Human Biomonitoring Data in Health Risk Assessments Published in Peer-Reviewed Journals between 2016 and 2021: Confronting Reality after a Preliminary Review
Abstract
:1. Introduction
- Are fundamental elements of HRA [5] considered in the publications on the practical integration of HBM data and HRA?
- In which HRA elements is the use of HBM data clearly demonstrated and reported?
2. Materials and Methods
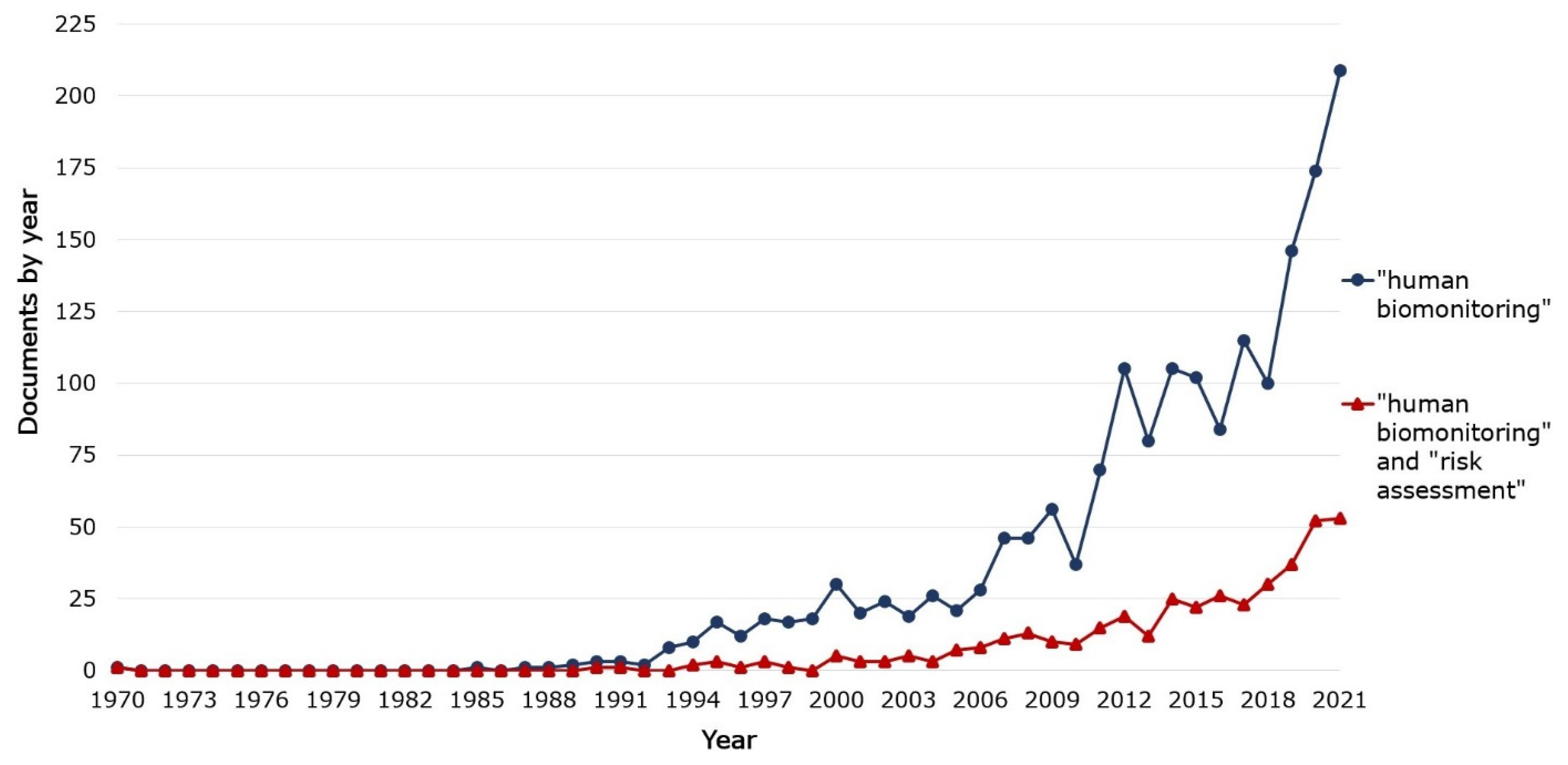
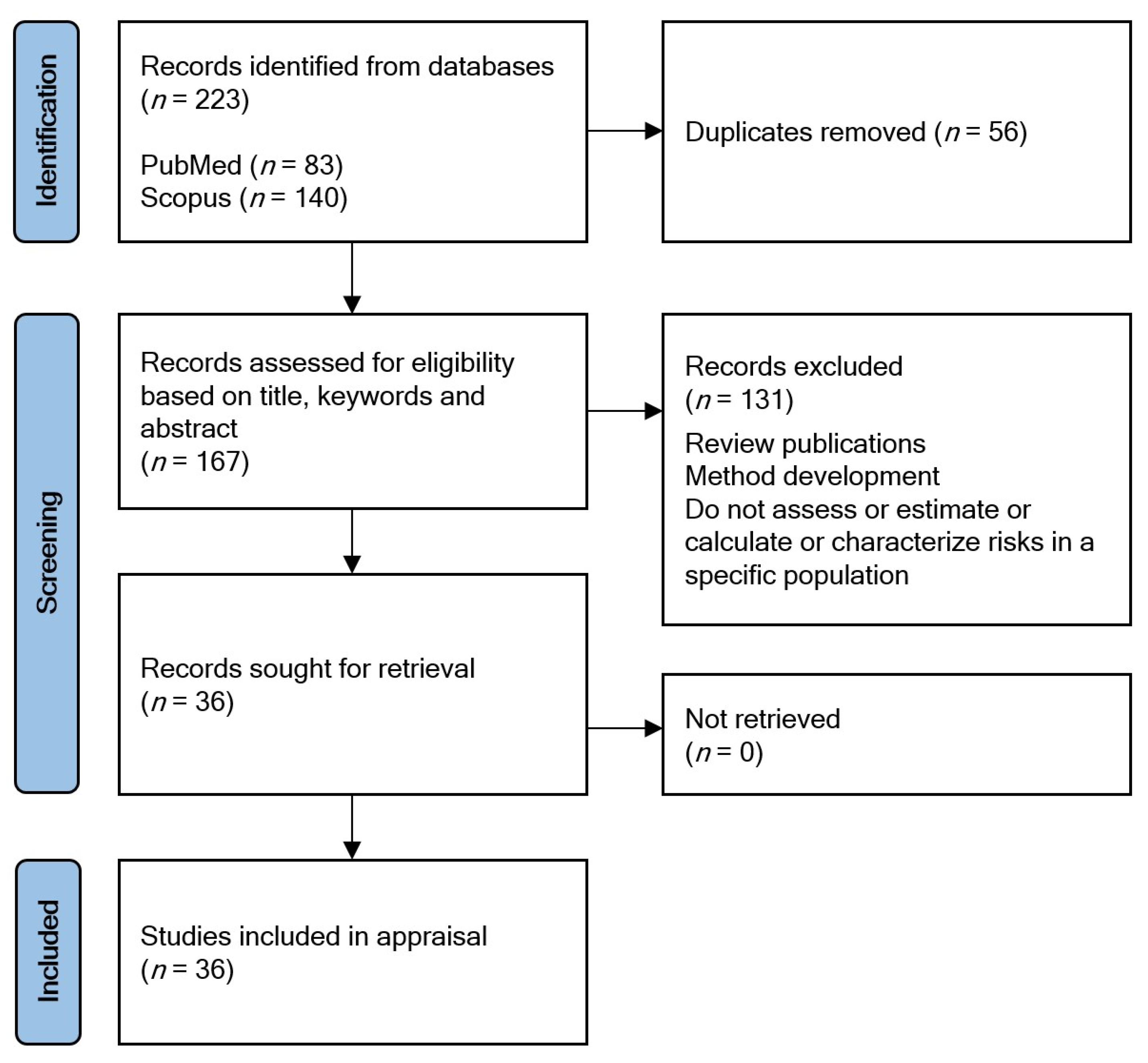
2.1. Publication Search
- PubMed: (((“risk assessment” [Title/Abstract] OR “HRA” [All Fields]) AND (“HBM” [Title/Abstract] OR “human biomonitoring” [Title/Abstract])) AND (y_5[Filter])) NOT (review [Title/Abstract]) Filters: in the last 5 years
- Scopus: (TITLE-ABS-KEY (“risk assessment” OR hra) AND TITLE-ABS-KEY (“Human biomonitoring” OR hbm) AND NOT TITLE-ABS-KEY (review)) AND (LIMIT-TO (PUBYEAR, 2021) OR LIMIT-TO (PUBYEAR, 2020) OR LIMIT-TO (PUBYEAR, 2019) OR LIMIT-TO (PUBYEAR, 2018) OR LIMIT-TO (PUBYEAR, 2017) OR LIMIT-TO (PUBYEAR, 2016)) AND (LIMIT-TO (DOCTYPE, “ar”)) AND (LIMIT-TO (EXACTKEYWORD, “Risk Assessment”)).
2.2. Appraisal Tool
3. Results
4. Discussion
4.1. Dose/Exposure-Resopnse and Exposure Assessment
4.2. Process and Results of Health Risk Assessment, and Risk Management
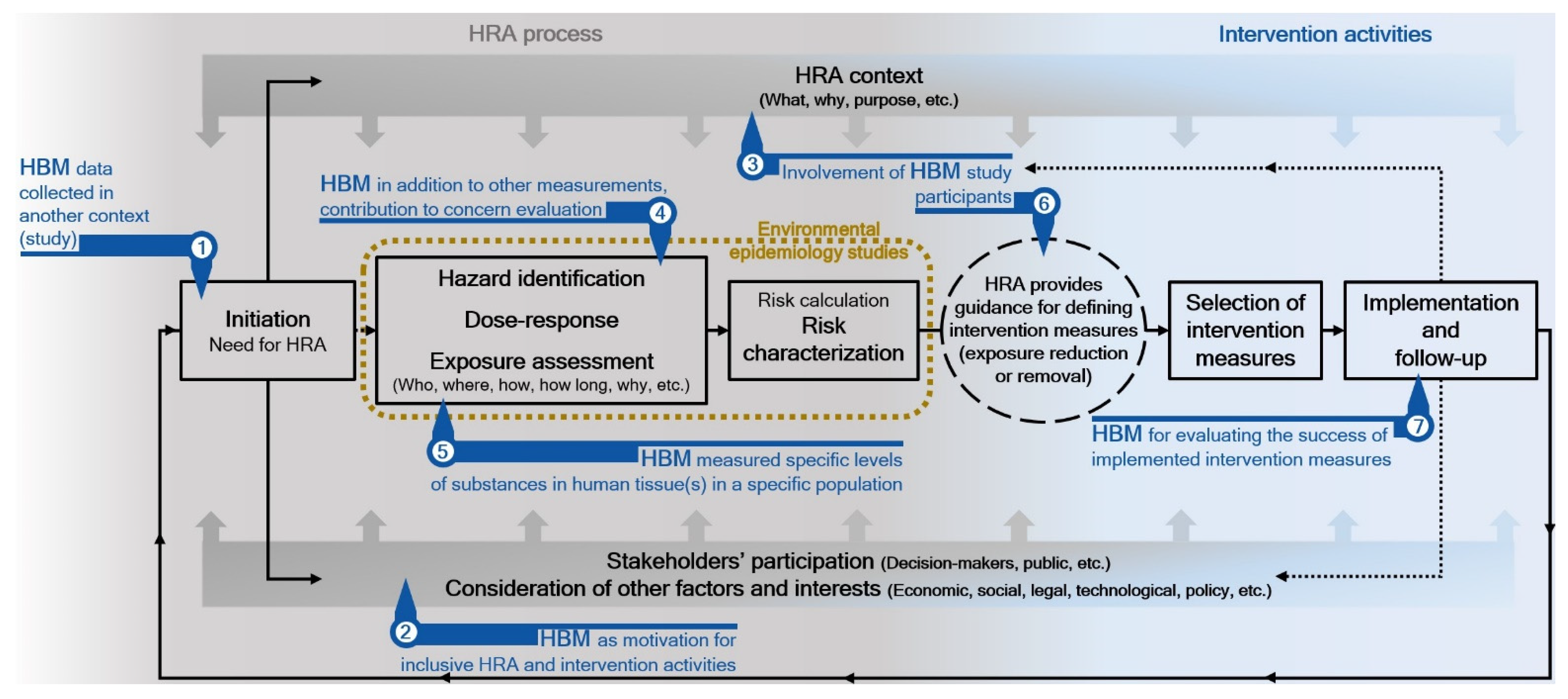
4.3. Value of Human Biomonitoring Data for Health Risk Assessment and Risk Management
4.4. Strengths and Weaknesses
5. Conclusions
- Stakeholder involvement in the early stages of HRA is crucial for the clarification of an assessment context. Clear assessment context assures that HRA can address the needs or concerns of decision makers or other stakeholders. HBM, if performed for the purpose of HRA, must acknowledge the assessment context in its planning stages.
- The lack of stakeholder involvement (e.g., when using existing databases) needs to be reported along with the discussion about the usefulness of obtained HRA results for specific purposes.
- The use of the term “risk assessment” creates confusion/false expectations among decision makers or other stakeholders if only parts of the HRA process are practiced.
- Underlying assumptions of HRA (e.g., related to HBM based exposure assessment, lacking pharmacokinetic knowledge, etc.) must be reported and thoroughly discussed, since they can be an important source of uncertainty or study limitations.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ganzleben, C.; Antignac, J.-P.; Barouki, R.; Castaño, A.; Fiddicke, U.; Klánová, J.; Lebret, E.; Olea, N.; Sarigiannis, D.; Schoeters, G.R.; et al. Human Biomonitoring as a Tool to Support Chemicals Regulation in the European Union. Int. J. Hyg. Environ. Health 2017, 220, 94–97. [Google Scholar] [CrossRef] [PubMed]
- Sexton, K.; Selevan, S.G.; Wagener, D.K.; Lybarger, J.A. Estimating Human Exposures to Environmental Pollutants: Availability and Utility of Existing Databases. Arch. Environ. Health 1992, 47, 398–407. [Google Scholar] [CrossRef]
- National Research Council. Risk Assessment in the Federal Government: Managing the Process; The National Academies Press: Washington, DC, USA, 1983; ISBN 978-0-309-03349-7. [Google Scholar]
- Krewski, D.; Westphal, M.; Andersen, M.E.; Paoli, G.M.; Chiu, W.A.; Al-Zoughool, M.; Croteau, M.C.; Burgoon, L.D.; Cote, I. A Framework for the Next Generation of Risk Science. Environ. Health Perspect. 2014, 122, 796–805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- United States Environmental Protection Agency. Framework for Human Health Risk Assessment to Inform Decision Making; United States Environmental Protection Agency: Washington, DC, USA, 2014; ISBN EPA/100/R-14/001. [Google Scholar]
- National Research Council. Science and Decisions: Advancing Risk Assessment; The National Academies Press: Washington, DC, USA, 2009; ISBN 0309120462. [Google Scholar]
- Society for Risk Analysis Core Subjects of Risk Analysis. Available online: https://www.sra.org/risk-analysis-overview/core-subjects/ (accessed on 4 August 2020).
- Engström, A.; Michaëlsson, K.; Vahter, M.; Julin, B.; Wolk, A.; Åkesson, A. Associations between Dietary Cadmium Exposure and Bone Mineral Density and Risk of Osteoporosis and Fractures among Women. Bone 2012, 50, 1372–1378. [Google Scholar] [CrossRef] [PubMed]
- Tassinari, R.; Tait, S.; Busani, L.; Martinelli, A.; Valeri, M.; Gastaldelli, A.; Deodati, A.; La Rocca, C.; Maranghi, F.; LIFE PERSUADED Project Group. Toxicological Assessment of Oral Co-Exposure to Bisphenol A (BPA) and Bis(2-Ethylhexyl) Phthalate (DEHP) in Juvenile Rats at Environmentally Relevant Dose Levels: Evaluation of the Synergic, Additive or Antagonistic Effects. Int. J. Environ. Res. Public Health 2021, 18, 4584. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.B.; Aggarwal, M.; Bartels, M.J.; Morriss, A.; Terry, C.; Lord, G.A.; Gant, T.W. PBTK Model for Assessment of Operator Exposure to Haloxyfop Using Human Biomonitoring and Toxicokinetic Data. Regul. Toxicol. Pharmacol. 2019, 102, 1–12. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Human Biomonitoring for Environmental Chemicals; The National Academies Press: Washington, DC, USA, 2006; ISBN 978-0-309-10272-8. [Google Scholar]
- Sexton, K.; Callahan, M.A.; Bryan, E.F. Estimating Exposure and Dose to Characterize Health Risks: The Role of Human Tissue Monitoring in Exposure Assessment. Environ. Health Perspect. 1995, 103, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Louro, H.; Heinälä, M.; Bessems, J.; Buekers, J.; Vermeire, T.; Woutersen, M.; van Engelen, J.; Borges, T.; Rousselle, C.; Ougier, E.; et al. Human Biomonitoring in Health Risk Assessment in Europe: Current Practices and Recommendations for the Future. Int. J. Hyg. Environ. Health 2019, 222, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Albertini, R.; Bird, M.; Doerrer, N.; Needham, L.; Robinson, S.; Sheldon, L.; Zenick, H. The Use of Biomonitoring Data in Exposure and Human Health Risk Assessments. Environ. Health Perspect. 2006, 114, 1755–1762. [Google Scholar] [CrossRef] [PubMed]
- Schulte, P.A.; Waters, M. Using Molecular Epidemiology in Assessing Exposure for Risk Assessment. Ann. N. Y. Acad. Sci. 1999, 895, 101–111. [Google Scholar] [CrossRef]
- World Health Organization; International Programme on Chemical Safety. Biomarkers in Risk Assessment: Validity and Validation; Environmental Health Criteria 222; World Health Organization: Geneva, Switzerland, 2001; ISBN 9241572221. [Google Scholar]
- Jung, D.H.; Lee, Y.J.; Park, B. Joint Effect of Hepatic Steatosis and Alanine Aminotransferase Within the Normal Range on Incident Ischemic Heart Disease: A Prospective Study in Koreans. Clin. Interv. Aging 2021, 16, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Bailey, L.A.; Kerper, L.E.; Goodman, J.E. Derivation of an Occupational Exposure Level for Manganese in Welding Fumes. Neurotoxicology 2018, 64, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Espín-Pérez, A.; Krauskopf, J.; de Kok, T.M.; Kleinjans, J.C. ‘OMICS-Based’ Biomarkers for Environmental Health Studies. Curr. Environ. Health Rep. 2014, 1, 353–362. [Google Scholar] [CrossRef] [Green Version]
- Elsevier B.V. Scopus—Analyze Search Results Human Biomonitoring and Risk Assessment. Available online: https://www.scopus.com/term/analyzer.uri?sid=57d074f41c8a4d07a01542e8d2c88ef1&origin=resultslist&src=s&s=TITLE-ABS-KEY%28%22human+biomonitoring%22+AND+%22risk+assessment%22%29&sort=plf-f&sdt=b&sot=b&sl=58&count=391&analyzeResults=Analyze+results&txGid=0c3 (accessed on 14 February 2022).
- Elsevier B.V. Scopus—Analyze Search Results Human Biomonitoring. Available online: https://www.scopus.com/term/analyzer.uri?sid=2f0ac94cfed964391780074281fdc10a&origin=resultslist&src=s&s=TITLE-ABS-KEY%28%22human+biomonitoring%22%29&sort=plf-f&sdt=b&sot=b&sl=36&count=1718&analyzeResults=Analyze+results&txGid=2a8680bf4a21eb55a5a79b73bfd8 (accessed on 14 February 2022).
- Fenner-Crisp, P.A.; Dellarco, V.L. Key Elements for Judging the Quality of a Risk Assessment. Environ. Health Perspect. 2016, 124, 1127–1135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Bruijn, J.; Hansen, B.; Johansson, S.; Luotamo, M.; Munn, S.; Musset, C.; Olsen, S.; Olsson, H.; Paya-Perez, A.; Pedersen, F.; et al. Technical Guidance Document on Risk Assessment. Part 1. European Commission Joint Research Centre EUR 20418 EN. JRC23785. 2002. Available online: https://publications.jrc.ec.europa.eu/repository/handle/JRC23785 (accessed on 11 March 2022).
- Faustman, E.M.; Omenn, G.S. Chapter 4 Risk Assessment. In Casarett/Doull’s Toxicology the Basic Science of Poisons; Klaassen, C.D., Ed.; McGraw-Hill Education, LLC: New York, NY, USA, 2013; pp. 123–149. ISBN 978-0-07-176923-5. [Google Scholar]
- Wild, C.P. The Exposome: From Concept to Utility. Int. J. Epidemiol. 2012, 41, 24–32. [Google Scholar] [CrossRef] [PubMed]
- European Commission CORDIS EU Research Results NEUROSOME Exploring the Neurological Exposome, Horizon 2020 Grant Agreement ID: 766251. Available online: https://cordis.europa.eu/project/id/766251 (accessed on 14 February 2022).
- Ali, M.U.; Liu, G.; Yousaf, B.; Ullah, H.; Abbas, Q.; Munir, M.A.M.; Irshad, S. Biomonitoring and Health Risks Assessment of Trace Elements in Various Age- and Gender-Groups Exposed to Road Dust in Habitable Urban-Industrial Areas of Hefei, China. Environ. Pollut. 2019, 244, 809–817. [Google Scholar] [CrossRef]
- Al-Saleh, I. Health Risk Assessment of Trace Metals Through Breast Milk Consumption in Saudi Arabia. Biol. Trace Elem. Res. 2021, 199, 4535–4545. [Google Scholar] [CrossRef] [PubMed]
- Bastiaensen, M.; Gys, C.; Colles, A.; Verheyen, V.; Koppen, G.; Govarts, E.; Bruckers, L.; Morrens, B.; Loots, I.; De Decker, A.; et al. Exposure Levels, Determinants and Risk Assessment of Organophosphate Flame Retardants and Plasticizers in Adolescents (14–15 Years) from the Flemish Environment and Health Study. Environ. Int. 2021, 147, 106368. [Google Scholar] [CrossRef]
- Berman, T.; Barnett-Itzhaki, Z.; Göen, T.; Hamama, Z.; Axelrod, R.; Keinan-Boker, L.; Shimony, T.; Goldsmith, R. Organophosphate Pesticide Exposure in Children in Israel: Dietary Associations and Implications for Risk Assessment. Environ. Res. 2020, 182, 108739. [Google Scholar] [CrossRef]
- Correia-Sá, L.; Schütze, A.; Norberto, S.; Calhau, C.; Domingues, V.F.; Koch, H.M. Exposure of Portuguese Children to the Novel Non-Phthalate Plasticizer Di-(Iso-Nonyl)-Cyclohexane-1,2-Dicarboxylate (DINCH). Environ. Int. 2017, 102, 79–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coscollà, C.; Sánchez, A.; Corpas-Burgos, F.; López, A.; Pérez, R.; Kuligowski, J.; Vento, M.; Yusà, V. Exposure and Risk Assessment of Hg, Cd, As, Tl, Se, and Mo in Women of Reproductive Age Using Urinary Biomonitoring. Environ. Toxicol. Chem. 2021, 40, 1477–1490. [Google Scholar] [CrossRef]
- Černá, M.; Krsková, A.; Šmíd, J.; Malý, M. Exposure and Risk Assessment of the Czech Population to Chlorinated Pesticides and Polychlorinated Biphenyls Using Archived Serum Samples from the Period 1970 to 1990. Int. J. Hyg. Environ. Health 2016, 219, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Li, C.; Zhou, S.; Wang, X.; Xu, H.; Wang, D.; Gong, Y.Y.; Routledge, M.N.; Zhao, Y.; Wu, Y. Risk Assessment of Deoxynivalenol in High-Risk Area of China by Human Biomonitoring Using an Improved High Throughput UPLC-MS/MS Method. Sci. Rep. 2018, 8, 3901. [Google Scholar] [CrossRef] [PubMed]
- Dualde, P.; Leon, N.; Pardo, O.; Coscollà, C.; Vento, M.; Pastor, A.; Yusà, V. Risk Assessment of Exposure to Phthalates in Breastfeeding Women Using Human Biomonitoring. Chemosphere 2020, 255, 127003. [Google Scholar] [CrossRef]
- Faure, S.; Noisel, N.; Werry, K.; Karthikeyan, S.; Aylward, L.L.; St-Amand, A. Evaluation of Human Biomonitoring Data in a Health Risk Based Context: An Updated Analysis of Population Level Data from the Canadian Health Measures Survey. Int. J. Hyg. Environ. Health 2020, 223, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Fernández, S.F.; Pardo, O.; Adam-Cervera, I.; Montesinos, L.; Corpas-Burgos, F.; Roca, M.; Pastor, A.; Vento, M.; Cernada, M.; Yusà, V. Biomonitoring of Non-Persistent Pesticides in Urine from Lactating Mothers: Exposure and Risk Assessment. Sci. Total Environ. 2020, 699, 134385. [Google Scholar] [CrossRef] [PubMed]
- Fernández, S.F.; Pardo, O.; Hernández, C.S.; Garlito, B.; Yusà, V. Children’s Exposure to Polycyclic Aromatic Hydrocarbons in the Valencian Region (Spain): Urinary Levels, Predictors of Exposure and Risk Assessment. Environ. Int. 2021, 153, 106535. [Google Scholar] [CrossRef] [PubMed]
- Giovanoulis, G.; Alves, A.; Papadopoulou, E.; Cousins, A.P.; Schütze, A.; Koch, H.M.; Haug, L.S.; Covaci, A.; Magnér, J.; Voorspoels, S. Evaluation of Exposure to Phthalate Esters and DINCH in Urine and Nails from a Norwegian Study Population. Environ. Res. 2016, 151, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Gracia-Lor, E.; Zuccato, E.; Hernández, F.; Castiglioni, S. Wastewater-Based Epidemiology for Tracking Human Exposure to Mycotoxins. J. Hazard. Mater. 2020, 382, 121108. [Google Scholar] [CrossRef] [PubMed]
- Hernández, C.S.; Pardo, O.; Corpas-Burgos, F.; Fernández, S.F.; López, A.; Coscollà, C.; Vento, M.; Yusà, V. Biomonitoring of Polychlorinated Dibenzo-p-Dioxins (PCDDs), Polychlorinated Dibenzofurans (PCDFs) and Dioxin-like Polychlorinated Biphenyls (Dl-PCBs) in Human Milk: Exposure and Risk Assessment for Lactating Mothers and Breastfed Children from Spain. Sci. Total Environ. 2020, 744, 140710. [Google Scholar] [CrossRef]
- Imo, D.; Dressel, H.; Byber, K.; Hitzke, C.; Bopp, M.; Maggi, M.; Bose-O’Reilly, S.; Held, L.; Muff, S. Predicted Mercury Soil Concentrations from a Kriging Approach for Improved Human Health Risk Assessment. Int. J. Environ. Res. Public Health 2018, 15, 1326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.H.; Lee, A.; Kim, S.K.; Moon, H.-B.; Park, J.; Choi, K.; Kim, S. Lead and Mercury Levels in Repeatedly Collected Urine Samples of Young Children: A Longitudinal Biomonitoring Study. Environ. Res. 2020, 189, 109901. [Google Scholar] [CrossRef] [PubMed]
- Lessmann, F.; Correia-Sá, L.; Calhau, C.; Domingues, V.F.; Weiss, T.; Brüning, T.; Koch, H.M. Exposure to the Plasticizer Di(2-Ethylhexyl) Terephthalate (DEHTP) in Portuguese Children—Urinary Metabolite Levels and Estimated Daily Intakes. Environ. Int. 2017, 104, 25–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.; Le, S.; Feng, C.; Qiu, X.; Xu, Q.; Jin, S.; Zhang, H.; Jin, Y.; Wen, Y.; Xu, H.; et al. Exposure and Health Risk Assessment of Secondary Contaminants Closely Related to Brominated Flame Retardants (BFRs): Polybrominated Dibenzo-p-Dioxins and Dibenzofurans (PBDD/Fs) in Human Milk in Shanghai. Environ. Pollut. 2021, 268, 115121. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-J.; Hsiao, J.-L.; Hsu, H.-T. Integration of Biomonitoring Data and Reverse Dosimetry Modeling to Assess Population Risks of Arsenic-Induced Chronic Kidney Disease and Urinary Cancer. Ecotoxicol. Environ. Saf. 2020, 206, 111212. [Google Scholar] [CrossRef] [PubMed]
- Martins, C.; Vidal, A.; De Boevre, M.; De Saeger, S.; Nunes, C.; Torres, D.; Goios, A.; Lopes, C.; Assunção, R.; Alvito, P. Exposure Assessment of Portuguese Population to Multiple Mycotoxins: The Human Biomonitoring Approach. Int. J. Hyg. Environ. Health 2019, 222, 913–925. [Google Scholar] [CrossRef] [PubMed]
- Nova, P.; Calheiros, C.S.C.; Silva, M. Glyphosate in Portuguese Adults—A Pilot Study. Environ. Toxicol. Pharmacol. 2020, 80, 103462. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Duarte, S.; Delerue-Matos, C.; Pena, A.; Morais, S. Exposure of Nursing Mothers to Polycyclic Aromatic Hydrocarbons: Levels of Un-Metabolized and Metabolized Compounds in Breast Milk, Major Sources of Exposure and Infants’ Health Risks. Environ. Pollut. 2020, 266. [Google Scholar] [CrossRef] [PubMed]
- Pérez, R.; Suelves, T.; Molina, Y.; Corpas-Burgos, F.; Yusà, V. Biomonitoring of Mercury in Hair of Children Living in the Valencian Region (Spain). Exposure and Risk Assessment. Chemosphere 2019, 217, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Quindroit, P.; Crépet, A.; Brochot, C. Estimating Human Exposure to Pyrethroids’ Mixtures from Biomonitoring Data Using Physiologically Based Pharmacokinetic Modeling. Environ. Res. 2021, 192, 110281. [Google Scholar] [CrossRef]
- Ratelle, M.; Li, X.; Laird, B.D. Cadmium Exposure in First Nations Communities of the Northwest Territories, Canada: Smoking Is a Greater Contributor than Consumption of Cadmium-Accumulating Organ Meats. Environ. Sci. Process. Impacts 2018, 20, 1441–1453. [Google Scholar] [CrossRef] [PubMed]
- Ratelle, M.; Skinner, K.; Laird, M.J.; Majowicz, S.; Brandow, D.; Packull-McCormick, S.; Bouchard, M.; Dieme, D.; Stark, K.D.; Henao, J.J.A.; et al. Implementation of Human Biomonitoring in the Dehcho Region of the Northwest Territories, Canada (2016–2017). Arch. Public Health 2018, 76, 73. [Google Scholar] [CrossRef] [Green Version]
- Rousis, N.I.; Gracia-Lor, E.; Reid, M.J.; Baz-Lomba, J.A.; Ryu, Y.; Zuccato, E.; Thomas, K.V.; Castiglioni, S. Assessment of Human Exposure to Selected Pesticides in Norway by Wastewater Analysis. Sci. Total Environ. 2020, 723, 138132. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, Y.; Coscollà, C.; Corpas-Burgos, F.; Vento, M.; Gormaz, M.; Yusà, V. Biomonitoring of Bisphenols A, F, S and Parabens in Urine of Breastfeeding Mothers: Exposure and Risk Assessment. Environ. Res. 2020, 185, 109481. [Google Scholar] [CrossRef]
- Sarigiannis, D.A.; Karakitsios, S.P.; Handakas, E.; Simou, K.; Solomou, E.; Gotti, A. Integrated Exposure and Risk Characterization of Bisphenol-A in Europe. Food Chem. Toxicol. 2016, 98, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Sarigiannis, D.A.; Tratnik, J.S.; Mazej, D.; Kosjek, T.; Heath, E.; Horvat, M.; Anesti, O.; Karakitsios, S.P. Risk Characterization of Bisphenol-A in the Slovenian Population Starting from Human Biomonitoring Data. Environ. Res. 2019, 170, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Scherer, M.; Petreanu, W.; Weber, T.; Scherer, G.; Pluym, N.; Kolossa-Gehring, M. Human Biomonitoring in Urine Samples from the Environmental Specimen Bank Reveals a Decreasing Trend over Time in the Exposure to the Fragrance Chemical Lysmeral from 2000 to 2018. Chemosphere 2021, 265, 128955. [Google Scholar] [CrossRef] [PubMed]
- Tschersich, C.; Murawski, A.; Schwedler, G.; Rucic, E.; Moos, R.K.; Kasper-Sonnenberg, M.; Koch, H.M.; Brüning, T.; Kolossa-Gehring, M. Bisphenol A and Six Other Environmental Phenols in Urine of Children and Adolescents in Germany—Human Biomonitoring Results of the German Environmental Survey 2014–2017 (GerES V). Sci. Total Environ. 2021, 763, 144615. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Guo, F.; Aamir, M.; Liu, Y.; Tang, M.; Liu, W. Multicenter Biomonitoring of Polybrominated Diphenyl Ethers (PBDEs) in Colostrum from China: Body Burden Profile and Risk Assessment. Environ. Res. 2019, 179, 108828. [Google Scholar] [CrossRef] [PubMed]
- Jeddi, M.Z.; Gorji, M.E.; Rietjens, I.M.C.M.; Louisse, J.; de Bruin, Y.B.; Liska, R. Biomonitoring and Subsequent Risk Assessment of Combined Exposure to Phthalates in Iranian Children and Adolescents. Int. J. Environ. Res. Public Health 2018, 15, 2336. [Google Scholar] [CrossRef]
- Zhu, Y.; Liu, K.; Zhang, J.; Liu, X.; Yang, L.; Wei, R.; Wang, S.; Zhang, D.; Xie, S.; Tao, F. Antibiotic Body Burden of Elderly Chinese Population and Health Risk Assessment: A Human Biomonitoring-Based Study. Environ. Pollut. 2020, 256, 113311. [Google Scholar] [CrossRef] [PubMed]
- Finkel, A.M.; Gray, G.M. The Pebble Remains in the Master’s Hand: Two Careers Spent Learning (Still) from John Evans. Risk Anal. 2021, 41, 678–693. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; International Programme on Chemical Safety. WHO Human Health Risk Assessment Toolkit: Chemical Hazards (IPCS Harmonization Project Document; No.8); World Health Organization: Geneva, Switzerland, 2010; ISBN 9789241548076. [Google Scholar]
- Greenberg, M.; Goldstein, B.D.; Anderson, E.; Dourson, M.; Landis, W.; North, D.W. Whither Risk Assessment: New Challenges and Opportunities a Third of a Century After the Red Book. Risk Anal. 2015, 35, 1959–1968. [Google Scholar] [CrossRef] [PubMed]
- IPCS IPCS Risk Assessment Terminology; World Health Organization: Geneva, Switzerland, 2004.
- Zartarian, V.; Bahadori, T.; McKone, T. Adoption of an Official ISEA Glossary. J. Expo. Anal. Environ. Epidemiol. 2005, 15, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aven, T.; Zio, E. Foundational Issues in Risk Assessment and Risk Management. Risk Anal. 2014, 34, 1164–1172. [Google Scholar] [CrossRef] [PubMed]
- Aven, T.; Flage, R. Foundational Challenges for Advancing the Field and Discipline of Risk Analysis. Risk Anal. 2020, 40, 2128–2136. [Google Scholar] [CrossRef] [PubMed]
- Serraino, A. Introduction to Risk Assessment Terminology. Ital. J. Food Saf. 2014, 3, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reij, M.W.; van Schothorst, M. Critical Notes on Microbiological Risk Assessment of Food. Brazilian J. Microbiol. 2000, 31, 01–08. [Google Scholar] [CrossRef]
- Buekers, J.; David, M.; Koppen, G.; Bessems, J.; Scheringer, M.; Lebret, E.; Sarigiannis, D.; Kolossa-Gehring, M.; Berglund, M.; Schoeters, G.; et al. Development of Policy Relevant Human Biomonitoring Indicators for Chemical Exposure in the European Population. Int. J. Environ. Res. Public Health 2018, 15, 2085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steckling, N.; Gotti, A.; Bose-O’Reilly, S.; Chapizanis, D.; Costopoulou, D.; De Vocht, F.; Garí, M.; Grimalt, J.O.; Heath, E.; Hiscock, R.; et al. Biomarkers of Exposure in Environment-Wide Association Studies—Opportunities to Decode the Exposome Using Human Biomonitoring Data. Environ. Res. 2018, 164, 597–624. [Google Scholar] [CrossRef]
- Radiation Health Management Division Environmental Health Department Minister’s Secretariat Ministry of the Environment Government of Japan; National Institutes for Quantum and Radiological Science and Technology. BOOKLET to Provide Basic Information Regarding Health Effects of Radiation, 1st ed.; Ministry of the Environment Government of Japan, 2019; Available online: https://www.env.go.jp/en/chemi/rhm/basic-info/1st/index.html (accessed on 11 March 2022).
- United States Environmental Protection Agency Human Exposure and Health. Available online: https://www.epa.gov/report-environment/human-exposure-and-health#note1 (accessed on 11 March 2022).
- Duffus, J. Glossary for Chemists of Terms Used in Toxicology (IUPAC Recommendations 1993). Pure Appl. Chem. 1993, 65, 2003–2122. [Google Scholar] [CrossRef]
- Thorp, H.H. Clarity in 2020. Science 2020, 367, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- International Atomic Energy Agency. “Reference Biospheres” for Solid Radioactive Waste Disposal. Report of BIOMASS Theme 1 of the BIOsphere Modelling and ASSessment (BIOMASS) Programme; Non-Serial Publications; International Atomic Energy Agency: Vienna, Austria, 2003; ISBN 92-0-106303-2. [Google Scholar]
- Osimitz, T.G.; Kacew, S.; Hayes, A.W. Assess Flame Retardants with Care. Science 2019, 365, 992–993. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Regional Office for Europe. Human Biomonitoring: Facts and Figures; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 2015. [Google Scholar]
- National Research Council. Exposure Science in the 21st Century: A Vision and a Strategy; National Academies Press: Washington, DC, USA, 2012; ISBN 0309264685. [Google Scholar]
- Tannenbaum, L.V.; Johnson, M.S.; Bazar, M. Application of the Hazard Quotient Method in Remedial Decisions: A Comparison of Human and Ecological Risk Assessments. Hum. Ecol. Risk Assess. 2003, 9, 387–401. [Google Scholar] [CrossRef]
- Jo, W.K.; Weisel, C.P.; Lioy, P.J. Routes of Chloroform Exposure and Body Burden from Showering with Chlorinated Tap Water. Risk Anal. 1990, 10, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Kontić, B. Why Are Some Experts More Credible than Others? Environ. Impact Assess. Rev. 2000, 20, 427–434. [Google Scholar] [CrossRef]
- Slovic, P. Trust, Emotion, Sex, Politics, and Science: Surveying the Risk-Assessment Battlefield. Risk Anal. 1999, 19, 689–701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Society for Risk Analysis Risk Analysis Quality Test Release 1.0. Available online: https://www.sra.org/resources/risk-analysis-quality-test/ (accessed on 14 February 2022).
| Appraised HRA Element * | Appraisal Question |
|---|---|
| Assessment context of HRA | Does the assessment clearly identify what is assessed and why at the start? Has assessment context been followed/applied in the HRA process? |
| Dose/exposure—response relationship | Is the applicability of the selected dose/exposure-response relationship for the assessment thoroughly discussed? |
| Exposure setting | Are the characteristics of the place of exposure clearly described? |
| Exposure sources | Are the major sources of hazardous material and/or activities causing the release(s) of hazardous material(s) into the environment identified? |
| Exposure duration | Is the duration and frequency of the exposure identified? |
| Exposed population | Is it clear who is really exposed (population/individuals, their number), and why are they exposed (e.g., their activities leading to exposure)? |
| Magnitude of risk | Are the types of the expected adverse outcomes, their severity and the probability of their occurrence identified clearly? |
| Uncertainty of HRA results | Are the major sources of uncertainty evaluated? |
| Options for mitigating/avoiding exposure | Are there any specific actions for avoiding or mitigating the exposure to the selected hazardous materials identified and/or proposed? |
| Transparency and clarity of the assessment process | Is it transparent and clear how was the assessment performed and its conclusions obtained? |
| Publication Title | Assessment context of HRA * | Dose/exposure—Response | Exposure Setting | Exposure Sources | Exposure Duration | Exposed Population | Magnitude of Risk | Uncertainty of HRA Results | Options for Mitigating Exposure | Transparency and Clarity |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. Biomonitoring and health risks assessment of trace elements in various age- and gender-groups exposed to road dust in habitable urban-industrial areas of Hefei, China [27] | No | No | Yes | No | No | Yes | No | No | Yes | Yes |
| X | ||||||||||
| 2. Health Risk Assessment of Trace Metals Through Breast Milk Consumption in Saudi Arabia [28] | Yes | No | No | Yes | No | Yes | Yes | Yes | No | Yes |
| X | X | |||||||||
| 3. Exposure levels, determinants and risk assessment of organophosphate flame retardants and plasticizers in adolescents (14–15 years) from the Flemish Environment and Health Study [29] | No | No | Yes | Yes | No | No | No | Yes | No | Yes |
| X | ||||||||||
| 4. Organophosphate pesticide exposure in children in Israel: Dietary associations and implications for risk assessment [30] | No | No | No | No | No | No | No | Yes | Yes | Yes |
| X | X | |||||||||
| 5. Exposure of Portuguese children to the novel non-phthalate plasticizer di-(iso-nonyl)-cyclohexane-1,2-dicarboxylate (DINCH) [31] | No | No | No | No | No | No | No | No | No | No |
| 6. Exposure and Risk Assessment of Hg, Cd, As, Tl, Se, and Mo in Women of Reproductive Age Using Urinary Biomonitoring [32] | No | No | No | No | No | No | No | No | No | No |
| 7. Exposure and risk assessment of the Czech population to chlorinated pesticides and polychlorinated biphenyls using archived serum samples from the period 1970 to 1990 [33] | Yes | Yes | Yes | Yes | No | No | No | Yes | No | Yes |
| X | X | X | X | X | X | X | ||||
| 8. Risk assessment of deoxynivalenol in high-risk area of China by human biomonitoring using an improved high throughput UPLC-MS/MS method [34] | No | No | No | No | No | No | No | No | No | Yes |
| X | ||||||||||
| 9. Risk assessment of exposure to phthalates in breastfeeding women using human biomonitoring [35] | Yes | Yes | No | Yes | No | Yes | No | Yes | No | Yes |
| X | X | X | X | |||||||
| 10. Evaluation of human biomonitoring data in a health risk based context: An updated analysis of population level data from the Canadian Health Measures Survey [36] | No | No | No | No | No | No | No | Yes | No | Yes |
| X | X | X | ||||||||
| 11. Biomonitoring of non-persistent pesticides in urine from lactating mothers: Exposure and risk assessment [37] | No | No | No | No | No | No | No | No | No | No |
| 12. Children’s exposure to polycyclic aromatic hydrocarbons in the Valencian Region (Spain): Urinary levels, predictors of exposure and risk assessment [38] | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | Yes |
| X | X | X | X | X | ||||||
| 13. Evaluation of exposure to phthalate esters and DINCH in urine and nails from a Norwegian study population [39] | Yes | Yes | No | Yes | No | Yes | No | No | No | Yes |
| X | X | X | ||||||||
| 14. Wastewater-based epidemiology for tracking human exposure to mycotoxins [40] | No | No | No | Yes | No | No | No | Yes | No | Yes |
| X | ||||||||||
| 15. Biomonitoring of polychlorinated dibenzo-p-dioxins (PCDDs), polychlorinated dibenzofurans (PCDFs) and dioxin-like polychlorinated biphenyls (dl-PCBs) in human milk: Exposure and risk assessment for lactating mothers and breastfed children from Spain [41] | No | Yes | No | No | No | No | No | Yes | No | No |
| X | ||||||||||
| 16. Predicted Mercury Soil Concentrations from a Kriging Approach for Improved Human Health Risk Assessment [42] | Yes | No | Yes | Yes | No | Yes | No | No | No | Yes |
| X | X | |||||||||
| 17. Lead and mercury levels in repeatedly collected urine samples of young children: A longitudinal biomonitoring study [43] | No | No | No | No | No | No | No | Yes | No | No |
| X | ||||||||||
| 18. Exposure to the plasticizer di(2-ethylhexyl) terephthalate (DEHTP) in Portuguese children–Urinary metabolite levels and estimated daily intakes [44] | Yes | Yes | Yes | No | No | Yes | No | No | No | Yes |
| X | X | X | ||||||||
| 19. Exposure and health risk assessment of secondary contaminants closely related to brominated flame retardants (BFRs): Polybrominated dibenzo-p-dioxins and dibenzofurans (PBDD/Fs) in human milk in Shanghai [45] | No | No | No | No | No | No | No | No | No | No |
| 20. Integration of biomonitoring data and reverse dosimetry modeling to assess population risks of arsenic-induced chronic kidney disease and urinary cancer [46] | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes |
| X | X | |||||||||
| 21. Exposure assessment of Portuguese population to multiple mycotoxins: The human biomonitoring approach [47] | No | No | No | No | No | No | No | Yes | No | Yes |
| X | X | |||||||||
| 22. Glyphosate in Portuguese Adults- A Pilot Study [48] | No | No | No | No | No | No | No | Yes | No | No |
| 23. Exposure of nursing mothers to polycyclic aromatic hydrocarbons: Levels of un-metabolized and metabolized compounds in breast milk, major sources of exposure and infants’ health risks [49] | Yes | No | Yes | No | Yes | Yes | No | No | No | Yes |
| X | ||||||||||
| 24. Biomonitoring of mercury in hair of children living in the Valencian Region (Spain). Exposure and risk assessment [50] | No | No | No | No | No | No | No | No | Yes | No |
| X | ||||||||||
| 25. Estimating human exposure to pyrethroids’ mixtures from biomonitoring data using physiologically based pharmacokinetic modeling [51] | Yes | Yes | Yes | No | No | Yes | No | Yes | No | Yes |
| X | X | |||||||||
| 26. Cadmium exposure in First Nations communities of the Northwest Territories, Canada: smoking is a greater contributor than consumption of cadmium-accumulating organ meats [52] | Yes | Yes | Yes | Yes | No | Yes | No | Yes | No | Yes |
| X | ||||||||||
| 27. Implementation of human biomonitoring in the Dehcho region of the Northwest Territories, Canada (2016–2017) [53] | Yes | Yes | Yes | Yes | No | No | No | Yes | No | Yes |
| X | X | X | X | X | X | |||||
| 28. Assessment of human exposure to selected pesticides in Norway by wastewater analysis [54] | No | No | No | Yes | No | No | No | Yes | No | Yes |
| X | ||||||||||
| 29. Biomonitoring of bisphenols A, F, S and parabens in urine of breastfeeding mothers: Exposure and risk assessment [55] | Yes | No | No | Yes | No | Yes | No | No | No | Yes |
| X | ||||||||||
| 30. Integrated exposure and risk characterization of bisphenol-A in Europe [56] | No | No | No | No | No | No | No | No | No | No |
| X | X | |||||||||
| 31. Risk characterization of bisphenol-A in the Slovenian population starting from human biomonitoring data [57] | Yes | Yes | Yes | No | Yes | No | No | Yes | No | Yes |
| X | ||||||||||
| 32. Human biomonitoring in urine samples from the Environmental Specimen Bank reveals a decreasing trend over time in the exposure to the fragrance chemical lysmeral from 2000 to 2018 [58] | Yes | Yes | No | Yes | No | Yes | No | Yes | No | Yes |
| X | X | X | ||||||||
| 33. Bisphenol A and six other environmental phenols in urine of children and adolescents in Germany-human biomonitoring results of the German Environmental Survey 2014–2017 (GerES V) [59] | Yes | No | Yes | Yes | No | Yes | No | No | No | Yes |
| X | ||||||||||
| 34. Multicenter biomonitoring of polybrominated diphenyl ethers (PBDEs) in colostrum from China: Body burden profile and risk assessment [60] | No | No | No | No | No | No | No | Yes | No | No |
| 35. Biomonitoring and Subsequent Risk Assessment of Combined Exposure to Phthalates in Iranian Children and Adolescents [61] | No | Yes | No | No | No | No | No | No | No | No |
| X | ||||||||||
| 36. Antibiotic body burden of elderly Chinese population and health risk assessment: A human biomonitoring-based study [62] | No | No | No | No | No | No | Yes | Yes | Yes | No |
| X | X | |||||||||
| Number of “Yes” (Yes proportion) | 16 (44%) | 12 (33%) | 13 (36%) | 15 (42%) | 3 (8%) | 14 (39%) | 4 (11%) | 20 (56%) | 5 (14%) | 24 (67%) |
| Number of “X” (X proportion) | 15 (42%) | 11 (31%) | 4 (11%) | 5 (14%) | 4 (11%) | 12 (33%) | 3 (8%) | 3 (8%) | 2 (6%) | 5 (14%) |
| Term | Definitions |
|---|---|
| Exposure | “Concentration or amount of a particular agent that reaches a target organism, system, or (sub)population in a specific frequency for a defined duration” [66] (p. 12). |
| “Contact between an agent and a target. Contact takes place at an exposure surface over an exposure period” [67] (p. 3). | |
| 1. “Concentration, amount, or intensity of a particular physical or chemical agent or environmental agent that reaches the target population, organism, organ, tissue or cell, usually expressed in numerical terms of substance concentration, duration, and frequency (for chemical agents and micro-organisms) or intensity (for physical agents such as radiation). 2. Process by which a substance becomes available for absorption by the target population, organism, organ, tissue or cell, by any route” [76] (p. 2047). | |
| “Exposure is defined as contact of a biologic, chemical, or physical agent with the outer part of the human body, such as the skin, mouth, or nostrils” [12] (p. 17). | |
| Dose | “Total amount of an agent administered to, taken up by, or absorbed by an organism, system, or (sub)population” [66] (p. 11). |
| “The amount of agent that enters a target after crossing an exposure surface. If the exposure surface is an absorption barrier, the dose is an absorbed dose/uptake dose; otherwise it is an intake dose” [67] (p. 3). | |
| “Total amount of a substance administered to, taken or absorbed by an organism” [76] (p. 2039). | |
| “Once the agent enters the body by either intake or uptake, it is described as a ‘dose’” [12] (p. 19). “Potential, or administered dose, is the amount of the agent that is actually ingested, inhaled, or applied to the skin” [12] (p. 19). “Applied dose is the amount of the agent directly in contact with the body’s absorption barriers, such as the skin, respiratory tract, and gastrointestinal tract, and therefore available for absorption” [12] (p. 19.). “The amount of the agent absorbed, and therefore available to undergo metabolism, transport, storage, or elimination, is referred to as the ‘internal’ or ‘absorbed dose’” [12] (p. 19). The portion of the internal (absorbed) dose that reaches a tissue of interest is called the ‘delivered dose’” [12] (p. 19). “The portion of the delivered dose that reaches the site or sites of toxic action is called the ‘biologically effective dose’” [12] (p. 19). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bizjak, T.; Capodiferro, M.; Deepika, D.; Dinçkol, Ö.; Dzhedzheia, V.; Lopez-Suarez, L.; Petridis, I.; Runkel, A.A.; Schultz, D.R.; Kontić, B. Human Biomonitoring Data in Health Risk Assessments Published in Peer-Reviewed Journals between 2016 and 2021: Confronting Reality after a Preliminary Review. Int. J. Environ. Res. Public Health 2022, 19, 3362. https://doi.org/10.3390/ijerph19063362
Bizjak T, Capodiferro M, Deepika D, Dinçkol Ö, Dzhedzheia V, Lopez-Suarez L, Petridis I, Runkel AA, Schultz DR, Kontić B. Human Biomonitoring Data in Health Risk Assessments Published in Peer-Reviewed Journals between 2016 and 2021: Confronting Reality after a Preliminary Review. International Journal of Environmental Research and Public Health. 2022; 19(6):3362. https://doi.org/10.3390/ijerph19063362
Chicago/Turabian StyleBizjak, Tine, Marco Capodiferro, Deepika Deepika, Öykü Dinçkol, Vazha Dzhedzheia, Lorena Lopez-Suarez, Ioannis Petridis, Agneta A. Runkel, Dayna R. Schultz, and Branko Kontić. 2022. "Human Biomonitoring Data in Health Risk Assessments Published in Peer-Reviewed Journals between 2016 and 2021: Confronting Reality after a Preliminary Review" International Journal of Environmental Research and Public Health 19, no. 6: 3362. https://doi.org/10.3390/ijerph19063362
APA StyleBizjak, T., Capodiferro, M., Deepika, D., Dinçkol, Ö., Dzhedzheia, V., Lopez-Suarez, L., Petridis, I., Runkel, A. A., Schultz, D. R., & Kontić, B. (2022). Human Biomonitoring Data in Health Risk Assessments Published in Peer-Reviewed Journals between 2016 and 2021: Confronting Reality after a Preliminary Review. International Journal of Environmental Research and Public Health, 19(6), 3362. https://doi.org/10.3390/ijerph19063362








