Adults with Intellectual Disability and Autism Spectrum Disorder: What Is the Evidence around the Use of Polypharmacy
Abstract
1. Introduction
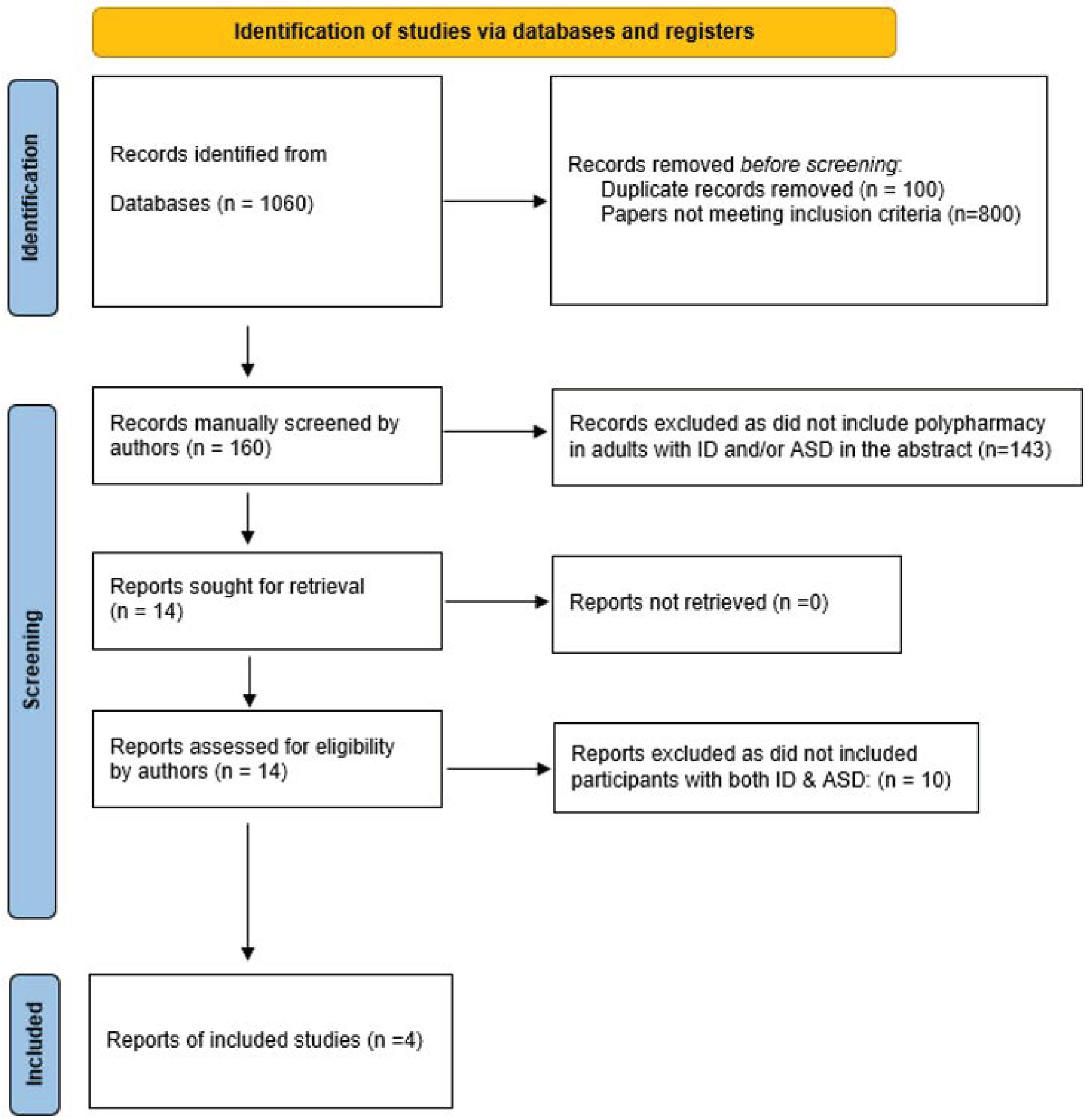
2. Materials and Methods
Inclusion and Exclusion Criteria
- Adults over the age of 18 years old;
- Research conducted between 2009 and 2021.
- Single case studies;
- Articles not written in English.
3. Results
3.1. Current Review
| No. | Author, Year | Participant Information | Location & Methods | Main Findings |
|---|---|---|---|---|
| 1 | Yoshida, K. et al. (2021) [21]. | 28 (Pharmacogenetics) PGx studies were identified through a systematic review. Three studies included adults with ASD/ID. The other studies focused on children and adolescents with ASD/ID. | A systematic review paper from Canada | There is limited data available from PGx studies in individuals with ASD/ID and in particular in adults. Given the potential for PGx testing in improving treatment outcomes, additional pharmacogenomic studies for psychotropic treatment in ASD/ID across age groups are warranted. |
| 2 | Espadas, C. et al. (2020) [24]. | Adults with ASD-ID (n = 83). | An observational, multicentre pharmacovigilance study in Spain of patients diagnosed with ASD and ID. | Risperidone and quetiapine were co-prescribed in 60% of the cases without any monitoring of adverse events being routine. The rates of multimorbidity and polypharmacy, among young adults with ASD and ID, were concerning. |
| 3 | Miot, S. et al. (2019) [25]. | Adults with ASD-ID (n = 63). | Detailed clinical examinations from nine specialist institutions in France. | A large range of comorbidities, gastrointestinal disorders and mental and neurological diseases were identified. Overall, 25% of ASD-ID sample had chronic kidney disease with associated increased cardiovascular risk factors. The comorbidity burden was high and comparable with that observed among older hospitalised patients in geriatric departments. Furthermore, the comorbidity burden positively correlated with age, decreased autonomy, and polypharmacy. |
| 4 | Mehta H & Glover G (2019) [23]. | Not stated. | A quantitative, descriptive study using data from The Health Improvement Network (THIN) database of Primary Care Practices in England. | Analyses looked separately at adults with ID and autistic children and young people. For adults with ID, favourable changes in trend were seen for the overall prescribing of antipsychotics, antidepressants and anxiolytics, but further analysis concerning the presence of recorded appropriate clinical indications produced a complex picture. Some measures of polypharmacy showed improvement. |
3.1.1. Research Methods
3.1.2. Potential of Pharmacogenetics
3.1.3. Trends in Prescribing in Response to National Policy
3.1.4. Multimorbidity and Patient Safety
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Underwood, L.; McCarthy, J.; Tsakanikos, E. Mental health of adults with autism spectrum disorders and intellectual disability. Curr. Opin. Psychiatry 2010, 23, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Payne, R.A. The epidemiology of polypharmacy. Clin. Med. 2016, 16, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Lunsky, Y.; Modi, M. Predictors of Psychotropic Polypharmacy Among Outpatients With Psychiatric Disorders and Intellectual Disability. Psychiatr. Serv. 2018, 69, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Deb, S.; Unwin, G.; Deb, T. Characteristics and the trajectory of psychotropic medication use in general and antipsychotics in particular among adults with an intellectual disability who exhibit aggressive behaviour. J. Intellect. Disabil. Res. 2014, 59, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Deb, S.S.; Bertelli, M.O.; Rossi, M. Psychopharmacology. In Textbook of Psychiatry for Intellectual Disability and Autism Spectrum Disorder; Bertelli, M.O., Deb, S.S., Munir, K., Hassiotis, A., Salvador-Carulla, L., Eds.; Springer Nature: New York, NY, USA, 2022. [Google Scholar]
- Furukawa, Y.; Hamza, T.; Cipriani, A.; Furukawa, T.A.; Salanti, G.; Ostinelli, E.G. Optimal dose of aripiprazole for augmentation therapy of antidepressant-refractory depression: Preliminary findings based on a systematic review and dose–effect meta-analysis. Br. J. Psychiatry 2021, 221, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Bertelli, M.; Bianco, A. New insights on psychopathological assessment in persons with intellectual disability and low-functioning autism spectrum disorder. In Improving Mental Health in Persons with Intellectual Disability—From Science to Practice; Sappok, T., Burtscher, R., Eds.; Loeper: Karlsruhe, Germany, 2021. [Google Scholar]
- Bertelli, M. (Ed.) New Insights on Psychopathological Assessment in Persons with Intellectual Disability and Low-Functioning Autism Spectrum Disorder; Wiley: Hoboken, NJ, USA, 2021. [Google Scholar]
- McCarthy, J.; Hemmings, C.; Kravariti, E.; Dworzynski, K.; Holt, G.; Bouras, N.; Tsakanikos, E. Challenging behavior and co-morbid psychopathology in adults with intellectual disability and autism spectrum disorders. Res. Dev. Disabil. 2010, 31, 362–366. [Google Scholar] [CrossRef]
- Baudewijns, L.; Ronsse, E.; Verstraete, V.; Sabbe, B.; Morrens, M.; Bertelli, M.O. Problem behaviours and Major Depressive Disorder in adults with intellectual disability and autism. Psychiatry Res. 2018, 270, 769–774. [Google Scholar] [CrossRef]
- Peña-Salazar, C.; Arrufat, F.; López, J.-M.S.; Fontanet, A.; Roura-Poch, P.; Gil-Girbau, M.; Carbonell-Ducastella, C.; Serrano-Blanco, A. Intellectual Disability, Autism Spectrum Disorders, Psychiatric Comorbidities and Their Relationship with Challenging Behavior. J. Ment. Health Res. Intellect. Disabil. 2022, 15, 77–94. [Google Scholar] [CrossRef]
- Underwood, L.; McCarthy, J.; Tsakanikos, E.; Howlin, P.; Bouras, N.; Craig, T.K.J. Health and Social Functioning of Adults With Intellectual Disability and Autism. J. Policy Pr. Intellect. Disabil. 2012, 9, 147–150. [Google Scholar] [CrossRef]
- Howes, O.D.; Rogdaki, M.; Findon, J.; Wichers, R.H.; Charman, T.; King, B.H.; Loth, E.; McAlonan, G.; McCracken, J.T.; Parr, J.R.; et al. Autism spectrum disorder: Consensus guidelines on assessment, treatment and research from the British Association for Psychopharmacology. J. Psychopharmacol. 2017, 32, 3–29. [Google Scholar] [CrossRef]
- Fung, V.; Price, M.; Busch, A.B.; Landrum, M.B.; Fireman, B.; Nierenberg, A.A.; Newhouse, J.P.; Hsu, J. The introduction of generic risperidone in Medicare Part D. Am. J. Manag. Care 2016, 22, 41–48. [Google Scholar] [PubMed]
- Tyrer, P.; Oliver-Africano, P.C.; Ahmed, Z.; Bouras, N.; Cooray, S.; Deb, S.; Murphy, D.; Hare, M.; Meade, M.; Reece, B.; et al. Risperidone, haloperidol, and placebo in the treatment of aggressive challenging behaviour in patients with intellectual disability: A randomised controlled trial. Lancet 2008, 371, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.; Charman, T.; Havdahl, A.; Carbone, P.; Anagnostou, E.; Boyd, B.; Carr, T.; de Vries, P.J.; Dissanayake, C.; Divan, G.; et al. The Lancet Commission on the future of care and clinical research in autism. Lancet 2021, 399, 271–334. [Google Scholar] [CrossRef] [PubMed]
- Perry, B.I.; Cooray, S.E.; Mendis, J.; Purandare, K.; Wijeratne, A.; Manjubhashini, S.; Dasari, M.; Esan, F.; Gunaratna, I.; Naseem, R.A.; et al. Problem behaviours and psychotropic medication use in intellectual disability: A multinational cross-sectional survey. J. Intellect. Disabil. Res. 2018, 62, 140–149. [Google Scholar] [CrossRef]
- Branford, D.; Shankar, R. Antidepressant prescribing for adult people with an intellectual disability living in England. Br. J. Psychiatry 2022, 221, 488–493. [Google Scholar] [CrossRef]
- Londoño, A.; Ballester, P.; Martínez, E.; Javaloyes, A.; Planelles, B.; Pérez, A.; Peiró, A. Comorbidities as predictor of polypharmacy in autism spectrum disorder. Eur. Neuropsychopharmacol. 2016, 26, S730–S731. [Google Scholar] [CrossRef]
- Matson, J.L.; Neal, D. Psychotropic medication use for challenging behaviors in persons with intellectual disabilities: An overview. Res. Dev. Disabil. 2009, 30, 572–586. [Google Scholar] [CrossRef]
- NICE. Developing NICE Guidelines: The Manual; National Institute for Health and Care Excellence: London, UK, 2014. [Google Scholar]
- Yoshida, K.; Koyama, E.; Zai, C.C.; Beitchman, J.H.; Kennedy, J.L.; Lunsky, Y.; Desarkar, P.; Müller, D.J. Pharmacogenomic Studies in Intellectual Disabilities and Autism Spectrum Disorder: A Systematic Review. Can. J. Psychiatry 2020, 66, 1019–1041. [Google Scholar] [CrossRef]
- Mehta, H.; Glover, G. Psychotropic Drugs and People with Learning Disabilities or Autism; Public Health: London, UK, 2019. [Google Scholar]
- Espadas, C.; Ballester, P.; Londoño, A.C.; Almenara, S.; Aguilar, V.; Belda, C.; Pérez, E.; Peiró, A.M. Multimorbidity and psychotropic polypharmacy among participants with autism spectrum disorder with intellectual disability. Psychiatry Res. 2020, 292, 113321. [Google Scholar] [CrossRef]
- Miot, S.; Akbaraly, T.; Michelon, C.; Couderc, S.; Crepiat, S.; Loubersac, J.; Picot, M.-C.; Pernon, É.; Gonnier, V.; Jeandel, C.; et al. Comorbidity Burden in Adults with Autism Spectrum Disorders and Intellectual Disabilities—A Report From the EFAAR (Frailty Assessment in Ageing Adults with Autism Spectrum and Intellectual Disabilities) Study. Front. Psychiatry 2019, 10, 617. [Google Scholar] [CrossRef]
- Sheehan, R.; Hassiotis, A.; Walters, K.; Osborn, D.; Strydom, A.; Horsfall, L. Mental illness, challenging behaviour, and psychotropic drug prescribing in people with intellectual disability: UK population based cohort study. BMJ 2015, 351, h4326. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, A.; Lake, J.K.; Lunsky, Y.; Liu, S.-K.; Desarkar, P. Psychopharmacological treatment of challenging behaviours in adults with autism and intellectual disabilities: A systematic review. Res. Autism Spectr. Disord. 2014, 8, 803–813. [Google Scholar] [CrossRef]
- Deb, S.; Nancarrow, T.; Limbu, B.; Sheehan, R.; Wilcock, M.; Branford, D.; Courtenay, K.; Perera, B.; Shankar, R. UK psychiatrists’ experience of withdrawal of antipsychotics prescribed for challenging behaviours in adults with intellectual disabilities and/or autism. BJPsych Open 2020, 6. [Google Scholar] [CrossRef] [PubMed]
- De Kuijper, G.M.; Hoekstra, P.J. An open-label discontinuation trial of long-term, off-label antipsychotic medication in people with intellectual disability: Determinants of success and failure. J. Clin. Pharmacol. 2018, 58, 1418–1426. [Google Scholar] [CrossRef]
- Shankar, R.; Wilcock, M.; Deb, S.; Goodey, R.; Corson, E.; Pretorius, C.; Praed, G.; Pell, A.; Vujkovic, D.; Wilkinson, E.; et al. A structured programme to withdraw antipsychotics among adults with intellectual disabilities: The Cornwall experience. J. Appl. Res. Intellect. Disabil. 2019, 32, 1389–1400. [Google Scholar] [CrossRef] [PubMed]
- Royal College of Psychiatrists. Psychotropic Drug Prescribing for People with Intellectual Disability, Mental Health Problems and/or Behaviours That Challenge: Practice Guidelines; Contract No.: FR/ID/09; Royal College of Psychiatrists: London, UK, 2016. [Google Scholar]
- Stortz, J.N.; Lake, J.K.; Cobigo, V.; Ouellette-Kuntz, H.M.; Lunsky, Y. Lessons learned from our elders: How to study polypharmacy in populations with intellectual and developmental disabilities. Ment. Retard. 2014, 52, 60–77. [Google Scholar] [CrossRef] [PubMed]
- Beumer, S.; Hamers, P.; Oppewal, A.; Maes-Festen, D. Antipsychotics withdrawal in adults with intellectual disability and challenging behaviour: Study protocol for a multicentre double-blind placebo-controlled randomised trial. BMC Psychiatry 2021, 21, 439. [Google Scholar] [CrossRef]
| Population | Autism Spectrum Disorder (ASD) and Intellectual Disability |
|---|---|
| Intervention | Psychopharmacology antipsychotics, antidepressants, mood stabilisers, anxiolytics, Polypharmacy |
| Comparison | High-functioning ASD |
| Outcome | Symptom improvement, Remission of psychiatric disorder, Quality of life |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCarthy, J.M.; Chaplin, E. Adults with Intellectual Disability and Autism Spectrum Disorder: What Is the Evidence around the Use of Polypharmacy. Int. J. Environ. Res. Public Health 2022, 19, 15974. https://doi.org/10.3390/ijerph192315974
McCarthy JM, Chaplin E. Adults with Intellectual Disability and Autism Spectrum Disorder: What Is the Evidence around the Use of Polypharmacy. International Journal of Environmental Research and Public Health. 2022; 19(23):15974. https://doi.org/10.3390/ijerph192315974
Chicago/Turabian StyleMcCarthy, Jane M., and Eddie Chaplin. 2022. "Adults with Intellectual Disability and Autism Spectrum Disorder: What Is the Evidence around the Use of Polypharmacy" International Journal of Environmental Research and Public Health 19, no. 23: 15974. https://doi.org/10.3390/ijerph192315974
APA StyleMcCarthy, J. M., & Chaplin, E. (2022). Adults with Intellectual Disability and Autism Spectrum Disorder: What Is the Evidence around the Use of Polypharmacy. International Journal of Environmental Research and Public Health, 19(23), 15974. https://doi.org/10.3390/ijerph192315974






