Benefits of Participation in Clinical Trials: An Umbrella Review
Abstract
1. Introduction
2. Material and Methods
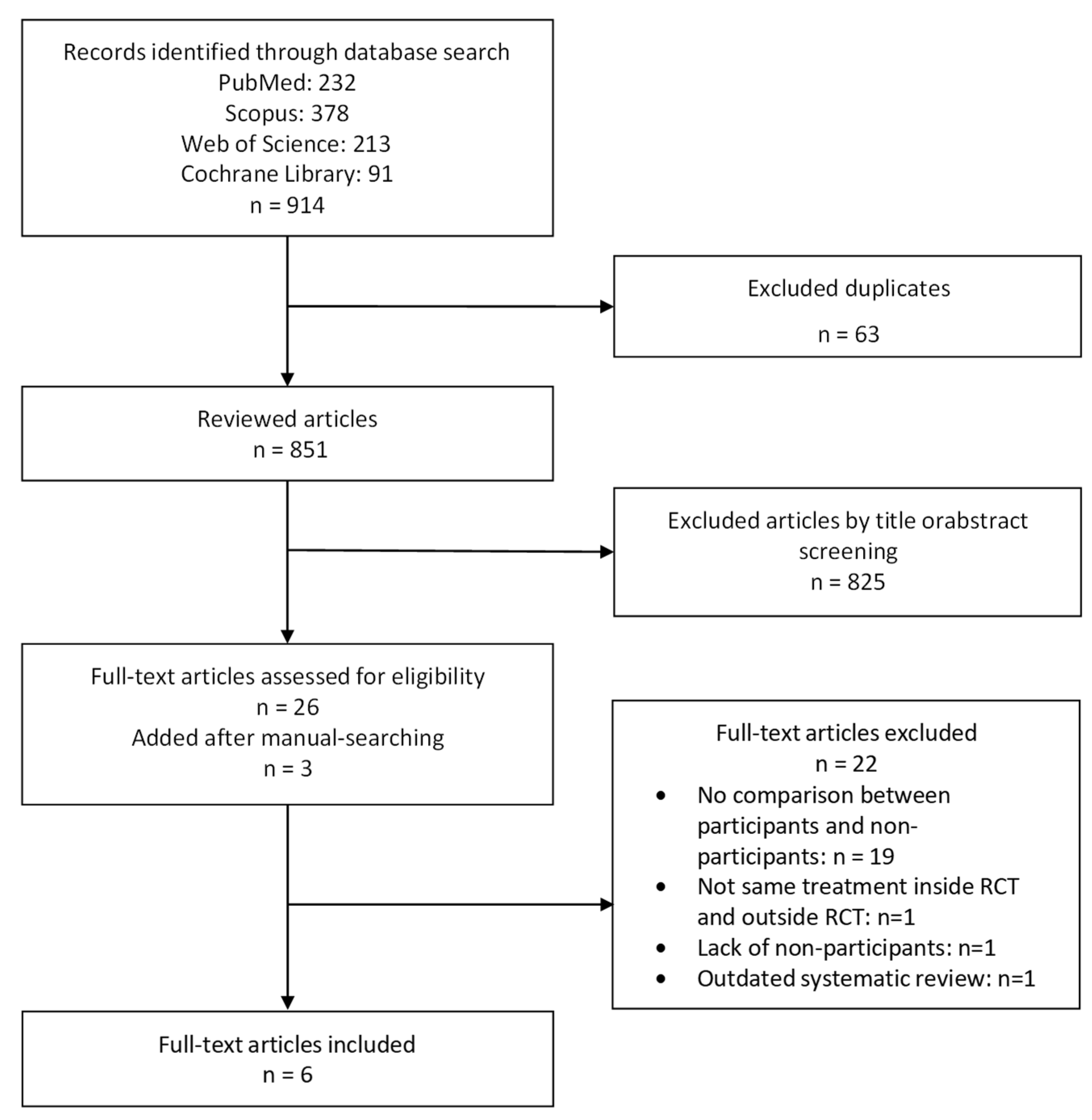
2.1. Literature Search and Selection
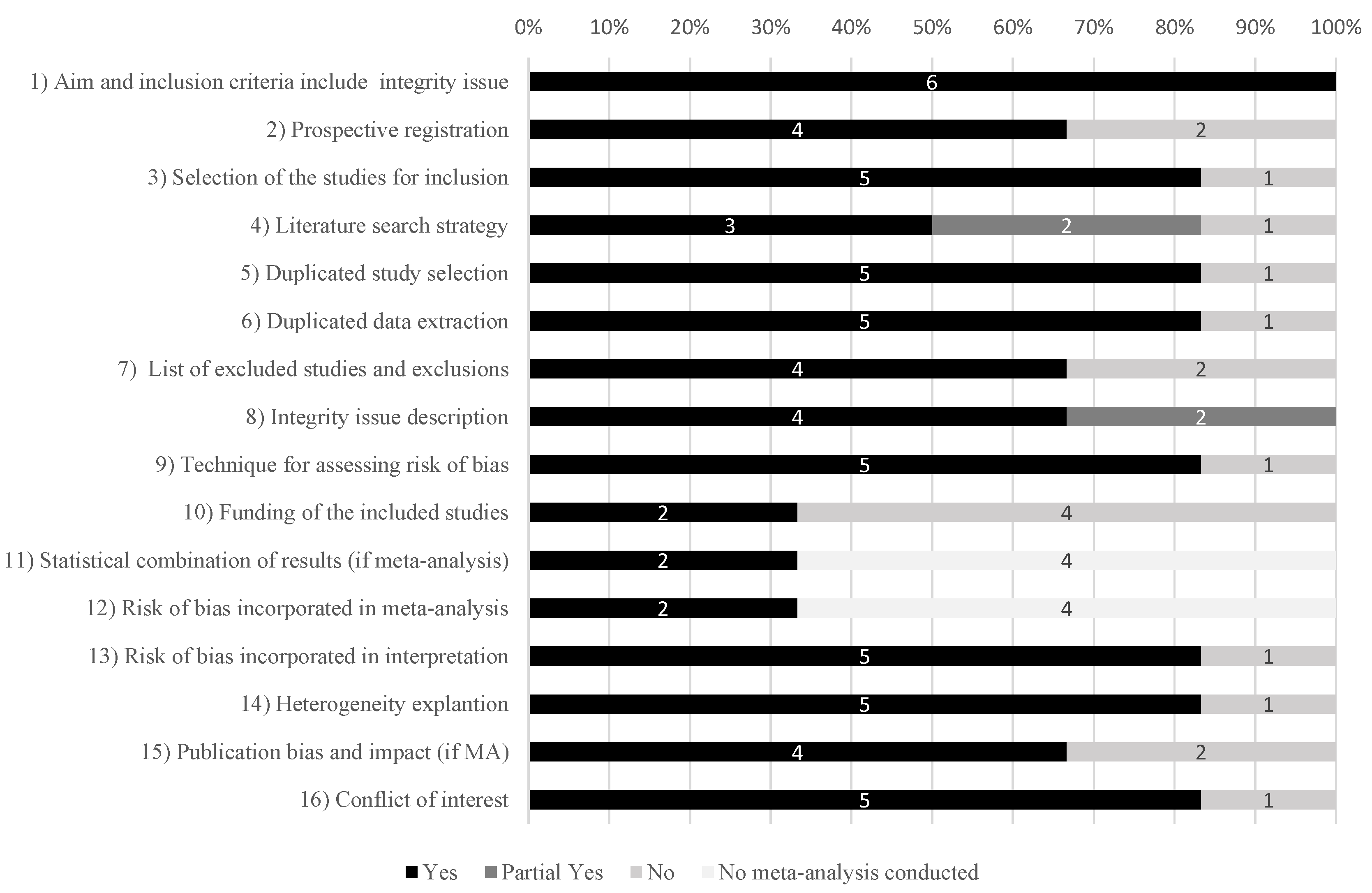
2.2. Data Extraction and Risk-of-Bias Assessment
2.3. Data Synthesis
3. Results
3.1. Selection, Characteristics and Quality of Studies
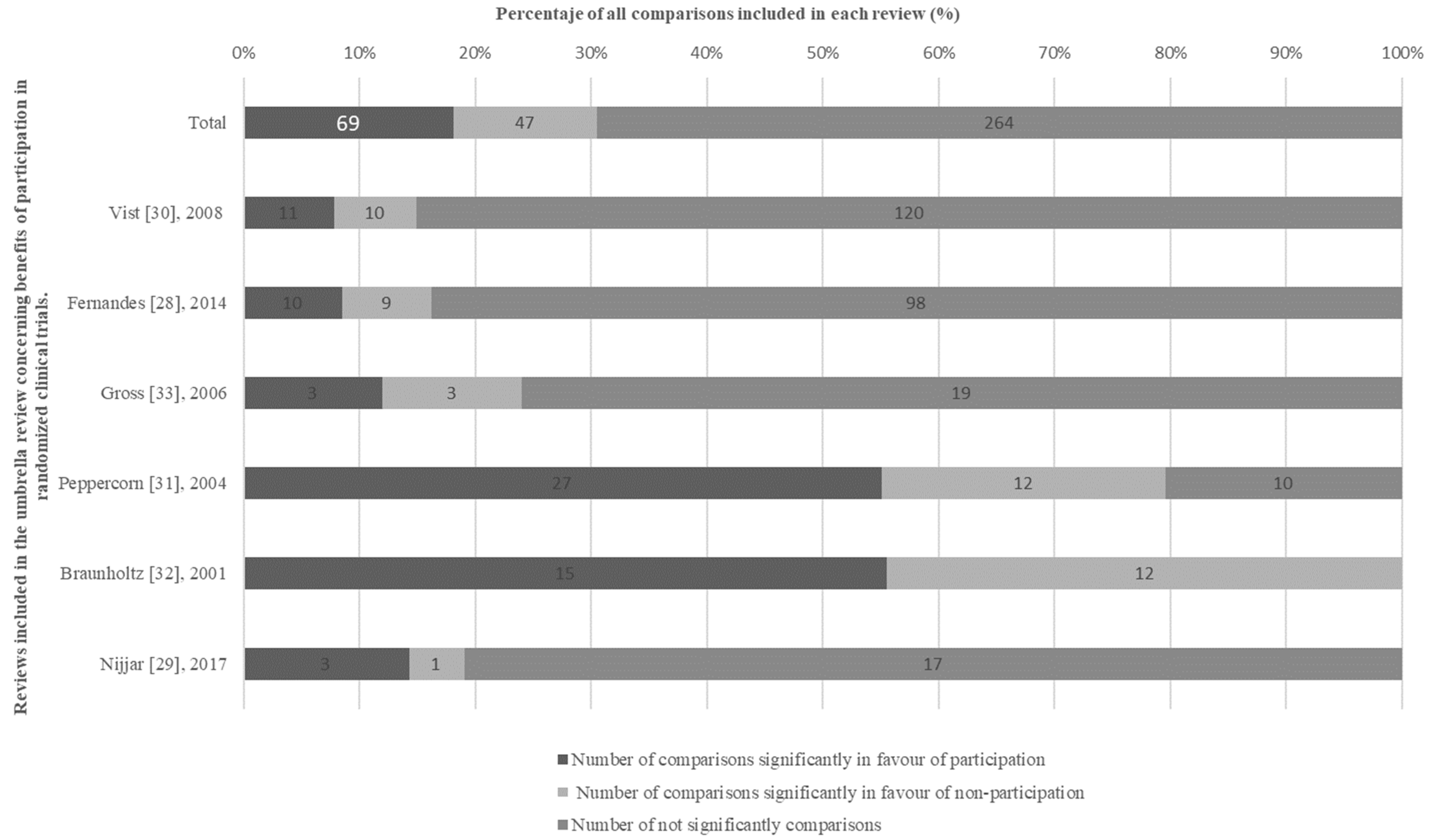
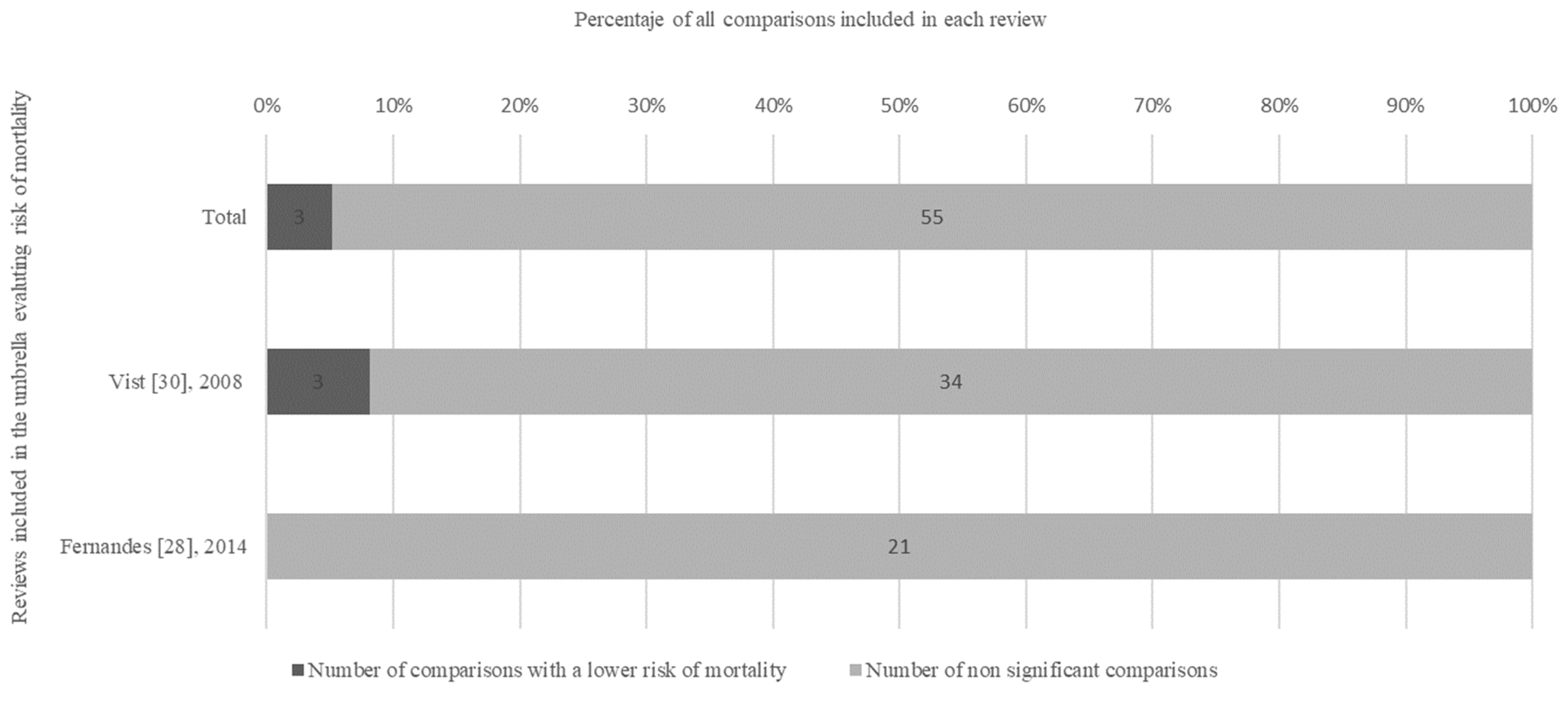
3.2. Synthesis of Findings
3.3. Findings from the High-Quality Subgroup of Reviews
3.4. Findings of the Subgroup of Reviews with Low and Critically Low Quality
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thangaratinam, S.; Khan, K.S. Participation in research as a means of improving quality of care: The role of a principal investigator in multicentre clinical trials. Obstet. Gynaecol. 2015, 17, 55–61. [Google Scholar] [CrossRef]
- Hellman, S.; Hellman, D.S. Of mice but not men. Problems of the randomized clinical trial. N. Engl. J. Med. 1991, 324, 1585–1589. [Google Scholar] [CrossRef]
- Clarke, M.; Loudon, K. Effects on patients of their healthcare practitioner’s or institution’s participation in clinical trials: A systematic review. Trials 2011, 12, 16. [Google Scholar] [CrossRef] [PubMed]
- Joosten, E.A.G.; DeFuentes-Merillas, L.; de Weert, G.H.; Sensky, T.; van der Staak, C.P.F.; de Jong, C.A.J. Systematic review of the effects of shared decision-making on patient satisfaction, treatment adherence and health status. Psychother. Psychosom. 2008, 77, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Waters, L.; George, A.S.; Chey, T.; Bauman, A. Weight change in control group participants in behavioural weight loss interventions: A systematic review and meta-regression study. BMC Med. Res. Methodol. 2012, 12, 120. [Google Scholar] [CrossRef]
- Bouzalmate Hajjaj, A.; Massó Guijarro, P.; Khan, K.S.; Bueno-Cavanillas, A.; Cano-Ibáñez, N. A systematic review and meta-analysis of weight loss in control group participants of lifestyle randomized trials. Sci. Rep. 2022, 12, 12252. [Google Scholar] [CrossRef]
- Wade, J.; Elliott, D.; Avery, K.N.L.; Gaunt, D.; Young, G.J.; Barnes, R.; Paramasivan, S.; Campbell, W.B.; Blazeby, J.M.; Birtle, A.J.; et al. Informed consent in randomised controlled trials: Development and preliminary evaluation of a measure of Participatory and Informed Consent (PIC). Trials 2017, 18, 327. [Google Scholar] [CrossRef]
- Koopmans, B.; Nielen, M.M.; Schellevis, F.G.; Korevaar, J.C. Non-participation in population-based disease prevention programs in general practice. BMC Public Health 2012, 12, 856. [Google Scholar] [CrossRef]
- Phelps, E.E.; Tutton, E.; Griffin, X.; Baird, J. A mixed-methods systematic review of patients’ experience of being invited to participate in surgical randomised controlled trials. Soc. Sci. Med. 2020, 253, 112961. [Google Scholar] [CrossRef]
- Unger, J.M.; Vaidya, R.; Hershman, D.L.; Minasian, L.M.; Fleury, M.E. Systematic Review and Meta-Analysis of the Magnitude of Structural, Clinical, and Physician and Patient Barriers to Cancer Clinical Trial Participation. Gynecol. Oncol. 2019, 111, 245–255. [Google Scholar] [CrossRef]
- Hillyer, G.C.; Beauchemin, M.; Hershman, D.L.; Kelsen, M.; Brogan, F.L.; Sandoval, R.; Schmitt, K.M.; Reyes, A.; Terry, M.B.; Lassman, A.B.; et al. Discordant attitudes and beliefs about cancer clinical trial participation between physicians, research staff, and cancer patients. Clin. Trials 2020, 17, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.R.; Sun, V.; George, K.; Liu, J.; Padam, S.; Chen, B.A.; George, T.; Amini, A.; Li, D.; Sedrak, M.S. Barriers to Participation in Therapeutic Clinical Trials as Perceived by Community Oncologists. JCO Oncol. Pract. 2020, 16, e849–e858. [Google Scholar] [CrossRef] [PubMed]
- Nipp, R.D.; Hong, K.; Paskett, E.D. Overcoming Barriers to Clinical Trial Enrollment. Am. Soc. Clin. Oncol. Educ. Book 2019, 39, 105–114. [Google Scholar] [CrossRef] [PubMed]
- García-Martín, M.; Amezcua-Prieto, C.; Al Wattar, B.H.; Jørgensen, J.S.; Bueno-Cavanillas, A.; Khan, K.S. Patient and Public Involvement in Sexual and Reproductive Health: Time to Properly Integrate Citizen’s Input into Science. Int. J. Environ. Res. Public Health 2020, 17, 8048. [Google Scholar] [CrossRef]
- Price, A.; Albarqouni, L.; Kirkpatrick, J.; Clarke, M.; Liew, S.M.; Roberts, N.; Burls, A. Patient and public involvement in the design of clinical trials: An overview of systematic reviews. J. Eval. Clin. Pract. 2017, 24, 240–253. [Google Scholar] [CrossRef]
- Knelson, L.P.; Cukras, A.R.; Savoie, J.; Agarwal, A.; Guo, H.; Hu, J.; Fell, G.; Lederman, R.; Hughes, M.E.; Winer, E.P.; et al. Barriers to Clinical Trial Accrual: Perspectives of Community-Based Providers. Clin. Breast Cancer 2020, 20, 395–401.e3. [Google Scholar] [CrossRef]
- McKinney, M.; Bell, R.; Samborski, C.; Attwood, K.; Dean, G.; Eakle, K.; Edge, S.B. Clinical Trial Participation: A Pilot Study of Patient-Identified Barriers. Clin. J. Oncol. Nurs. 2021, 25, 647–654. [Google Scholar] [CrossRef]
- Rogers, M.; Lemstra, M.; Bird, Y.; Nwankwo, C.; Moraros, J. Weight-loss intervention adherence and factors promoting adherence: A meta-analysis. Patient Prefer. Adherence 2016, 2016, 1547–1559. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Aromataris, E.; Fernandez, R.; Godfrey, C.M.; Holly, C.; Khalil, H.; Tungpunkom, P. Summarizing systematic reviews: Methodological development, conduct and reporting of an umbrella review approach. Int. J. Evid. Based Healthc. 2015, 13, 132–140. [Google Scholar] [CrossRef]
- Gates, M.; Gates, A.; Pieper, D.; Fernandes, R.M.; Tricco, A.C.; Moher, D.; E Brennan, S.; Li, T.; Pollock, M.; Lunny, C.; et al. Reporting guideline for overviews of reviews of healthcare interventions: Development of the PRIOR statement. BMJ 2022, 378, e070849. [Google Scholar] [CrossRef]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Lu, T.; Ge, L.; Yang, N.; Yan, P.; Yang, K. Use of AMSTAR-2 in the methodological assessment of systematic reviews: Protocol for a methodological study. Ann. Transl. Med. 2020, 8, 652. [Google Scholar] [CrossRef] [PubMed]
- Perry, R.; Whitmarsh, A.; Leach, V.; Davies, P. A comparison of two assessment tools used in overviews of systematic reviews: ROBIS versus AMSTAR-2. Syst. Rev. 2021, 10, 273. [Google Scholar] [CrossRef]
- Rikke, B.A.; Wynes, M.W.; Rozeboom, L.M.; Barón, A.E.; Hirsch, F.R. Independent validation test of the vote-counting strategy used to rank biomarkers from published studies. Biomark. Med. 2015, 9, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Pieper, D.; Antoine, S.-L.; Mathes, T.; Neugebauer, E.A.; Eikermann, M. Systematic review finds overlapping reviews were not mentioned in every other overview. J. Clin. Epidemiol. 2014, 67, 368–375. [Google Scholar] [CrossRef]
- Pollock, M.; Fernandes, R.M.; Newton, A.S.; Scott, S.D.; Hartling, L. A decision tool to help researchers make decisions about including systematic reviews in overviews of reviews of healthcare interventions. Syst. Rev. 2019, 8, 29. [Google Scholar] [CrossRef]
- Fernandes, N.; Bryant, D.; Griffith, L.; El-Rabbany, M.; Fernandes, N.M.; Kean, C.; Marsh, J.; Mathur, S.; Moyer, R.; Reade, C.J.; et al. Outcomes for patients with the same disease treated inside and outside of randomized trials: A systematic review and meta-analysis. Can. Med. Assoc. J. 2014, 186, E596–E609. [Google Scholar] [CrossRef]
- Nijjar, S.K.; D’Amico, M.I.; Wimalaweera, N.A.; Cooper, N.A.M.; Zamora, J.; Khan, K.S. Participation in clinical trials improves outcomes in women’s health: A systematic review and meta-analysis. BJOG Int. J. Obstet. Gynaecol. 2017, 124, 863–871. [Google Scholar] [CrossRef]
- Vist, G.E.; Bryant, D.; Somerville, L.; Birminghem, T.; Oxman, A.D. Outcomes of patients who participate in randomized controlled trials compared to similar patients receiving similar interventions who do not participate. Cochrane Database Syst. Rev. 2008, 3, MR000009. [Google Scholar] [CrossRef]
- Gross, C.P.; Krumholz, H.M.; Van Wye, G.; Emanuel, E.J.; Wendler, D. Does random treatment assignment cause harm to research participants? PLoS Med. 2006, 3, e188. [Google Scholar] [CrossRef]
- Peppercorn, J.M.; Weeks, J.C.; Cook, E.F.; Joffe, S. Comparison of outcomes in cancer patients treated within and outside clinical trials: Conceptual framework and structured review. Lancet 2004, 363, 263–270. [Google Scholar] [CrossRef]
- Braunholtz, D.A.; Edwards, S.J.; Lilford, R.J. Are randomized clinical trials good for us (in the short term)? Evidence for a “trial effect”. J. Clin. Epidemiol. 2001, 54, 217–224. [Google Scholar] [CrossRef]
- Stiller, C.A.; Draper, G.J. Treatment centre size, entry to trials, and survival in acute lymphoblastic leukaemia. Arch. Dis. Child. 1989, 64, 657–661. [Google Scholar] [CrossRef]
- Unger, J.M.; Barlow, W.E.; Martin, D.P.; Ramsey, S.D.; Leblanc, M.; Etzioni, R.; Hershman, D.L. Comparison of Survival Outcomes Among Cancer Patients Treated In and Out of Clinical Trials. JNCI J. Natl. Cancer Inst. 2014, 106, dju002. [Google Scholar] [CrossRef] [PubMed]
- Lilford, R.J.; Edwards, S.J.; Braunholtz, D.A.; Jackson, J.; Thornton, J.; Hewison, J.; Stevens, A.; Abrams, K.; Brazier, J.; Fitzpatrick, R. Ethical Issues in the Design and Conduct of Randomised Controlled Trials. In The Advanced Handbook of Methods in Evidence Based Healthcare; Stevens, A., Abrams, K., Brazier, J., Fitzpatrick, R., Lilford, R., Eds.; SAGE Publications Ltd.: Thousand Oaks, CA, USA, 2001; pp. 1–132. [Google Scholar]
- Dellson, P.; Nilsson, K.; Jernström, H.; Carlsson, C. Patients’ reasoning regarding the decision to participate in clinical cancer trials: An interview study. Trials 2018, 19, 528. [Google Scholar] [CrossRef]
- Kao, C.Y.; Aranda, S.; Krishnasamy, M.; Hamilton, B. Interventions to improve patient understanding of cancer clinical trial participation: A systematic review. Eur. J. Cancer Care 2016, 26, e12424. [Google Scholar] [CrossRef]
- Domecq, J.P.; Prutsky, G.; Elraiyah, T.; Wang, Z.; Nabhan, M.; Shippee, N.; Brito, J.P.; Boehmer, K.; Hasan, R.; Firwana, B.; et al. Patient engagement in research: A systematic review. BMC Health Serv. Res. 2014, 14, 89. [Google Scholar] [CrossRef]
- Goldstein, C.E.; Weijer, C.; Brehaut, J.C.; Fergusson, D.A.; Grimshaw, J.M.; Horn, A.R.; Taljaard, M. Ethical issues in pragmatic randomized controlled trials: A review of the recent literature identifies gaps in ethical argumentation. BMC Med. Ethics 2018, 19, 14. [Google Scholar] [CrossRef]
- Odgaard-Jensen, J.; E Vist, G.; Timmer, A.; Kunz, R.; A Akl, E.; Schünemann, H.; Briel, M.; Nordmann, A.J.; Pregno, S.; Oxman, A.D. Randomisation to protect against selection bias in healthcare trials. Cochrane Database Syst. Rev. 2011, 2, MR000012. [Google Scholar] [CrossRef]
- Wendler, D.; Kington, R.; Madans, J.; Van Wye, G.; Christ-Schmidt, H.; Pratt, L.A.; Brawley, O.W.; Gross, C.P.; Emanuel, E. Are racial and ethnic minorities less willing to participate in health research? PLoS Med. 2006, 3, e19. [Google Scholar] [CrossRef] [PubMed]
- Sacristán, J.A.; Aguaron, A.; Avendaño-Solá, C.; Garrido, P.; Carrion, J.; Gutierrez, A.; Kroes, R.; Flores, A. Patient involvement in clinical research: Why, when, and how. Patient Prefer. Adherence 2016, 10, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, B.; Gheorghe, A.; Moore, D.; Wilson, S.; Damery, S. Data from: Improving the recruitment activity of clinicians in randomised controlled trials: A systematic review. BMJ Open 2012, 2, e000496. [Google Scholar] [CrossRef] [PubMed]
- McCambridge, J.; Witton, J.; Elbourne, D.R. Systematic review of the Hawthorne effect: New concepts are needed to study research participation effects. J. Clin. Epidemiol. 2014, 67, 267–277. [Google Scholar] [CrossRef]
- Ward, M.M. Primer: Measuring the effects of treatment in clinical trials. Nat. Clin. Pract. Rheumatol. 2007, 3, 291–297. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Objective | No. of Databases Searched, Date Range of Searching, and Publication Data Range (PDR) of Primary Studies | QUALITY ASSESSMent on Primary Studies | RCT Participant Characteristics (No. Comparisons) | Non-Participant Characteristics (No. Comparisons) | Outcomes (Measurement) |
|---|---|---|---|---|---|---|
| Vist [30], 2008 | To assess the effects of patient participation in RCTs (’trial effects’) independent both of the effects of the clinical treatments being compared (’treatment effects’) and any differences between patients who participated in RCTs and those who did not | 5 databases: The Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE, The Cochrane Methodology Register, SciSearch and PsycINFO. Up to March 2007. PDR: 1978–2006 | Per review. No validated tool used. The criteria followed to assess the validity of comparisons was scored as: “met”, part, “partially met”, “not imbalance”, “not met”, “unclear”. | Patients in different clinical areas and interventions: oncology (31), cardiology (22), other internal medicine subspecialties (27), obstetrics and gynaecology (29), psychology or drug abuse (15), and paediatrics (12), surgery or other procedures (33), drug therapy (28), radiotherapy (15), counselling or education (9), usual care (45), and active monitoring/watchful waiting (6). | Patients receiving similar treatment outside of RCTs (80), eligible refusers (1), patients not invited to participate (2), eligible non-participants who do not meet the above categories (2). | Mortality (dichotomous) morbidity and changes in self-reported pain, quality of life, and function (continuous). |
| Fernandes [28], 2014 | To compare outcomes for patients with the same diagnoses who did (“insiders”) and did not (“outsiders”) enter RCTs, without regard to the specific therapies received for their respective diagnoses. | MEDLINE (1966 to November 2010), Embase (1980 to November 2010), Cochrane Central Register of Controlled Trials (CENTRAL; 1960 to last quarter of 2010) and PsycINFO (1880 to November 2010). From 1880 to 2010. PDR:1979–2009 | Per review. No validated tool used. | Patients in different clinical areas and interventions: surgery or medical procedures (46); drug therapy (57); radiotherapy (5); counselling or education (27); other therapy (12). | Patients with the same diagnoses who did not enter RCTs known as “outsiders” (147). | Mortality (dichotomous), patient-reported or other clinically important outcomes (continuous outcomes) |
| Gross [31], 2006 | To quantify the differences in health outcomes between randomized trial participants and eligible non-participants. | Medline, the Web of Science citation database, and manuscript references. From 1984 to 2002. PDR: 1084–2002. | Per review. No validated tool used. | Patients in different clinical areas (oncology, cardiovascular diseases, obstetrics and gynaecology) and interventions: diagnostics (2), medical (14) or surgical (9). | Patients sharing healthcare settings at recruitment, participants recruited in a similar way, eligible non-participants, non-participants allowed to access agents used in a trial. | Mortality, treatment acceptability, and proportion of time or number of days with a given condition. |
| Peppercorn [32], 2004 | To assess the empirical evidence that patients with cancer who enrols in clinical trials have better outcomes than those who do not enrol. | Medline. Search range nor defined. PDR: 1971–2002. | Per review. Not validated tool. Pilot-tested forms recording potential sources of bias. | Cancer patients (24). | Eligible refusers (4), patients in retrospective cohort (21), participants in natural experiment (1). | Health benefits in cancer patients in RCTs (trial effect) |
| Braunholtz [33], 2001 | To assess whether there is evidence that randomized controlled trials are systematically beneficial, or harmful, for patients. | Databases not defined. Up to August 1996. PDR: 1879–1996. | Per review. Not validated tool. Sources of bias of concern were conceptualized as: “patient selection bias”, clinician selection bias”, “detection bias”, “transfer bias” and “study induced bias”. | Patients in cancer therapy (10), cardiovascular (2), other medical interventions (2). | Indirect comparisons (2); patients in at least one concurrent non-trial control group (11): eligible refusers (3), refusers and eligible non-recruited patients (4), all non-randomized patients of recruiting clinicians (1), all non-recruited patients of recruiting and non-recruiting clinicians (3); no control group (1). | Health benefits in cancer patients in RCTs, |
| Nijjar [29], 2017 | To determine whether participation in randomised controlled trials (RCTs), compared with non-participation, has a beneficial effect on women’s health. | MEDLINE (1966 to December 2015), Embase (1980 to December 2015), Cochrane Central Register of Controlled Trials (CENTRAL; 1960 to last quarter of 2015) and PsycINFO (1880 to December 2015). PDR:1981–2015. | Per review. Jadad scale and Newcastle Ottawa score (NOS). | Women in obstetrics-gynaecology interventions: medical (12), surgical (6), and other (3). | Comparable non-participants cohort (21). | Health benefits in women, fetuses or new-borns (dichotomous). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouzalmate-Hajjaj, A.; Massó Guijarro, P.; Khan, K.S.; Bueno-Cavanillas, A.; Cano-Ibáñez, N. Benefits of Participation in Clinical Trials: An Umbrella Review. Int. J. Environ. Res. Public Health 2022, 19, 15368. https://doi.org/10.3390/ijerph192215368
Bouzalmate-Hajjaj A, Massó Guijarro P, Khan KS, Bueno-Cavanillas A, Cano-Ibáñez N. Benefits of Participation in Clinical Trials: An Umbrella Review. International Journal of Environmental Research and Public Health. 2022; 19(22):15368. https://doi.org/10.3390/ijerph192215368
Chicago/Turabian StyleBouzalmate-Hajjaj, Amira, Paloma Massó Guijarro, Khalid Saeed Khan, Aurora Bueno-Cavanillas, and Naomi Cano-Ibáñez. 2022. "Benefits of Participation in Clinical Trials: An Umbrella Review" International Journal of Environmental Research and Public Health 19, no. 22: 15368. https://doi.org/10.3390/ijerph192215368
APA StyleBouzalmate-Hajjaj, A., Massó Guijarro, P., Khan, K. S., Bueno-Cavanillas, A., & Cano-Ibáñez, N. (2022). Benefits of Participation in Clinical Trials: An Umbrella Review. International Journal of Environmental Research and Public Health, 19(22), 15368. https://doi.org/10.3390/ijerph192215368







