An Examination of US COVID-19 Vaccine Distribution in New Jersey, Pennsylvania, and New York
Abstract
1. Introduction
2. Materials and Methods
2.1. Case Study Approach
2.2. Government Website Review
2.3. Key Informant Interviews and Survey
2.4. Social Media Search
3. Results
3.1. Government Websites
3.2. Key Informant Interviews and Survey
“Registration for vaccine interest was completed using the internet, almost exclusively, marginalized communities (i.e., people of color, the poor, the transient) may not have had the opportunity to register for their vaccine interest. We did not go door to door to identify people’s interests and register them (with acknowledgment of their limitations—no transportation, etc.).”
“Not equitable, Philadelphia missed the mark with partnering with diverse organizations to ensure equitable access and distribution of the vaccine to the most vulnerable populations”
“The Latino population was receiving mixed messages “Operation Warp Speed” was not clearly, explained…Very unreliable source President Trump contradicting the experts on national television…Materials not available in Spanish for low literacy levels…There did not appear to be great interest in providing, clear information to the Latinx population. Mega sites were usually not located in poor communities, limited hours, and roll out was inefficient for example in [name] county the Mega Site was open 3 days from 9am-3pm....Outrageous!”
“Understanding the ever changing tiers” (phases)
“Vaccine delivery chain issues plagued the rollout”
“No rollout of public education from federal, state or local government…this generated mistrust and fear”
“Provide vaccines to local health departments that best know their community/populations. Local health departments providing vaccine/medication to the community in times of crises/emergency/pandemics has been the overarching plan for decades. COVID-19 vaccine deployment in New Jersey has not followed that model.”
“Follow the plans... They were there for a reason, but abandoned without explanation.”
“Public health efforts must start with public education. There must also be a plan for distribution that is equitable. Federal, state, and local governments have to work together.”
“the government must work with a diverse group of organizations that work within the community. Partnering with these organizations helps vaccinate the most vulnerable populations.”
3.3. Vaccine Availability via Social Media
4. Discussion
4.1. Historical Lessons Learned and Community “Buy-In”
4.2. COVID-19 Vaccination Distribution and Logistics Challenges
4.3. The Phased Approach
4.4. Communication
4.5. Accessibility
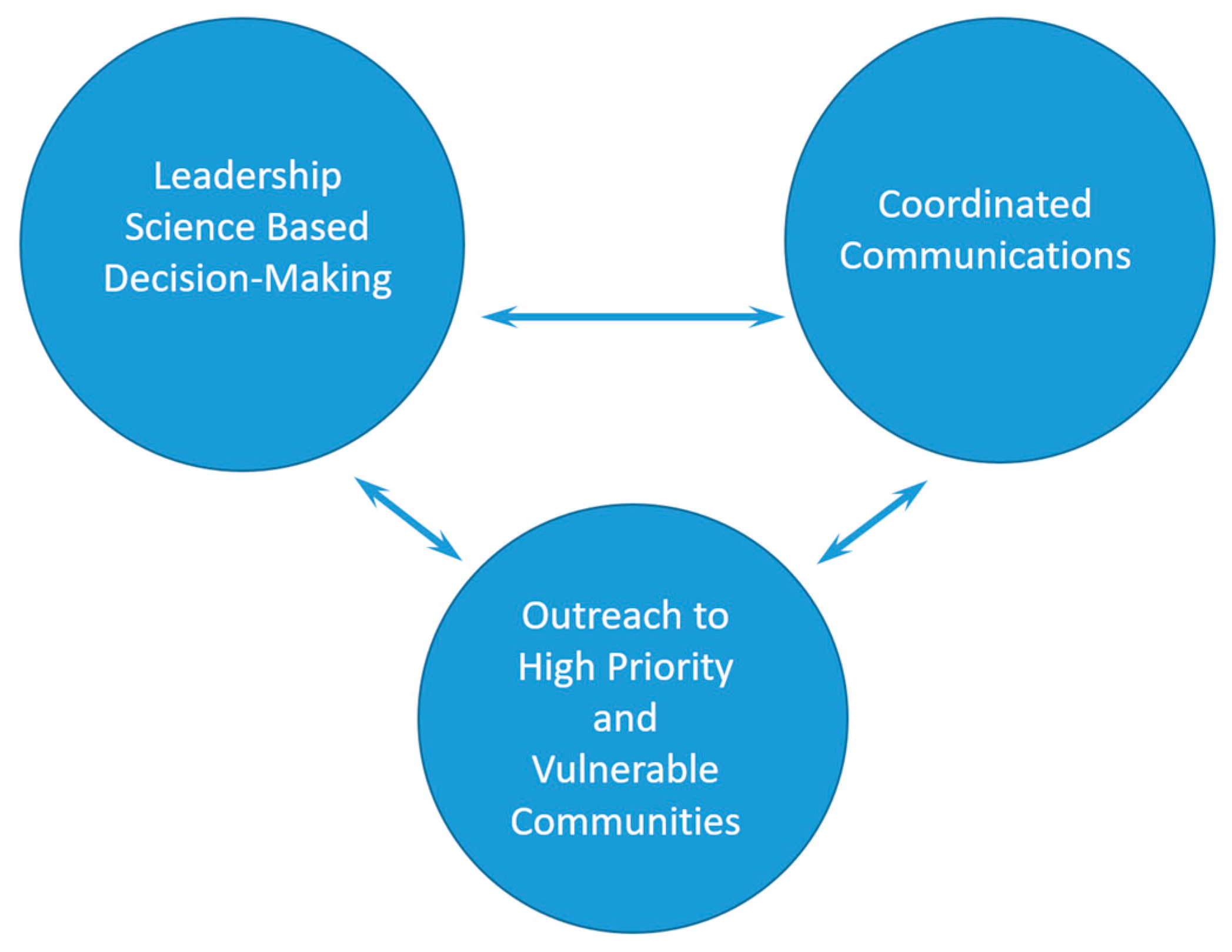
4.6. Policy Implications and Framework for the Future
5. Limitations to the Study
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Center for Disease Control and Prevention. COVID Data Tracker; Center for Disease Control and Prevention: Atlanta, GA, USA, 2020.
- Center for Disease Control and Prevention. December 19 and 20, 2020 Presentation Slides. 2020. Available online: https://www.cdc.gov/vaccines/acip/meetings/slides-2020-12-19-20.html (accessed on 1 September 2022).
- Rattner, N. COVID Cases Dropping by 5% or More in Nearly Every U.S. State, Vaccinations Continue to Fall. Available online: https://www.cnbc.com/2021/05/18/covid-19-cases-deaths-vaccinations-daily-update.html (accessed on 30 September 2022).
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard. 2021. Available online: https://covid19.who.int (accessed on 1 September 2022).
- Robinson, E.; Jones, A.; Lesser, I.; Daly, M. International Estimates of Intended Uptake and Refusal of COVID-19 Vaccines: A Rapid Systematic Review and Meta-Analysis of Large Nationally Representative Samples. Vaccine 2021, 39, 2024–2034. [Google Scholar] [CrossRef] [PubMed]
- Yasmin, F.; Najeeb, H.; Moeed, A.; Naeem, U.; Asghar, M.S.; Chughtai, N.U.; Yousaf, Z.; Seboka, B.T.; Ullah, I.; Lin, C.-Y.; et al. COVID-19 Vaccine Hesitancy in the United States: A Systematic Review. Front. Public Health 2021, 9, 770985. [Google Scholar] [CrossRef] [PubMed]
- Al-Amer, R.; Maneze, D.; Everett, B.; Montayre, J.; Villarosa, A.R.; Dwekat, E.; Salamonson, Y. COVID-19 Vaccination Intention in the First Year of the Pandemic: A Systematic Review. J. Clin. Nurs. 2022, 31, 62–86. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Tu, P.; Beitsch, L.M. Confidence and Receptivity for COVID-19 Vaccines: A Rapid Systematic Review. Vaccines 2020, 9, 16. [Google Scholar] [CrossRef] [PubMed]
- Washburn, K.J.; LeBrón, A.M.W.; Reyes, A.S.; Becerra, I.; Bracho, A.; Ahn, E.; Urzúa, A.S.; Foo, M.A.; Zárate, S.; Tanjasiri, S.P.; et al. Orange County, California COVID-19 Vaccine Equity Best Practices Checklist: A Community-Centered Call to Action for Equitable Vaccination Practices. Health Equity 2022, 6, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Polaha, J.; Johnson, L.; Quinn, M.; Sunderji, N. Lessons from the Team Care Playbook: Recommendations for COVID-19 Vaccination Dissemination and Uptake. Fam. Syst. Health 2020, 38, 341–345. [Google Scholar] [CrossRef]
- Pierce, H.H. An Ace Up Our Sleeves: The COVID-19 Vaccine Rollout Revealed Our Strengths and Our Neglected Public Health Infrastructure. J. Public Health Manag. Pract. 2021, 27, 223–225. [Google Scholar] [CrossRef]
- Benjamin-Chung, J.; Reingold, A. Measuring the Success of the US COVID-19 Vaccine Campaign–Itâ€TMs Time to Invest in and Strengthen Immunization Information Systems. AJPH 2021, 111, 1078–1080. [Google Scholar] [CrossRef]
- Snowdon, A.W.; Wright, A.; Saunders, M. An Evidence-Based Strategy to Scale Vaccination in Canada. Healthc. Q. 2021, 24, 28–35. [Google Scholar] [CrossRef]
- Halperin, D.T.; Hearst, N.; Hodgins, S.; Bailey, R.C.; Klausner, J.D.; Jackson, H.; Wamai, R.G.; Ladapo, J.A.; Over, M.; Baral, S.; et al. Revisiting COVID-19 Policies: 10 Evidence-Based Recommendations for Where to Go from Here. BMC Public Health 2021, 21, 2084. [Google Scholar] [CrossRef]
- Bayati, M.; Noroozi, R.; Ghanbari-Jahromi, M.; Jalali, F.S. Inequality in the Distribution of COVID-19 Vaccine: A Systematic Review. Int. J. Equity Health 2022, 21, 122. [Google Scholar] [CrossRef] [PubMed]
- Renson, A.; Dennis, A.C.; Noppert, G.; McClure, E.S.; Aiello, A.E. Interventions on Socioeconomic and Racial Inequities in Respiratory Pandemics: A Rapid Systematic Review. Curr. Epidemiol. Rep. 2022, 9, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Reñosa, M.D.C.; Landicho, J.; Wachinger, J.; Dalglish, S.L.; Bärnighausen, K.; Bärnighausen, T.; McMahon, S.A. Nudging toward Vaccination: A Systematic Review. BMJ Glob. Health 2021, 6, e006237. [Google Scholar] [CrossRef] [PubMed]
- Batteux, E.; Mills, F.; Jones, L.F.; Symons, C.; Weston, D. The Effectiveness of Interventions for Increasing COVID-19 Vaccine Uptake: A Systematic Review. Vaccines 2022, 10, 386. [Google Scholar] [CrossRef]
- Jean-Jacques, M.; Bauchner, H. Vaccine Distribution—Equity Left Behind? JAMA 2021, 325, 829–830. [Google Scholar] [CrossRef]
- LSU Manship School News Service. COVID Vaccines Present a Challenge for Smaller Pharmacies; New Orleans CityBusiness: Metairie, LA, USA, 2021. [Google Scholar]
- COVID-19 Vaccination Program-Centers for Disease Control and Prevention 2020. Available online: https://www.cdc.gov/vaccines/covid-19/covid19-vaccination-guidance.html (accessed on 1 September 2022).
- Disparities in COVID-19 Vaccination Coverage Between Urban and Rural Counties—United States, December 14, 2020–April 10, 2021. CDC MMWR 2021, 70, 759–764.
- Crowe, S.; Cresswell, K.; Robertson, A.; Huby, G.; Avery, A.; Sheikh, A. The Case Study Approach. BMC Med. Res. Methodol. 2011, 11, 100. [Google Scholar] [CrossRef]
- U.S. Census Bureau QuickFacts: Pennsylvania; New Jersey; New York. Available online: https://www.census.gov/quickfacts/fact/table/PA,NJ,NY/PST045221 (accessed on 3 October 2022).
- Russell, R. Grocery Retailers Ramp Up COVID-19 Vaccination Distribution; Supermarket News: New York, NY, USA, 2021. [Google Scholar]
- Villanova University Presents 2022 Praxis Award in Professional Ethics to Black Doctors COVID-19 Consortium Founder Dr. Ala Stanford. Villanova University. Available online: https://www1.villanova.edu/villanova/media/pressreleases/2022/villanova-university-presents-2022-praxis-award-in-professional-ethics.html (accessed on 3 October 2022).
- Skippack Pharmacy. Available online: https://www.facebook.com/skippackrx/ (accessed on 3 October 2022).
- United States Government Accountability Office. GAO-11-632 Influenza Pandemic: Lessons from the H1N1 Pandemic Should be 2011; United States Government Accountability Office: Washington, DC, USA, 2011.
- Radcliffe, S. Here’s How the Last Pandemic Vaccine Was Rolled out in the US 2020. Available online: https://www.healthline.com/health-news/what-happened-the-last-time-we-had-a-vaccine-during-a-pandemic (accessed on 1 September 2022).
- Prati, G. Intention to Receive a Vaccine against SARS-CoV-2 in Italy and Its Association with Trust, Worry and Beliefs about the Origin of the Virus. Health Educ. Res. 2020, 35, 505–511. [Google Scholar] [CrossRef]
- Paris, C.; Bénézit, F.; Geslin, M.; Polard, E.; Baldeyrou, M.; Turmel, V.; Tadié, É.; Garlantezec, R.; Tattevin, P. COVID-19 Vaccine Hesitancy among Healthcare Workers. Infect. Dis. Now 2021, 51, 484–487. [Google Scholar] [CrossRef]
- Bungaro, M.; Passiglia, F.; Scagliotti, G.V. COVID-19 and Lung Cancer: A Comprehensive Overview from Outbreak to Recovery. Biomedicines 2022, 10, 776. [Google Scholar] [CrossRef]
- Ahmed, N.; Boxley, C.; Dixit, R.; Krevat, S.; Fong, A.; Ratwani, R.M.; Wesley, D.B. Evaluation of a Text Message-Based COVID-19 Vaccine Outreach Program Among Older Patients: Cross-Sectional Study. JMIR Form. Res. 2022, 6, e33260. [Google Scholar] [CrossRef] [PubMed]
- Dobrina, R.; Bicego, L. COVID-19 Vaccine Campaign Has Now Opened up for Children Aged 5–11. How Are Kids Going to Live It? J. Pediatr. Nurs. 2022, 65, e19. [Google Scholar] [CrossRef] [PubMed]
- Ramprasad, A.; Qureshi, F.; Lee, B.R.; Jones, B.L. The Relationship between Structural Racism and COVID-19 Related Health Disparities across 10 Metropolitan Cities in the United States. J. Natl. Med. Assoc. 2022, 114, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Scherer, M.; Kamler, A.; Weiss, L.; Blacksher, E.; Jeavons, J.; Gold, M.R. Using Public Deliberation to Set Priorities: The Case of COVID-19 Vaccine Access in New York City. J. Public Health Manag. Pract. 2022, 28, 86–94. [Google Scholar] [CrossRef]
- Hodge, J.G.; Piatt, J.L.; Barraza, L.; Freed, R.; Ghaith, S.; Wells, N. Legal Challenges Underlying COVID-19 Vaccinations. J. Law Med. Ethics 2021, 49, 495–499. [Google Scholar] [CrossRef]
- Islam, M.S.; Kamal, A.M.; Kabir, A.; Southern, D.L.; Khan, S.H.; Hasan, S.M.M.; Sarkar, T.; Sharmin, S.; Das, S.; Roy, T.; et al. COVID-19 Vaccine Rumors and Conspiracy Theories: The Need for Cognitive Inoculation against Misinformation to Improve Vaccine Adherence. PLoS ONE 2021, 16, e0251605. [Google Scholar] [CrossRef]
- Center for Disease Control and Prevention 2022. Available online: https://www.cdc.gov/vaccines/programs/vfc/index.html (accessed on 1 September 2022).
- Marlow, M.A.; Moore, K.; DeBolt, C.; Patel, M.; Marin, M. Health Departments’ Experience With Mumps Outbreak Response and Use of a Third Dose of Measles, Mumps, and Rubella Vaccine. J. Public Health Manag. Pract. 2020, 26, 101–108. [Google Scholar] [CrossRef]
- Lo, N.C.; Hotez, P.J. Public Health and Economic Consequences of Vaccine Hesitancy for Measles in the United States. JAMA Pediatr. 2017, 171, 887–892. [Google Scholar] [CrossRef]
- Pfizer Pfizer and BioNTech Submit COVID-19 Vaccine Stability Data at Standard Temperature to the US FDA 2021. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-submit-covid-19-vaccine-stability-data#.YwJKgulN66Y.link (accessed on 1 September 2022).
- Eaton, J. The US Has Wasted over 82 Million COVID Vaccine Doses. NBC News, 6 June 2022. [Google Scholar]
- Burgos, R.M.; Badowski, M.E.; Drwiega, E.; Ghassemi, S.; Griffith, N.; Herald, F.; Johnson, M.; Smith, R.O.; Michienzi, S.M. The Race to a COVID-19 Vaccine: Opportunities and Challenges in Development and Distribution. Drugs Context 2021, 10, 1–10. [Google Scholar] [CrossRef]
- Hass, K. Being Human during COVID; University of Michigan Press: Ann Arbor, MI, USA, 2021; ISBN 978-0-472-03878-7. [Google Scholar]
- Geneviève, L.D.; Martani, A.; Wangmo, T.; Elger, B.S. Precision Public Health and Structural Racism in the United States: Promoting Health Equity in the COVID-19 Pandemic Response. JMIR Public Health Surveill. 2022, 8, e33277. [Google Scholar] [CrossRef]
- House, T.W. Priorities. COVID-19. The Biden-Harrison Plan to Beat COVID 2022. Available online: https://www.whitehouse.gov/priorities/covid-19/ (accessed on 1 September 2022).
- Tolbert, J.; Artiga, S.; Kates, J.; Rudowitz, J. COVID-19 Funding on Access to COVID-19 Testing, Treatment and Vaccines 2022. Available online: https://www.kff.org/coronavirus-covid-19/issue-brief/implications-of-the-lapse-in-federal-covid-19-funding-on-access-to-covid-19-testing-treatment-and-vaccines/ (accessed on 1 September 2022).
- Hennink, M.M.; Kaiser, B.N.; Weber, M.B. What Influences Saturation? Estimating Sample Sizes in Focus Group Research. Qual. Health Res. 2019, 29, 1483–1496. [Google Scholar] [CrossRef] [PubMed]
- Karafillakis, E.; Dinca, I.; Apfel, F.; Cecconi, S.; Wűrz, A.; Takacs, J.; Suk, J.; Celentano, L.P.; Kramarz, P.; Larson, H.J. Vaccine Hesitancy among Healthcare Workers in Europe: A Qualitative Study. Vaccine 2016, 34, 5013–5020. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.S.; Corbett, K.S. Prototype Pathogen Approach for Pandemic Preparedness: World on Fire. J. Clin. Investig. 2020, 130, 3348–3349. [Google Scholar] [CrossRef] [PubMed]
- Sessa, F.; Salerno, M.; Esposito, M.; Di Nunno, N.; Zamboni, P.; Pomara, C. Autopsy Findings and Causality Relationship between Death and COVID-19 Vaccination: A Systematic Review. J. Clin. Med. 2021, 10, 5876. [Google Scholar] [CrossRef] [PubMed]
- Cirillo, N. Reported Orofacial Adverse Effects of COVID-19 Vaccines: The Knowns and the Unknowns. J. Oral Pathol. Med. 2021, 50, 424–427. [Google Scholar] [CrossRef]
- Moeti, M.; Gao, G.F.; Herrman, H. Global Pandemic perspectives: Public health, mental health and lessons for the future. Lancet 2022, 400, e3–e7. [Google Scholar] [CrossRef]
| Online Tools | Languages? | Call/Email For Appt for Computer Literacy/Accessibility | Sign Language Available for the Press Conference | |
|---|---|---|---|---|
| New Jersey | NJ VSS QR Code needed | English Spanish | Call Center | Yes |
| New York | “Am I eligible” app and State form/waitlist QR Code used | Translations available | Call Center | Yes |
| NYC | “Am I eligible” app and State form/waitlist QR Code used | Translations available | Call Center | Unknown |
| Pennsylvania | Provider map | Translations available | Not seen | Yes |
| Philadelphia | Provider map | Translations available | Not seen | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moss, E.; Patterson, N.A.; Seals, B.F. An Examination of US COVID-19 Vaccine Distribution in New Jersey, Pennsylvania, and New York. Int. J. Environ. Res. Public Health 2022, 19, 15629. https://doi.org/10.3390/ijerph192315629
Moss E, Patterson NA, Seals BF. An Examination of US COVID-19 Vaccine Distribution in New Jersey, Pennsylvania, and New York. International Journal of Environmental Research and Public Health. 2022; 19(23):15629. https://doi.org/10.3390/ijerph192315629
Chicago/Turabian StyleMoss, Ethan, Natasha A. Patterson, and Brenda F. Seals. 2022. "An Examination of US COVID-19 Vaccine Distribution in New Jersey, Pennsylvania, and New York" International Journal of Environmental Research and Public Health 19, no. 23: 15629. https://doi.org/10.3390/ijerph192315629
APA StyleMoss, E., Patterson, N. A., & Seals, B. F. (2022). An Examination of US COVID-19 Vaccine Distribution in New Jersey, Pennsylvania, and New York. International Journal of Environmental Research and Public Health, 19(23), 15629. https://doi.org/10.3390/ijerph192315629









