Sexual Function in Women with Breast Cancer: An Evidence Map of Observational Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Eligibility Criteria
2.3. Search Strategy
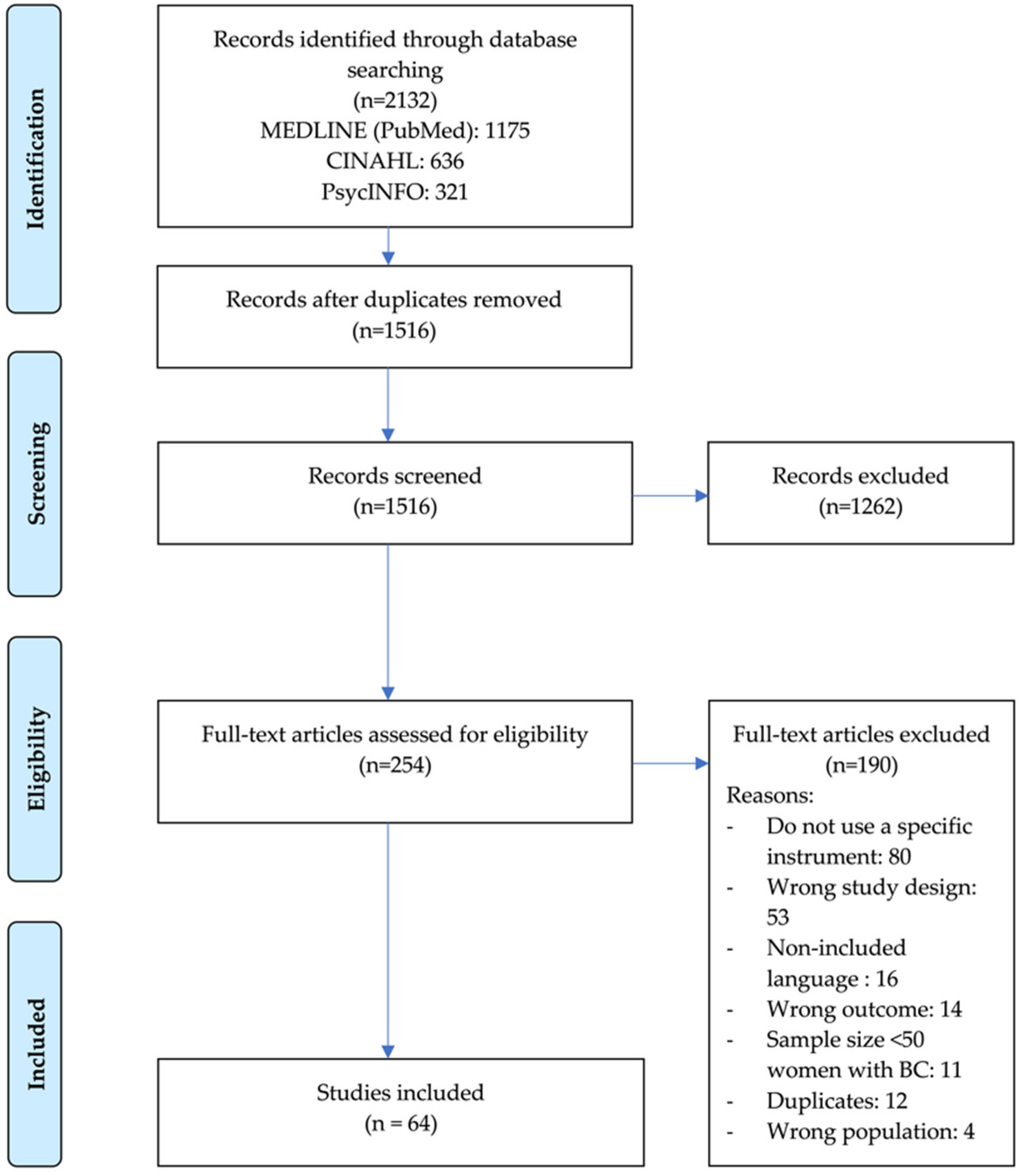
2.4. Study Selection
2.5. Data Extraction
3. Results
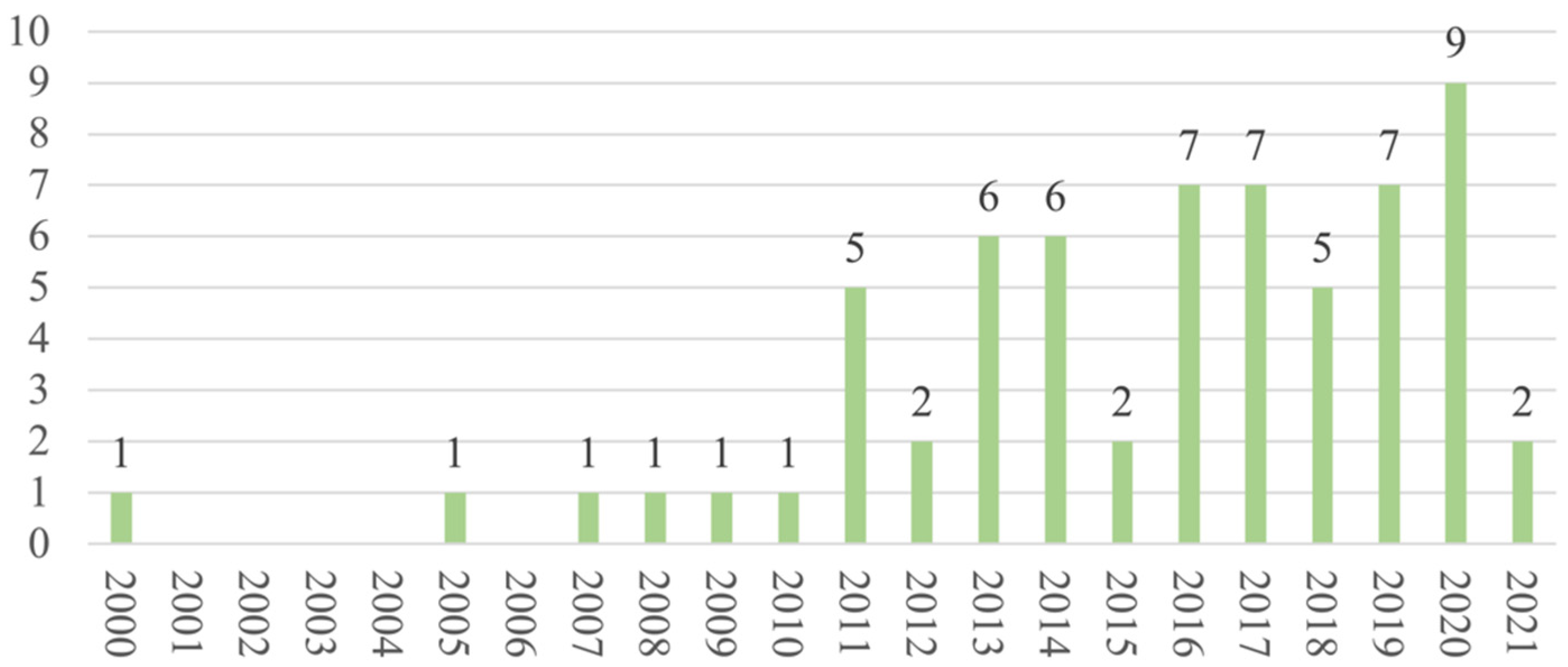
3.1. Selected Studies
3.2. Study Characteristics
3.3. SF Assessment
4. Discussion
4.1. Study Limitations
4.2. Clinical Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Es-timates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, O.; Yip, C.; Brooks, A.; Cabanes, A.; Caleffi, M.; Yataco, J.A.D.; Gyawali, B.; McCormack, V.; de Anderson, M.M.; Mehrotra, R.; et al. Breast cancer early detection: A phased approach to implementation. Cancer 2020, 126, 2379–2393. [Google Scholar] [CrossRef] [PubMed]
- Carreira, H.; Williams, R.; Müller, M.; Harewood, R.; Stanway, S.; Bhaskaran, K. Associations Between Breast Cancer Survivorship and Adverse Mental Health Outcomes: A Systematic Review. J. Natl. Cancer Inst. 2018, 110, 1311–1327. [Google Scholar] [CrossRef] [PubMed]
- Jing, L.; Zhang, C.; Li, W.; Jin, F.; Wang, A. Incidence and severity of sexual dysfunction among women with breast cancer: A me-ta-analysis based on female sexual function index. Support. Care Cancer 2019, 27, 1171–1180. [Google Scholar] [CrossRef] [PubMed]
- Cobo-Cuenca, A.I.; Espínosa, N.M.M.; Sampietro-Crespo, A.; Rodríguez-Borrego, M.A.; Carmona-Torres, J.M. Sexual dysfunction in Spanish women with breast cancer. PLoS ONE 2018, 13, e0203151. [Google Scholar] [CrossRef]
- Ljungman, L.; Ahlgren, J.; Petersson, L.-M.; Flynn, K.; Weinfurt, K.; Gorman, J.R.; Wettergren, L.; Lampic, C. Sexual dysfunction and reproductive concerns in young women with breast cancer: Type, prevalence, and predictors of problems. Psycho-Oncology 2018, 27, 2770–2777. [Google Scholar] [CrossRef]
- Masià, J.; Merchán-Galvis, Á.; Salas, K.; Requeijo, C.; Cánovas, E.; Quintana, M.J.; Bonfill, X. Socio-economic impact on women diagnosed and treated for breast cancer: A cross-sectional study. Clin. Transl. Oncol. 2019, 21, 1736–1745. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Boswell, E.N.; Dizon, D.S. Breast cancer and sexual function. Transl. Androl. Urol. 2015, 4, 160–168. [Google Scholar] [CrossRef]
- Bragge, P.; Clavisi, O.; Turner, T.; Tavender, E.; Collie, A.; Gruen, R.L. The Global Evidence Mapping Initiative: Scoping research in broad topic areas. BMC Med. Res. Methodol. 2011, 11, 92. [Google Scholar] [CrossRef]
- Miake-Lye, I.M.; Hempel, S.; Shanman, R.; Shekelle, P.G. What is an evidence map? A systematic review of published evidence maps and their definitions, methods, and products. Syst. Rev. 2016, 5, 1–21. [Google Scholar] [CrossRef]
- Haddaway, N.R.; Bernes, C.; Jonsson, B.-G.; Hedlund, K. The benefits of systematic mapping to evidence-based environmental management. Ambio 2016, 45, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Snilstveit, B.; Vojtkova, M.; Bhavsar, A.; Stevenson, J.; Gaarder, M. Evidence & Gasp Maps: A tool for promoting evidence informed policy and strategic research agendas. J. Clin. Epidemiol. 2016, 79, 120–129. [Google Scholar] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Abasher, S.M. Sexual health issues in Sudanese women before and during hormonal treatment for breast cancer. Psycho-Oncology 2009, 18, 858–865. [Google Scholar] [CrossRef] [PubMed]
- Aerts, L.; Christiaens, M.; Enzlin, P.; Neven, P.; Amant, F. Sexual functioning in women after mastectomy versus breast conserving therapy for early-stage breast cancer: A prospective controlled study. Breast 2014, 23, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Alacacioglu, A.; Ulger, E.; Varol, U.; Yildiz, I.; Salman, T.; Bayoglu, V.; Dirican, A.; Demir, L.; Akyol, M.; Yildiz, Y.; et al. Depression, Anxiety and Sexual Satisfaction in Breast Cancer Patients and their Partners-Izmir Oncology Group Study. Asian Pac. J. Cancer Prev. 2014, 15, 10631–10636. [Google Scholar] [CrossRef]
- Archangelo S de, C.V.; Sabino, M.; Veiga, D.F.; Garcia, E.B.; Ferreira, L.M. Sexuality, depression and body image after breast recon-struction. Clinics 2019, 74, e883. [Google Scholar] [CrossRef]
- Assogba, E.L.; Mamguem Kamga, A.; Costaz, H.; Jankowski, C.; Dumas, A.; Roignot, P.; Jolimoy, G.; Coutant, C.; Arveux, P.; Dabakuyo-Yonli, T.S. What Are Young Women Living Conditions after Breast Cancer? Health-Related Quality of Life, Sexual and Fertility Issues, Professional Reinsertion. Cancers 2020, 12, 1564. [Google Scholar] [CrossRef]
- Bober, S.L.; Giobbie-Hurder, A.; Emmons, K.M.; Winer, E.; Partridge, A. Psychosexual Functioning and Body Image Following a Diagnosis of Ductal Carcinoma In Situ. J. Sex. Med. 2013, 10, 370–377. [Google Scholar] [CrossRef]
- Boehmer, U.; Ozonoff, A.; Timm, A.; Winter, M.; Potter, J. After Breast Cancer: Sexual Functioning of Sexual Minority Survivors. J. Sex Res. 2014, 51, 681–689. [Google Scholar] [CrossRef]
- Brédart, A.; Dolbeault, S.; Savignoni, A.; Besancenet, C.; This, P.; Giami, A.; Michaels, S.; Flahault, C.; Falcou, M.-C.; Asselain, B.; et al. Prevalence and associated factors of sexual problems after early-stage breast cancer treatment: Results of a French exploratory survey. Psycho-Oncology 2011, 20, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Robles, L.S.B.; Soto-Lesmes, V.I. Salud sexual y alteraciones emocionales en mujeres colombianas con cáncer de mama. Psicooncol. Investig. Clín. Biopsicosoc. Oncol. 2015, 12, 405–416. [Google Scholar] [CrossRef][Green Version]
- Córdoba-de Juan, C.; Arranz-Martín, B.; Torres-Lacomba, M. Disfunción sexual en mujeres diagnosticadas y tratadas de cáncer de mama. Estudio descriptivo longitudinal. Fisioterapia 2019, 41, 73–82. [Google Scholar] [CrossRef]
- Cornell, L.F.; Mussallem, D.M.; Gibson, T.C.; Diehl, N.N.; Bagaria, S.P.; McLaughlin, S.A. Trends in Sexual Function After Breast Cancer Surgery. Ann. Surg. Oncol. 2017, 24, 2526–2538. [Google Scholar] [CrossRef]
- Cortés-Flores, A.O.; Vargas-Meza, A.; Morgan-Villela, G.; Jiménez-Tornero, J.; Del Valle, C.J.Z.-F.; Solano-Genesta, M.; Miranda-Ackerman, R.C.; Vázquez-Reyna, I.; García-González, L.A.; Cervantes-Cardona, G.A.; et al. Sexuality Among Women Treated for Breast Cancer: A Survey of Three Surgical Procedures. Aesthetic Plast. Surg. 2017, 41, 1275–1279. [Google Scholar] [CrossRef]
- Davis, S.C.; Meneses, K.; Messias, D.K.H. Exploring sexuality & quality of life in women after breast cancer surgery. Nurse Pract. 2010, 35, 25–31. [Google Scholar] [CrossRef]
- Ellouz, F.; Marrakchi, N.; Raies, H.; Masmoudi, S.; Mezlini, A.; M’Rad, M. Dysfonction sexuelle chez 100 femmes tunisiennes atteintes d’un cancer du sein. Sexologies 2019, 28, 43–48. [Google Scholar] [CrossRef]
- Elmas, Ö.; ÇAkmak, G.K.; Bakkal, B.H. A Comparison between Breast-Conserving Surgery and Modified Radical Mastectomy Concerning the Female Sexual Function in Breast Cancer Patients under 50 Years of Age. Turk. J. Oncol. 2020, 35, 26–30. [Google Scholar]
- Farthmann, J.; Hanjalicbeck, A.; Veit, J.J.; Rautenberg, B.; Stickeler, E.; Erbes, T.; Földi, M.; Hasenburg, A. The impact of chemotherapy for breast cancer on sexual function and health-related quality of life. Support. Care Cancer 2016, 24, 2603–2609. [Google Scholar] [CrossRef]
- Fogh, M.; Højgaard, A.; Rotbøl, C.B.; Jensen, A.B. The majority of Danish breast cancer survivors on adjuvant endocrine therapy have clinically relevant sexual dysfunction: A cross-sectional study. Acta Oncol. 2021, 60, 61–68. [Google Scholar] [CrossRef]
- Fouladi, N.; Feizi, I.; Mehriar, N.; Mehrara, E.; Adldoosti, R.; Alimohammadi, S. The Predictors of Sexual Satisfaction among Iranian Women with Breast Cancer. Asian Pac. J. Cancer Prev. 2021, 22, 391–396. [Google Scholar] [CrossRef]
- Frechette, D.; Paquet, L.; Verma, S.; Clemons, M.; Wheatley-Price, P.; Gertler, S.Z.; Song, X.; Graham, N.; Dent, S. The impact of endocrine therapy on sexual dysfunction in postmenopausal women with early stage breast cancer: Encouraging results from a prospective study. Breast Cancer Res. Treat. 2013, 141, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, A.; Esposito, D.; Accardo, G.; Taddeo, M.; Letizia, A.; Tagliafierro, R.; Esposito, K.; Pasquali, D. Sexual function and sex hormones in breast cancer patients. Endocrine 2018, 60, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, C.; Butler, E.; Pesek, S.; Kwait, R.; Edmonson, D.; Raker, C.; Clark, M.A.; Stuckey, A.; Gass, J. Sexual Dysfunction in Breast Cancer Survivors. Am. J. Clin. Oncol. 2019, 42, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Harirchi, I.; Montazeri, A.; Bidokhti, F.Z.; Mamishi, N.; Zendehdel, K. Sexual function in breast cancer patients: A prospective study from Iran. J. Exp. Clin. Cancer Res. 2012, 31, 20–26. [Google Scholar] [CrossRef][Green Version]
- Herbenick, D.; Reece, M.; Hollub, A.; Satinsky, S.; Dodge, B. Young Female Breast Cancer Survivors. Cancer Nurs. 2008, 31, 417–425. [Google Scholar] [CrossRef]
- İzci, F.; Özdem, G.; İlgün, A.S.; Ağaçayak, F.; Duymaz, T.; Erdoğan, Z.; Alço, G.; Elbüken, F.; Öztürk, A.; Ordu, Ç.; et al. Pre-Treatment and Post-Treatment Anxiety, Depression, Sleep and Sexual Function Levels in Patients with Breast Cancer. Eur. J. Breast Health 2020, 16, 219–225. [Google Scholar]
- Kedde, H.; Van De Wiel, H.B.M.; Schultz, W.C.M.W.; Wijsen, C. Sexual dysfunction in young women with breast cancer. Support. Care Cancer 2013, 21, 271–280. [Google Scholar] [CrossRef]
- Kowalczyk, R.; Nowosielski, K.; Cedrych, I.; Krzystanek, M.; Glogowska, I.; Streb, J.; Kucharz, J.; Lew-Starowicz, Z. Factors Affecting Sexual Function and Body Image of Early-Stage Breast Cancer Survivors in Poland: A Short-Term Observation. Clin. Breast Cancer 2019, 19, e30–e39. [Google Scholar] [CrossRef]
- Landi, S.N.; Doll, K.M.; Bensen, J.T.; Hendrix, L.; Anders, C.K.; Wu, J.M.; Nichols, H.B. Endocrine therapy and urogenital outcomes among women with a breast cancer diagnosis. Cancer Causes Control 2016, 27, 1325–1332. [Google Scholar] [CrossRef]
- Lashani, F.; Rohani, C.; Estebsari, F.; Nasiri, M. Exploring the relationship between sexual function, sense of coherence, and well-being in a sample of Iranian breast cancer survivors. Support. Care Cancer 2020, 29, 3191–3199. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Kim, Y.H.; Jeon, M.J. Risk factors for negative impacts on sexual activity and function in younger breast cancer survivors. Psycho-Oncology 2015, 24, 1097–1103. [Google Scholar] [CrossRef] [PubMed]
- Manganiello, A.; Hoga, L.A.K.; Reberte, L.M.; Miranda, C.M.; Rocha, C.A.M. Sexuality and quality of life of breast cancer patients post mastectomy. Eur. J. Oncol. Nurs. 2011, 15, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Mayer, S.; Iborra, S.; Grimm, D.; Steinsiek, L.; Mahner, S.; Bossart, M.; Woelber, L.; Voss, P.J.; Gitsch, G.; Hasenburg, A. Sexual activity and quality of life in patients after treatment for breast and ovarian cancer. Arch. Gynecol. Obstet. 2019, 299, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, K.A.; Semple, J.; Quan, M.-L.; Vadaparampil, S.T.; Holloway, C.; Brown, M.; Bower, B.; Sun, P.; Narod, S.A. Changes in Psychosocial Functioning 1 Year After Mastectomy Alone, Delayed Breast Reconstruction, or Immediate Breast Reconstruction. Ann. Surg. Oncol. 2012, 19, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Notari, S.C.; Favez, N.; Notari, L.; Panes-Ruedin, B.; Antonini, T.; Delaloye, J.-F. Women’s experiences of sexual functioning in the early weeks of breast cancer treatment. Eur. J. Cancer Care 2018, 27, e12607. [Google Scholar] [CrossRef]
- Oberguggenberger, A.; Martini, C.; Huber, N.; Fallowfield, L.; Hubalek, M.; Daniaux, M.; Sperner-Unterweger, B.; Holzner, B.; Sztankay, M.; Gamper, E.; et al. Self-reported sexual health: Breast cancer survivors compared to women from the general population—An observational study. BMC Cancer 2017, 17, 1–9. [Google Scholar] [CrossRef]
- Ztürk, D.; Akyolcu, N. Assessing sexual function and dysfunction in Turkish women undergoing surgical breast cancer treatment. Jpn. J. Nurs. Sci. 2016, 13, 220–228. [Google Scholar] [CrossRef]
- Paiva, C.E.; Rezende, F.F.; Paiva, B.S.R.; Mauad, E.C.; Zucca-Matthes, G.; Carneseca, E.C.; Syrjänen, K.J.; Schover, L.R. Associations of Body Mass Index and Physical Activity with Sexual Dysfunction in Breast Cancer Survivors. Arch. Sex. Behav. 2016, 45, 2057–2068. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Yoon, H.G. Menopausal symptoms, sexual function, depression, and quality of life in Korean patients with breast cancer receiving chemotherapy. Support. Care Cancer 2013, 21, 2499–2507. [Google Scholar] [CrossRef]
- Parker, P.A.; Youssef, A.; Walker, S.; Basen-Engquist, K.; Cohen, L.; Gritz, E.R.; Wei, Q.X.; Robb, G.L. Short-Term and Long-Term Psychosocial Ad-justment and Quality of Life in Women Undergoing Different Surgical Procedures for Breast Cancer. Ann. Surg. Oncol. 2007, 14, 3078–3089. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.; Sharma, K.; Thornton, B.M.; Myckatyn, T.M.; Tenenbaum, M.M. Vaginal Laxity, Sexual Distress, and Sexual Dysfunction: A Cross-Sectional Study in a Plastic Surgery Practice. Aesthetic Surg. J. 2018, 38, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Raggio, G.A.; Butryn, M.L.; Arigo, D.; Mikorski, R.; Palmer, S.C. Prevalence and correlates of sexual morbidity in long-term breast cancer survivors. Psychol. Health 2014, 29, 632–650. [Google Scholar] [CrossRef] [PubMed]
- Reese, J.B.; Sorice, K.A.; Pollard, W.; Zimmaro, L.A.; Beach, M.C.; Handorf, E.; Lepore, S.J. Understanding Sexual Help-Seeking for Women with Breast Cancer: What Distinguishes Women Who Seek Help From Those Who Do Not? J. Sex. Med. 2020, 17, 1729–1739. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.J.; Bell, R.J.; Christakis, M.K.; Ivezic, S.R.; Davis, S.R. Aromatase Inhibitors are Associated with Low Sexual Desire Causing Distress and Fecal Incontinence in Women: An Observational Study. J. Sex. Med. 2017, 14, 1566–1574. [Google Scholar] [CrossRef]
- Rojas, K.; Onstad, M.; Raker, C.; Clark, M.; Stuckey, A.; Gass, J. The impact of mastectomy type on the Female Sexual Function Index (FSFI), satisfaction with appearance, and the reconstructed breast’s role in intimacy. Breast Cancer Res. Treat. 2017, 163, 273–279. [Google Scholar] [CrossRef]
- Rosenberg, S.M.; Dominici, L.S.; Gelber, S.; Poorvu, P.D.; Ruddy, K.J.; Wong, J.S.; Tamimi, R.M.; Schapira, L.; Come, S.; Peppercorn, J.M.; et al. Association of Breast Cancer Surgery with Quality of Life and Psychosocial Well-being in Young Breast Cancer Survivors. JAMA Surg. 2020, 155, 1035. [Google Scholar] [CrossRef]
- Rottmann, N.; Hansen, D.G.; Christensen, R.D.; Hagedoorn, M.; Frisch, M.; Nicolaisen, A.; Kroman, N.; Flyger, H.; Johansen, C. Satisfaction with sex life in sexually active heterosexual couples dealing with breast cancer: A nationwide longitudinal study. Acta Oncol. 2017, 56, 212–219. [Google Scholar] [CrossRef]
- Rowland, J.H.; Desmond, K.A.; Meyerowitz, B.E.; Belin, T.R.; Wyatt, G.E.; Ganz, P.A. Role of Breast Reconstructive Surgery in Physical and Emotional Outcomes Among Breast Cancer Survivors. J. Natl. Cancer Inst. 2000, 92, 1422–1429. [Google Scholar] [CrossRef]
- Safarinejad, M.R.; Shafiei, N.; Safarinejad, S. Quality of life and sexual functioning in young women with early-stage breast cancer 1 year after lumpectomy. Psycho-Oncology 2013, 22, 1242–1248. [Google Scholar] [CrossRef]
- Sayakhot, P.; Vincent, A.; Deeks, A.; Teede, H. Potential adverse impact of ovariectomy on physical and psychological function of younger women with breast cancer. Menopause 2011, 18, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Sbitti, Y.; Kadiri, H.; Essaidi, I.; Fadoukhair, Z.; Kharmoun, S.; Slimani, K.; Ismaili, N.; Ichou, M.; Errihani, H. Breast cancer treatment and sexual dysfunction: Moroccan women’s perception. BMC Women’s Health 2011, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Schover, L.R.; Baum, G.P.; Fuson, L.A.; Brewster, A.; Melhem-Bertrandt, A. Sexual Problems During the First 2 Years of Adjuvant Treatment with Aromatase Inhibitors. J. Sex. Med. 2014, 11, 3102–3111. [Google Scholar] [CrossRef] [PubMed]
- Shandiz, F.H.; Karimi, F.Z.; Rahimi, N.; Abdolahi, M.; Anbaran, Z.K.; Ghasemi, M.; Mazlom, S.R.; Kheirabadi, A.N. Investigating Sexual Function and Affecting Factors in Women with Breast Cancer in Iran. Asian Pac. J. Cancer Prev. 2016, 17, 3583–3586. [Google Scholar]
- Soldera, S.V.; Ennis, M.; Lohmann, A.E.; Goodwin, P.J. Sexual health in long-term breast cancer survivors. Breast Cancer Res Treat. 2018, 172, 159–166. [Google Scholar] [CrossRef]
- Sorouri, F.; Yaghubi, H. Comparing the Negative Emotions, Body Image, Sexual Schemas and Sexual Function in Women with Breast Cancer and Healthy Women. Arch. Psychiatry Res. 2019, 55, 49–60. [Google Scholar] [CrossRef]
- Speer, J.J.; Hillenberg, B.; Sugrue, D.P.; Blacker, C.; Kresge, C.L.; Decker, V.B.; Zakalik, D.; Decker, D.A. Study of Sexual Functioning Determinants in Breast Cancer Survivors. Breast J. 2005, 11, 440–447. [Google Scholar] [CrossRef]
- Tahir, K.; Khan, N. Mediating role of body image between sexual functioning and marital intimacy in Pakistani women with breast cancer. Psycho-Oncology 2020, 30, 260–266. [Google Scholar] [CrossRef]
- Tucker, P.; Cohen, P.A.; Bulsara, M.K.; Jeffares, S.; Saunders, C. The impact of bilateral salpingo-oophorectomy on sexuality and quality of life in women with breast cancer. Support. Care Cancer 2020, 29, 369–375. [Google Scholar] [CrossRef]
- Tucker, P.E.; Saunders, C.; Bulsara, M.K.; Tan, J.J.-S.; Salfinger, S.G.; Green, H.; Cohen, P.A. Sexuality and quality of life in women with a prior diagnosis of breast cancer after risk-reducing salpingo-oophorectomy. Breast 2016, 30, 26–31. [Google Scholar] [CrossRef]
- Usta, O.Y.; Gokcol, D. Sexual Dysfunction in Women with Breast Cancer Receiving Chemotherapy. Int. J. Caring Sci. 2017, 10, 1439–1446. [Google Scholar]
- Vaidakis, D.; Panoskaltsis, T.; Poulakaki, N.; Kouloura, A.; Kassanos, D.; Papadimitriou, G.; Salamalekis, E. Female sexuality after female cancer treatment: A clinical issue. Eur. J. Gynaecol. Oncol. 2014, 35, 635–640. [Google Scholar] [PubMed]
- Webber, K.; Mok, K.; Bennett, B.; Lloyd, A.R.; Friedlander, M.; Juraskova, I.; Goldstein, D. FolCan study group If I Am in the Mood, I Enjoy It: An Exploration of Cancer-Related Fatigue and Sexual Functioning in Women with Breast Cancer. Oncologist 2011, 16, 1333–1344. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Wang, J.; Bender, C.M.; Zhang, N.; Yuan, C. Patterns of sexual health in patients with breast cancer in China: A latent class analysis. Support. Care Cancer 2020, 28, 5147–5156. [Google Scholar] [CrossRef]
- Zaied, S.; Fatma, L.B.; Laadhari, A.; Boudegga, M.Z.; Hochlef, M.; Chabchoub, I.; Ezzairi, F.; Gharbi, O.; Gaha, L.; Ahmed, S.B. Étude de la sexualité chez les femmes tuni-siennes en rémission complète d’un cancer du sein non métastatique, à propos de 100 femmes. Bull. Cancer 2013, 100, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Del Pup, L.; Villa, P.; Amar, I.D.; Bottoni, C.; Scambia, G. Approach to sexual dysfunction in women with cancer. Int. J. Gynecol. Cancer 2019, 29, 630–634. [Google Scholar] [CrossRef]
- Studd, J.; Schwenkhagen, A. The historical response to female sexuality. Maturitas 2009, 63, 107–111. [Google Scholar] [CrossRef]
- Hill, G.; Holborn, C. Sexual minority experiences of cancer care: A systematic review. J. Cancer Policy 2015, 6, 11–22. [Google Scholar] [CrossRef]
- Kamen, C.S.; Smith-Stoner, M.; Heckler, C.E.; Flannery, M.; Margolies, L. Social Support, Self-Rated Health, and Lesbian, Gay, Bisexual, and Transgender Identity Disclosure to Cancer Care Providers. Oncol. Nurs. Forum 2015, 42, 44–51. [Google Scholar] [CrossRef]
- Wandrey, R.L.; Qualls, W.D.; Mosack, K.E. Rejection of Breast Reconstruction Among Lesbian Breast Cancer Patients. LGBT Health 2016, 3, 74–78. [Google Scholar] [CrossRef]
- Mattingly, A.E.; Kiluk, J.V.; Lee, M.C. Clinical Considerations of Risk, Incidence, and Outcomes of Breast Cancer in Sexual Mi-norities. Cancer Control 2016, 23, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Pratt-Chapman, M.L.; Alpert, A.B.; Castillo, D.A. Health outcomes of sexual and gender minorities after cancer: A systematic review. Syst. Rev. 2021, 10, 1–30. [Google Scholar] [CrossRef]
- Rosen, C.; Brown, J.; Heiman, S.; Leiblum, C.; Meston, R.; Shabsigh, D.; Ferguson, R.; D’Agostino, R. The Female Sexual Function Index (FSFI): A Multidi-mensional Self-Report Instrument for the Assessment of Female Sexual Function. J. Sex Marital Ther. 2000, 26, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Bartula, I.; Sherman, K.A. The Female Sexual Functioning Index (FSFI): Evaluation of acceptability, reliability, and validity in women with breast cancer. Support. Care Cancer 2015, 23, 2633–2641. [Google Scholar] [CrossRef] [PubMed]
- Thirlaway, K.; Fallowfield, L.; Cuzick, J. The Sexual Activity Questionnaire: A measure of women’s sexual functioning. Qual. Life Res. 1996, 5, 81–90. [Google Scholar] [CrossRef]
- Atkins, L.; Fallowfield, L. Fallowfield’s Sexual Activity Questionnaire in women with without and at risk of cancer. Menopause Int. 2007, 13, 103–109. [Google Scholar] [CrossRef]
- da Costa, F.A.; Ribeiro, M.C.; Braga, S.; Carvalho, E.; Francisco, F.; da Costa Miranda, A.; Moreira, A.; Fallowfield, L. Sexual Dysfunction in Breast Cancer Sur-vivors: Cross-Cultural Adaptation of the Sexual Activity Questionnaire for Use in Portugal. Acta Med. Port. 2016, 29, 533. [Google Scholar] [CrossRef]
- Oppenheimer, A.; Panel, P.; Rouquette, A.; du Cheyron, J.; Deffieux, X.; Fauconnier, A. Validation of the Sexual Activity Question-naire in women with endometriosis. Hum. Reprod. 2019, 34, 824–833. [Google Scholar] [CrossRef]
- Vistad, I.; Fosså, S.D.; Kristensen, G.B.; Mykletun, A.; Dahl, A.A. The Sexual Activity Questionnaire: Pychometric Properties and Normative Data in A Norwegian Population Sample. J. Women’s Health 2007, 16, 139–148. [Google Scholar] [CrossRef]
- Flynn, K.E.; Lin, L.; Cyranowski, J.M.; Reeve, B.B.; Reese, J.B.; Jeffery, D.D.; Smith, A.W.; Porter, L.S.; Dombeck, C.B.; Bruner, D.W.; et al. Development of the NIH PROMIS® Sexual Function and Satisfaction Measures in Patients with Cancer. J. Sex. Med. 2013, 10, 43–52. [Google Scholar] [CrossRef]
- McCabe, M.P.; Sharlip, I.D.; Atalla, E.; Balon, R.; Fisher, A.D.; Laumann, E.; Lee, S.W.; Lewis, R.; Segraves, R.T. Definitions of Sexual Dysfunctions in Women and Men: A Consensus Statement From the Fourth International Consultation on Sexual Medicine 2015. J. Sex. Med. 2016, 13, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-H.; Lin, Y.-C.; Chiu, L.-H.; Chu, Y.-H.; Ruan, F.-F.; Liu, W.-M.; Wang, P.-H. Female sexual dysfunction: Definition, classification, and debates. Taiwan. J. Obstet. Gynecol. 2013, 52, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Park, B.-W.; Lee, S.; Lee, A.R.; Lee, K.-H.; Hwang, S.Y. Quality of Life Differences between Younger and Older Breast Cancer Patients. J. Breast Cancer 2011, 14, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-C.; Chang, S.-R.; Chiu, S.-C. Sexual Problems of Patients with Breast Cancer after Treatment. Cancer Nurs. 2019, 42, 418–425. [Google Scholar] [CrossRef]
- Mokhtari-Hessari, P.; Montazeri, A. Health-related quality of life in breast cancer patients: Review of reviews from 2008 to 2018. Health Qual. Life Outcomes 2020, 18, 338. [Google Scholar] [CrossRef]
- Zehra, S.; Doyle, F.; Barry, M.; Walsh, S.; Kell, M.R. Health-related quality of life following breast reconstruction compared to total mastectomy and breast-conserving surgery among breast cancer survivors: A systematic review and meta-analysis. Breast Cancer 2020, 27, 534–566. [Google Scholar] [CrossRef]
- Peddie, N.; Agnew, S.; Crawford, M.; Dixon, D.; MacPherson, I.; Fleming, L. The impact of medication side effects on adherence and persistence to hormone therapy in breast cancer survivors: A qualitative systematic review and thematic synthesis. Breast 2021, 58, 147–159. [Google Scholar] [CrossRef]
- Valpey, R.; Kucherer, S.; Nguyen, J. Sexual dysfunction in female cancer survivors: A narrative review. Gen. Hosp. Psychiatry 2019, 60, 141–147. [Google Scholar] [CrossRef]

| Author, Year | Journal | Study Design | Country | Objective | Participants (Age in Years) |
|---|---|---|---|---|---|
| Abasher, 2009 [15] | Psycho-Oncology | Case–control | Sudan | To assess whether there is any decrease in sexual function as a result of breast cancer treatment. | 100 BC/100 CG (range: 25–55) |
| Aerts et al., 2014 [16] | The Breast | Prospective | Belgium | To prospectively investigate the presence of sexual dysfunctions and the psychological and relational functioning in women after mastectomy and breast-conserving therapy, and to compare the sexual functioning of breast cancer patients with a control group of healthy women. | 149 BC/81 CG (mean ME: 54.51/mean BCT: 57.26/mean CG: 56.12) |
| Alacacioglu et al., 2014 [17] | Asian Pacific Journal of Cancer Prevention | Cross-sectional | Turkey | To investigate the anxiety, depression, and sexual satisfaction levels of Turkish breast cancer patients and their partners. | 100 BC (mean: 44.7) |
| Archangelo et al., 2019 [18] | Clinics | Cross-sectional | Brazil | To evaluate the impact of breast reconstruction after mastectomy on specific aspects of the patient quality of life, including sexual function, body image, and depression. | 90 BC (mean RG: 47.5/mean ME: 48/mean CG: 47) |
| Assogba et al., 2020 [19] | Cancers | Cross-sectional | France | To identify the clinical, social, and economic determinants of health-related quality of life, and to describe other living conditions of young long-term breast cancer survivors. | 218 BC (mean: 47.5) |
| Bober et al., 2013 [20] | Journal of Sexual Medicine | Prospective | USA | To characterise sexual functioning in women recently diagnosed with ductal carcinoma in situ, as well as at 9 and 18 months of follow-up. | 304 BC (median: 50.3) |
| Boehmer et al., 2014 [21] | The Journal of Sex Research | Case–control | USA | To compare the sexual function of breast cancer survivors (cases) to an age- and partner-status-matched control group of sexual minority women without cancer. | 85 BC/85 CG (mean BC: 51.6/mean CG: 50.9) |
| Brédart et al., 2011 [22] | Psycho-Oncology | Cross-sectional | France | To assess the prevalence and associated factors of the sexual activity, sexual problems, and sexual satisfaction of French early-stage breast cancer survivors. | 378 BC (mean: 53) |
| Bueno-Robles et al., 2015 [23] | Psicooncología | Cross-sectional | Colombia | To determine the effect of mood, anxiety, and depression on sexual health, and the impact on Colombian women who have undergone treatment for breast cancer. | 103 BC (mean: 48) |
| Cobo-Cuenca et al., 2018 [5] | Plos One | Cross-sectional | Spain | To determine whether there are changes in the sexuality of women after breast cancer, to understand the sexual function of women with breast cancer in Spain, and to describe the relationship between the sociodemographic and clinical variables and sexual dysfunction. | 514 BC (mean: 46.34) |
| Cordoba-de Juan et al., 2019 [24] | Fisioterapia | Cross-sectional | Spain | To describe sexual function state in women treated for breast cancer over a year after the diagnosis of breast cancer. | 109 BC (median: 56.89) |
| Cornell et al., 2017 [25] | Annals of Surgical Oncology | Prospective | USA | To evaluate the trends in sexual function in women with breast cancer from the time of diagnosis to designated follow-up surgery using a validated sexual questionnaire. | 226 BC (median: 56) |
| Cortés-Flores et al., 2017 [26] | Aesthetic Plastic Surgery | Cross-sectional | Mexico | To use the Female Sexual Function Index questionnaire to evaluate and compare the sexuality of women who underwent conservative mastectomy, mastectomy alone, and breast reconstruction after cancer treatment in private practice in Mexico. | 74 BC (mean CM: 46/mean MRM: 50.1/mean MRMR: 45.2) |
| Davis et al., 2010 [27] | The Nurse Practitioner | Cross-sectional | USA | To better understand the relationship between sexuality and quality of life in women who have undergone surgical treatment for breast cancer. | 72 BC (mean: 61.03) |
| Ellouz et al., 2019 [28] | Sexologies | Cross-sectional | Tunisia | To investigate the prevalence of sexual dysfunction in a population of women followed for breast cancer, and the factors associated with it. | 100 BC (mean: 42.6) |
| Elmas et al., 2020 [29] | Turkish Journal of Oncology | Prospective | Turkey | To compare the differences in the sexual function of breast cancer patients undergoing breast cancer surgery and modified radical mastectomy followed by chemotherapy and radiation therapy. | 71 BC (median: 43) |
| Farthmann et al., 2016 [30] | Supportive Care in Cancer | Prospective | Germany | To evaluate the influence of chemotherapy for breast cancer on women’s health-related quality of life, sexual function, and depression. | 79 BC (mean: 47.46) |
| Fogh et al., 2021 [31] | Acta Oncologica | Cross-sectional | Denmark | To explore the prevalence of clinically relevant sexual dysfunction among breast cancer survivors on adjuvant endocrine therapy, determine the associated factors of sexual dysfunction, explore the extent of the distress caused by specific impairments in sexual function, and analyse whether these were perceived as consequences of breast cancer treatment by breast cancer survivors. | 333 BC (mean: 58.74) |
| Fouladi et al., 2021 [32] | Asian Pacific Journal of Cancer Prevention | Cross-sectional | Iran | Not stated | 144 BC (mean: 31.4) |
| Frechette et al., 2013 [33] | Breast Cancer Research and Treatment | Prospective | Canada | To document changes in the gynaecological symptoms, sexual problems, and sexual dysfunction (as per DMS-IV criteria) among postmenopausal women with early-stage breast cancer over a 6-month period following the initiation of endocrine therapy, and to identify the predictors of sexual dysfunction 6 months after endocrine treatment. | 66 BC (mean: 61) |
| Gambadrella et al., 2018 [34] | Endocrine | Cross-sectional | Italy | To evaluate the impact of different treatment strategies and steroid hormone levels on sexual function in 122 breast cancer women. | 122 BC (mean: 46.9) |
| Gandhi et al., 2019 [35] | American Journal of Clinical Oncology | Cross-sectional | USA | To assess the associations of the breast cancer surgical modality and adjuvant therapy on women’s sexual dysfunction in survivorship. | 278 BC |
| Harirchi et al., 2012 [36] | Journal of Experimental & Clinical Cancer Research | Prospective | Iran | To elucidate the issue and contribute to the existing knowledge on the topic, and to provide necessary information for implementing possible future interventions to improve the quality of life of breast cancer patients. | 216 BC (mean: 44.3) |
| Herbenick et al., 2008 [37] | Cancer Nursing | Cross-sectional | USA | To use a reliable and valid measure to examine the sexual function of women younger than 50 years at the time of their breast cancer diagnosis, and to explore their interest in sexual enhancement products. | 115 BC (median: 37.8) |
| İzci et al., 2020 [38] | European Journal of Breast Health | Prospective | Turkey | To examine the pretreatment and post-treatment anxiety, depression, and sleep and sexual function levels in patients with breast cancer. | 56 BC/52 CG (mean BC: 53/mean CG: 52.5) |
| Kedde et al., 2013 [39] | Supportive Care in Cancer | Case–control | Netherlands | To determine the prevalence of sexual dysfunction in young women with breast cancer, and to assess the relationship between the treatment administered for breast cancer and sexual dysfunction. | 332 BC/1430 CG (mean BC: 38.7/CG range: 22–49) |
| Kowalczyk et al., 2019 [40] | Clinical Breast Cancer | Retrospective | Poland | To evaluate the correlates and impact factors of the sexual function, prevalence of sexual dysfunction, quality of sexual life, and body image of female breast cancer survivors. | 128 BC (median: 52.5) |
| Landi et al., 2016 [41] | Cancer Causes & Control | Cross-sectional | USA | To examine the association between the use of endocrine therapy and incident urinary incontinence and sexual dysfunction. | 548 BC (mean: 58.1) |
| Lashani et al., 2020 [42] | Supportive Care in Cancer | Cross-sectional | Iran | To explore the types and roles of the relationships between the sexual function, sense of coherence, and wellbeing in a sample of Iranian female breast cancer survivors. | 181 BC (mean: 47.04) |
| Lee et al., 2015 [43] | Psycho-Oncology | Cross-sectional | South Korea | To examine the changes in the sexual activity and function of younger breast cancer survivors who were sexually active before diagnosis, and to explore the risk factors that have negative impacts on them. | 304 BC (median: 46) |
| Ljungman et al., 2018 [6] | Psycho-Oncology | Prospective | Sweden | To investigate the sexual dysfunction and reproductive concerns in women under the age of 40 years at breast cancer diagnosis, and to identify predictors of high levels of problems and potential interdependence between sexual dysfunction and reproductive concerns. | 181 BC (mean: 36.5) |
| Manganiello et al., 2011 [44] | European Journal of Oncology Nursing | Cross-sectional | Brazil | To evaluate the sexual functioning of postmasectomy breast cancer patients and its associations with their quality of life, the personal characteristics of women and their partners, breast reconstruction, cancer staging, and adjuvant therapies. | 100 BC (-) |
| Mayer et al., 2019 [45] | Archives of Gynecology and Obstetrics | Retrospective | Germany | To analyse the sexual activity, sexual functioning, and quality of life in patients after the completion of treatment for breast cancer and ovarian cancer. | 183 BC/62 OC/60 HG (median BC: 56/median OC: 53/median CG: 46) |
| Metcalfe et al., 2012 [46] | Annals of Surgical Oncology | Prospective | Canada | To report on the changes in the psychosocial functioning over 1 year following breast cancer surgery in three groups of women, including those with mastectomy alone, those with mastectomy and immediate reconstruction, and those with delayed reconstruction. | 190 BC (mean MA: 53.5/mean MIR: 46.2/mean DR: 51.6) |
| Notari et al., 2018 [47] | European Journal of Cancer Care | Cross-sectional | Switzerland | To fill in a gap in the present literature by describing women’s sexual functioning in the early weeks of active treatment for breast cancer. | 75 BC (mean: 52.55) |
| Oberguggenberger et al., 2017 [48] | BMC Cancer | Cross-sectional | Germany | To investigate the self-reported sexual health outcomes of breast cancer survivors in routine after-care in comparison with women with no previous or current breast cancer. | 105 BC/97 NBC (mean BC: 49/mean NBC: 49) |
| Öztürk et al., 2016 [49] | Japan Journal of Nursing Science | Cross-sectional | Turkey | To assess the sexual function of Turkish women undergoing a surgical procedure, and to determine whether there were differences between Turkish women undergoing postmastectomy breast reconstruction and those undergoing breast-conserving surgery or mastectomy alone. | 100 BC (mean: 47) |
| Paiva et al., 2016 [50] | Archives of Sexual Behavior | Cross-sectional | Brazil | To investigate the prevalence of the sexual dysfunction and identify the associated conditions in a patient population of Brazilian breast cancer survivors, focusing on obesity-related conditions. | 216 BC (mean: 51.9) |
| Park et al., 2013 [51] | Supportive Care in Cancer | Cross-sectional | South Korea | To study the relationships among menopausal symptoms, sexual function, depression, and quality of life in women with breast cancer undergoing chemotherapy. | 200 BC (mean: 45.64) |
| Parker et al., 2007 [52] | Annals of Surgical Oncology | Prospective | USA | To prospectively examine the short- and long-term effects of mastectomy with reconstruction, mastectomy without reconstruction, and breast-conserving therapy on aspects of psychosocial adjustment and quality of life in a sample of 258 women with breast cancer. | 258 BC (mean MWR: 49.2/mean MA: 52.8/mean BCT: 53.7) |
| Qureshi et al., 2018 [53] | Aesthetic Surgery Journal | Cross-sectional | USA | To assess the prevalence of sexual health issues in a plastic surgery patient population including breast cancer survivors and women without breast cancer. | 90 BC/149 NBC (mean BC: 51.2/mean NBC: 47) |
| Raggio et al., 2014 [54] | Psychology & Health | Cross-sectional | USA | To assess four self-reported sexual morbidity domains, including sexual function, sexual distress, body change stress, and body satisfaction, in a sample of long-term breast cancer survivors, and to evaluate the influence of select psychosocial and medical factors based on the extant literature, including age, treatment modality (e.g., mastectomy, specific treatment effects (e.g., weight gain and premature menopause), and psychosocial factors (e.g., depression, marital/relationship status and satisfaction, and quality of life)), within and across four sexual morbidity domains. | 83 BC (mean: 56.21) |
| Reese et al., 2020 [55] | The Journal of Sexual Medicine | Cross-sectional | USA | To determine, in a sample of BC outpatients, how commonly women sought help for sexual concerns from a healthcare provider, from other individuals, or from alternate sources, and to examine whether the help-seeking was associated with women’s sexual function/activity, self-efficacy for clinical communication about sexual health, or sociodemographic/medical characteristics. | 144 BC (mean: 56) |
| Robinson et al., 2017 [56] | The Journal of Sexual Medicine | Cross-sectional | Australia | To document the prevalence of and factors associated with low desire, sexually related personal distress, hypoactive sexual desire dysfunction, and pelvic-floor dysfunction in women 10 years after breast cancer diagnosis. | 625 BC (median: 65.1) |
| Rojas et al., 2017 [57] | Breast Cancer Research and Treatment | Cross-sectional | USA | To explore the impact of mastectomy type on sexual function, as measured by the Female Sexual Function Index, satisfaction with appearance, and the reconstructed breast’s role in intimacy. | 60 BC (median TMRM: 52/median SSM: 50.57 median NSM: 46.5) |
| Rosenberg et al., 2020 [58] | Jama Surgery | Prospective | USA | To describe the changes in these outcomes from 1 to 5 years following diagnosis comparing bilateral mastectomy vs. breast-conserving surgery and unilateral mastectomy, as well as to examine the differences by primary surgery type, receipt of radiation, and reconstruction. | 826 BC (mean BCS: 35.9/mean UM: 36.4/mean BM: 36.1) |
| Rottmann et al., 2017 [59] | Acta Oncologica | Longitudinal | Denmark | To examine whether individual and partner sexual functioning, affectionate behaviour, emotional closeness, and depressive symptoms are associated with change over time in the satisfaction with the sex lives of sexually active heterosexual couples dealing with BC, and to explore whether the associations differ between patients and partners. | 287 BC (mean: 55.77) |
| Rowland et al., 2000 [60] | Journal of the National Cancer Institute | Cross-sectional | USA | To examine the characteristics of women undergoing lumpectomy, mastectomy with reconstruction, and mastectomy alone, and the relationship of the different surgical treatments to specific aspects of the health-related quality of life, body image, and physical and sexual functioning. | 1957 B (mean MWR: 50.3/mean LG: 55.9/mean MA: 58.9) |
| Safarinejad et al., 2013 [61] | Psycho-oncology | Cross-sectional | Iran | To compare sexual function, self-esteem, and quality of life in young women with breast cancer by lumpectomy with those from the age-matched general female population. | 186 BC/204 CG (mean BC: 37.7/mean CG: 37.2) |
| Sayakhot et al., 2011 [62] | Menopause | Cross-sectional | Australia | To investigate menopausal symptoms, psychological symptoms (including anxiety and depression), sexual function, and body image experienced by younger Australian women with breast cancer, and to examine the effect of different breast cancer treatments (chemotherapy, adjuvant endocrine therapy, and ovariectomy) on the menopausal, physical, and psychological symptoms. | 114 BC (mean: 47.2) |
| Sbitti et al., 2011 [63] | BMC Women’s Health | Cross-sectional | Morocco | To prospectively evaluate the body image scale and impact of breast cancer therapy on the sexuality of women from Morocco. | 120 BC (mean: 45.3) |
| Schover et al., 2014 [64] | The Journal of Sexual Medicine | Cross-sectional | USA | To provide a more detailed picture of the sexual problems during the first two years on aromatase inhibitor therapy, to provide a benchmark for a prospective intervention trial. | 129 BC (mean: 63.4) |
| Shandiz et al., 2016 [65] | Asian Pacific Journal of Cancer Prevention | Cross-sectional | Iran | To investigate sexual function and factors affecting it in women with breast cancer. | 94 BC (mean: 45.20) |
| Soldera et al., 2018 [66] | Breast Cancer Research and Treatment | Prospective | Canada | To evaluate sexual health in long-term breast cancer survivors compared with aged-matched controls, determine the impact of chemotherapy and endocrine therapy on the sexual functioning, and compare the related symptoms, such as the gynaecological, vasomotor, and bladder complaints, between groups to potentially explain the source of any differences in sexual function. | 248 BC/159 CG (mean BC: 62/mean CG: 59) |
| Sorouri et al., 2019 [67] | Archives of Psychiatry Research | Cross-sectional | Iran | To compare negative emotions, body image, sexual schemas, and sexual function in women with breast cancer after mastectomy and healthy women. | 105 BC/100 CG (mean BC: 41.09/mean CG: 41.5) |
| Speer et al., 2005 [68] | The Breast Journal | Case–control | USA | To investigate how testosterone levels, mood, body image, depression, relationship quality, and age influence the sexual function of female BCSs who have been treated with surgery, radiation, chemotherapy, or adjunctive hormone therapy. | 55 BC (mean: 53.4) |
| Tahir et al., 2020 [69] | Psycho-Oncology | Cross-sectional | Pakistan | To explore the mediating role of body image (dissatisfaction) between sexual functioning (SF) and marital intimacy in Pakistani women with breast cancer. | 118 BC (mean: 39.58) |
| Tucker et al., 2020 [70] | Supportive Care in Cancer | Cross-sectional | Australia | To compare the sexual function and quality of life in female breast cancer survivors with and without a history of bilateral salpingo-oophorectomy. | 172 BC (mean BSO: 53/mean no BSO: 58) |
| Tucker et al., 2016 [71] | The Breast | Cross-sectional | Australia | To investigate the prevalence of the sexual dysfunction in women with a prior history of breast cancer following risk-reducing salpingo-oophorectomy (RRSO), and to compare this to women without a previous diagnosis of breast cancer. The secondary objectives were to describe the effects of mastectomy, breast reconstruction, and antioestrogen therapy on the sexual function and quality-of-life outcomes of women with a previous diagnosis of breast cancer after RRSO. | 60 BC (mean: 50) |
| Usta et al., 2017 [72] | International Journal of Caring Sciences | Cross-sectional | Turkey | To determine the frequency of sexual dysfunction and factors affecting sexual dysfunction in women with breast cancer receiving chemotherapy. | 118 BC (mean: 47.74) |
| Vaidakis et al., 2014 [73] | European Journal of Gynaecological Oncology | Prospective | Greece | To record how the treatment of breast and gynaecological cancer may affect the subjective perception of the interpersonal relation of the couple with respect to female sexuality. The authors also attempted to associate and evaluate disorders of desire, arousal, orgasm, and pain, as well as problems of the sexual relationship, with cancer diagnosis and treatment. | 67 BC (mean: 50.5) |
| Webber et al., 2011 [74] | The Oncologist | Prospective | Australia | To examine the incidence of sexual problems after breast cancer in a real-world setting, the predictors of sexual problems over 12 months after adjuvant therapy, and the potential impact of sexual problems on the quality of life. | 92 BC (mean: 49.8) |
| Yuan et al., 2020 [75] | Supportive Care in Cancer | Cross-sectional | China | To identify the unobserved distinct latent classes/subgroups of breast cancer patients in China with respect to various sexual health measures, and to examine the association of the latent membership with the individual characteristics. | 123 BC (mean: 42.80) |
| Zaied et al., 2013 [76] | Bulletin du Cancer | Cross-sectional | Tunisia | To evaluate the frequency and type of sexual dysfunction in 100 patients treated for nonmetastatic breast cancer in post-treatment monitoring in external consultation (Department of Oncological Medicine, CHU Farhat Hached, Sousse), and to identify the predictive factors of these disorders. | 100 BC (mean: 47.24) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues-Machado, N.; Quintana, M.J.; Gómez-Gómez, R.; Bonfill-Cosp, X. Sexual Function in Women with Breast Cancer: An Evidence Map of Observational Studies. Int. J. Environ. Res. Public Health 2022, 19, 13976. https://doi.org/10.3390/ijerph192113976
Rodrigues-Machado N, Quintana MJ, Gómez-Gómez R, Bonfill-Cosp X. Sexual Function in Women with Breast Cancer: An Evidence Map of Observational Studies. International Journal of Environmental Research and Public Health. 2022; 19(21):13976. https://doi.org/10.3390/ijerph192113976
Chicago/Turabian StyleRodrigues-Machado, Nelson, M. Jesús Quintana, Raquel Gómez-Gómez, and Xavier Bonfill-Cosp. 2022. "Sexual Function in Women with Breast Cancer: An Evidence Map of Observational Studies" International Journal of Environmental Research and Public Health 19, no. 21: 13976. https://doi.org/10.3390/ijerph192113976
APA StyleRodrigues-Machado, N., Quintana, M. J., Gómez-Gómez, R., & Bonfill-Cosp, X. (2022). Sexual Function in Women with Breast Cancer: An Evidence Map of Observational Studies. International Journal of Environmental Research and Public Health, 19(21), 13976. https://doi.org/10.3390/ijerph192113976






