalisation of degraded urban green areas. In Microbial Biotechnology in the Laboratory and Practice; Długoński, J., Ed.; Łódź University Press & Jagellonian University Press: Łódź-Kraków, Poland, 2021; pp. 297–309. [Google Scholar]
- Baran, A.; Tarnawski, M. Phytotoxkit/Phytotestkit and Microtox® as tools for toxicity assessment of sediments. Ecotoxicol. Environ. Saf. 2013, 98, 19–27. [Google Scholar] [CrossRef]
- Antonkiewicz, J.; Baran, A.; Pełka, R.; Wisła-Świder, A.; Nowak, E.; Konieczka, P. A mixture of cellulose production waste with municipal sewage as new material for an ecological management of wastes. Ecotoxicol. Environ. Saf. 2019, 169, 607–614. [Google Scholar] [CrossRef]
- Szara, M.; Baran, A.; Klimkowicz-Pawlas, A.; Tarnawski, M. Ecotoxicological and chemical properties of the rożnów reservoir bottom sediment amended with various waste materials. J. Environ. Manag. 2020, 273, 111176. [Google Scholar] [CrossRef] [PubMed]
- Gworek, B.; Baczewska Dabrowska, A.H.; Kalinowski, R.; Gorska, E.B.; Rekosz-Burlaga, H.; Gozdowski, D.; Olejniczak, I.; Graniewska, M.; Dmuchowski, W. Ecological risk assessment for land contaminated by petrochemical industry. PLoS ONE 2018, 13, e0204852. [Google Scholar] [CrossRef] [PubMed]
- Sha, H.; Nie, J.; Lian, L.; Yan, S.; Song, W. Phototransformation of an emerging cyanotoxin (Aerucyclamide A) in simulated natural waters. Water Res. 2021, 201, 117339. [Google Scholar] [CrossRef] [PubMed]
- Dries, J.; Daens, D.; Geuens, L.; Blust, R. Evaluation of acute ecotoxicity removal from industrial wastewater using a battery of rapid bioassays. Water Sci. Technol. 2014, 70, 2056–2061. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Ávila, L.; Peidro-Guzmán, H.; Pérez-Llano, Y.; Moreno-Perlín, T.; Sánchez-Reyes, A.; Aranda, E.; Ángeles de Paz, G.; Fernández-Silva, A.; Folch-Mallol, J.L.; Cabana, H.; et al. Tracking gene expression, metabolic profiles, and biochemical analysis in the halotolerant basidiomycetous yeast Rhodotorula mucilaginosa EXF-1630 during benzo[a]pyrene and phenanthrene biodegradation under hypersaline conditions. Environ. Pollut. 2021, 271, 116358. [Google Scholar] [CrossRef] [PubMed]
- Stella, T.; Covino, S.; Čvančarová, M.; Filipová, A.; Petruccioli, M.; D’Annibale, A.; Cajthaml, T. Bioremediation of long-term PCB-contaminated soil by white-rot fungi. J. Hazard. Mater. 2017, 324, 701–710. [Google Scholar] [CrossRef]
- Górny, D.; Guzik, U.; Hupert-Kocurek, K.; Wojcieszyńska, D. Naproxen ecotoxicity and biodegradation by Bacillus thuringiensis B1(2015b) strain. Ecotoxicol. Environ. Saf. 2019, 167, 505–512. [Google Scholar] [CrossRef]
- Baran, W.; Adamek, E.; Jajko, M.; Sobczak, A. Removal of veterinary antibiotics from wastewater by electrocoagulation. Chemosphere 2018, 194, 381–389. [Google Scholar] [CrossRef]
- Park, J.S.; Lee, B.J.; Kang, K.S.; Tai, J.H.; Cho, J.J.; Cho, M.H.; Inoue, T.; Lee, Y.S. Hormonal effects of several chemicals in recombinant yeast, MCF-7 cells and uterotrophic assays in mice. J. Microbiol. Biotechnol. 2000, 10, 293–299. [Google Scholar]
- Preuss, T.G.; Gehrhardt, J.; Schirmer, K.; Coors, A.; Rubach, M.; Russ, A.; Jones, P.D.; Giesy, J.P.; Ratte, H.T. Nonylphenol isomers differ in estrogenic activity. Environ. Sci. Technol. 2006, 40, 5147–5153. [Google Scholar] [CrossRef]
- Uchiyama, T.; Makino, M.; Saito, H.; Katase, T.; Fujimoto, Y. Syntheses and estrogenic activity of 4-nonylphenol isomers. Chemosphere 2008, 73, S60–S65. [Google Scholar] [CrossRef] [PubMed]
- Sohoni, P.; Sumpter, J.P. Several environmental oestrogens are also anti-androgens. J. Endocrinol. 1998, 158, 327–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paris, F.; Balaguer, P.; Térouanne, B.; Servant, N.; Lacoste, C.; Cravedi, J.P.; Nicolas, J.C.; Sultan, C. Phenylphenols, biphenols, bisphenol-A and 4-tert-octylphenol exhibit α and β estrogen activities and antiandrogen activity in reporter cell lines. Mol. Cell. Endocrinol. 2002, 193, 43–49. [Google Scholar] [CrossRef]
- Li, J.; Ma, M.; Wang, Z. In vitro profiling of endocrine disrupting effects of phenols. Toxicol. Vitr. 2010, 24, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Chattopadhyay, S.; Gong, E.Y.; Ahn, R.S.; Lee, K. Antiandrogenic effects of bisphenol A and nonylphenol on the function of androgen receptor. Toxicol. Sci. 2003, 75, 40–46. [Google Scholar] [CrossRef] [Green Version]
- Janas, M.; Zawadzka, A. Assessment of the monitoring of an industrial waste landfill. Ecol. Chem. Eng. S 2018, 25, 659–669. [Google Scholar] [CrossRef] [Green Version]
- Góralczyk-Bińkowska, A.; Długoński, A.; Bernat, P.; Długoński, J.; Jasińska, A. Environmental and molecular approach to dye industry waste degradation by the ascomycete fungus Nectriella pironii. Sci. Rep. 2021, 11, 23829. [Google Scholar] [CrossRef]
- Schieber, A.; Wüst, M. Volatile phenols-important contributors to the aroma of plant-derived foods. Molecules 2020, 25, 4529. [Google Scholar] [CrossRef]
- Guo, Y.; Jud, W.; Weikl, F.; Ghirardo, A.; Junker, R.R.; Polle, A.; Benz, J.P.; Pritsch, K.; Schnitzler, J.-P.; Rosenkranz, M. Volatile organic compound patterns predict fungal trophic mode and lifestyle. Commun. Biol. 2021, 4, 673. [Google Scholar] [CrossRef]
- Kennes, C.; Abubackar, H.N.; Chen, J.; Veiga, M.C. Microorganisms Application for Volatile Compounds Degradation. In Microbial Biodegradation: From Omics to Function and Application; Długoński, J., Ed.; Caister Academic Press: Norfolk, UK, 2016; pp. 183–196. [Google Scholar]
- de Almeida, N.C.; de Faria, A.U.; de Oliveira, V.J.A.; Govone, J.S.; de Angelis, D.d.F. Biodegradation and toxicity of byproducts from the treatment of landfileachate with hydrotalcite. Desalin. Water Treat. 2018, 118, 281–293. [Google Scholar] [CrossRef] [Green Version]
- Paskuliakova, A.; McGowan, T.; Tonry, S.; Touzet, N. Phycoremediation of landfill leachate with the chlorophyte Chlamydomonas sp. SW15aRL and evaluation of toxicity pre and post treatment. Ecotoxicol. Environ. Saf. 2018, 147, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Kalka, J. Landfill leachate toxicity removal in combined treatment with municipal wastewater. Sci. World J. 2012, 2012, 202897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalčíková, G.; Babič, J.; Pavko, A.; Gotvajn, A.Ž. Fungal and enzymatic treatment of mature municipal landfill leachate. Waste Manag. 2014, 34, 798–803. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Adil, M.; Ehtisham-ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Shar, G.A.; Asif Tahir, M.; Iqbal, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar] [CrossRef]
- Melnyk, A.; Kuklińska, K.; Wolska, L.; Namieśnik, J. Chemical pollution and toxicity of water samples from stream receiving leachate from controlled municipal solid waste (MSW) landfill. Environ. Res. 2014, 135, 253–261. [Google Scholar] [CrossRef]
- Kalcikova, G.; Vavrova, M.; Zagorc-Koncan, J.; Zgajnar Gotvajn, A. Evaluation of the hazardous impact of landfill leachates by toxicity and biodegradability tests. Environ. Technol. 2011, 32, 1345–1353. [Google Scholar] [CrossRef]
- Sackey, L.N.A.; Kočí, V.; van Gestel, C.A.M. Ecotoxicological effects on Lemna minor and Daphnia magna of leachates from differently aged landfills of Ghana. Sci. Total Environ. 2020, 698, 134295. [Google Scholar] [CrossRef]
- Schiavo, S.; Oliviero, M.; Chiavarini, S.; Manzo, S. Adverse effects of oxo-degradable plastic leachates in freshwater environment. Environ. Sci. Pollut. Res. 2020, 27, 8586–8595. [Google Scholar] [CrossRef]
- Šourková, M.; Adamcová, D.; Zloch, J.; Skutnik, Z.; Vaverková, M.D. Evaluation of the phytotoxicity of leachate from a municipal solid waste landfill: The case study of bukov landfill. Environments 2020, 7, 111. [Google Scholar] [CrossRef]
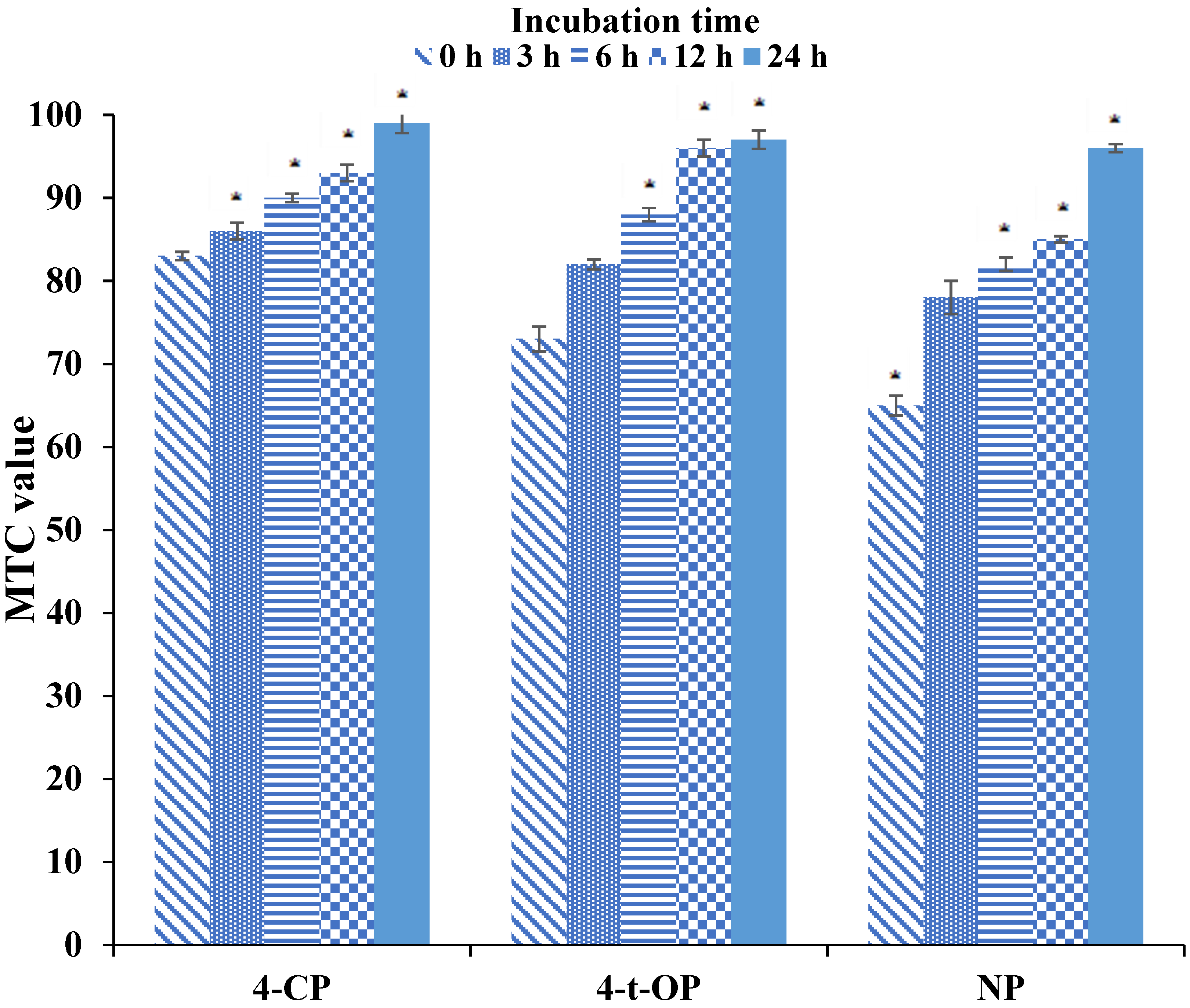

| Xenobiotic | Time (h) | P. tricornutum | R. subcapitata | T. platyurus | A. fischeri |
|---|---|---|---|---|---|
| EC50 | |||||
| 4-CP | 0 | 10.1 ± 0.3 | 22.2 ± 0.5 | 3.6 ± 0.3 | 46.2 ± 1.6 |
| 3 | 9.5 ± 0.5 | 19.7 ± 0.4 | 4.0 ± 0.5 | 59.0 ± 1.8 | |
| 6 | 10.7 ± 0.2 | 20.1 ± 0.4 | 5.0 ± 0.4 | 63.1 ± 2.1 | |
| 12 | 10.9 ± 0.7 | 19.8 ± 0.3 | 7.6 ± 0.5 | 70.4 ± 1.6 | |
| 24 | 16.1 ± 0.7 | 28.6 ± 0.5 | 9.8 ± 0.4 | 74.1 ± 1.5 | |
| 4-t-OP | 0 | 3.3 ± 0.3 | 4.4 ± 0.2 | 10.1 ± 0.7 | 52.6 ± 2.0 |
| 3 | 4.2 ± 0.5 | 4.3 ± 0.3 | 13.2 ± 0.3 | 62.1 ± 1.1 | |
| 6 | 5.1 ± 0.3 | 4.5 ± 0.2 | 14.3 ± 0.6 | 71.1 ± 1.7 | |
| 12 | 3.8 ± 0.4 | 5.8 ± 0.3 | 14.7 ± 0.3 | 75.7 ± 2.0 | |
| 24 | 7.8 ± 0.5 | 6.2 ± 0.1 | 17.9 ± 0.7 | 74.2 ± 1.4 | |
| NP | 0 | 6.5 ± 0.7 | 4.9 ± 0.2 | 4.5 ± 0.4 | 58.3 ± 1.5 |
| 3 | 6.3 ± 0.3 | 5.1 ± 0.3 | 5.9 ± 0.4 | 70.8 ± 1.3 | |
| 6 | 6.0 ± 0.6 | 6.4 ± 0.3 | 5.6 ± 0.2 | 74.2 ± 1.5 | |
| 12 | 7.4 ± 0.4 | 9.1 ± 0.2 | 5.4 ± 0.2 | 65.2 ± 1.7 | |
| 24 | 19.6 ± 0.7 | 12 ± 0.4 | 7.9 ± 0.5 | 72.8 ± 2.1 | |
| Xenobiotic | Incubation Time (h) | Germination Inhibition (PE%) | Root Growth Inhibition (PE%) | Germination Index (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sa 1 | Ss 2 | Ls 3 | Sa 1 | Ss 2 | Ls 3 | Sa 1 | Ss 2 | Ls 3 | ||
| NP | 0 | 100 ± 0 | 0 ± 0 | 0 ± 0 | 100 ± 0 | 59.5 ± 4.0 | 72.5 ± 3.0 | 0 ± 0 | 40.5 ± 1.8 | 27.5 ± 2.2 |
| 3 | 80 ± 5 | 0 ± 0 | 0 ± 0 | 78.9 ± 3.5 | 42.8 ± 2.6 | 67.1 ± 2.9 | 4.2 ± 1.1 | 57.2 ± 2.5 | 32.9 ± 1.9 | |
| 6 | 90 ± 3 | 10 ± 3 | 0 ± 0 | 88.6 ± 2.8 | 25.3 ± 2.7 | 60.3 ± 3.3 | 1.4 ± 0.2 | 67.0 ± 3.0 | 39.8 ± 2.5 | |
| 12 | 100 ± 0 | 0 ± 0 | 0 ± 0 | 100 ± 0 | 25.5 ± 4.5 | 54.5 ± 2.0 | 0 ± 0 | 74.7 ± 3.6 | 45.6 ± 2.8 | |
| 24 | 90 ± 3 | 0 ± 0 | 0 ± 0 | 90.1 ± 3.1 | 21.0 ± 3.5 | 54.1 ± 3.5 | 1.0 ± 0.2 | 79.0 ± 2.8 | 45.9 ± 2.2 | |
| 4-t-OP | 0 | 100 ± 0 | 0 ± 0 | 0 ± 0 | 100 ± 0 | 65.3 ± 3.1 | 92.1 ± 3.5 | 0 ± 0 | 34.8 ± 1.7 | 11.7 ± 1.0 |
| 3 | 90 ± 5 | 0 ± 0 | 0 ± 0 | 99.2 ± 1.0 | 65.8 ± 2.5 | 87.1 ± 4.1 | 0.07 ± 0.1 | 34.2 ± 1.1 | 12.9 ± 1.7 | |
| 6 | 100 ± 0 | 0 ± 0 | 0 ± 0 | 100 ± 0 | 59.9 ± 2.6 | 87.6 ± 2.6 | 0 ± 0 | 40.1 ± 1.0 | 12.4 ± 1.5 | |
| 12 | 90 ± 5 | 0 ± 0 | 0 ± 0 | 97.7 ± 1.0 | 56.0 ± 2.6 | 83.2 ± 3.0 | 0.23 ± 0.1 | 44.0 ± 1.5 | 16.7 ± 1.8 | |
| 24 | 100 ± 0 | 0 ± 0 | 0 ± 0 | 100 ± 0 | 51.1 ± 2.0 | 81.4 ± 2.2 | 0 ± 0 | 48.9 ± 1.8 | 18.5 ± 1.5 | |
| 4-CP | 0 | 90 ± 3 | 0 ± 0 | 0 ± 0 | 98.9 ± 1.2 | 59.9 ± 4.2 | 84.4 ± 4.1 | 0.22 ± 0.1 | 40.6 ± 3.6 | 18.6 ± 2.2 |
| 3 | 90 ± 3 | 0 ± 0 | 10 ± 3 | 95.8 ± 2.7 | 55.5 ± 3.0 | 81.1 ± 3.6 | 0.42 ± 0.1 | 44.5 ± 2.1 | 18.9 ± 3.0 | |
| 6 | 90 ± 0 | 0 ± 0 | 0 ± 0 | 95.2 ± 3.8 | 55.5 ± 3.5 | 77.3 ± 3.0 | 0.48 ± 0.1 | 44.5 ± 2.1 | 22.8 ± 2.5 | |
| 12 | 100 ± 0 | 0 ± 0 | 0 ± 0 | 100 ± 0 | 48.5 ± 4.0 | 76.9 ± 3.3 | 0 ± 0 | 51.5 ± 3.8 | 23.1 ± 2.6 | |
| 24 | 90 ± 3 | 0 ± 0 | 0 ± 0 | 95.3 ± 3.4 | 36.5 ± 3.2 | 63.9 ± 3.6 | 0.45 ± 0.1 | 63.5 ± 4.2 | 36.0 ± 3.1 | |
| Xenobiotic | YES | YAS | Anti-YES | Anti-YAS |
|---|---|---|---|---|
| 4-CP (concentration range: 5000–15 µg L−1) | Estrogenic activity (about 10.000 times lower than estradiol) | Lack of androgenic properties | Lack of anti-estrogenic properties | Weak anti-androgenic activity |
| 4-t-OP (concentration range: 5000–15 µg L−1) | Lack of estrogenic activity | Lack of androgenic properties | Lack of anti-estrogenic properties | Weak anti-androgenic activity |
| NP (concentration range: 200,000–1500 µg L−1) | Estrogenic activity (about 1.000 times lower than estradiol) | Lack of androgenic properties | Lack of anti-estrogenic properties | Lack of anti-androgenic properties |
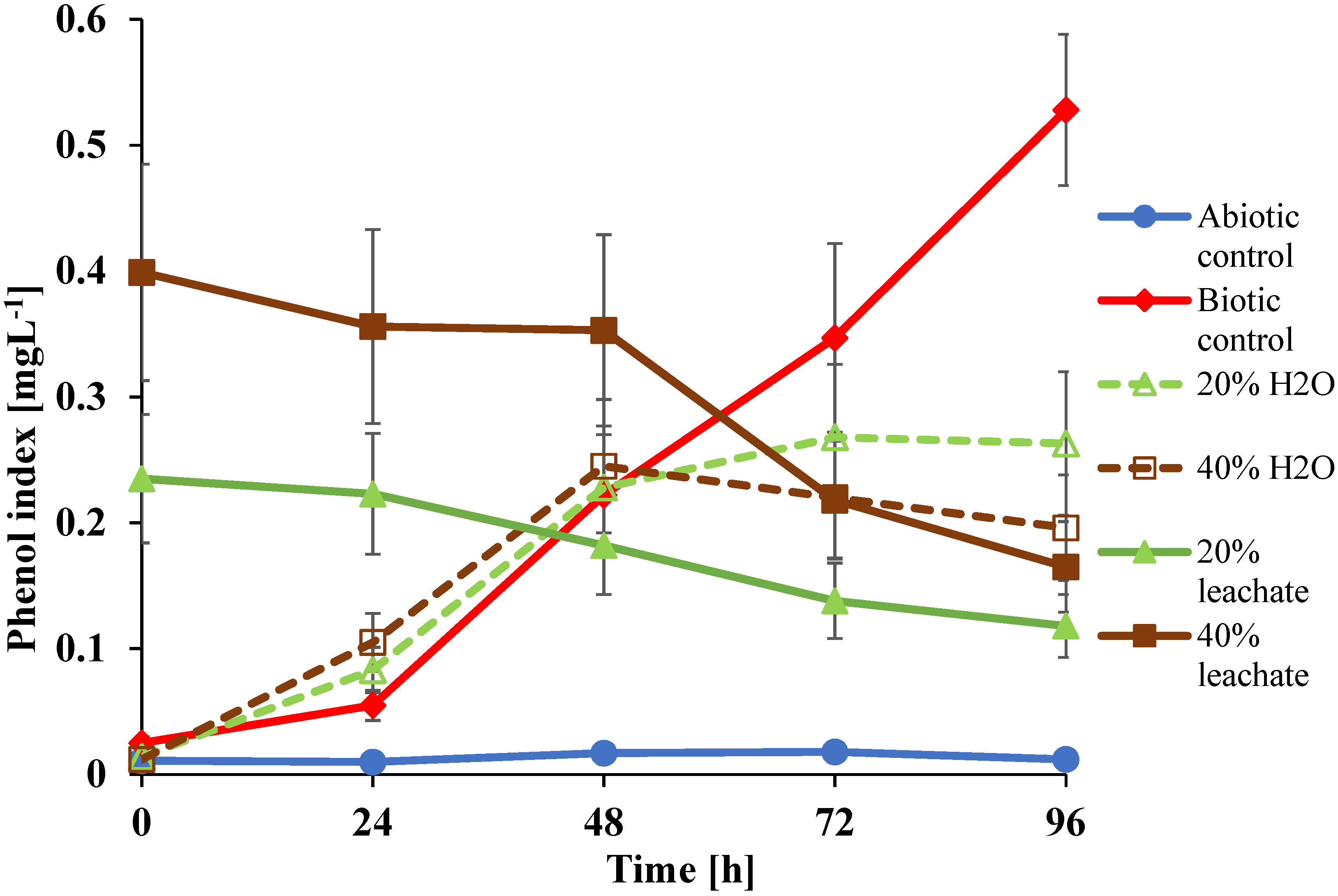
| Landfill Leachate (%) | Time (h) | A. fischeri | A. fransciscana | D. magna |
|---|---|---|---|---|
| 20 | 0 | 34.2 ± 0.6 | 13.4 ± 0.6 | 9.1 ± 0.6 |
| 24 | 40.1 ± 0.8 | 15.6 ± 0.9 | 14.5 ± 1.1 | |
| 48 | 45.1 ± 1.0 | 19.7 ± 1.1 | 26.9 ± 1.4 | |
| 72 | 56.4 ± 0.9 | 25.6 ± 0.5 | 30.4 ± 1.0 | |
| 96 | 66.9 ± 1.3 | 50.4 ± 1.2 | 44.1 ± 1.5 | |
| 40 | 0 | 15.9 ± 0.4 | 7.8 ± 1.0 | 4.4 ± 0.2 |
| 24 | 24.7 ± 0.5 | 10.1 ± 0.9 | 7.2 ± 0.9 | |
| 48 | 28.2 ± 0.9 | 15.2 ± 1.0 | 10.0 ± 0.5 | |
| 72 | 31.1 ± 0.5 | 17.4 ± 1.0 | 11.5 ± 0.3 | |
| 96 | 34.0 ± 1.0 | 28.6 ± 1.5 | 20.2 ± 1.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janicki, T.; Długoński, A.; Felczak, A.; Długoński, J.; Krupiński, M. Ecotoxicological Estimation of 4-Cumylphenol, 4-t-Octylphenol, Nonylphenol, and Volatile Leachate Phenol Degradation by the Microscopic Fungus Umbelopsis isabellina Using a Battery of Biotests. Int. J. Environ. Res. Public Health 2022, 19, 4093. https://doi.org/10.3390/ijerph19074093
Janicki T, Długoński A, Felczak A, Długoński J, Krupiński M. Ecotoxicological Estimation of 4-Cumylphenol, 4-t-Octylphenol, Nonylphenol, and Volatile Leachate Phenol Degradation by the Microscopic Fungus Umbelopsis isabellina Using a Battery of Biotests. International Journal of Environmental Research and Public Health. 2022; 19(7):4093. https://doi.org/10.3390/ijerph19074093
Chicago/Turabian StyleJanicki, Tomasz, Andrzej Długoński, Aleksandra Felczak, Jerzy Długoński, and Mariusz Krupiński. 2022. "Ecotoxicological Estimation of 4-Cumylphenol, 4-t-Octylphenol, Nonylphenol, and Volatile Leachate Phenol Degradation by the Microscopic Fungus Umbelopsis isabellina Using a Battery of Biotests" International Journal of Environmental Research and Public Health 19, no. 7: 4093. https://doi.org/10.3390/ijerph19074093
APA StyleJanicki, T., Długoński, A., Felczak, A., Długoński, J., & Krupiński, M. (2022). Ecotoxicological Estimation of 4-Cumylphenol, 4-t-Octylphenol, Nonylphenol, and Volatile Leachate Phenol Degradation by the Microscopic Fungus Umbelopsis isabellina Using a Battery of Biotests. International Journal of Environmental Research and Public Health, 19(7), 4093. https://doi.org/10.3390/ijerph19074093






