Effects of Environmental and Socioeconomic Inequalities on Health Outcomes: A Multi-Region Time-Series Study
Highlights
- The maximum temperature and relative humidity are the variables of greatest risk for cardiovascular diseases.
- The minimum temperature represents the highest risk variable for respiratory diseases.
- Mental diseases are influenced by extreme temperatures.
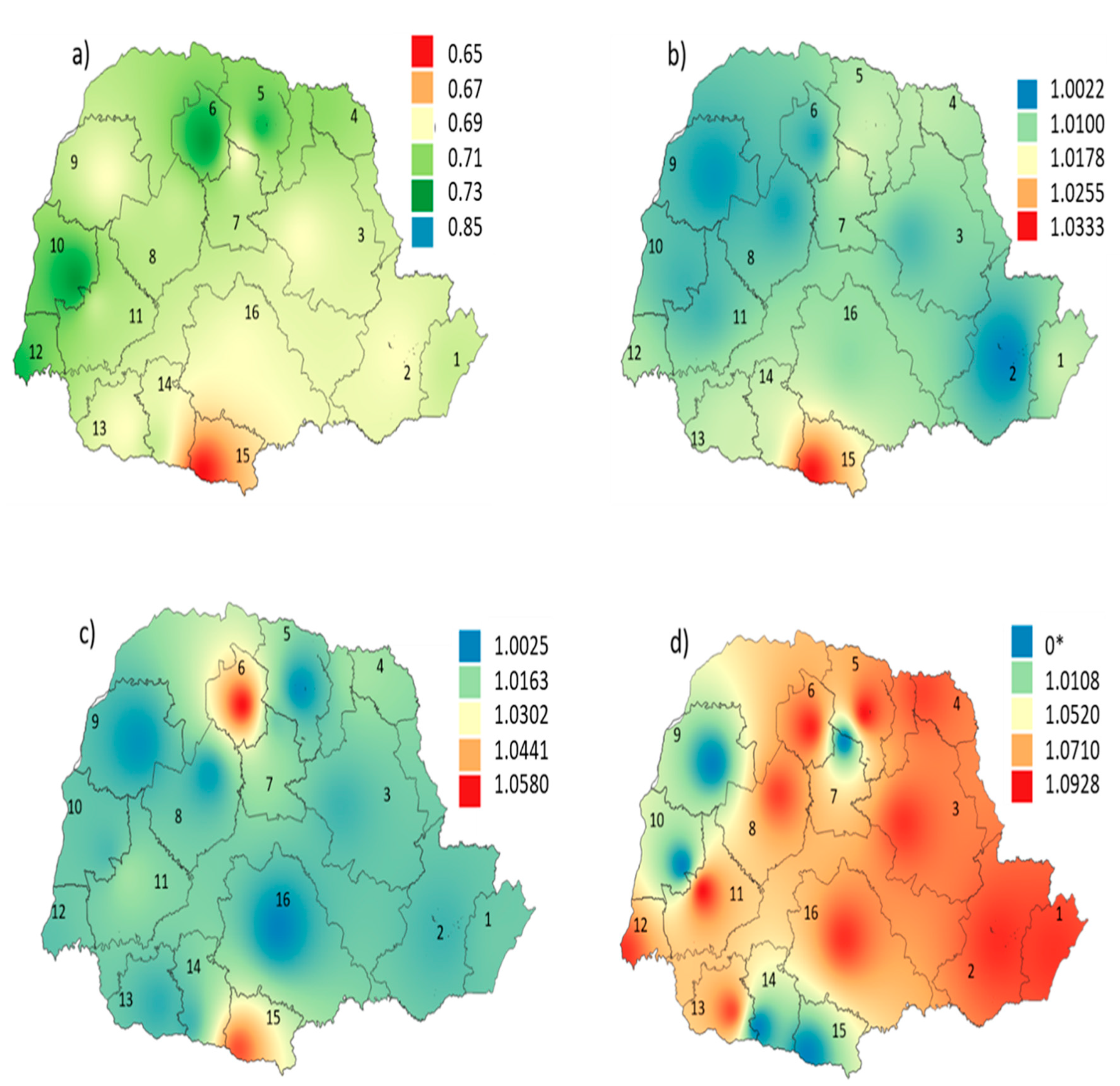
- The relative risk varied among regions as a function of the socioeconomic conditions and climate.
Abstract
1. Introduction
2. Materials and Methods
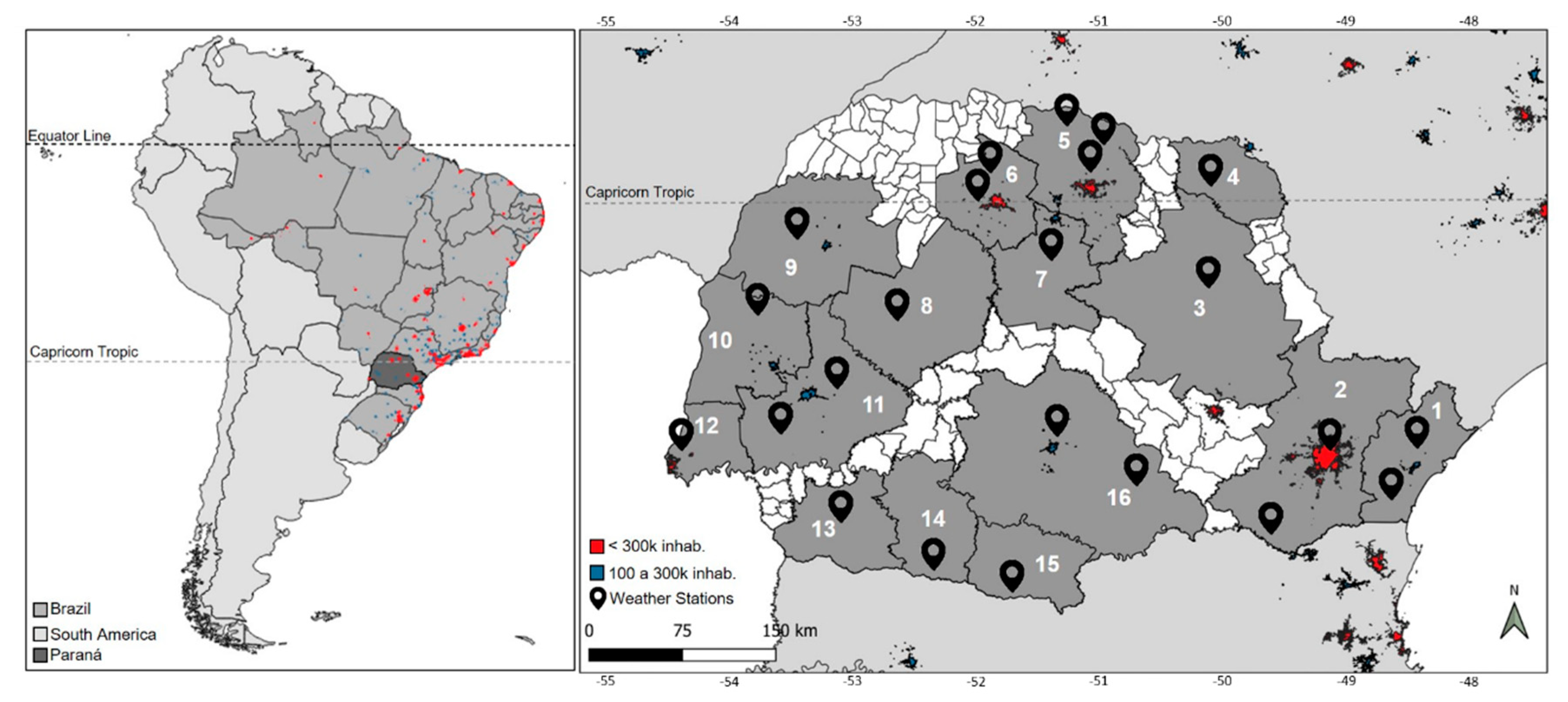
2.1. Study Area
2.2. Data Collection
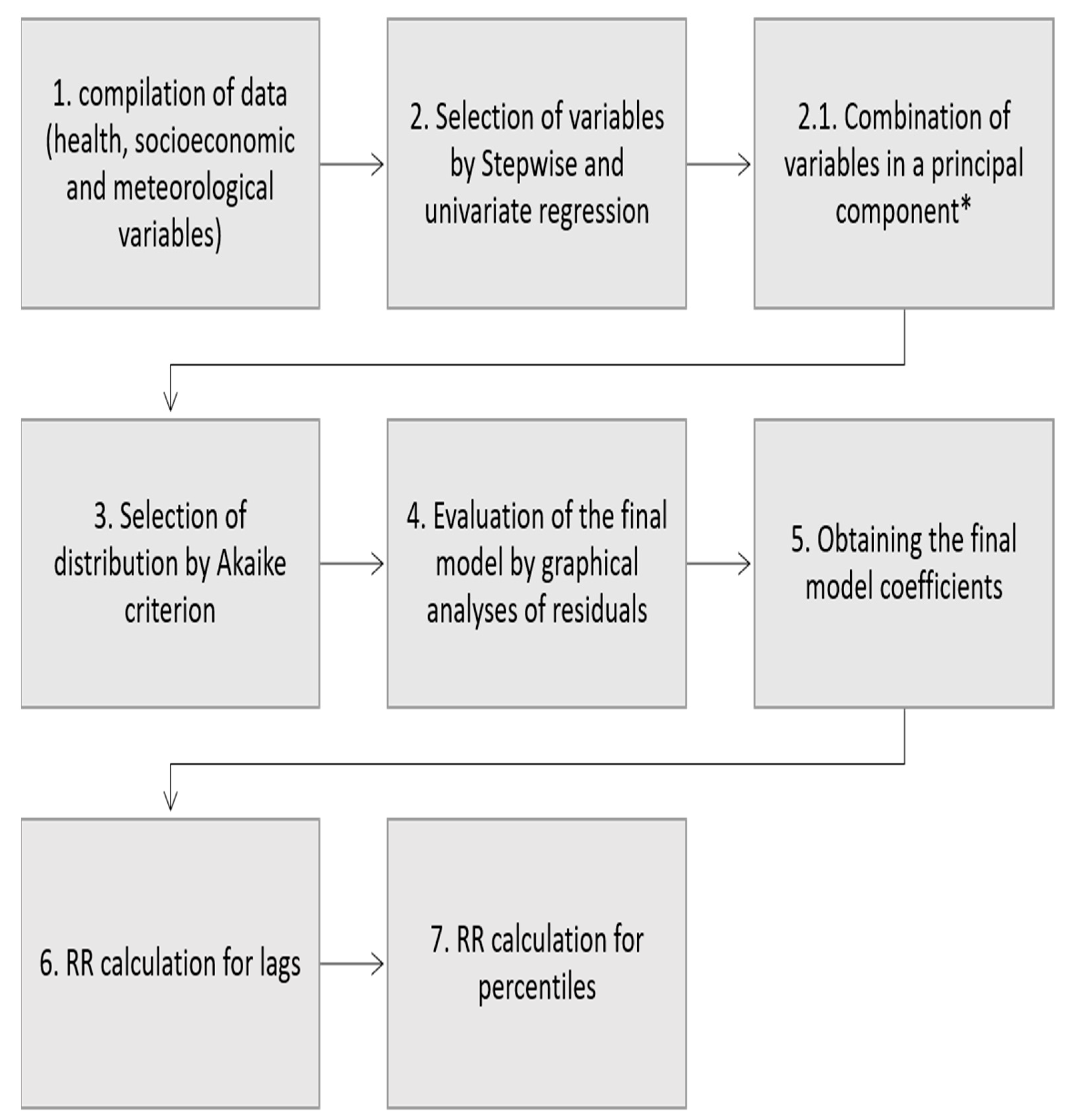
2.3. Statistical Methods
3. Results
3.1. Description and Characteristics of the Study Variables
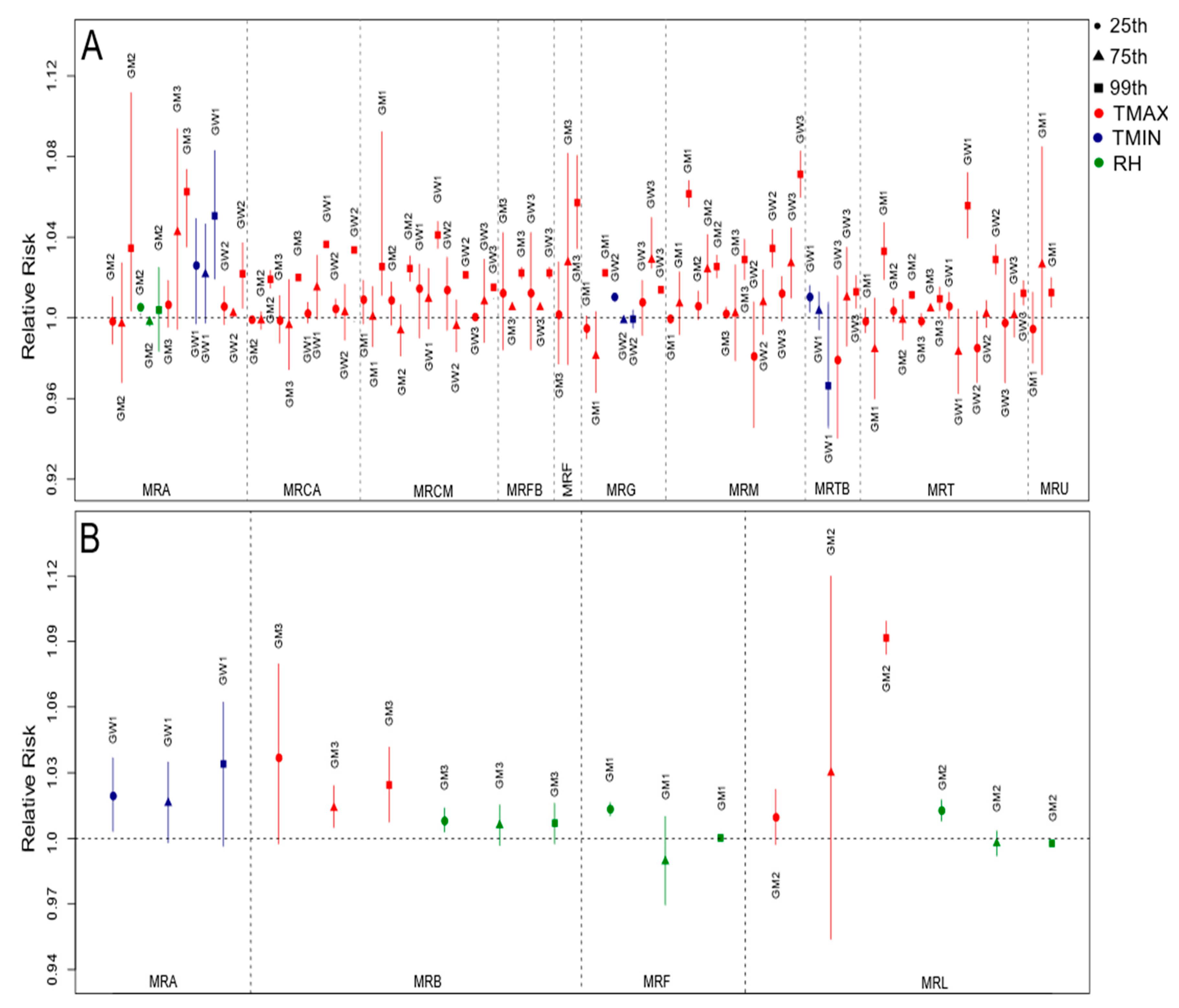
3.2. Regional Variability in Short- and Long-Term Effects of the Environmental and Socioeconomic Conditions on Health
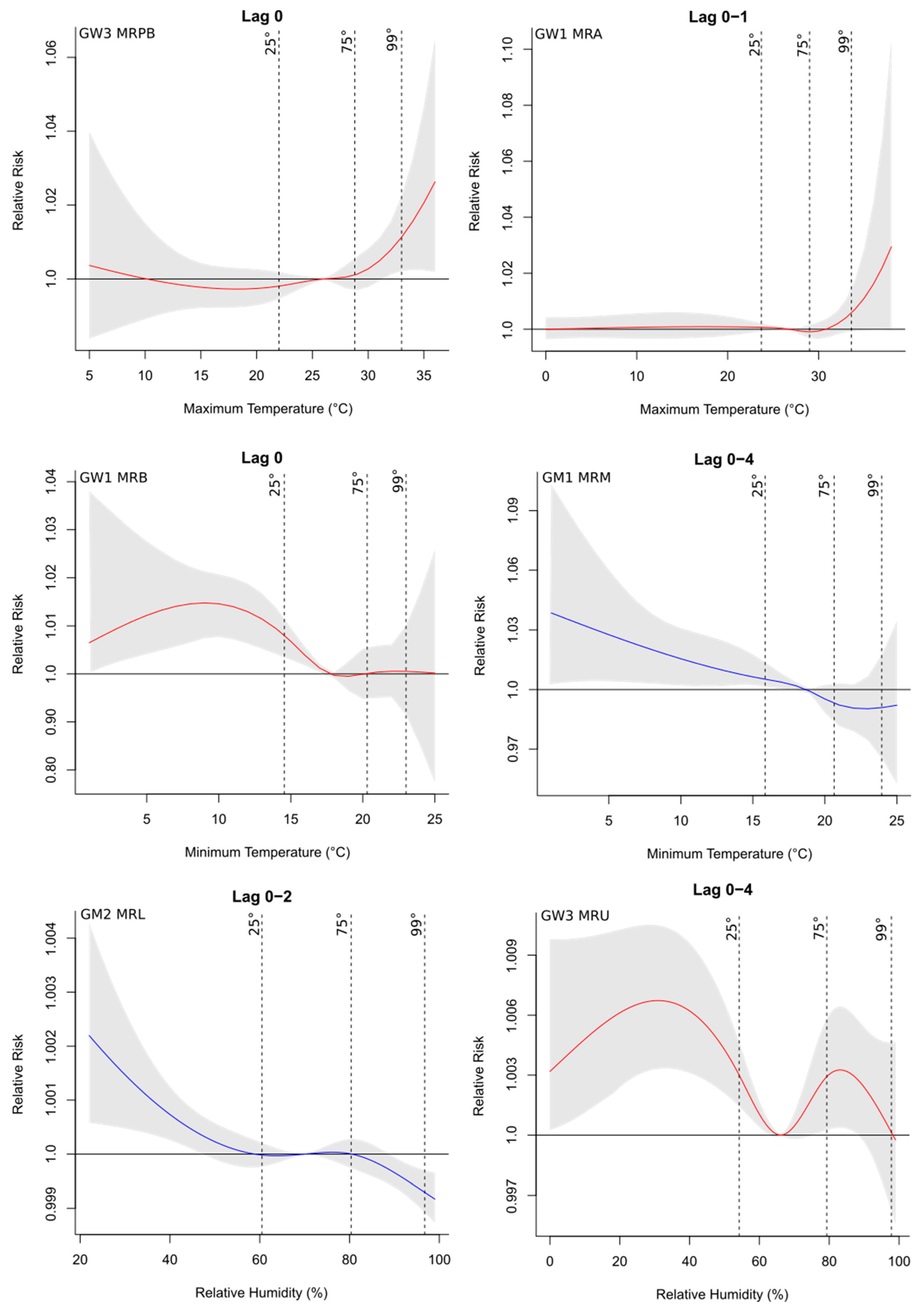
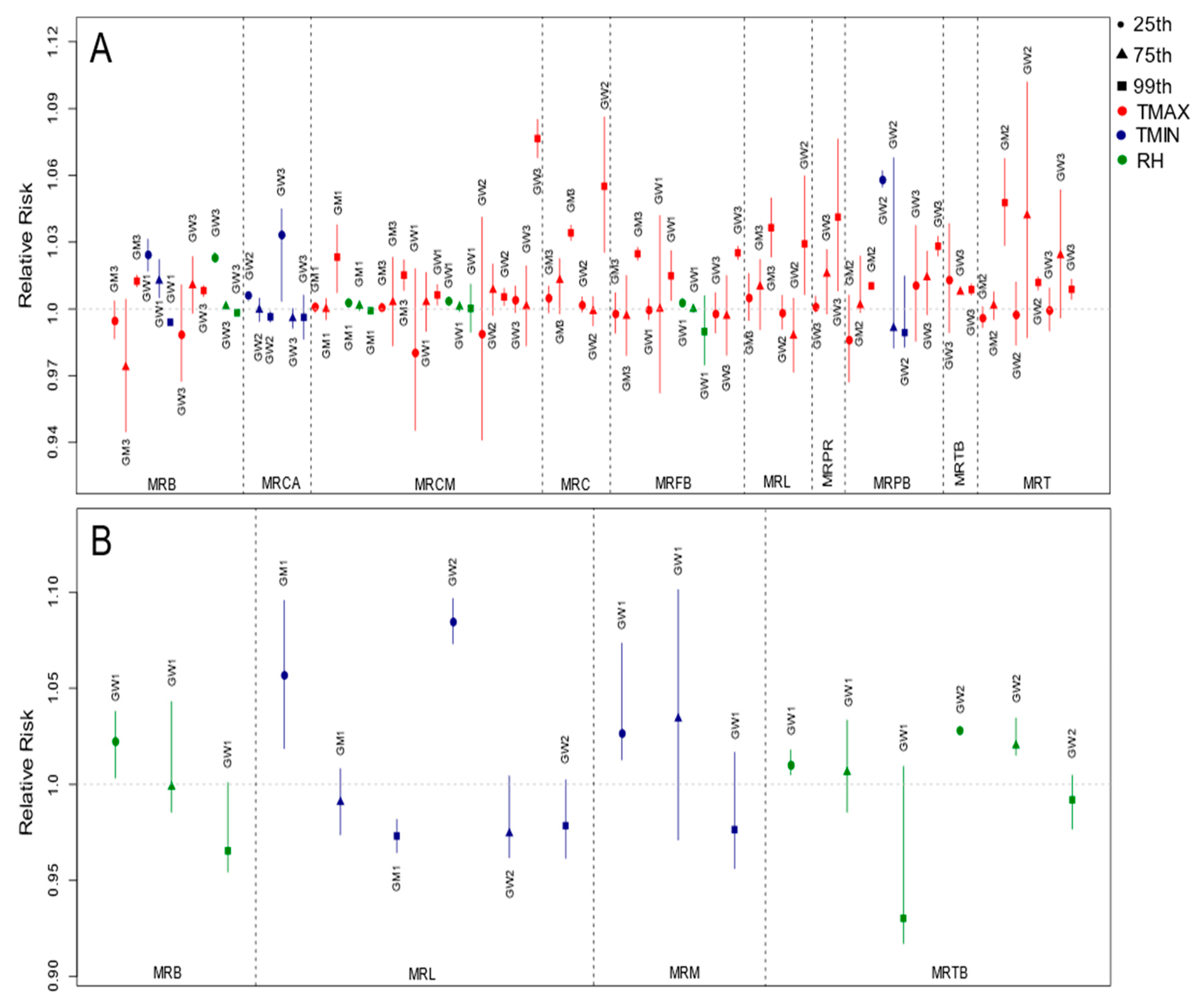
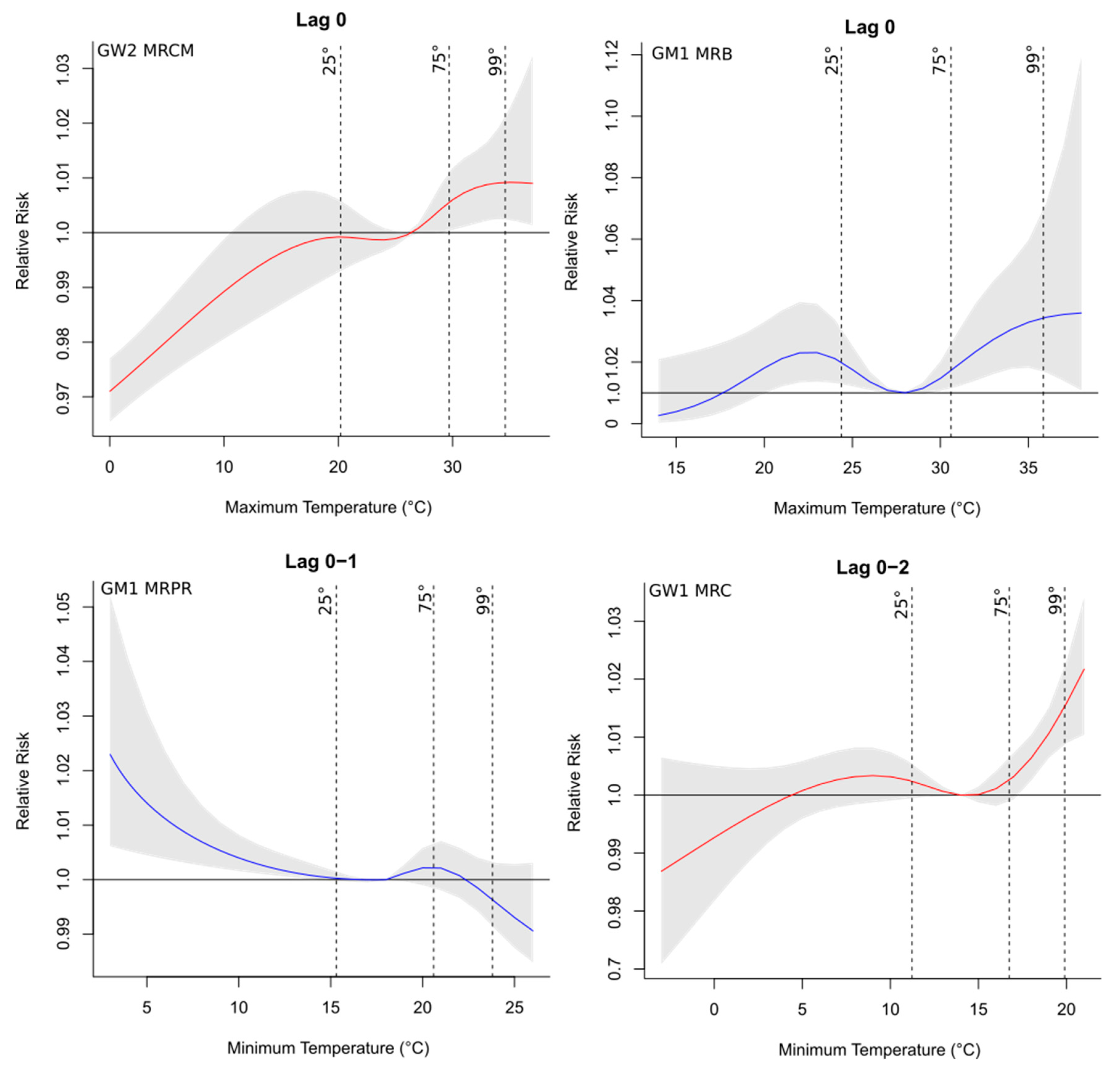
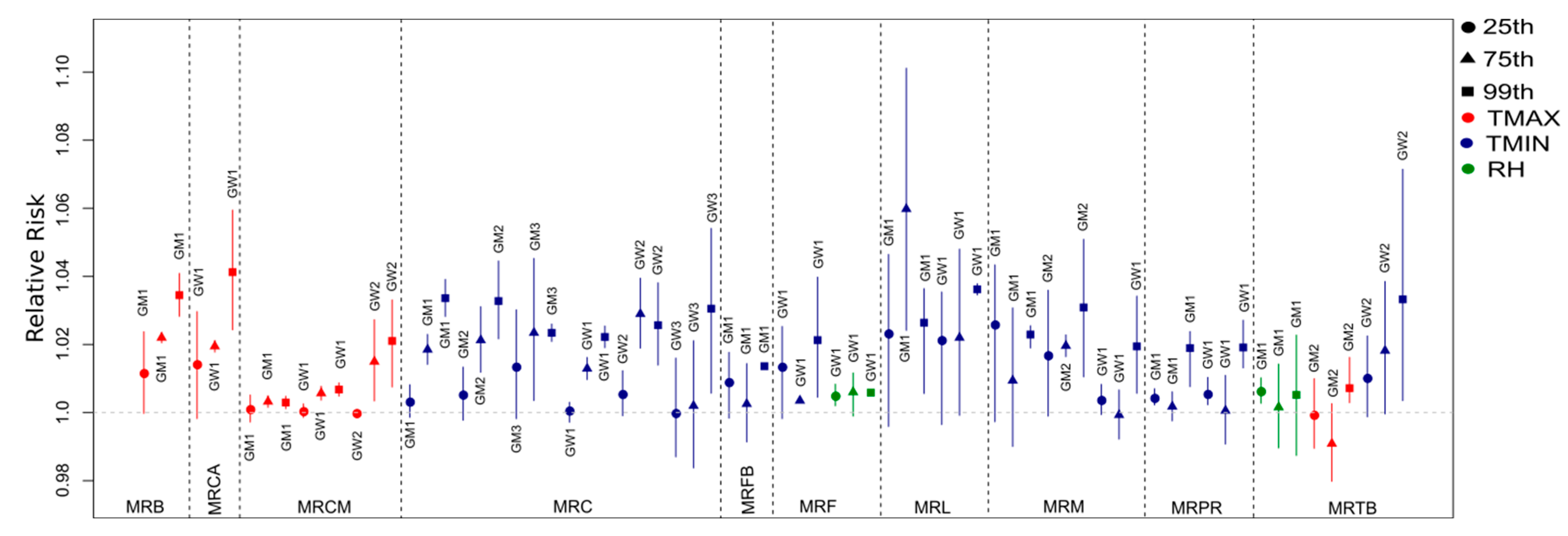
3.2.1. Cardiovascular Diseases
3.2.2. Respiratory Diseases
3.2.3. Mental Illnesses and Disorders
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chakrabarty, D. The Politics of Climate Change Is More Than the Politics of Capitalism. Theory Cult. Soc. 2017, 34, 25–37. [Google Scholar] [CrossRef]
- Lade, S.J.; Fetzer, I.; Cornell, S.E.; Crona, B. A prototype Earth system impact metric that accounts for cross-scale interactions. Environ. Res. Lett. 2021, 16, 115005. [Google Scholar] [CrossRef]
- O’Brien, K.; Leichenko, R.; Kelkar, U.; Venema, H.; Aandahl, G.; Tompkins, H.; Javed, A.; Bhadwal, S.; Barg, S.; Nygaard, L.; et al. Mapping vulnerability to multiple stressors: Climate change and globalization in India. Glob. Environ. Chang. 2004, 14, 303–313. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014. Synthesis Report. Versión Inglés. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014; ISBN 9789291691432.
- Zhao, Q.; Zhao, Y.; Li, S.; Zhang, Y.; Wang, Q.; Zhang, H.; Qiao, H.; Li, W.; Huxley, R.; Williams, G.; et al. Impact of ambient temperature on clinical visits for cardio-respiratory diseases in rural villages in northwest China. Sci. Total Environ. 2018, 612, 379–385. [Google Scholar] [CrossRef]
- Ebi, K.L.; Capon, A.; Berry, P.; Broderick, C.; de Dear, R.; Havenith, G.; Honda, Y.; Kovats, R.S.; Ma, W.; Malik, A.; et al. Hot weather and heat extremes: Health risks. Lancet 2021, 398, 698–708. [Google Scholar] [CrossRef]
- Lawson, C.R.; Vindenes, Y.; Bailey, L.; van de Pol, M. Environmental variation and population responses to global change. Ecol. Lett. 2015, 18, 724–736. [Google Scholar] [CrossRef]
- Sinclair, B.J.; Marshall, K.E.; Sewell, M.A.; Levesque, D.L.; Willett, C.S.; Slotsbo, S.; Dong, Y.; Harley, C.D.G.; Marshall, D.J.; Helmuth, B.S.; et al. Can we predict ectotherm responses to climate change using thermal performance curves and body temperatures? Ecol. Lett. 2016, 19, 1372–1385. [Google Scholar] [CrossRef]
- Gilman, S.E.; Wethey, D.S.; Helmuth, B. Variation in the sensitivity of organismal body temperature to climate change over local and geographic scales. Proc. Natl. Acad. Sci. USA 2006, 103, 9560–9565. [Google Scholar] [CrossRef]
- Qi, L.; Wu, Q.; Zhang, L.; Liu, J. Effect of environmental parameters on heat transfer characteristics and thermal sensation of human body with step change of temperature. Heat Mass Transf. Stoffuebertragung 2022, 58, 873–885. [Google Scholar] [CrossRef]
- Franchini, M.; Mannucci, P.M. Impact on human health of climate changes. Eur. J. Intern. Med. 2015, 26, 1–5. [Google Scholar] [CrossRef]
- Hamdi, M.; Lachiver, G.; Michaud, F. A new predictive thermal sensation index of human response. Energy Build. 2002, 29, 167–178. [Google Scholar] [CrossRef]
- Fiala, D.; Lomas, K.J.; Stohrer, M. First Principles Modeling of Thermal Sensation Responses in Steady-State and Transient Conditions. ASHRAE Trans. 2003, 109, 179–186. [Google Scholar]
- Yang, J.; Yin, P.; Zhou, M.; Ou, C.Q.; Guo, Y.; Gasparrini, A.; Liu, Y.; Yue, Y.; Gu, S.; Sang, S.; et al. Cardiovascular mortality risk attributable to ambient temperature in China. Heart 2015, 101, 1966–1972. [Google Scholar] [CrossRef] [PubMed]
- Bunker, A.; Wildenhain, J.; Vandenbergh, A.; Henschke, N.; Rocklöv, J.; Hajat, S.; Sauerborn, R. Effects of Air Temperature on Climate-Sensitive Mortality and Morbidity Outcomes in the Elderly; a Systematic Review and Meta-analysis of Epidemiological Evidence. EBioMedicine 2016, 6, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Gasparrini, A.; Guo, Y.; Hashizume, M.; Lavigne, E.; Zanobetti, A.; Schwartz, J.; Tobias, A.; Tong, S.; Rocklöv, J.; Forsberg, B.; et al. Mortality risk attributable to high and low ambient temperature: A multicountry observational study. Lancet 2015, 386, 369–375. [Google Scholar] [CrossRef]
- Phung, D.; Guo, Y.; Thai, P.; Rutherford, S.; Wang, X.; Nguyen, M.; Do, C.M.; Nguyen, N.H.; Alam, N.; Chu, C. The effects of high temperature on cardiovascular admissions in the most populous tropical city in Vietnam. Environ. Pollut. 2016, 208, 33–39. [Google Scholar] [CrossRef]
- Song, X.; Wang, S.; Hu, Y.; Yue, M.; Zhang, T.; Liu, Y.; Tian, J.; Shang, K. Impact of ambient temperature on morbidity and mortality: An overview of reviews. Sci. Total Environ. 2017, 586, 241–254. [Google Scholar] [CrossRef]
- Sherbakov, T.; Malig, B.; Guirguis, K.; Gershunov, A.; Basu, R. Ambient temperature and added heat wave effects on hospitalizations in California from 1999 to 2009. Environ. Res. 2018, 160, 83–90. [Google Scholar] [CrossRef]
- Cui, L.; Geng, X.; Ding, T.; Tang, J.; Xu, J.; Zhai, J. Impact of ambient temperature on hospital admissions for cardiovascular disease in Hefei City, China. Int. J. Biometeorol. 2019, 63, 723–734. [Google Scholar] [CrossRef]
- Yin, Q.; Wang, J.; Ren, Z.; Li, J.; Guo, Y. Mapping the increased minimum mortality temperatures in the context of global climate change. Nat. Commun. 2019, 10, 4640. [Google Scholar] [CrossRef]
- Trang, P.M.; Rocklöv, J.; Giang, K.B.; Kullgren, G.; Nilsson, M. Heatwaves and hospital admissions for mental disorders in Northern Vietnam. PLoS ONE 2016, 11, e0155609. [Google Scholar] [CrossRef]
- Peng, Z.; Wang, Q.; Kan, H.; Chen, R.; Wang, W. Effects of ambient temperature on daily hospital admissions for mental disorders in Shanghai, China: A time-series analysis. Sci. Total Environ. 2017, 590–591, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Santana, P.; Rocha, A. Effects of extreme temperatures on cerebrovascular mortality in Lisbon: A distributed lag non-linear model. Int. J. Biometeorol. 2019, 63, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Thackeray, S.J.; Henrys, P.A.; Hemming, D.; Bell, J.R.; Botham, M.S.; Burthe, S.; Helaouet, P.; Johns, D.G.; Jones, I.D.; Leech, D.I.; et al. Phenological sensitivity to climate across taxa and trophic levels. Nature 2016, 535, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Ghisletta, P.; Renaud, O.; Fagot, D.; Lecerf, T.; de Ribaupierre, A. Age and sex differences in intra-individual variability in a simple reaction time task. Int. J. Behav. Dev. 2018, 42, 294–299. [Google Scholar] [CrossRef]
- Pörtner, H.O.; Farrell, A.P. Ecology: Physiology and climate change. Science 2008, 322, 690–692. [Google Scholar] [CrossRef] [PubMed]
- Huey, R.B.; Kearney, M.R.; Krockenberger, A.; Holtum, J.A.M.; Jess, M.; Williams, S.E. Predicting organismal vulnerability to climate warming: Roles of behaviour, physiology and adaptation. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 1665–1679. [Google Scholar] [CrossRef] [PubMed]
- Giorgini, P.; Di Giosia, P.; Petrarca, M.; Lattanzio, F.; Stamerra, C.A.; Ferri, C. Climate Changes and Human Health: A Review of the Effect of Environmental Stressors on Cardiovascular Diseases Across Epidemiology and Biological Mechanisms. Curr. Pharm. Des. 2017, 23, 3247–3261. [Google Scholar] [CrossRef]
- Romanello, M.; McGushin, A.; Di Napoli, C.; Drummond, P.; Hughes, N.; Jamart, L.; Kennard, H.; Lampard, P.; Solano Rodriguez, B.; Arnell, N.; et al. The 2021 report of the Lancet Countdown on health and climate change: Code red for a healthy future. Lancet 2021, 398, 1619–1662. [Google Scholar] [CrossRef]
- McCartney, G.; Hart, C.; Watt, G. How can socioeconomic inequalities in hospital admissions be explained? a cohort study. BMJ Open 2013, 3, e002433. [Google Scholar] [CrossRef]
- World Health Organization. WHO Global Air Quality Guidelines; WHO: Geneva, Switzerland, 2021; pp. 1–360. [Google Scholar]
- IBGE. Projeções da população: Brasil e unidades da federação. In Coordenação de População e Indicadores Sociais; IBGE: Rio de Janeiro, Brazil, 2018. [Google Scholar]
- IPARDES. Cadernos Ipardes. 2018. Available online: http://www.ipardes.pr.gov.br/ojs/index.php/caderno (accessed on 1 March 2020).
- IBGE. Panorama Curitiba. 2018. Available online: https://cidades.ibge.gov.br/brasil/pr/curitiba/panorama (accessed on 25 March 2019).
- Alvares, C.A.; Stape, J.L.; Sentelhas, P.C.; De Moraes Gonçalves, J.L.; Sparovek, G. Köppen’s climate classification map for Brazil. Meteorol. Z. 2013, 22, 711–728. [Google Scholar] [CrossRef] [PubMed]
- IAP—Instituto Ambiental do Paraná. Inventário Estadual de Emissões Atmosféricas de Poluentes (MP, CO, NOx, SOx) e Propostas Para Revisão e Ampliação da Rede de Monitoramento da Qualidade do ar do Estado do Paraná—Relatório Final; IAP: São José, Brazil, 2013.
- de Oliveira Aparecido, L.E.; Rolim, G.D.S.; Richetti, J.; de Souza, P.S.; Johann, J.A. Köppen, Thornthwaite and Camargo climate classifications for climatic zoning in the State of Paraná, Brazil. Cienc. Agrotecnol. 2016, 40, 405–417. [Google Scholar] [CrossRef]
- Blair, J.; Lacy, M.G. From the SAGE Social Science Collections. Rights Reserved. Sociol. Methods Res. 2000, 28, 251–280. [Google Scholar] [CrossRef]
- Krüger, E.L.; Rossi, F.A. Effect of personal and microclimatic variables on observed thermal sensation from a field study in southern Brazil. Build. Environ. 2011, 46, 690–697. [Google Scholar] [CrossRef]
- Wachter, K.; Finch, C. Biodemography of Aging. 1997, pp. 1–18. Available online: http://scholar.google.es/scholar?q=The+Biodemography+of+Aging&btnG=&hl=es&as_sdt=0#6 (accessed on 1 September 2021).
- Xiong, J.; Ma, T.; Lian, Z.; de Dear, R. Perceptual and physiological responses of elderly subjects to moderate temperatures. Build. Environ. 2019, 156, 117–122. [Google Scholar] [CrossRef]
- IPARDES. Índice de Desenvolvimento Humano Municipal segundo os municípios do Paraná. Inst. Parana. Desenvolv. Econômico Soc. Indicadores Sociais. Índice Desenvolv. Hum. Munic. 2010; pp. 1–11. Available online: http://www.ipardes.gov.br/pdf/indices/IDHM_municipios_pr.pdf (accessed on 1 July 2022).
- IPARDES. Índice Ipardes de Desempenho Municipal (IPDM). 2019. Available online: https://www.ipardes.pr.gov.br/sites/ipardes/arquivos_restritos/files/documento/2022-01/IPDM_2018_2019_comentários.pdf (accessed on 1 July 2022).
- Aikaike, H. A New Look at the Statistical Model Identification. IEEE Trans. Automat. Contr. 1974, AC-19, 716–723. [Google Scholar] [CrossRef]
- Zhang, Z. Variable selection with stepwise and best subset approaches. Ann. Transl. Med. 2016, 4, 136. [Google Scholar] [CrossRef]
- Kalnins, A. Multicollinearity: How common factors cause Type 1 errors in multivariate regression. Strateg. Manag. J. 2018, 39, 2362–2385. [Google Scholar] [CrossRef]
- Fox, J.; Monette, G. Generalized collinearity diagnostics. J. Am. Stat. Assoc. 1992, 87, 178–183. [Google Scholar] [CrossRef]
- Wold, S.; Esbensen, K.; Geladi, P. Principal Component Analysis. Chemom. Intell. Lab. Syst. 1987, 2, 37–52. Available online: https://www.sciencedirect.com/science/article/abs/pii/0169743987800849 (accessed on 1 August 2022). [CrossRef]
- Aït-Sahalia, Y.; Xiu, D. Principal Component Analysis of High-Frequency Data. J. Am. Stat. Assoc. 2019, 114, 287–303. [Google Scholar] [CrossRef]
- Walker, A.S.; Mason, A.; Quan, T.P.; Fawcett, N.J.; Watkinson, P.; Llewelyn, M.; Stoesser, N.; Finney, J.; Davies, J.; Wyllie, D.H.; et al. Mortality risks associated with emergency admissions during weekends and public holidays: An analysis of electronic health records. Lancet 2017, 390, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Ardiles, L.G.; Tadano, Y.S.; Costa, S.; Urbina, V.; Capucim, M.N.; da Silva, I.; Braga, A.; Martins, J.A.; Martins, L.D. Negative Binomial regression model for analysis of the relationship between hospitalization and air pollution. Atmos. Pollut. Res. 2017, 9, 333–341. [Google Scholar] [CrossRef]
- de Souza Tadano, Y.; Ugaya, C.M.L.; Franco, A.T. Método de regressão de Poisson: Metodologia para avaliação do impacto da poluição atmosférica na saúde populacional. Ambient. Soc. 2010, 12, 241–255. [Google Scholar] [CrossRef]
- Bakonyi, S.M.C.; Danni-Oliveira, I.M.; Martins, L.C.; Braga, A.L.F. Air pollution and respiratory diseases among children in Brazil. Rev. Saude Publica J. Public Health 2004, 38, 695–700. Available online: http://www.popline.org/node/242055 (accessed on 1 November 2021). [CrossRef]
- Cheng, J.; Zhu, R.; Xu, Z.; Xu, X.; Wang, X.; Li, K.; Su, H. Temperature variation between neighboring days and mortality: A distributed lag non-linear analysis. Int. J. Public Health 2014, 59, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Gasparrini, A. Distributed Lag Linear and Non-Linear Models in R: The Package dlnm. J. Stat. Softw. 2011, 43, 1–20. [Google Scholar] [CrossRef]
- Gasparrini, A.; Armstrong, B.; Kenward, M.G. Distributed lag non-linear models. Stat. Med. 2010, 29, 2224–2234. [Google Scholar] [CrossRef]
- Uematsu, H.; Kunisawa, S.; Yamashita, K.; Fushimi, K.; Imanaka, Y. Impact of weekend admission on in-hospital mortality in severe community-acquired pneumonia patients in Japan. Respirology 2016, 21, 905–910. [Google Scholar] [CrossRef]
- Luan, G.; Yin, P.; Wang, L.; Zhou, M. The temperature–mortality relationship: An analysis from 31 Chinese provincial capital cities. Int. J. Environ. Health Res. 2018, 28, 192–201. [Google Scholar] [CrossRef]
- Louie, A.P.; Rowe, J.D.; Love, W.J.; Lehenbauer, T.W.; Aly, S.S. Effect of the environment on the risk of respiratory disease in preweaning dairy calves during summer months. J. Dairy Sci. 2018, 101, 10230–10247. [Google Scholar] [CrossRef] [PubMed]
- Mäkinen, T.M.; Juvonen, R.; Jokelainen, J.; Harju, T.H.; Peitso, A.; Bloigu, A.; Silvennoinen-Kassinen, S.; Leinonen, M.; Hassi, J. Cold temperature and low humidity are associated with increased occurrence of respiratory tract infections. Respir. Med. 2009, 103, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Huang, Z.; Wang, S.; Hu, J.; Xiao, J.; Li, X.; Liu, T.; Zeng, W.; Guo, L.; Du, Q.; et al. Morbidity burden of respiratory diseases attributable to ambient temperature: A case study in a subtropical city in China. Environ. Health A Glob. Access Sci. Source 2019, 18, 89. [Google Scholar] [CrossRef] [PubMed]
- Krendl, A.C.; Pescosolido, B.A. Countries and Cultural Differences in the Stigma of Mental Illness: The East–West Divide. J. Cross. Cult. Psychol. 2020, 51, 149–167. [Google Scholar] [CrossRef]
- Melo, L.C.; Sanquetta, C.R.; Corte, A.P.D.; Virgens Filho, J.S. Cenários Climáticos Futuros Para O Paraná: Oportunidades Para O Setor Florestal. Rev. Bras. Climatol. 2015, 16. [Google Scholar] [CrossRef]
- Neves, G.; Virgens Filho, J.; Leite, M.L.; Santos, E.N. Trendof air temperature in the state of Paraná, Brazil. Rev. Bras. Climatol. 2016, 18, 361–376. [Google Scholar]
- da Costa Santos, L.; José, J.V.; Bender, F.D.; Alves, D.S.; Nitsche, P.R.; dos Reis, E.F.; Coelho, R.D. Climate change in the Paraná state, Brazil: Responses to increasing atmospheric CO2 in reference evapotranspiration. Theor. Appl. Climatol. 2020, 140, 55–68. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, H.; Gasparrini, A.; Armstrong, B.; Bell, M.L.; Sera, F.; Lavigne, E.; Abrutzky, R.; Tong, S.; Coelho, M.D.S.Z.S.; et al. Predicted temperature-increase-induced global health burden and its regional variability. Environ. Int. 2019, 131, 105027. [Google Scholar] [CrossRef]
- Dadbakhsh, M.; Khanjani, N.; Bahrampour, A.; Haghighi, P.S. Death from respiratory diseases and temperature in Shiraz, Iran (2006–2011). Int. J. Biometeorol. 2017, 61, 239–246. [Google Scholar] [CrossRef]
- Sun, S.; Laden, F.; Hart, J.E.; Qiu, H.; Wang, Y.; Wong, C.M.; Lee, R.S.Y.; Tian, L. Seasonal temperature variability and emergency hospital admissions for respiratory diseases: A population-based cohort study. Thorax 2018, 73, 951–958. [Google Scholar] [CrossRef]
- Parker, G.B.; Graham, R.K. Seasonal variations in rates of hospitalisation for mania and hypomania in psychiatric hospitals in NSW. J. Affect. Disord. 2016, 191, 289–291. [Google Scholar] [CrossRef] [PubMed]
- Wirz-Justice, A. Seasonality in affective disorders. Gen. Comp. Endocrinol. 2018, 258, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.L.; Yang, L.; Chan, K.P.; Chiu, S.S.; Chan, K.H.; Peiris, J.S.M.; Wong, C.M. Model selection in time series studies of influenza-associated mortality. PLoS ONE 2012, 7, e39423. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Wolff, R.; Yu, W.; Vaneckova, P.; Pan, X.; Tong, S. Ambient temperature and morbidity: A review of epidemiological evidence. Environ. Health Perspect 2012, 120, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Lewer, D.; Jayatunga, W.; Aldridge, R.W.; Edge, C.; Marmot, M.; Story, A.; Hayward, A. Premature mortality attributable to socioeconomic inequality in England between 2003 and 2018: An observational study. Lancet Public Health 2020, 5, e33–e41. [Google Scholar] [CrossRef]
- Polak, M.; Genowska, A.; Szafraniec, K.; Fryc, J.; Jamiołkowski, J.; Pająk, A. Area-based socio-economic inequalities in mortality from lung cancer and respiratory diseases. Int. J. Environ. Res. Public Health 2019, 16, 1791. [Google Scholar] [CrossRef]
- Kysely, J.; Pokorna, L.; Kyncl, J.; Kriz, B. Excess cardiovascular mor- tality associated with cold spells in the Czech Republic. BMC Public Health 2009, 2, 19. [Google Scholar]
- De’Donato, F.K.; Leone, M.; Noce, D.; Davoli, M.; Michelozzi, P. The impact of the February 2012 cold spell on health in Italy using surveillance data. PLoS ONE 2013, 8, e61720. [Google Scholar] [CrossRef]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; Amann, M.; Anderson, H.R.; Andrews, K.G.; Aryee, M.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Eikelis, N.; Marques, F.Z.; Hering, D.; Marusic, P.; Head, G.A.; Walton, A.S.; Lambert, E.A.; Esler, M.D.; Sari, C.I.; Schlaich, M.P.; et al. A polymorphism in the noradrenaline transporter gene is associated with increased blood pressure in patients with resistant hypertension. J. Hypertens. 2018, 36, 1571–1577. [Google Scholar] [CrossRef]
- Wu, P.; Vaseghi, M. The autonomic nervous system and ventricular arrhythmias in myocardial infarction and heart failure. PACE Pacing Clin. Electrophysiol. 2020, 43, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yavar, Z.; Sun, Q. Cardiovascular response to ther- moregulatory challenges. Physiol. Heart Circ Physiol 2015, 309, H1793–H1812. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lavigne, E.; Ouellette-Kuntz, H.; Chen, B.E. Acute impacts of extreme temperature exposure on emergency room admissions related to mental and behavior disorders in Toronto, Canada. J. Affect. Disord. 2014, 155, 154–161. [Google Scholar] [CrossRef]
- Flouris, A.D.; McGinn, R.; Poirier, M.P.; Louie, J.C.; Ioannou, L.G.; Tsoutsoubi, L.; Sigal, R.J.; Boulay, P.; Hardcastle, S.G.; Kenny, G.P. Screening criteria for increased susceptibility to heat stress during work or leisure in hot environments in healthy individuals aged 31–70 years. Temperature 2018, 5, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Achebak, H.; Devolder, D.; Ballester, J. Trends in temperature-related age-specific and sex-specific mortality from cardiovascular diseases in Spain: A national time-series analysis. Lancet Planet. Health 2019, 3, e297–e306. [Google Scholar] [CrossRef]
- Dawa; Bai, L.; Cirendunzhu; Liu, Q.; Woodward, A.; Chen, B.; Zhaxisangmu. Temperature, hospital admissions and emergency room visits in Lhasa, Tibet: A time-series analysis. Sci. Total Environ. 2014, 490, 838–848. [Google Scholar]
- Onozuka, D.; Hagihara, A. Extreme temperature and out-of-hospital cardiac arrest in Japan: A nationwide, retrospective, observational study. Sci. Total Environ. 2017, 575, 258–264. [Google Scholar] [CrossRef]
- Zeng, J.; Zhang, X.; Yang, J.; Bao, J.; Xiang, H.; Dear, K.; Liu, Q.; Lin, S.; Lawrence, W.R.; Lin, A.; et al. Humidity may modify the relationship between temperature and cardiovascular mortality in Zhejiang province, China. Int. J. Environ. Res. Public Health 2017, 14, 1383. [Google Scholar] [CrossRef]
- Bai, L.; Li, Q.; Wang, J.; Lavigne, E.; Gasparrini, A.; Copes, R.; Yagouti, A.; Burnett, R.T.; Goldberg, M.S.; Cakmak, S.; et al. Increased coronary heart disease and stroke hospitalisations from ambient temperatures in Ontario. Heart 2018, 104, 673–679. [Google Scholar] [CrossRef]
- McGeehin, M.A.; Mirabelli, M. The potential impacts of climate variability and change on temperature-related morbidity and mortality in the United States. Environ. Health Perspect. 2001, 109, 185–189. [Google Scholar]
- Huynen, M.-M.; Martens, P.; Schram, D.; Weijenberg, M.P.; Kunst, A. The impact of heat waves and cold spells on mortality rates in the Dutch population. Environ. Health Perspect. 2001, 109, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Itani, M.; Ghaddar, N.; Ghali, K.; Laouadi, A. Bioheat modeling of elderly and young for prediction of physiological and thermal responses in heat-stressful conditions. J. Therm. Biol. 2020, 88, 102533. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Molecular, F. Exercise for Cardiovascular Disease Prevention and Treatment; Springer: Cham, Switzerland, 2017; ISBN 9789811043031. [Google Scholar]
- Hagström, L.; Henein, M.Y.; Karp, K.; Waldenström, A.; Lindqvist, P. Impact of age and sex on normal left heart structure and function. Clin. Physiol. Funct. Imaging 2017, 37, 759–766. [Google Scholar] [CrossRef]
- Choi, S.; Kim, K.; Kim, S.M.; Lee, G.; Jeong, S.M.; Park, S.Y.; Kim, Y.Y.; Son, J.S.; Yun, J.M.; Park, S.M. Association of obesity or weight change with coronary heart disease among young adults in South Korea. JAMA Intern. Med. 2018, 178, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Wåhlin, A.; Nyberg, L. At the Heart of Cognitive Functioning in Aging. Trends Cogn. Sci. 2019, 23, 717–720. [Google Scholar] [CrossRef] [PubMed]
- Iñiguez, C.; Royé, D.; Tobías, A. Contrasting patterns of temperature related mortality and hospitalization by cardiovascular and respiratory diseases in 52 Spanish cities. Environ. Res. 2021, 192, 110191. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, H.; Cheng, B.; Shen, J.; Li, H.; Guo, Y.; Cheng, Y. Health risk of extreme low temperature on respiratory diseases in western China. Environ. Sci. Pollut. Res. 2022, 29, 35760–35767. [Google Scholar] [CrossRef] [PubMed]
- da Silva Viana Jacobson, L.; de Oliveira, B.F.A.; Schneider, R.; Gasparrini, A.; de Souza Hacon, S. Mortality risk from respiratory diseases due to non-optimal temperature among Brazilian elderlies. Int. J. Environ. Res. Public Health 2021, 18, 5550. [Google Scholar] [CrossRef]
- Su, Q.; Liu, H.; Yuan, X.; Xiao, Y.; Zhang, X.; Sun, R.; Dang, W.; Zhang, J.; Qin, Y.; Men, B.; et al. The Interaction Effects of Temperature and Humidity on Emergency Room Visits for Respiratory Diseases in Beijing, China. Cell Biochem. Biophys. 2014, 70, 1377–1384. [Google Scholar] [CrossRef]
- Wolkoff, P. Indoor air humidity, air quality, and health—An overview. Int. J. Hyg. Environ. Health 2018, 221, 376–390. [Google Scholar] [CrossRef]
- Kudo, E.; Song, E.; Yockey, L.J.; Rakib, T.; Wong, P.W.; Homer, R.J.; Iwasaki, A. Low ambient humidity impairs barrier function and innate resistance against influenza infection. Proc. Natl. Acad. Sci. USA 2019, 166, 10905–10910. [Google Scholar] [CrossRef] [PubMed]
- Kenny, G.P.; Yardley, J.; Brown, C.; Sigal, R.J.; Jay, O. Heat stress in older individuals and patients with common chronic diseases. CMAJ 2010, 182, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Shaun, F.; Morrison, K.N. Central neural pathways for thermoregulation. Front. Biosci. 2011, 16, 74–104. [Google Scholar]
- Smith, C.J.; Johnson, J.M. Responses to hyperthermia. Optimizing heat dissipation by convection and evaporation: Neural control of skin blood flow and sweating in humans. Auton. Neurosci. Basic Clin. 2016, 196, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Yablonskiy, D.A.; Ackerman, J.J.H.; Raichle, M.E. Coupling between changes in human brain temperature and oxidative metabolism during prolonged visual stimulation. Proc. Natl. Acad. Sci. USA 2000, 97, 7603–7608. [Google Scholar] [CrossRef] [PubMed]
- Lõhmus, M. Possible Biological Mechanisms Linking Mental Health and Heat—A Contemplative Review. Int. J. Environ. Res. Public Health 2018, 15, 1515. [Google Scholar] [CrossRef]
- Vida, S.; Durocher, M.; Ouarda, T.B.M.J.; Gosselin, P. Relationship Between Ambient Temperature and Humidity and Visits to Mental Health Emergency Departments in Québec. Psychiatr. Serv. 2012, 63, 1150–1153. [Google Scholar] [CrossRef]
- Lee, S.; Lee, H.; Myung, W.; Kim, E.J.; Kim, H. Mental disease-related emergency admissions attributable to hot temperatures. Sci. Total Environ. 2018, 616–617, 688–694. [Google Scholar] [CrossRef]
- Moreira, J.; Peixoto, A. Mental illnesses in tropical climates (1906). Int. Rev. Psychiatry 2017, 29, 216–224. [Google Scholar] [CrossRef]
- da Silva, I.; Martins, L.D.; de Almeida, D.S.; Hashimoto, E.M. Risk assessment of temperature and air pollutants on hospitalizations for mental nd behavioral disorders in Curitiba, Brazil. Environ. Health 2019, 19, 79. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, Y.; Xie, X.H.; Li, H.; Han, R.; Hou, J.; Sun, J.; Qian, Z.; Wu, S.; Huang, C.; et al. The effect of temperature on cause-specific mental disorders in three subtropical cities: A case-crossover study in China. Environ. Int. 2020, 143, 105938. [Google Scholar] [CrossRef] [PubMed]
- Mullins, J.T.; White, C. Temperature and mental health: Evidence from the spectrum of mental health outcomes. J. Health Econ. 2019, 68, 102240. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.L.; Bi, P.; Ryan, P.; Nitschke, M.; Pisaniello, D.; Tucker, G. The effect of heat waves on hospital admissions for renal disease in a temperate city of Australia. Int. J. Epidemiol. 2008, 37, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Wong, E.C.; Collins, R.L.; Cerully, J.L.; Yu, J.W.; Seelam, R. Effects of contact-based mental illness stigma reduction programs: Age, gender, and Asian, Latino, and White American differences. Soc. Psychiatry Psychiatr. Epidemiol. 2018, 53, 299–308. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, I.d.; Wikuats, C.F.H.; Hashimoto, E.M.; Martins, L.D. Effects of Environmental and Socioeconomic Inequalities on Health Outcomes: A Multi-Region Time-Series Study. Int. J. Environ. Res. Public Health 2022, 19, 16521. https://doi.org/10.3390/ijerph192416521
Silva Id, Wikuats CFH, Hashimoto EM, Martins LD. Effects of Environmental and Socioeconomic Inequalities on Health Outcomes: A Multi-Region Time-Series Study. International Journal of Environmental Research and Public Health. 2022; 19(24):16521. https://doi.org/10.3390/ijerph192416521
Chicago/Turabian StyleSilva, Iara da, Caroline Fernanda Hei Wikuats, Elizabeth Mie Hashimoto, and Leila Droprinchinski Martins. 2022. "Effects of Environmental and Socioeconomic Inequalities on Health Outcomes: A Multi-Region Time-Series Study" International Journal of Environmental Research and Public Health 19, no. 24: 16521. https://doi.org/10.3390/ijerph192416521
APA StyleSilva, I. d., Wikuats, C. F. H., Hashimoto, E. M., & Martins, L. D. (2022). Effects of Environmental and Socioeconomic Inequalities on Health Outcomes: A Multi-Region Time-Series Study. International Journal of Environmental Research and Public Health, 19(24), 16521. https://doi.org/10.3390/ijerph192416521





