Relationship between Pain Intensity, Physical Factors, Pronociceptive Pain Modulation Profile and Psychological Vulnerability on Upper Limb Disability in Older Patients with Chronic Shoulder Pain
Abstract
1. Introduction
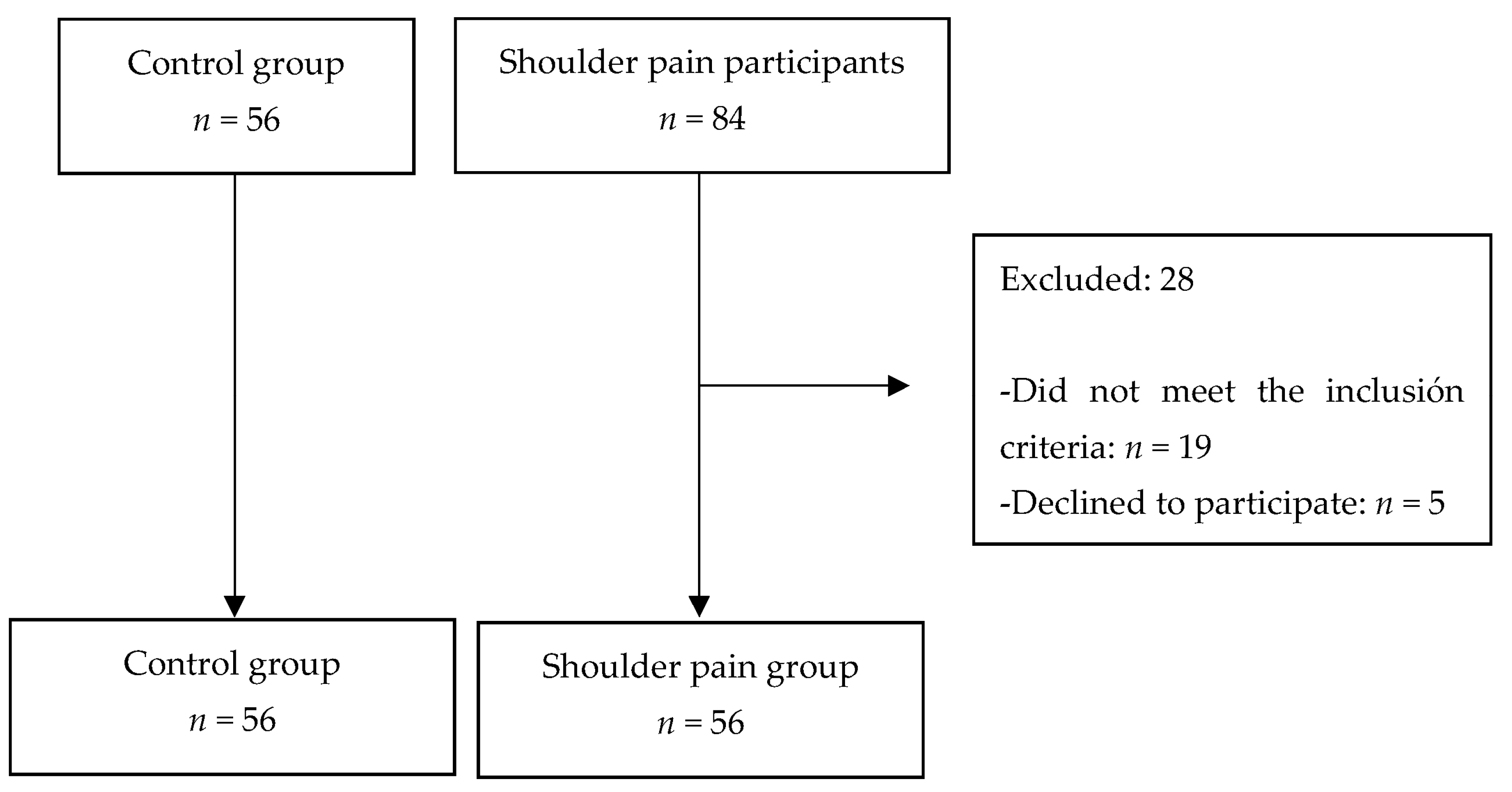
2. Methods
2.1. Measurements and Procedures
2.1.1. Pain Intensity
2.1.2. Physical Factors
2.1.3. Psychological Vulnerability
2.1.4. Pronociceptive Pain Modulation Profile
2.1.5. Disability
2.2. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McBeth, J.; Jones, K. Epidemiology of chronic musculoskeletal pain. Best Pract. Res. Clin. Rheumatol. 2007, 21, 403–425. [Google Scholar] [CrossRef] [PubMed]
- Jordan, K.P.; Kadam, U.T.; Hayward, R.; Porcheret, M.; Young, C.; Croft, P. Annual consultation prevalence of regional musculoskeletal problems in primary care: An observational study. BMC Musculoskelet. Disord. 2010, 11, 144. [Google Scholar] [CrossRef]
- Gaskin, D.J.; Richard, P. The economic costs of pain in the United States. J. Pain. 2012, 13, 715–724. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Calderon, J.; Struyf, F.; Meeus, M.; Morales-Ascencio, J.M.; Luque-Suarez, A. Influence of psychological factors on the prognosis of chronic shoulder pain: Protocol for a prospective cohort study. BMJ. Open 2017, 7, e012822. [Google Scholar] [CrossRef]
- Lobo, C.C.; Morales, C.R.; Sanz, D.R.; Corbalán, I.S.; Romero, E.A.S.; Carnero, J.F.; López, D.L. Comparison of hand grip strength and upper limb pressure pain threshold between older adults with or without non-specific shoulder pain. PeerJ 2017, 5, e2995. [Google Scholar] [CrossRef] [PubMed]
- Camargo, P.R.; Alburquerque-Sendín, F.; Avila, M.A.; Haik, M.N.; Vieira, A.; Salvini, T.F. Effects of Stretching and Strengthening Exercises, with and without Manual Therapy, on Scapular Kinematics, Function, and Pain in Individuals with Shoulder Impingement: A Randomized Controlled Trial. J. Orthop. Sports Phys. Ther. 2015, 45, 984–997. [Google Scholar] [CrossRef] [PubMed]
- Haik, M.N.; Alburquerque-Sendín, F.; Moreira, R.F.; Pires, E.D.; Camargo, P.R. Effectiveness of physical therapy treatment of clearly defined subacromial pain: A systematic review of randomised controlled trials. Br. J. Sports. Med. 2016, 50, 1124–1134. [Google Scholar] [CrossRef] [PubMed]
- Dinnes, J.; Loveman, E.; McIntyre, L.; Waugh, N. The effectiveness of diagnostic tests for the assessment of shoulder pain due to soft tissue disorders: A systematic review. Health Technol. Assess. 2003, 7, 1–166. [Google Scholar] [CrossRef]
- Giamberardino, M.A.; Tana, C.; Costantini, R. Pain thresholds in women with chronic pelvic pain. Curr. Opin. Obstet. Gynecol. 2014, 26, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Nijs, J.; Goubert, D.; Ickmans, K. Recognition and Treatment of Central Sensitization in Chronic Pain Patients: Not Limited to Specialized Care. J. Orthop. Sports. Phys. Ther. 2016, 46, 1024–1028. [Google Scholar] [CrossRef]
- Grinberg, K.; Granot, M.; Lowenstein, L.; Abramov, L.; Weissman-Fogel, I. A common pronociceptive pain modulation profile typifying subgroups of chronic pelvic pain syndromes is interrelated with enhanced clinical pain. Pain. 2017, 158, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Granot, M.; Lavee, Y. Psychological factors associated with perception of experimental pain in vulvar vestibulitis syndrome. J. Sex. Marital. Ther. 2005, 31, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Klintberg, I.H.; Cools, A.M.; Holmgren, T.M.; Holzhausen, A.C.G.; Johansson, K.; Maenhout, A.G.; Ginn, K. Consensus for physiotherapy for shoulder pain. Int. Orthop. 2015, 39, 715–720. [Google Scholar] [CrossRef]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152, S2–S15. [Google Scholar] [CrossRef] [PubMed]
- General Assembly of the World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. J. Am. Coll. Dent. 2014, 81, 14–18. [Google Scholar]
- Roberts, H.C.; Denison, H.J.; Martin, H.J.; Patel, H.P.; Syddall, H.; Cooper, C.; Sayer, A.A. A review of the measurement of grip strength in clinical and epidemiological studies: Towards a standardised approach. Age Ageing 2011, 40, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Peolsson, A.; Hedlund, R.; Oberg, B. Intra- and inter-tester reliability and reference values for hand strength. J. Rehabil. Med. 2001, 33, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W.; Schaubert, K.L. Test-retest reliability of grip-strength measures obtained over a 12-week interval from community-dwelling elders. J. Hand. Ther. 2005, 18, 426–427. [Google Scholar] [CrossRef]
- Sporrong, H.; Palmerud, G.; Herberts, P. Hand grip increases shoulder muscle activity, An EMG analysis with static hand contractions in 9 subjects. Acta Orthop. Scand. 1996, 67, 485–490. [Google Scholar] [CrossRef]
- Gómez-Pérez, L.; López-Martínez, A.E.; Ruiz-Párraga, G.T. Psychometric Properties of the Spanish Version of the Tampa Scale for Kinesiophobia (TSK). J. Pain 2011, 12, 425–435. [Google Scholar] [CrossRef]
- Kovacs, F.M.; Muriel, A.; Medina, J.M.; Abraira, V.; Sánchez, M.D.; Jaúregui, J.O. Spanish Back Pain Research Network. Psychometric characteristics of the Spanish version of the FAB questionnaire. Spine 2006, 31, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Andersen, H.; Ge, H.Y.; Arendt-Nielsen, L.; Danneskiold-Samsøe, B.; Graven-Nielsen, T. Increased trapezius pain sensitivity is not associated with increased tissue hardness. J. Pain 2010, 11, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Chesterton, L.S.; Sim, J.; Wright, C.C.; Foster, N.E. Interrater reliability of algometry in measuring pressure pain thresholds in healthy humans, using multiple raters. Clin. J. Pain 2007, 23, 760–766. [Google Scholar] [CrossRef] [PubMed]
- Granges, G.; Littlejohn, G. Pressure pain threshold in pain-free subjects, in patients with chronic regional pain syndromes, and in patients with fibromyalgia syndrome. Arthritis Rheum. 1993, 36, 642–646. [Google Scholar] [CrossRef]
- Mintken, P.E.; Glynn, P.; Cleland, J.A. Psychometric properties of the shortened disabilities of the Arm, Shoulder, and Hand Questionnaire (QuickDASH) and Numeric Pain Rating Scale in patients with shoulder pain. J. Shoulder. Elbow. Surg. 2009, 18, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Hervás, M.T.; Collado, M.J.N.; Peiró, S.; Pérez, J.L.R.; Matéu, P.L.; Tello, I.M. Versión española del cuestionario DASH. Adaptación transcultural, fiabilidad, validez y sensibilidad a los cambios [Spanish version of the DASH questionnaire. Cross-cultural adaptation, reliability, validity and responsiveness]. Med. Clin. 2006, 127, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Polson, K.; Reid, D.; McNair, P.J.; Larmer, P. Responsiveness, minimal importance difference and minimal detectable change scores of the shortened disability arm shoulder hand (QuickDASH) questionnaire. Man. Ther. 2010, 15, 404–407. [Google Scholar] [CrossRef]
- Portney, L.G.; Watkins, M.P. Foundations of Clinical Research: Applications to Practice, 2nd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2000. [Google Scholar]
- Bang, M.D.; Deyle, G.D. Comparison of supervised exercise with and without manual physical therapy for patients with shoulder impingement syndrome. J. Orthop. Sports. Phys. Ther. 2000, 30, 126–137. [Google Scholar] [CrossRef]
- Ebenbichler, G.R.; Erdogmus, C.B.; Resch, K.L.; Funovics, M.A.; Kainberger, F.; Barisani, G.; Fialka-Moser, V.; Baghestanian, M.; Preisinger, E.; Weinstabl, R.; et al. Ultrasound therapy for calcific tendinitis of the shoulder. N. Engl. J. Med. 1999, 340, 1533–1538. [Google Scholar] [CrossRef]
- Ginn, K.A.; Herbert, R.D.; Khouw, W.; Lee, R. A randomized, controlled clinical trial of a treatment for shoulder pain. Phys. Ther. 1997, 77, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Nicassio, P.M.; Moxham, E.G.; Schuman, C.E.; Gevirtz, R.N. The contribution of pain, reported sleep quality, and depressive symptoms to fatigue in fibromyalgia. Pain 2002, 100, 271–279. [Google Scholar] [CrossRef]
- Buer, N.; Linton, S.J. Fear-avoidance beliefs and catastrophizing: Occurrence and risk factor in back pain and ADL in the general population. Pain 2002, 99, 485–491. [Google Scholar] [CrossRef]
- Grotle, M.; Vøllestad, N.K.; Veierød, M.B.; Brox, J.I. Fear-avoidance beliefs and distress in relation to disability in acute and chronic low back pain. Pain 2004, 112, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Vernon, H.; Guerriero, R.; Soave, D.; Kavanaugh, S.; Puhl, A.; Reinhart, C. The relationship between self-rated disability, fear-avoidance beliefs, and nonorganic signs in patients with chronic whiplash-associated disorder. J. Manip. Physiol. Ther. 2011, 34, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Keefe, F.J.; Lefebvre, J.C.; Egert, J.R.; Affleck, G.; Sullivan, M.J.; Caldwell, D.S. The relationship of gender to pain, pain behavior, and disability in osteoarthritis patients: The role of catastrophizing. Pain 2000, 87, 325–334. [Google Scholar] [CrossRef]
- Andersen, L.L.; Andersen, C.H.; Skotte, J.H.; Suetta, C.; Søgaard, K.; Saltin, B.; Sjøgaard, G. High-intensity strength training improves function of chronically painful muscles: Case-control and RCT studies. BioMed Res. Int. 2014, 2014, 187324. [Google Scholar] [CrossRef]
- Brandt, M.; Sundstrup, E.; Jakobsen, M.D.; Jay, K.; Colado, J.C.; Wang, Y.; Andersen, L.L. Association between Neck/Shoulder Pain and Trapezius Muscle Tenderness in Office Workers. Pain Res. Treat. 2014, 2014, 352735. [Google Scholar] [CrossRef]
- Crombez, G.; Vlaeyen, J.W.; Heuts, P.H.; Lysens, R. Pain-related fear is more disabling than pain itself: Evidence on the role of pain-related fear in chronic back pain disability. Pain 1999, 80, 329–339. [Google Scholar] [CrossRef]
- George, S.Z.; Dover, G.C.; Fillingim, R.B. Fear of pain influences outcomes after exercise-induced delayed onset muscle soreness at the shoulder. Clin. J. Pain 2007, 23, 76–84. [Google Scholar] [CrossRef]
- George, S.Z.; Hirsh, A.T. Psychologic influence on experimental pain sensitivity and clinical pain intensity for patients with shoulder pain. J. Pain 2009, 10, 293–299. [Google Scholar] [CrossRef]
- Rodriguez-Raecke, R.; Doganci, B.; Breimhorst, M.; Stankewitz, A.; Büchel, C.; Birklein, F.; May, A. Insular cortex activity is associated with effects of negative expectation on nociceptive long-term habituation. J. Neurosci. 2010, 30, 11363–11368. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.M.; Turk, D.C. Central sensitization and the biopsychosocial approach to understanding pain. J. Appl. Biobehav. Res. 2018, 23, e12125. [Google Scholar] [CrossRef]
- Adams, L.M.; Turk, D.C. Psychosocial factors and central sensitivity syndromes. Curr. Rheumatol. Rev. 2015, 11, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Domenech, J.; Sánchez-Zuriaga, D.; Segura-Ortí, E.; Espejo-Tort, B.; Lisón, J.F. Impact of biomedical and biopsychosocial training sessions on the attitudes, beliefs, and recommendations of health care providers about low back pain: A randomised clinical trial. Pain 2011, 152, 2557–2563. [Google Scholar] [CrossRef]
- Anwer, S.; Alghadir, A.H.; Al-Eisa, E.S.; Iqbal, Z.A. The relationships between shoulder pain, range of motion, and disability in patients with shoulder dysfunction. J. Back Musculoskelet. Rehabil. 2018, 31, 163–167. [Google Scholar] [CrossRef]
- Haik, M.N.; Evans, K.; Smith, A.; Henríquez, L.; Bisset, L. People with musculoskeletal shoulder pain demonstrate no signs of altered pain processing. Musculoskelet. Sci. Pract. 2019, 39, 32–38. [Google Scholar] [CrossRef]
| Shoulder Pain Group (n = 56) | Control Group (n = 56) | p | |
|---|---|---|---|
| Sex (% men) | 61.1 | 35.2 | 0.127 |
| Age (years) | 54.35 ± 9.23 | 66.94 ± 11.50 | 0.001 * |
| BMI (kg/m2) | 26.09 ± 3.08 | 25.24 ± 3.51 | 0.452 |
| Occupation (%) | |||
| Physical jobs Not physical jobs | 61.1 38.8 | 35.2 64.7 | 0.127 |
| Pain duration | 3.39 ± 0.92 | - | - |
| Initial side of pain (% right) | 76.4 | - | - |
| Shoulder Pain Group (n = 56) | Control Group (n = 56) | p | |
|---|---|---|---|
| Pain intensity | |||
| VAS right | 4.36 ± 2.26 | 1.9 ± 2 | 0.003 * |
| VAS left | 3.39 ± 3.67 | 0.53 ± 1.33 | 0.006 * |
| Physical factors | |||
| Dynamometry right | 25.4 ± 9.29 | 29.95 ± 12.98 | 0.240 |
| Dynamometry left | 24.94 ± 8.29 | 29.87 ± 13.41 | 0.197 |
| Pronociceptive pain modulation profile | |||
| Shoulder algometry right | 2.54 ± 1.04 | 4.71 ± 1.67 | <0.001 ** |
| Shoulder algometry left | 2.68 ± 1.35 | 4.85 ± 1.99 | 0.001 * |
| Trapezium algometry right | 3.63 ± 1.96 | 5.13 ± 1.99 | 0.032 * |
| Trapezium algometry left | 3.49 ± 1.68 | 5.55 ± 1.73 | 0.001 * |
| Psychological vulnerability | |||
| FABQ 1 | 15.56 ± 7.22 | 10.76 ± 8.59 | 0.083 |
| FABQ 2 | 17.44 ± 11.61 | 9 ± 11.27 | 0.036 * |
| FABQ total | 33 ± 17.87 | 19.29 ± 14.23 | <0.001 ** |
| TSK | 45.94 ± 7.5 | 32.53 ± 11.98 | <0.001 ** |
| Disability | |||
| QuickDASH | 42.8 ± 20.09 | 18.24 ± 16.83 | <0.001 ** |
| Disability | Psychological Vulnerability | Physical Factors | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| QuickDASH | FABQ1 | FABQ2 | FABQ Total | TSK | VAS Right | VAS Left | Dynamometry Right | Dynamometry Left | ||
| Pain intensity | ||||||||||
| Physical factors | VAS right | 0.779 ** | 0.674 ** | 0.182 | 0.455 ** | 0.440 * | 1 | 0.346 | −0.480 ** | −0.510 ** |
| VAS left | 0.396 * | 0.211 | 0.462 * | 0.427 * | 0.194 | 0.346 | 1 | −0.176 | −0.225 | |
| Dynamometry right | −0.403 * | −0.403 ** | −0.147 | −0.286 | −0.420 * | −0.480 ** | −0.176 | 1 | 0.957 ** | |
| Dynamometry left | −0.444 ** | −0.330 | −0.179 | −0.272 | −0.364 * | −0.510 ** | −0.225 | 0.957 ** | 1 | |
| Pronociceptive pain variables | Shoulder algometry right | −0.770 ** | −0.729 ** | −0.397 ** | −0.626 ** | −0.555 ** | −0.658 ** | −0.452 * | 0.450 ** | 0.456 ** |
| Shoulder algometry left | −0.759 ** | −0.630 ** | −0.353 ** | 0.544 ** | −0.481 ** | −0.680 ** | −0.491 ** | 0.602 ** | 0.643 ** | |
| Trapezium algometry right | −0.659 ** | −0.543 ** | −0.270 | −0.438 ** | −0.236 | −0.540 ** | −0.412 * | 0.477 ** | 0.461 ** | |
| Trapezium algometry left | −0.668 ** | −0.552 ** | −0.287 | −0.454 ** | −0.268 | −0.667 ** | −0.553 ** | 0.518 ** | 0.542 ** | |
| Disability | QuickDASH | 1 | 0.781 ** | 0.406 * | 0.657 ** | 0.414 * | 0.779 ** | 0.369 ** | −0.403 * | −0.444 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valenza-Peña, G.; Martín-Núñez, J.; Heredia-Ciuró, A.; Granados-Santiago, M.; López-López, L.; Valenza, M.C.; Cabrera-Martos, I. Relationship between Pain Intensity, Physical Factors, Pronociceptive Pain Modulation Profile and Psychological Vulnerability on Upper Limb Disability in Older Patients with Chronic Shoulder Pain. Int. J. Environ. Res. Public Health 2022, 19, 15006. https://doi.org/10.3390/ijerph192215006
Valenza-Peña G, Martín-Núñez J, Heredia-Ciuró A, Granados-Santiago M, López-López L, Valenza MC, Cabrera-Martos I. Relationship between Pain Intensity, Physical Factors, Pronociceptive Pain Modulation Profile and Psychological Vulnerability on Upper Limb Disability in Older Patients with Chronic Shoulder Pain. International Journal of Environmental Research and Public Health. 2022; 19(22):15006. https://doi.org/10.3390/ijerph192215006
Chicago/Turabian StyleValenza-Peña, Geraldine, Javier Martín-Núñez, Alejandro Heredia-Ciuró, María Granados-Santiago, Laura López-López, Marie Carmen Valenza, and Irene Cabrera-Martos. 2022. "Relationship between Pain Intensity, Physical Factors, Pronociceptive Pain Modulation Profile and Psychological Vulnerability on Upper Limb Disability in Older Patients with Chronic Shoulder Pain" International Journal of Environmental Research and Public Health 19, no. 22: 15006. https://doi.org/10.3390/ijerph192215006
APA StyleValenza-Peña, G., Martín-Núñez, J., Heredia-Ciuró, A., Granados-Santiago, M., López-López, L., Valenza, M. C., & Cabrera-Martos, I. (2022). Relationship between Pain Intensity, Physical Factors, Pronociceptive Pain Modulation Profile and Psychological Vulnerability on Upper Limb Disability in Older Patients with Chronic Shoulder Pain. International Journal of Environmental Research and Public Health, 19(22), 15006. https://doi.org/10.3390/ijerph192215006








