Assessing the Effects of Nature on Physiological States Using Wearable Technologies
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of the Nature Break Activity
2.1.1. L’Université dans la Nature
2.1.2. Nature Break
2.2. Participants
2.3. Study Site
2.4. Procedure
2.5. Data Collection
2.5.1. Psychological Measurements
2.5.2. Physiological Measurements
Electrodermal Activity (EDA)
Fingertip Skin Temperature
Heart Rate (HR)
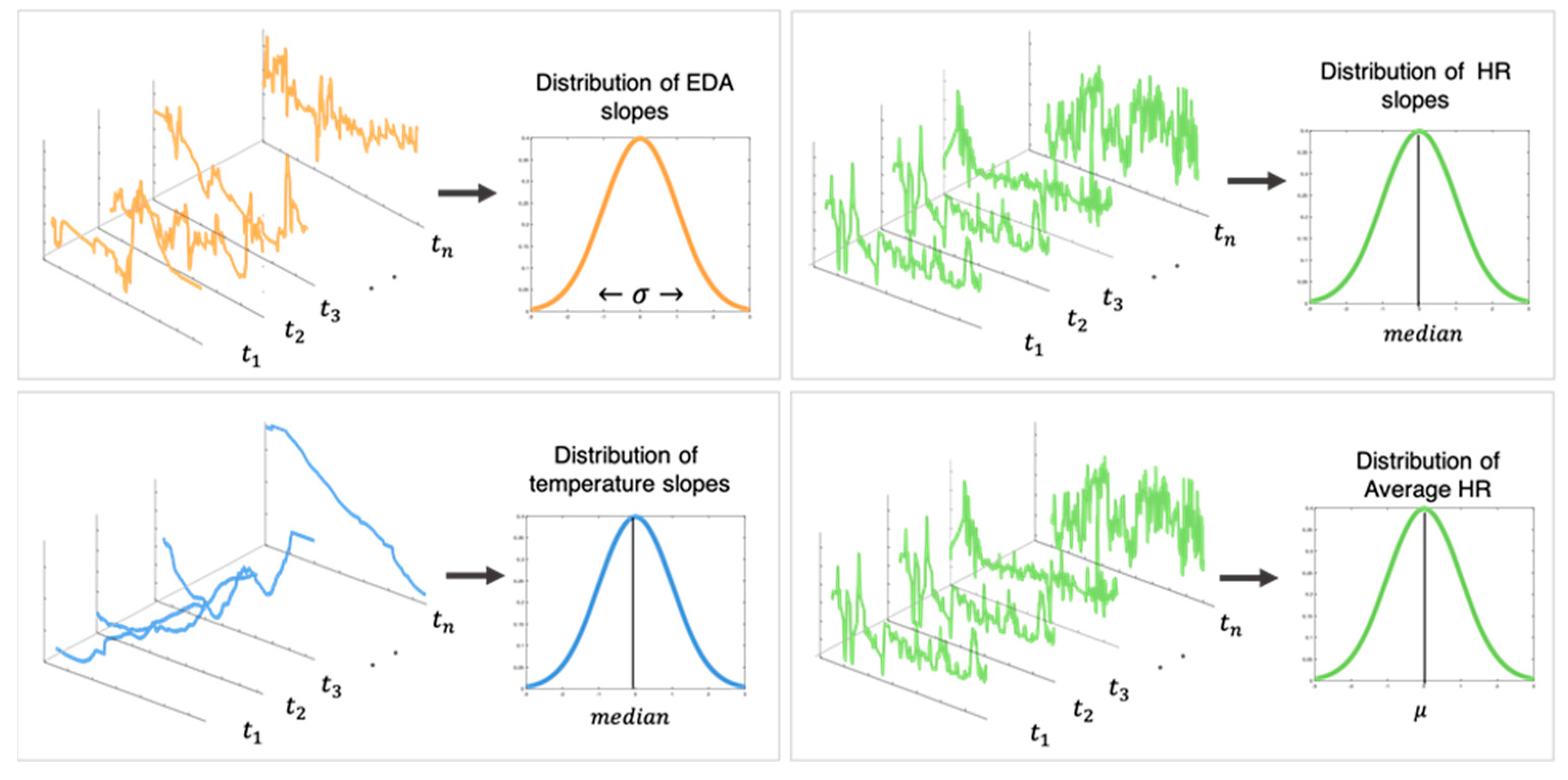
2.6. Data Analysis
2.6.1. Psychological Measures Analysis
2.6.2. Physiological Measures Analysis
2.6.3. Determining the Physiological Effect of Nature Break
Control Analysis 1: Ambient Temperature
Control Analysis 2: Time of Day
3. Results
3.1. Forest Bathing Increases Positive Mood States and Decreases Negative Mood States
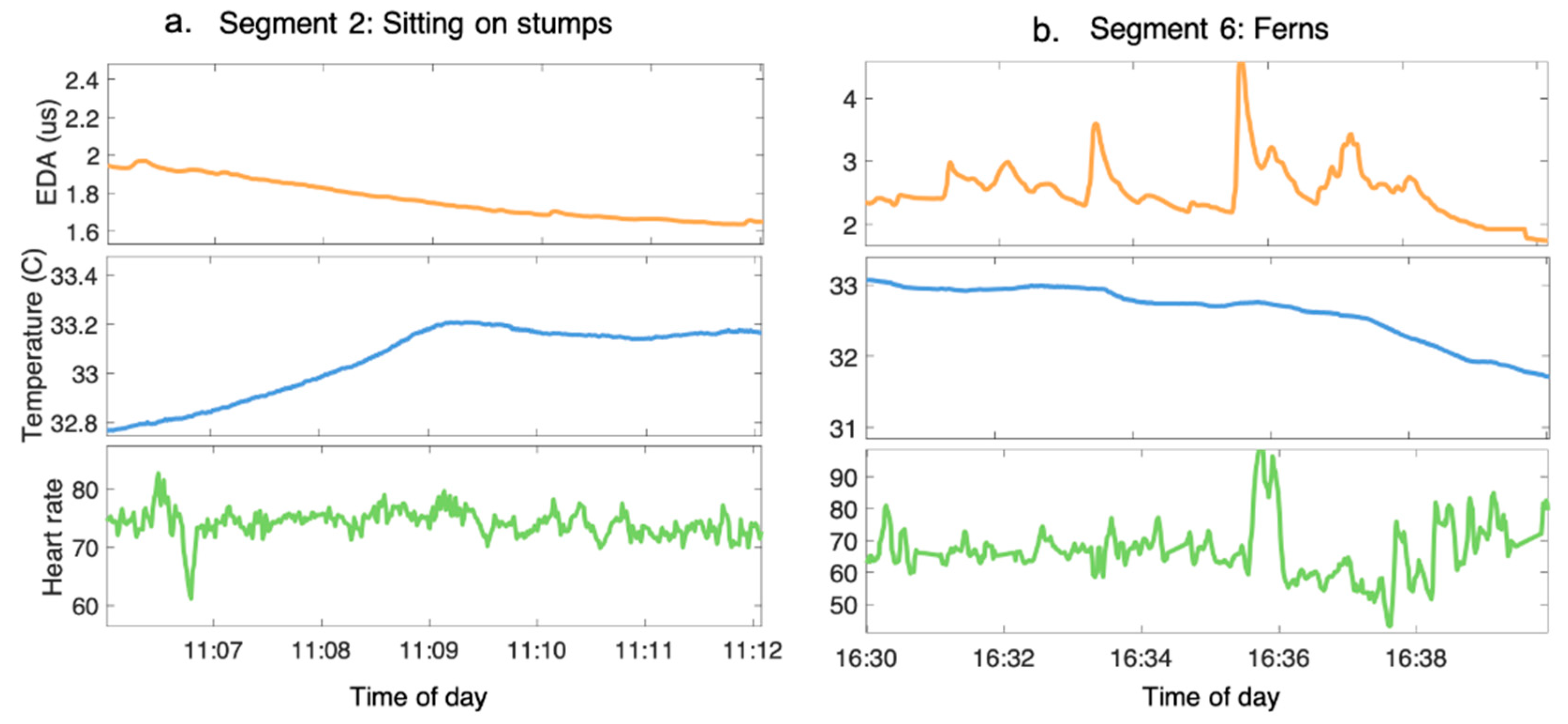
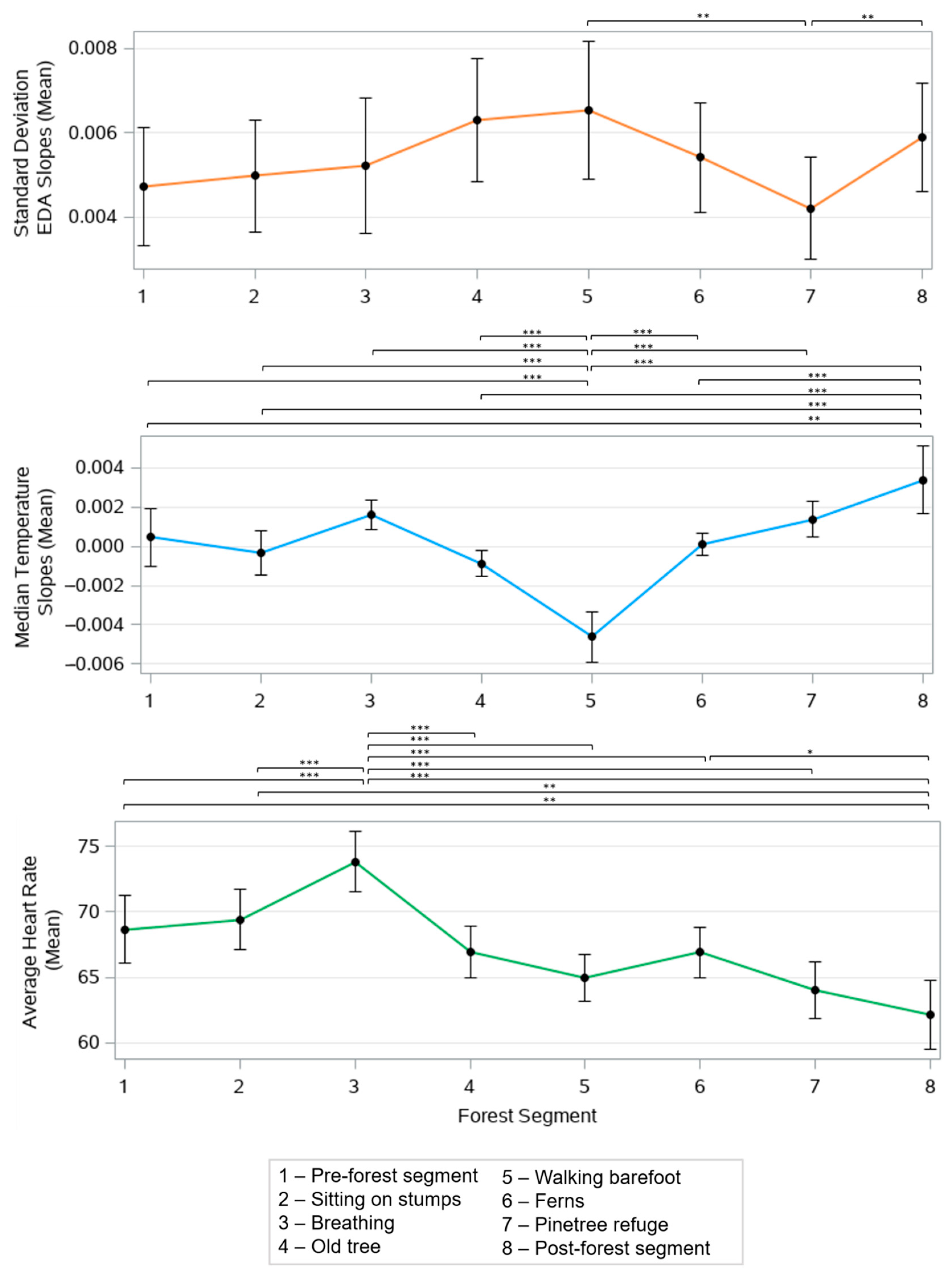
3.2. Purposeful Engagement with the Forest Induces Different Physiological Response Profiles
3.3. Ambient Temperature Has an Effect on Skin Temperature and Heart Rate Response
3.4. Circadian Rhythms Affect Skin Temperature Response to Forest Bathing
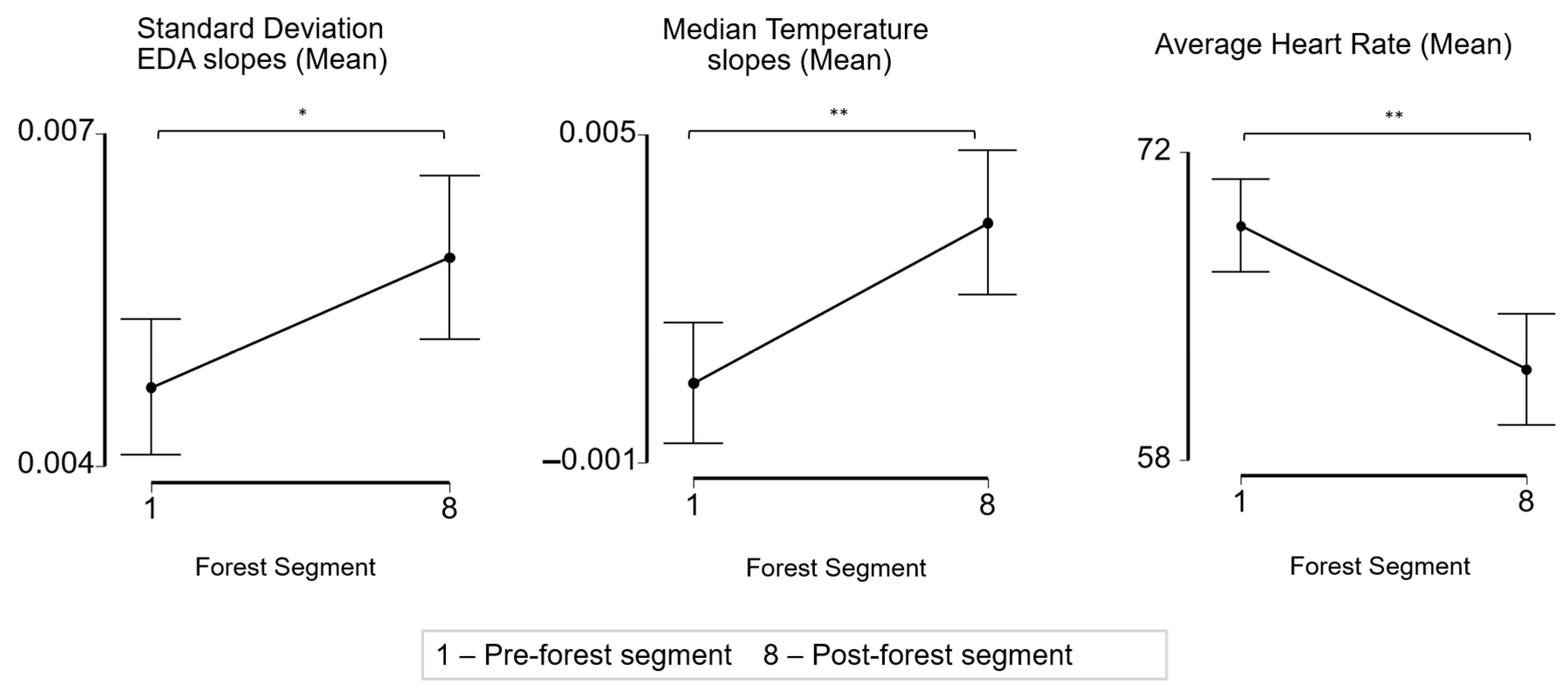
3.5. Participants Have Significant Physiological Differences Pre- and Post-Forest Bathing
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations; Department of Economic and Social Affairs; Population Division. World Urbanization Prospects: The 2018 Revision; United Nations: New York, NY, USA, 2019; ISBN 978-92-1-148319-2. [Google Scholar]
- Vlahov, D. Urbanization, Urbanicity, and Health. J. Urban Health Bull. N. Y. Acad. Med. 2002, 79, S1–S12. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.M.; Jones, R.; Tocchini, K. Shinrin-Yoku (Forest Bathing) and Nature Therapy: A State-of-the-Art Review. Int. J. Environ. Res. Public Health 2017, 14, 851. [Google Scholar] [CrossRef]
- Park, B.J.; Tsunetsugu, Y.; Kasetani, T.; Kagawa, T.; Miyazaki, Y. The Physiological Effects of Shinrin-Yoku (Taking in the Forest Atmosphere or Forest Bathing): Evidence from Field Experiments in 24 Forests across Japan. Environ. Health Prev. Med. 2009, 15, 18. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Yan, Q.; Pan, Y.; Gu, X.; Liu, Y. Medical Empirical Research on Forest Bathing (Shinrin-Yoku): A Systematic Review. Environ. Health Prev. Med. 2019, 24, 70. [Google Scholar] [CrossRef]
- Kondo, M.C.; Jacoby, S.F.; South, E.C. Does Spending Time Outdoors Reduce Stress? A Review of Real-Time Stress Response to Outdoor Environments. Health Place 2018, 51, 136–150. [Google Scholar] [CrossRef]
- Oh, B.; Lee, K.J.; Zaslawski, C.; Yeung, A.; Rosenthal, D.; Larkey, L.; Back, M. Health and Well-Being Benefits of Spending Time in Forests: Systematic Review. Environ. Health Prev. Med. 2017, 22, 71. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.-X.; Cao, Y.-B.; Lan, X.-G.; He, Z.-H.; Chen, Z.-M.; Wang, Y.-Z.; Hu, X.-L.; Lv, Y.-D.; Wang, G.-F.; Yan, J. Therapeutic Effect of Forest Bathing on Human Hypertension in the Elderly. J. Cardiol. 2012, 60, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Hunter, M.R.; Gillespie, B.W.; Chen, S.Y.-P. Urban Nature Experiences Reduce Stress in the Context of Daily Life Based on Salivary Biomarkers. Front. Psychol. 2019, 10, 722. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Park, B.-J.; Tsunetsugu, Y.; Ohira, T.; Kagawa, T.; Miyazaki, Y. Effect of Forest Bathing on Physiological and Psychological Responses in Young Japanese Male Subjects. Public Health 2011, 125, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Bielinis, E.; Takayama, N.; Boiko, S.; Omelan, A.; Bielinis, L. The Effect of Winter Forest Bathing on Psychological Relaxation of Young Polish Adults. Urban For. Urban Green. 2018, 29, 276–283. [Google Scholar] [CrossRef]
- Horiuchi, M.; Endo, J.; Akatsuka, S.; Hasegawa, T.; Yamamoto, E.; Uno, T.; Kikuchi, S. An Effective Strategy to Reduce Blood Pressure after Forest Walking in Middle-Aged and Aged People. J. Phys. Ther. Sci. 2015, 27, 3711–3716. [Google Scholar] [CrossRef][Green Version]
- Morita, E.; Fukuda, S.; Nagano, J.; Hamajima, N.; Yamamoto, H.; Iwai, Y.; Nakashima, T.; Ohira, H.; Shirakawa, T. Psychological Effects of Forest Environments on Healthy Adults: Shinrin-Yoku (Forest-Air Bathing, Walking) as a Possible Method of Stress Reduction. Public Health 2007, 121, 54–63. [Google Scholar] [CrossRef]
- Mitten, D.; Overholt, J.R.; Haynes, F.I.; D’Amore, C.C.; Ady, J.C. Hiking. Am. J. Lifestyle Med. 2016, 12, 302–310. [Google Scholar] [CrossRef]
- Kobayashi, H.; Song, C.; Ikei, H.; Kagawa, T.; Miyazaki, Y. Analysis of Individual Variations in Autonomic Responses to Urban and Forest Environments . Available online: https://www.hindawi.com/journals/ecam/2015/671094/ (accessed on 29 January 2021).
- Lee, M.; Lee, J.; Park, B.-J.; Miyazaki, Y. Interaction with Indoor Plants May Reduce Psychological and Physiological Stress by Suppressing Autonomic Nervous System Activity in Young Adults: A Randomized Crossover Study. J. Physiol. Anthropol. 2015, 34, 21. [Google Scholar] [CrossRef] [PubMed]
- Tsunetsugu, Y.; Park, B.-J.; Ishii, H.; Hirano, H.; Kagawa, T.; Miyazaki, Y. Physiological Effects of Shinrin-Yoku (Taking in the Atmosphere of the Forest) in an Old-Growth Broadleaf Forest in Yamagata Prefecture, Japan. J. Physiol. Anthropol. 2007, 26, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Tsunetsugu, Y.; Park, B.-J.; Miyazaki, Y. Trends in Research Related to “Shinrin-Yoku” (Taking in the Forest Atmosphere or Forest Bathing) in Japan. Environ. Health Prev. Med. 2010, 15, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, M.; Miwa, M.; Ikei, H.; Song, C.; Takagaki, M.; Miyazaki, Y. Physiological and Psychological Effects of Viewing a Kiwifruit (Actinidia Deliciosa ‘Hayward’) Orchard Landscape in Summer in Japan. Int. J. Environ. Res. Public. Health 2015, 12, 6657–6668. [Google Scholar] [CrossRef]
- Jia, B.B.; Yang, Z.X.; Mao, G.X.; Lyu, Y.D.; Wen, X.L.; Xu, W.H.; Lyu, X.L.; Cao, Y.B.; Wang, G.F. Health Effect of Forest Bathing Trip on Elderly Patients with Chronic Obstructive Pulmonary Disease. Biomed. Environ. Sci. BES 2016, 29, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Song, C.; Ikei, H.; Park, B.-J.; Lee, J.; Kagawa, T.; Miyazaki, Y. Population-Based Study on the Effect of a Forest Environment on Salivary Cortisol Concentration. Int. J. Environ. Res. Public Health 2017, 14, 931. [Google Scholar] [CrossRef] [PubMed]
- Im, S.G.; Choi, H.; Jeon, Y.-H.; Song, M.-K.; Kim, W.; Woo, J.-M. Comparison of Effect of Two-Hour Exposure to Forest and Urban Environments on Cytokine, Anti-Oxidant, and Stress Levels in Young Adults. Int. J. Environ. Res. Public Health 2016, 13, 625. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.X.; Cao, Y.B.; Yang, Y.; Chen, Z.M.; Dong, J.H.; Chen, S.S.; Wu, Q.; Lyu, X.L.; Jia, B.B.; Yan, J.; et al. Additive Benefits of Twice Forest Bathing Trips in Elderly Patients with Chronic Heart Failure. Biomed. Environ. Sci. 2018, 31, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Tsao, T.-M.; Tsai, M.-J.; Hwang, J.-S.; Cheng, W.-F.; Wu, C.-F.; Chou, C.-C.K.; Su, T.-C. Health Effects of a Forest Environment on Natural Killer Cells in Humans: An Observational Pilot Study. Oncotarget 2018, 9, 16501–16511. [Google Scholar] [CrossRef]
- Chen, Z.; Schulz, S.; Qiu, M.; Yang, W.; He, X.; Wang, Z.; Yang, L. Assessing Affective Experience of In-Situ Environmental Walk via Wearable Biosensors for Evidence-Based Design. Cogn. Syst. Res. 2018, 52, 970–977. [Google Scholar] [CrossRef]
- Elsadek, M.; Liu, B.; Lian, Z. Green Façades: Their Contribution to Stress Recovery and Well-Being in High-Density Cities. Urban For. Urban Green. 2019, 46, 126446. [Google Scholar] [CrossRef]
- Reeves, J.P.; Knight, A.T.; Strong, E.A.; Heng, V.; Neale, C.; Cromie, R.; Vercammen, A. The Application of Wearable Technology to Quantify Health and Wellbeing Co-Benefits From Urban Wetlands. Front. Psychol. 2019, 10, 1840. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, R.S.; Simons, R.F.; Losito, B.D.; Fiorito, E.; Miles, M.A.; Zelson, M. Stress Recovery during Exposure to Natural and Urban Environments. J. Environ. Psychol. 1991, 11, 201–230. [Google Scholar] [CrossRef]
- Duvall, J. Enhancing the Benefits of Outdoor Walking with Cognitive Engagement Strategies. J. Environ. Psychol. 2011, 31, 27–35. [Google Scholar] [CrossRef]
- Duvall, J. Using Engagement-Based Strategies to Alter Perceptions of the Walking Environment. Environ. Behav. 2013, 45, 303–322. [Google Scholar] [CrossRef]
- Korpela, K.; Savonen, E.-M.; Anttila, S.; Pasanen, T.; Ratcliffe, E. Enhancing Wellbeing with Psychological Tasks along Forest Trails. Urban For. Urban Green. 2017, 26, 25–30. [Google Scholar] [CrossRef]
- Hartig, T.; Böök, A.; Garvill, J.; Olsson, T.; Gärling, T. Environmental Influences on Psychological Restoration. Scand. J. Psychol. 1996, 37, 378–393. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.; Tao, J.; Li, G.; Jiang, M.; Aii, L.; Zhihui, J.; Zongfang, L.; Qibing, C. Effects of Walking in Bamboo Forest and City Environments on Brainwave Activity in Young Adults. Available online: https://www.hindawi.com/journals/ecam/2018/9653857/ (accessed on 14 January 2021).
- Ochiai, H.; Ikei, H.; Song, C.; Kobayashi, M.; Miura, T.; Kagawa, T.; Li, Q.; Kumeda, S.; Imai, M.; Miyazaki, Y. Physiological and Psychological Effects of a Forest Therapy Program on Middle-Aged Females. Int. J. Environ. Res. Public Health 2015, 12, 15222–15232. [Google Scholar] [CrossRef]
- Ulrich, R.S. View through a Window May Influence Recovery from Surgery. Science 1984, 224, 420–421. [Google Scholar] [CrossRef] [PubMed]
- Curran, S.L.; Andrykowski, M.A.; Studts, J.L. Short Form of the Profile of Mood States (POMS-SF): Psychometric Information. Psychol. Assess. 1995, 7, 80–83. [Google Scholar] [CrossRef]
- Cheung, S.; Han, E.; Kushki, A.; Anagnostou, E.; Biddiss, E. Biomusic: An Auditory Interface for Detecting Physiological Indicators of Anxiety in Children. Front. Neurosci. 2016, 10, 401. [Google Scholar] [CrossRef] [PubMed]
- Blain-Moraes, S.; Chau, T.; Mihailidis, A. Peripheral Autonomic Signals as Access Pathways for Individuals with Severe Disabilities: A Literature Appraisal. Open Rehabil. J. 2008, 1, 27–37. [Google Scholar] [CrossRef]
- Noyes, F.R.; Barber-Westin, S.D. 40-Diagnosis and Treatment of Complex Regional Pain Syndrome. In Noyes’ Knee Disorders: Surgery, Rehabilitation, Clinical Outcomes, 2nd ed.; Noyes, F.R., Barber-Westin, S.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1122–1160. ISBN 978-0-323-32903-3. [Google Scholar]
- Blain, S.; McKeever, P. Revealing Personhood through Biomusic of Individuals without Communicative Interaction Ability. Augment. Altern. Commun. 2011, 27, 1–4. [Google Scholar] [CrossRef]
- Shu, L.; Xie, J.; Yang, M.; Li, Z.; Li, Z.; Liao, D.; Xu, X.; Yang, X. A Review of Emotion Recognition Using Physiological Signals. Sensors 2018, 18, 2074. [Google Scholar] [CrossRef] [PubMed]
- Kistler, A.; Mariauzouls, C.; von Berlepsch, K. Fingertip Temperature as an Indicator for Sympathetic Responses. Int. J. Psychophysiol. 1998, 29, 35–41. [Google Scholar] [CrossRef]
- Kaushik, R.M.; Kaushik, R.; Mahajan, S.K.; Rajesh, V. Effects of Mental Relaxation and Slow Breathing in Essential Hypertension. Complement. Ther. Med. 2006, 14, 120–126. [Google Scholar] [CrossRef]
- Bruning, N.S.; Frew, D.R. Effects of Exercise, Relaxation, and Management Skills Training on Physiological Stress Indicators: A Field Experiment. J. Appl. Psychol. 1987, 72, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Khanna, A.; Paul, M.; Sandhu, J.S. Efficacy of two relaxation techniques in reducing pulse rate among highly stressed females. Calicut Med. J. 2007, 5, E3. [Google Scholar]
- Lai Kwan, C.; Mahdid, Y.; Motta Ochoa, R.; Lee, K.; Park, M.; Blain-Moraes, S. Wearable Technology for Detecting Significant Moments in Individuals with Dementia. BioMed Res. Int. 2019, 2019, e6515813. [Google Scholar] [CrossRef] [PubMed]
- Blain, S.; Mihailidis, A.; Chau, T. Assessing the Potential of Electrodermal Activity as an Alternative Access Pathway. Med. Eng. Phys. 2008, 30, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Olafsdottir, G.; Cloke, P.; Schulz, A.; van Dyck, Z.; Eysteinsson, T.; Thorleifsdottir, B.; Vögele, C. Health Benefits of Walking in Nature: A Randomized Controlled Study Under Conditions of Real-Life Stress. Environ. Behav. 2020, 52, 248–274. [Google Scholar] [CrossRef]
- Song, C.; Ikei, H.; Kobayashi, M.; Miura, T.; Taue, M.; Kagawa, T.; Li, Q.; Kumeda, S.; Imai, M.; Miyazaki, Y. Effect of Forest Walking on Autonomic Nervous System Activity in Middle-Aged Hypertensive Individuals: A Pilot Study. Int. J. Environ. Res. Public Health 2015, 12, 2687–2699. [Google Scholar] [CrossRef]
- Cox, D.T.C.; Hudson, H.L.; Shanahan, D.F.; Fuller, R.A.; Gaston, K.J. The Rarity of Direct Experiences of Nature in an Urban Population. Landsc. Urban Plan. 2017, 160, 79–84. [Google Scholar] [CrossRef]
- Cox, D.T.C.; Shanahan, D.F.; Hudson, H.L.; Plummer, K.E.; Siriwardena, G.M.; Fuller, R.A.; Anderson, K.; Hancock, S.; Gaston, K.J. Doses of Neighborhood Nature: The Benefits for Mental Health of Living with Nature. BioScience 2017, 67, 147–155. [Google Scholar] [CrossRef]
- Li, Q.; Kobayashi, M.; Kumeda, S.; Ochiai, T.; Miura, T.; Kagawa, T.; Imai, M.; Wang, Z.; Otsuka, T.; Kawada, T. Effects of Forest Bathing on Cardiovascular and Metabolic Parameters in Middle-Aged Males. Evid. Based Complement. Alternat. Med. 2016, 2016, 2587381. [Google Scholar] [CrossRef]
- Song, C.; Ikei, H.; Park, B.-J.; Lee, J.; Kagawa, T.; Miyazaki, Y. Psychological Benefits of Walking through Forest Areas. Int. J. Environ. Res. Public Health 2018, 15, 2804. [Google Scholar] [CrossRef]
- Williams, G.P.; Gold, L.W. Ground Temperatures. Can. Build. Dig. 1976, 6, 101. [Google Scholar] [CrossRef]
- Korhonen, I. Blood Pressure and Heart Rate Responses in Men Exposed to Arm and Leg Cold Pressor Tests and Whole-Body Cold Exposure. Int. J. Circumpolar Health 2006, 65, 178–184. [Google Scholar] [CrossRef]
- Venables, P.H.; Mitchell, D.A. The Effects of Age, Sex and Time of Testing on Skin Conductance Activity. Biol. Psychol. 1996, 43, 87–101. [Google Scholar] [CrossRef]
- Park, B.-J.; Furuya, K.; Kasetani, T.; Takayama, N.; Kagawa, T.; Miyazaki, Y. Relationship between Psychological Responses and Physical Environments in Forest Settings. Landsc. Urban Plan. 2011, 102, 24–32. [Google Scholar] [CrossRef]

| Forest Activity Segment | Description | Length (min.) |
|---|---|---|
| 1: Pre-forest | The participants fill out pre-forest POMS and don the wearable sensor. | ~10–20 |
| 2: Sitting on stumps | The UdN guide invites the participants to understand the course of time in forests and invites comparisons with the speed of life in the city. | ~10–15 |
| 3: Breathing | The participants are standing. The UdN guide invites them to do a US Army breathing exercise and to focus on smells. The guide explains what scientists have found about certain particles emitted by trees and their healing benefits. S/he will also discuss facts about the air that is breathed in the city. The participants will practice a breathing exercise that they can repeat on a daily basis. | ~10–15 |
| 4: Old tree | The participants are invited to lie down directly on the ground. The UdN guide explains the scientific discoveries that are related to the body’s contact with the ground. The participants practice an exercise that they can repeat on a daily basis. The participants are also invited to grab a handful of dirt and to observe and smell the contents. | ~20 |
| 5: Walking barefoot | The participants are invited to remove their shoes and socks for the duration of the walk to the next stop. | ~5 |
| 6: Ferns | The participants are invited to observe and listen to their immediate environment. The UdN guide explains Dr. Ulrich’s “View through the window” study [35] and the uses of nature in hospitals since this discovery. The participants practice an exercise that they can repeat on a daily basis. | ~20–30 |
| 7: Pine tree refuge | The guide explains the discoveries that demonstrate the positive impact of nature on mental health and invites participants to find their own refuge in this clearing. They stay here for a couple of minutes and then meet the guide. The participants are invited to taste some resin from the pine trees. | ~10–15 |
| 8: Post-forest | the guides lead a group discussion about the participants’ forest experience. The participants fill out post-forest POMS. | ~15–20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, D.; Serra, N.I.; Mansion, H.; Mansion, E.T.; Blain-Moraes, S. Assessing the Effects of Nature on Physiological States Using Wearable Technologies. Int. J. Environ. Res. Public Health 2022, 19, 1231. https://doi.org/10.3390/ijerph19031231
Fu D, Serra NI, Mansion H, Mansion ET, Blain-Moraes S. Assessing the Effects of Nature on Physiological States Using Wearable Technologies. International Journal of Environmental Research and Public Health. 2022; 19(3):1231. https://doi.org/10.3390/ijerph19031231
Chicago/Turabian StyleFu, Dannie, Natalia Incio Serra, Hubert Mansion, Emilia Tamko Mansion, and Stefanie Blain-Moraes. 2022. "Assessing the Effects of Nature on Physiological States Using Wearable Technologies" International Journal of Environmental Research and Public Health 19, no. 3: 1231. https://doi.org/10.3390/ijerph19031231
APA StyleFu, D., Serra, N. I., Mansion, H., Mansion, E. T., & Blain-Moraes, S. (2022). Assessing the Effects of Nature on Physiological States Using Wearable Technologies. International Journal of Environmental Research and Public Health, 19(3), 1231. https://doi.org/10.3390/ijerph19031231






