Factors Associated with Polyp Detection Rate in European Colonoscopy Practice: Findings of The European Colonoscopy Quality Investigation (ECQI) Group
Abstract
1. Introduction
2. Materials and Methods
2.1. Questionnaire Development
2.2. Recruitment
2.3. Ethics
2.4. Dataset
2.5. Polyp Detection Rate
2.6. Statistical Analysis
3. Results
Polyp Detection Rate
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Murchie, B.; Tandon, K.; Zackria, S.; Wexner, S.D.; O’Rourke, C.; Castro, F.J. Can polyp detection rate be used prospectively as a marker of adenoma detection rate? Surg. Endosc. 2018, 32, 1141–1148. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, M.F.; Wieszczy, P.; Kolacz, A.; Rupinski, M.; Franczyk, R.; Rupinska, M.; Bretthauer, M.; Regula, J. Tu1006 Comparison of Quality Measures for Detection of Neoplasia at Screening Colonoscopy. Gastrointest. Endosc. 2016, 83, AB527. [Google Scholar] [CrossRef]
- Spada, C.; Koulaouzidis, A.; Hassan, C.; Amaro, P.; Agrawal, A.; Brink, L.; Fischbach, W.; Hünger, M.; Jover, R.; Kinnunen, U.; et al. Colonoscopy quality across Europe: A report of the European Colonoscopy Quality Investigation (ECQI) Group. Endosc. Int. Open 2021, 9, E1456–E1462. [Google Scholar] [CrossRef]
- Kaminski, M.F.; Thomas-Gibson, S.; Bugajski, M.; Bretthauer, M.; Rees, C.J.; Dekker, E.; Hoff, G.; Jover, R.; Suchanek, S.; Ferlitsch, M.; et al. Performance measures for lower gastrointestinal endoscopy: A European Society of Gastrointestinal Endoscopy (ESGE) quality improvement initiative. United Eur. Gastroenterol. J. 2017, 5, 309–334. [Google Scholar] [CrossRef] [PubMed]
- Rembacken, B.; Hassan, C.; Riemann, J.F.; Chilton, A.; Rutter, M.; Dumonceau, J.M.; Omar, M.; Ponchon, T. Quality in screening colonoscopy: Position statement of the European Society of Gastrointestinal Endoscopy (ESGE). Endoscopy 2012, 44, 957–968. [Google Scholar] [CrossRef] [PubMed]
- Riemann, J.F.; Agrawal, A.; Amaro, P.; Brink, L.; Fischbach, W.; Hünger, M.; Jover, R.; Ono, A.; Toth, E.; Spada, C. Adoption of colonoscopy quality measures across Europe: The European Colonoscopy Quality Investigation (ECQI) Group experience. United Eur. Gastroenterol. J. 2018, 6, 1106–1107. [Google Scholar] [CrossRef] [PubMed]
- Tribonias, G.; Theodoropoulou, A.; Konstantinidis, K.; Vardas, E.; Karmiris, K.; Chroniaris, N.; Chlouverakis, G.; Paspatis, G.A. Comparison of standard vs. high-definition, wide-angle colonoscopy for polyp detection: A randomized controlled trial. Color. Dis. 2010, 12, e260–e266. [Google Scholar] [CrossRef]
- Mills, D.; Spyratos, T.; Haider, A.; Sulo, S.; Laurie, T. Use of wide-screen, high-definition monitors for improving adenoma detection: A prospective, randomized, controlled trial. Dig. Endosc. 2016, 28, 577–582. [Google Scholar] [CrossRef]
- East, J.E.; Stavrindis, M.; Thomas-Gibson, S.; Guenther, T.; Tekkis, P.P.; Saunders, B.P. A comparative study of standard vs. high definition colonoscopy for adenoma and hyperplastic polyp detection with optimized withdrawal technique. Aliment. Pharm. Ther. 2008, 28, 768–776. [Google Scholar] [CrossRef]
- Pellisé, M.; Fernández-Esparrach, G.; Cárdenas, A.; Sendino, O.; Ricart, E.; Vaquero, E.; Gimeno-García, A.Z.; de Miguel, C.R.; Zabalza, M.; Ginès, A.; et al. Impact of wide-angle, high-definition endoscopy in the diagnosis of colorectal neoplasia: A randomized controlled trial. Gastroenterology 2008, 135, 1062–1068. [Google Scholar] [CrossRef]
- Subramanian, V.; Mannath, J.; Hawkey, C.J.; Ragunath, K. High definition colonoscopy vs. standard video endoscopy for the detection of colonic polyps: A meta-analysis. Endoscopy 2011, 43, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Roelandt, P.; Demedts, I.; Willekens, H.; Bessissow, T.; Braeye, L.; Coremans, G.; Cuyle, P.J.; Ferrante, M.; Gevers, A.M.; Hiele, M.; et al. Impact of endoscopy system, high definition, and virtual chromoendoscopy in daily routine colonoscopy: A randomized trial. Endoscopy 2019, 51, 237–243. [Google Scholar] [CrossRef]
- Buchner, A.M.; Shahid, M.W.; Heckman, M.G.; McNeil, R.B.; Cleveland, P.; Gill, K.R.; Schore, A.; Ghabril, M.; Raimondo, M.; Gross, S.A.; et al. High-definition colonoscopy detects colorectal polyps at a higher rate than standard white-light colonoscopy. Clin. Gastroenterol. Hepatol. 2010, 8, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Banks, M.R.; Haidry, R.; Butt, M.A.; Whitley, L.; Stein, J.; Langmead, L.; Bloom, S.L.; O’Bichere, A.; McCartney, S.; Basherdas, K.; et al. High resolution colonoscopy in a bowel cancer screening program improves polyp detection. World J. Gastroenterol. 2011, 17, 4308–4313. [Google Scholar] [CrossRef]
- Jrebi, N.Y.; Hefty, M.; Jalouta, T.; Ogilvie, J.; Davis, A.T.; Asgeirsson, T.; Luchtefeld, M. High-definition colonoscopy increases adenoma detection rate. Surg. Endosc. 2017, 31, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.; Thaventhiran, A.; Mackenzie, H.; Stubbs, B. The use of high definition colonoscopy versus standard definition: Does it affect polyp detection rate? Surg. Endosc. 2018, 32, 2676–2682. [Google Scholar] [CrossRef] [PubMed]
- Burke, C.A.; Choure, A.G.; Sanaka, M.R.; Lopez, R. A comparison of high-definition versus conventional colonoscopes for polyp detection. Dig. Dis Sci. 2010, 55, 1716–1720. [Google Scholar] [CrossRef] [PubMed]
- Waldmann, E.; Britto-Arias, M.; Gessl, I.; Heinze, G.; Salzl, P.; Sallinger, D.; Trauner, M.; Weiss, W.; Ferlitsch, A.; Ferlitsch, M. Endoscopists with low adenoma detection rates benefit from high-definition endoscopy. Surg. Endosc. 2015, 29, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Pioche, M.; Denis, A.; Allescher, H.D.; Andrisani, G.; Costamagna, G.; Dekker, E.; Fockens, P.; Gerges, C.; Groth, S.; Kandler, J.; et al. Impact of 2 generational improvements in colonoscopes on adenoma miss rates: Results of a prospective randomized multicenter tandem study. Gastrointest. Endosc. 2018, 88, 107–116. [Google Scholar] [CrossRef]
- Zimmermann-Fraedrich, K.; Groth, S.; Sehner, S.; Schubert, S.; Aschenbeck, J.; Mayr, M.; Aminalai, A.; Schröder, A.; Bruhn, J.P.; Bläker, M.; et al. Effects of two instrument-generation changes on adenoma detection rate during screening colonoscopy: Results from a prospective randomized comparative study. Endoscopy 2018, 50, 878–885. [Google Scholar] [CrossRef]
- Lee, J.Y.; Koh, M.; Lee, J.H. Latest Generation High-Definition Colonoscopy Increases Adenoma Detection Rate by Trainee Endoscopists. Dig. Dis Sci. 2021, 66, 2756–2762. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ou, Y.; Yue, H.; Sun, H.; Chen, P.; Liu, S.; He, F.; Peng, Q.; Deng, S. Comparison of the detection of colorectal lesions in different endoscopic modalities: A network meta-analysis and systematic review. Exp. Ther. Med. 2019, 18, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Facciorusso, A.; Triantafyllou, K.; Murad, M.H.; Prokop, L.J.; Tziatzios, G.; Muscatiello, N.; Singh, S. Compared Abilities of Endoscopic Techniques to Increase Colon Adenoma Detection Rates: A Network Meta-analysis. Clin. Gastroenterol. Hepatol. 2019, 17, 2439–2454.e2425. [Google Scholar] [CrossRef]
- Di Caro, S.; Fini, L.; Vega, R.; Fragkos, K.C.; Dolwani, S.; Green, J.; Smith, L.A.; Beckett, C.; Cameron, E.; Banks, M. Multicentre randomised controlled trial comparing standard and high resolution optical technologies in colorectal cancer screening. Front. Gastroenterol. 2019, 10, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, A.; Early, D.S.; Gupta, N.; Bansal, A.; Singh, V.; Ansstas, M.; Jonnalagadda, S.S.; Hovis, C.E.; Gaddam, S.; Wani, S.B.; et al. Randomized, controlled trial of standard-definition white-light, high-definition white-light, and narrow-band imaging colonoscopy for the detection of colon polyps and prediction of polyp histology. Gastrointest. Endosc. 2011, 74, 593–602. [Google Scholar] [CrossRef]
- Neumann, H.; Rerknimitr, R. Does the “polyp paradox” really exist? Dig. Dis Sci. 2014, 59, 2611–2612. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gómez, V.; Racho, R.G.; Heckman, M.G.; Diehl, N.N.; Wallace, M.B. High-definition white-light (HDWL) colonoscopy and higher adenoma detection rate and the potential for paradoxical over surveillance. Dig. Dis Sci. 2014, 59, 2749–2756. [Google Scholar] [CrossRef] [PubMed]
- Kamiński, M.F.; Hassan, C.; Bisschops, R.; Pohl, J.; Pellisé, M.; Dekker, E.; Ignjatovic-Wilson, A.; Hoffman, A.; Longcroft-Wheaton, G.; Heresbach, D.; et al. Advanced imaging for detection and differentiation of colorectal neoplasia: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2014, 46, 435–449. [Google Scholar] [CrossRef] [PubMed]
- Bisschops, R.; East, J.E.; Hassan, C.; Hazewinkel, Y.; Kamiński, M.F.; Neumann, H.; Pellisé, M.; Antonelli, G.; Bustamante Balen, M.; Coron, E.; et al. Advanced imaging for detection and differentiation of colorectal neoplasia: European Society of Gastrointestinal Endoscopy (ESGE) Guideline—Update 2019. Endoscopy 2019, 51, 1155–1179. [Google Scholar] [CrossRef]
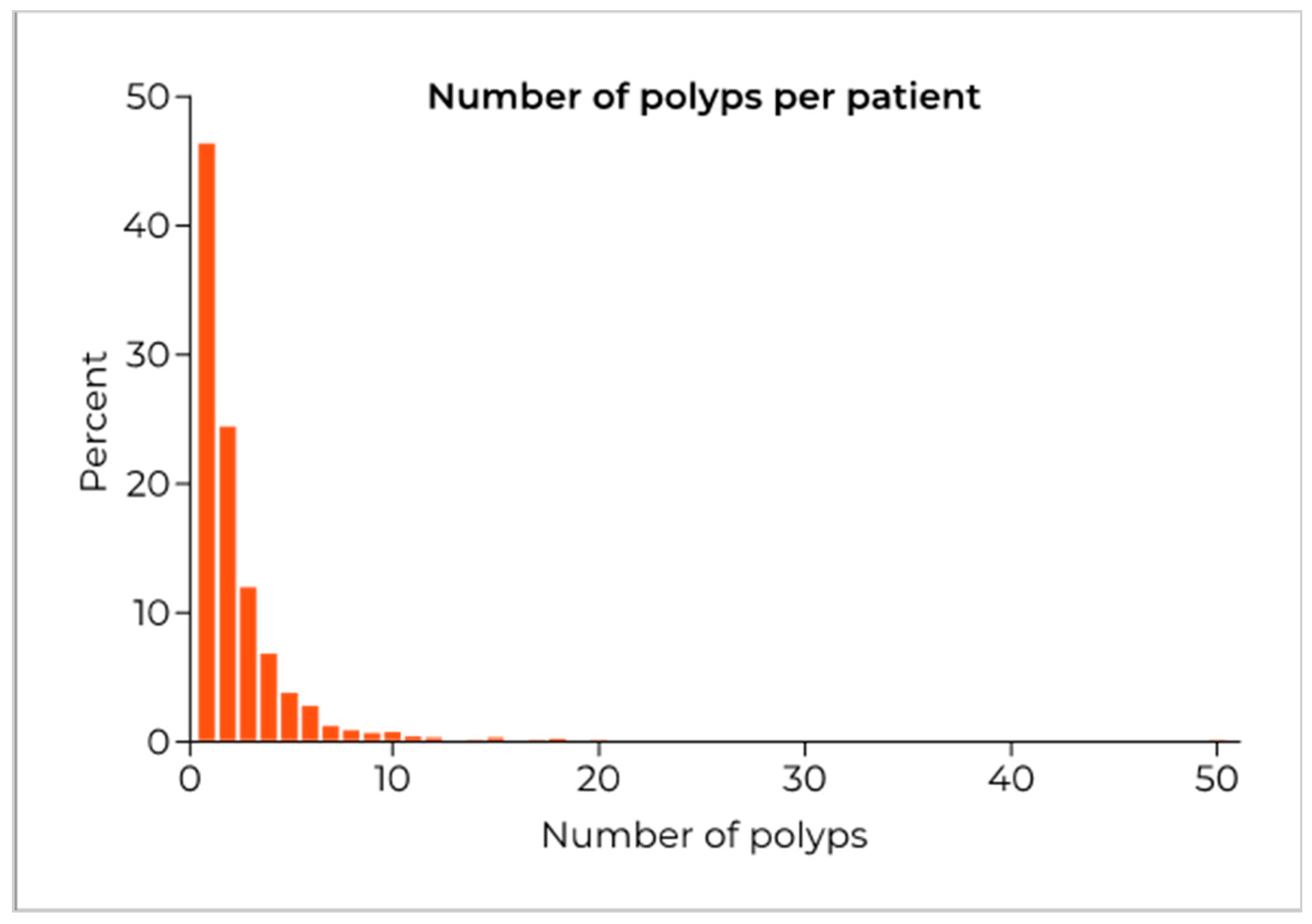
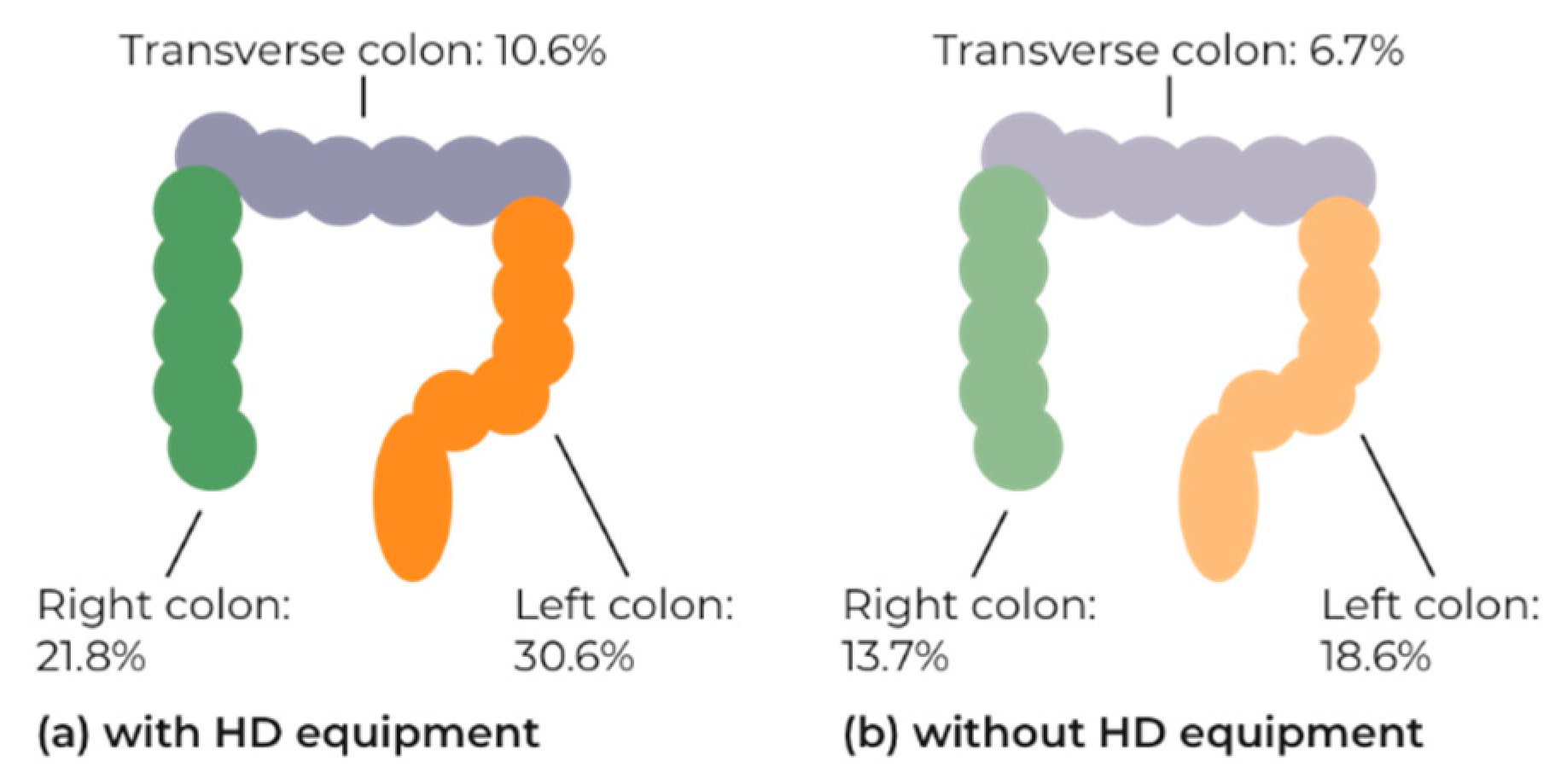
| Variable | Number with Polyp Detected (%) | Odds Ratio (95%CI) | p Value | p Value for Variable |
|---|---|---|---|---|
| Age, years | <0.001 | |||
| 50–59 | 319/1007 (31.7) | Reference | ||
| 60–69 | 556/1197 (46.4) | 1.871 (1.571, 2.228) | <0.001 | |
| 70–79 | 385/905 (42.5) | 1.597 (1.324, 1.925) | <0.001 | |
| ≥80 | 103/256 (40.2) | 1.452 (1.094, 1.927) | 0.01 | |
| Missing data | None | |||
| BMI, kg/m2 | <0.001 | |||
| <18.5 | 17/58 (29.3) | 0.688 (0.386, 1.228) | 0.206 | |
| 18.5–25 | 402/1069 (37.6) | Reference | ||
| 25–30 | 476/1040 (45.8) | 1.400 (1.177, 1.666) | <0.001 | |
| 30–35 | 188/344 (54.7) | 2.000 (1.564, 2.556) | <0.001 | |
| >35 | 53/108 (49.1) | 1.599 (1.075, 2.378) | 0.021 | |
| Missing data | 746 | |||
| Gender | <0.001 | |||
| Female | 613/1771 (34.6) | Reference | ||
| Male | 750/1594 (47.1) | 1.679 (1.461, 1.929) | <0.001 | |
| Missing data | None | |||
| Patient type | 0.368 | |||
| Outpatient | 1043/2561 (40.7) | Reference | ||
| Inpatient | 166/432 (38.4) | 0.908 (0.737, 1.120) | 0.368 | |
| Missing data | 372 | |||
| Reason for procedure | <0.001 | |||
| Diagnosis | 821/2304 (35.6) | Reference | ||
| Screening | 542/1061 (51.1) | 1.886 (1.628, 2.186) | <0.001 | |
| Missing data | None | |||
| BBPS | 0.246 | |||
| BBPS < 6 (inadequate) | 172/465 (38.7) | Reference | ||
| BBPS ≥ 6 (adequate) | 1166/2805 (41.6) | 1.129 (0.920, 1.386) | 0.246 | |
| Missing data | 95 | |||
| Previous total colonoscopy in last 5 years | 0.914 | |||
| No | 981/2431 (40.4) | Reference | ||
| Yes | 227/566 (40.1) | 0.990 (0.821, 1.193) | 0.914 | |
| Missing data | 368 | |||
| Sedation used | 0.019 | |||
| No | 381/1012 (37.6) | Reference | ||
| Yes | 980/2334 (42.0) | 1.199 (1.030, 1.395) | 0.019 | |
| Missing data | 19 | |||
| High-definition equipment used | <0.001 | |||
| No | 250/820 (30.5) | Reference | ||
| Yes | 959/2172 (44.2) | 1.802 (1.519, 2.139) | <0.001 | |
| Missing data | 373 | |||
| Assistive technology used | 0.015 | |||
| No | 1100/2774 (39.7) | Reference | ||
| Yes | 136/289 (47.1) | 1.353 (1.061, 1.725) | 0.015 | |
| Missing data | 302 | |||
| Colonoscopy endpoint | 0.089 | |||
| Terminal ileum/neo terminal ileum | 354/806 (43.9) | Reference | ||
| Anastomosis | 5/17 (29.4) | 0.532 (0.186, 1.524) | 0.24 | |
| Caecum | 997/2494 (40.0) | 0.850 (0.724, 0.998) | 0.048 | |
| Missing data | 48 | |||
| Colonoscopy endpoint reached | <0.001 | |||
| No | 37/159 (23.3) | Reference | ||
| Yes | 1309/3147 (41.6) | 2.348 (1.615, 3.415) | <0.001 | |
| Missing data | 59 | |||
| Time colonoscopy performed | 0.622 | |||
| 07:00–11:59 (morning) | 504/1261 (40.0) | Reference | ||
| 12:00–17:59 (afternoon) | 434/1035 (41.9) | 1.085 (0.918, 1.282) | 0.34 | |
| 18:00–19:59 (evening) | 33/83 (39.8) | 0.991 (0.630, 1.561) | 0.97 | |
| Missing data | 986 | |||
| Rank | Variable | Response (n) | Proportion Reporting Polyp | Odds Ratio (95%CI) | p Value |
|---|---|---|---|---|---|
| 1 | Use of HD equipment | No (441) | 30.8% | Reference | <0.0001 |
| Yes (1007) | 52.7% | 2.501 (1.973, 3.170) | |||
| 2 | BMI category | <18.5 kg/m2 (33) | 30.3% | 0.632 (0.296, 1.350) | <0.0001 |
| 18.5 < BMI ≤ 25 kg/m2 (618) | 40.8% | Reference | |||
| 25 < BMI ≤ 30 kg/m2 (565) | 46.4% | 1.256 (0.997, 1.581) | |||
| 30 < BMI ≤ 35 kg/m2 (176) | 63.6% | 2.542 (1.797, 3.594) | |||
| >35 kg/m2 (56) | 55.4% | 1.801 (1.038, 3.124) | |||
| 3 | Gender | Female (760) | 40.7% | Reference | <0.0001 |
| Male (688) | 52.0% | 1.583 (1.286, 1.950) | |||
| 4 | Age group | 50–59 years (446) | 36.1% | Reference | <0.0001 |
| 60–69 years (540) | 53.0% | 1.993 (1.542, 2.576) | |||
| 70–79 years (355) | 48.2% | 1.645 (1.238, 2.185) | |||
| ≥80 years (107) | 45.8% | 1.495 (0.976, 2.291) | |||
| 5 | Reason for procedure | Diagnosis (839) | 40.9% | Reference | 0.0002 |
| Screening (609) | 53.2% | 1.644 (1.322, 2.029) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spada, C.; Koulaouzidis, A.; Hassan, C.; Amaro, P.; Agrawal, A.; Brink, L.; Fischbach, W.; Hünger, M.; Jover, R.; Kinnunen, U.; et al. Factors Associated with Polyp Detection Rate in European Colonoscopy Practice: Findings of The European Colonoscopy Quality Investigation (ECQI) Group. Int. J. Environ. Res. Public Health 2022, 19, 3388. https://doi.org/10.3390/ijerph19063388
Spada C, Koulaouzidis A, Hassan C, Amaro P, Agrawal A, Brink L, Fischbach W, Hünger M, Jover R, Kinnunen U, et al. Factors Associated with Polyp Detection Rate in European Colonoscopy Practice: Findings of The European Colonoscopy Quality Investigation (ECQI) Group. International Journal of Environmental Research and Public Health. 2022; 19(6):3388. https://doi.org/10.3390/ijerph19063388
Chicago/Turabian StyleSpada, Cristiano, Anastasios Koulaouzidis, Cesare Hassan, Pedro Amaro, Anurag Agrawal, Lene Brink, Wolfgang Fischbach, Matthias Hünger, Rodrigo Jover, Urpo Kinnunen, and et al. 2022. "Factors Associated with Polyp Detection Rate in European Colonoscopy Practice: Findings of The European Colonoscopy Quality Investigation (ECQI) Group" International Journal of Environmental Research and Public Health 19, no. 6: 3388. https://doi.org/10.3390/ijerph19063388
APA StyleSpada, C., Koulaouzidis, A., Hassan, C., Amaro, P., Agrawal, A., Brink, L., Fischbach, W., Hünger, M., Jover, R., Kinnunen, U., Ono, A., Patai, Á., Pecere, S., Petruzziello, L., Riemann, J. F., Staines, H., Stringer, A. L., Toth, E., Antonelli, G., ... on behalf of the ECQI Group. (2022). Factors Associated with Polyp Detection Rate in European Colonoscopy Practice: Findings of The European Colonoscopy Quality Investigation (ECQI) Group. International Journal of Environmental Research and Public Health, 19(6), 3388. https://doi.org/10.3390/ijerph19063388









