Impact of Exposome Factors on Epidermal Barrier Function in Patients with Obstructive Sleep Apnea Syndrome
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design
2.2. Study Population
- –
- Patients with OSAS were patients aged between 18 and 65 years, newly diagnosed with OSAS according to American Academy of Sleep Medicine [21], without previous treatment of this disease.
- –
- Healthy subjects were people matched by age (+/− 3 years) and sex with OSAS patients that did not have any history of sleep disturbances or inflammatory skin disease.
2.3. Study Variables
2.4. Statistical Analysis
2.5. Ethics
3. Results
3.1. Subject Characteristics
3.2. Skin Barrier Function between Patients with OSAS and Healthy Individual
3.3. The Impact of Exposome Factors on Skin Homeostasis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Larcher, F.; Espada, J.; Diaz-Ley, B.; Jaen, P.; Juarranz, A.; Quintanilla, M. New experimental models of skin homeostasis and diseases. Actas Dermosifiliogr 2015, 106, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Rueda, M.I.; Montero-Vilchez, T.; Martinez-Lopez, A.; Molina-Leyva, A.; Sierra-Sanchez, A.; Arias-Santiago, S.; Buendia-Eisman, A. Cutaneous homeostasis and epidermal barrier function in a young healthy Caucasian population. Eur. J. Dermatol. 2021, 31, 176–182. [Google Scholar] [CrossRef]
- Clark, R.A.; Ghosh, K.; Tonnesen, M.G. Tissue engineering for cutaneous wounds. J. Investig. Dermatol. 2007, 127, 1018–1029. [Google Scholar] [CrossRef] [Green Version]
- Montero-Vilchez, T.; Martinez-Lopez, A.; Cuenca-Barrales, C.; Rodriguez-Tejero, A.; Molina-Leyva, A.; Arias-Santiago, S. Impact of Gloves and Mask Use on Epidermal Barrier Function in Health Care Workers. Dermatitis 2021, 32, 57–62. [Google Scholar] [CrossRef]
- Akdeniz, M.; Gabriel, S.; Lichterfeld-Kottner, A.; Blume-Peytavi, U.; Kottner, J. Transepidermal water loss in healthy adults: A systematic review and meta-analysis update. Br. J. Dermatol 2018, 179, 1049–1055. [Google Scholar] [CrossRef] [PubMed]
- Montero-Vilchez, T.; Segura-Fernandez-Nogueras, M.V.; Perez-Rodriguez, I.; Soler-Gongora, M.; Martinez-Lopez, A.; Fernandez-Gonzalez, A.; Molina-Leyva, A.; Arias-Santiago, S. Skin Barrier Function in Psoriasis and Atopic Dermatitis: Transepidermal Water Loss and Temperature as Useful Tools to Assess Disease Severity. J. Clin. Med. 2021, 10, 359. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J. Dermatol Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef] [Green Version]
- Buendia-Eisman, A.; Prieto, L.; Abarquero, M.; Arias-Santiago, S. Study of the Exposome Ageing-related Factors in the Spanish Population. Acta Derm. Venereol. 2020, 100, adv00153. [Google Scholar] [CrossRef] [PubMed]
- Ekiz, O.; Yuce, G.; Ulasli, S.S.; Ekiz, F.; Yuce, S.; Basar, O. Factors influencing skin ageing in a Mediterranean population from Turkey. Clin. Exp. Dermatol. 2012, 37, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Oyetakin-White, P.; Suggs, A.; Koo, B.; Matsui, M.S.; Yarosh, D.; Cooper, K.D.; Baron, E.D. Does poor sleep quality affect skin ageing? Clin. Exp. Dermatol. 2015, 40, 17–22. [Google Scholar] [CrossRef]
- Passeron, T.; Zouboulis, C.C.; Tan, J.; Andersen, M.L.; Katta, R.; Lyu, X.; Aguilar, L.; Kerob, D.; Morita, A.; Krutmann, J.; et al. Adult skin acute stress responses to short-term environmental and internal aggression from exposome factors. J. Eur. Acad. Dermatol. Venereol. 2021, 35, 1963–1975. [Google Scholar] [CrossRef]
- Jang, S.I.; Han, J.; Lee, M.; Seo, J.; Kim, B.J.; Kim, E. A study of skin characteristics according to humidity during sleep. Ski. Res. Technol. 2019, 25, 456–460. [Google Scholar] [CrossRef]
- Patel, S.R. Obstructive Sleep Apnea. Ann. Intern. Med. 2019, 171, ITC81–ITC96. [Google Scholar] [CrossRef]
- Gottlieb, D.J.; Punjabi, N.M. Diagnosis and Management of Obstructive Sleep Apnea: A Review. JAMA 2020, 323, 1389–1400. [Google Scholar] [CrossRef] [PubMed]
- Clinton, S.K.; Giovannucci, E.L.; Hursting, S.D. The World Cancer Research Fund/American Institute for Cancer Research Third Expert Report on Diet, Nutrition, Physical Activity, and Cancer: Impact and Future Directions. J. Nutr. 2020, 150, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, J.I.; Lee-Wong, M.; Silverberg, N.B. Complementary and alternative medicines and childhood eczema: A US population-based study. Dermatitis 2014, 25, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Molina-Leyva, A.; Cuenca-Barrales, C.; Vega-Castillo, J.J.; Ruiz-Carrascosa, J.C.; Ruiz-Villaverde, R. Adherence to Mediterranean diet in Spanish patients with psoriasis: Cardiovascular benefits? Dermatol. Ther. 2019, 32, e12810. [Google Scholar] [CrossRef]
- Almohanna, H.M.; Ahmed, A.A.; Tsatalis, J.P.; Tosti, A. The Role of Vitamins and Minerals in Hair Loss: A Review. Dermatol. Ther. (Heidelb) 2019, 9, 51–70. [Google Scholar] [CrossRef] [Green Version]
- Cao, C.; Xiao, Z.; Wu, Y.; Ge, C. Diet and Skin Aging-From the Perspective of Food Nutrition. Nutrients 2020, 12. [Google Scholar] [CrossRef] [Green Version]
- Passeron, T.; Krutmann, J.; Andersen, M.L.; Katta, R.; Zouboulis, C.C. Clinical and biological impact of the exposome on the skin. J. Eur. Acad. Dermatol. Venereol. 2020, 34 (Suppl. S4), 4–25. [Google Scholar] [CrossRef]
- Kapur, V.K.; Auckley, D.H.; Chowdhuri, S.; Kuhlmann, D.C.; Mehra, R.; Ramar, K.; Harrod, C.G. Clinical Practice Guideline for Diagnostic Testing for Adult Obstructive Sleep Apnea: An American Academy of Sleep Medicine Clinical Practice Guideline. J. Clin. Sleep Med. 2017, 13, 479–504. [Google Scholar] [CrossRef]
- Delgado-Vargas, B.; Acle-Cervera, L.; Narciso Lopez, G. Validation of the Spanish Version of the STOP-Bang Questionnaire: Usefulness as a Screening Tool for Obstructive Sleep Apnea in Adults. Ear Nose Throat J. 2020, 145561320932334. [Google Scholar] [CrossRef] [PubMed]
- Nagappa, M.; Liao, P.; Wong, J.; Auckley, D.; Ramachandran, S.K.; Memtsoudis, S.; Mokhlesi, B.; Chung, F. Validation of the STOP-Bang Questionnaire as a Screening Tool for Obstructive Sleep Apnea among Different Populations: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0143697. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Sharma, V.K. Skin typing: Fitzpatrick grading and others. Clin. Dermatol. 2019, 37, 430–436. [Google Scholar] [CrossRef]
- Hita-Contreras, F.; Martinez-Lopez, E.; Latorre-Roman, P.A.; Garrido, F.; Santos, M.A.; Martinez-Amat, A. Reliability and validity of the Spanish version of the Pittsburgh Sleep Quality Index (PSQI) in patients with fibromyalgia. Rheumatol. Int. 2014, 34, 929–936. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A.; Garcia-Arellano, A.; Toledo, E.; Salas-Salvado, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.I.; Schroder, H.; Aros, F.; Gomez-Gracia, E.; et al. A 14-item Mediterranean diet assessment tool and obesity indexes among high-risk subjects: The PREDIMED trial. PLoS ONE 2012, 7, e43134. [Google Scholar] [CrossRef] [Green Version]
- Herrero, M.J.; Blanch, J.; Peri, J.M.; De Pablo, J.; Pintor, L.; Bulbena, A. A validation study of the hospital anxiety and depression scale (HADS) in a Spanish population. Gen. Hosp. Psychiatry 2003, 25, 277–283. [Google Scholar] [CrossRef]
- Remor, E. Psychometric properties of a European Spanish version of the Perceived Stress Scale (PSS). Span. J. Psychol. 2006, 9, 86–93. [Google Scholar] [CrossRef] [Green Version]
- Yoshizaki, T.; Kimira, Y.; Mano, H.; Ota, M.; Iwatsuki, K.; Oishi, Y.; Yamane, T. Association between Skin Condition and Sleep Efficiency in Japanese Young Adults. J. Nutr. Sci. Vitaminol. 2017, 63, 15–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, M.; Kim, E.; Kang, B.; Lee, H. The Effects of Sleep Deprivation on the Biophysical Properties of Facial Skin. J. Cosmet. Dermatol. Sci. Appl. 2017, 7, 34–47. [Google Scholar] [CrossRef] [Green Version]
- Jang, S.I.; Lee, M.; Han, J.; Kim, J.; Kim, A.R.; An, J.S.; Park, J.O.; Kim, B.J.; Kim, E. A study of skin characteristics with long-term sleep restriction in Korean women in their 40s. Skin Res. Technol. 2020, 26, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.I.; Jung, Y.; Lee, M.; Kim, J.; Kim, B.J.; Suh, B.F.; Kim, E. Evaluation of changes in skin characteristics due to the poor quality of sleep caused by smartphone usage. J. Cosmet. Dermatol. 2021. [Google Scholar] [CrossRef]
- Kahan, V.; Andersen, M.L.; Tomimori, J.; Tufik, S. Stress, immunity and skin collagen integrity: Evidence from animal models and clinical conditions. Brain Behav. Immun. 2009, 23, 1089–1095. [Google Scholar] [CrossRef]
- Kahan, V.; Andersen, M.L.; Tomimori, J.; Tufik, S. Can poor sleep affect skin integrity? Med. Hypotheses 2010, 75, 535–537. [Google Scholar] [CrossRef]
- Te Lindert, B.H.W.; Van Someren, E.J.W. Skin temperature, sleep, and vigilance. Handb. Clin. Neurol. 2018, 156, 353–365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angelhoff, C.; Blomqvist, Y.T.; Sahlen Helmer, C.; Olsson, E.; Shorey, S.; Frostell, A.; Morelius, E. Effect of skin-to-skin contact on parents’ sleep quality, mood, parent-infant interaction and cortisol concentrations in neonatal care units: Study protocol of a randomised controlled trial. BMJ Open 2018, 8, e021606. [Google Scholar] [CrossRef]
- Purba, M.B.; Kouris-Blazos, A.; Wattanapenpaiboon, N.; Lukito, W.; Rothenberg, E.M.; Steen, B.C.; Wahlqvist, M.L. Skin wrinkling: Can food make a difference? J. Am. Coll. Nutr. 2001, 20, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Latreille, J.; Kesse-Guyot, E.; Malvy, D.; Andreeva, V.; Galan, P.; Tschachler, E.; Hercberg, S.; Guinot, C.; Ezzedine, K. Association between dietary intake of n-3 polyunsaturated fatty acids and severity of skin photoaging in a middle-aged Caucasian population. J. Dermatol. Sci. 2013, 72, 233–239. [Google Scholar] [CrossRef]
- Boelsma, E.; van de Vijver, L.P.; Goldbohm, R.A.; Klopping-Ketelaars, I.A.; Hendriks, H.F.; Roza, L. Human skin condition and its associations with nutrient concentrations in serum and diet. Am. J. Clin. Nutr. 2003, 77, 348–355. [Google Scholar] [CrossRef] [Green Version]
- Maarouf, M.; Maarouf, C.L.; Yosipovitch, G.; Shi, V.Y. The impact of stress on epidermal barrier function: An evidence-based review. Br. J. Dermatol. 2019, 181, 1129–1137. [Google Scholar] [CrossRef]
- Etindele Sosso, F.A.; Holmes, S.D.; Weinstein, A.A. Influence of socioeconomic status on objective sleep measurement: A systematic review and meta-analysis of actigraphy studies. Sleep Health 2021, 7, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Etindele-Sosso, F.A. Insomnia, excessive daytime sleepiness, anxiety, depression and socioeconomic status among customer service employees in Canada. Sleep Sci. 2020, 13, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Rexbye, H.; Petersen, I.; Johansens, M.; Klitkou, L.; Jeune, B.; Christensen, K. Influence of environmental factors on facial ageing. Age Ageing 2006, 35, 110–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choe, S.J.; Kim, D.; Kim, E.J.; Ahn, J.S.; Choi, E.J.; Son, E.D.; Lee, T.R.; Choi, E.H. Psychological Stress Deteriorates Skin Barrier Function by Activating 11beta-Hydroxysteroid Dehydrogenase 1 and the HPA Axis. Sci. Rep. 2018, 8, 6334. [Google Scholar] [CrossRef]

| KERRYPNX | Study Population (n = 86) | Healthy Participants (n = 30) | Patients with OSAS (n = 56) | p |
|---|---|---|---|---|
| Age (years) | 48.87 (SD 11.63) | 47.37 (SD 11.08) | 49.68 (SD 11.93) | 0.383 |
| Sex, n (%) | 0.576 | |||
| Male | 55 (64%) | 18 (60%) | 37 (66.1%) | |
| Female | 31 (36%) | 12 (40%) | 19 (33.9%) | |
| Residential environment, n (%) | 0.011 * | |||
| Urban | 47(54.7%) | 22 (73.3%) | 25 (44.6%) | |
| Rural | 39 (45.3%) | 8 (26.7%) | 31 (55.4%) | |
| Current occupation, n (%) | 0.111 | |||
| Employed | 66 (76.7%) | 26 (86.7%) | 40 (71.4%) | |
| Unemployed | 20 (23.3%) | 4 (13.3%) | 16 (28.6%) | |
| Phototype, n (%) | 0.578 | |||
| I | ||||
| II | 15 (17.4%) | 7 (23.3%) | 8 (14.3%) | |
| III | 58 (67.4%) | 20 (66.7%) | 38 (67.9%) | |
| IV | 12 (14.0%) | 3 (10.0%) | 9 (16.1%) | |
| V | ||||
| VI | 1 (1.2%) | 0 (0%) | 1 (1.8%) | |
| Marital status, n (%) | 0.562 | |||
| Single | 21 (24.4%) | 9 (30.0%) | 12 (21.4%) | |
| Married | 54 (62.8%) | 16 (53.3%) | 38 (67.9%) | |
| Divorced | 8 (9.3%) | 4 (13.3%) | 4 (7.1%) | |
| Widowed | 3 (3.5%) | 1 (3.3%) | 2 (3.6%) | |
| Educational Level, n (%) | 0.061 | |||
| None | 2 (2.3%) | 0 (0%) | 2 (3.6%) | |
| Primary or Equivalent | 13 (15.1%) | 1 (3.3%) | 12 (21.4%) | |
| Secondary or Equivalent | 21 (24.4%) | 6 (20.0%) | 15 (26.8%) | |
| High school/Vocational training | 31 (36.0%) | 13 (43.3%) | 18 (32.1%) | |
| University or higher | 19 (22.1%) | 10 (33.3%) | 9 (16.1%) | |
| Smokers | 0.598 | |||
| n (%) | 26 (30.2%) | 8 (26.7%) | 18 (32,1%) | |
| Mean cigarette per day | 4.44 (SD 8.21) | 3.23 (SD 6.59) | 5.09 (SD 8.95) | 0.278 |
| Drinkers, n (%) | 0.679 | |||
| n (%) | 37 (43.0%) | 12 (40.0%) | 25 (44.6%) | |
| Mean alcohol g/week | 29.01 (SD 53.59) | 27.16 (SD 58.67) | 30.0 (SD 51.20) | 0.817 |
| Solar exposure (h/week) | 21.53 (SD 16.02) | 17.03 (SD 10.59) | 23.94 (SD 17.91) | 0.027 * |
| Skincare | 0.199 0.486 | |||
| Moisturizing use | ||||
| -Yes, n (%) | 35 (40.7%) | 15 (50.0%) | 20 (35.7%) | |
| -Mean moisturizing use per week | 1.83 (SD 2.65) | 2.10 (SD 2.73) | 1.68 (SD 2.62) | |
| Sun lotion use, n (%) | <0.001 * | |||
| -Never | 39 (45.3%) | 5 (16.7%) | 34 (60.7%) | |
| -Sometimes | 33 (38.4%) | 14 (46.7%) | 19 (33.9%) | |
| -Always | 14 (16.3%) | 11 (36.7%) | 3 (5.4%) | |
| Weight (kg) | 87.42 (SD 20.04) | 71.67 (SD 12.93) | 95.86 (SD 18.02) | <0.001 * |
| Height (cm) | 170.44 (SD 8.90) | 169.83 (SD 9.09) | 170.77 (SD 8.86) | 0.645 |
| BMI (kg/m2) | 29.99 (SD 6.23) | 24.72 (SD 3.44) | 32.81 (SD 5.53) | <0.001 * |
| Healthy (n = 30) | OSAS Patients (n = 56) | p * | |
|---|---|---|---|
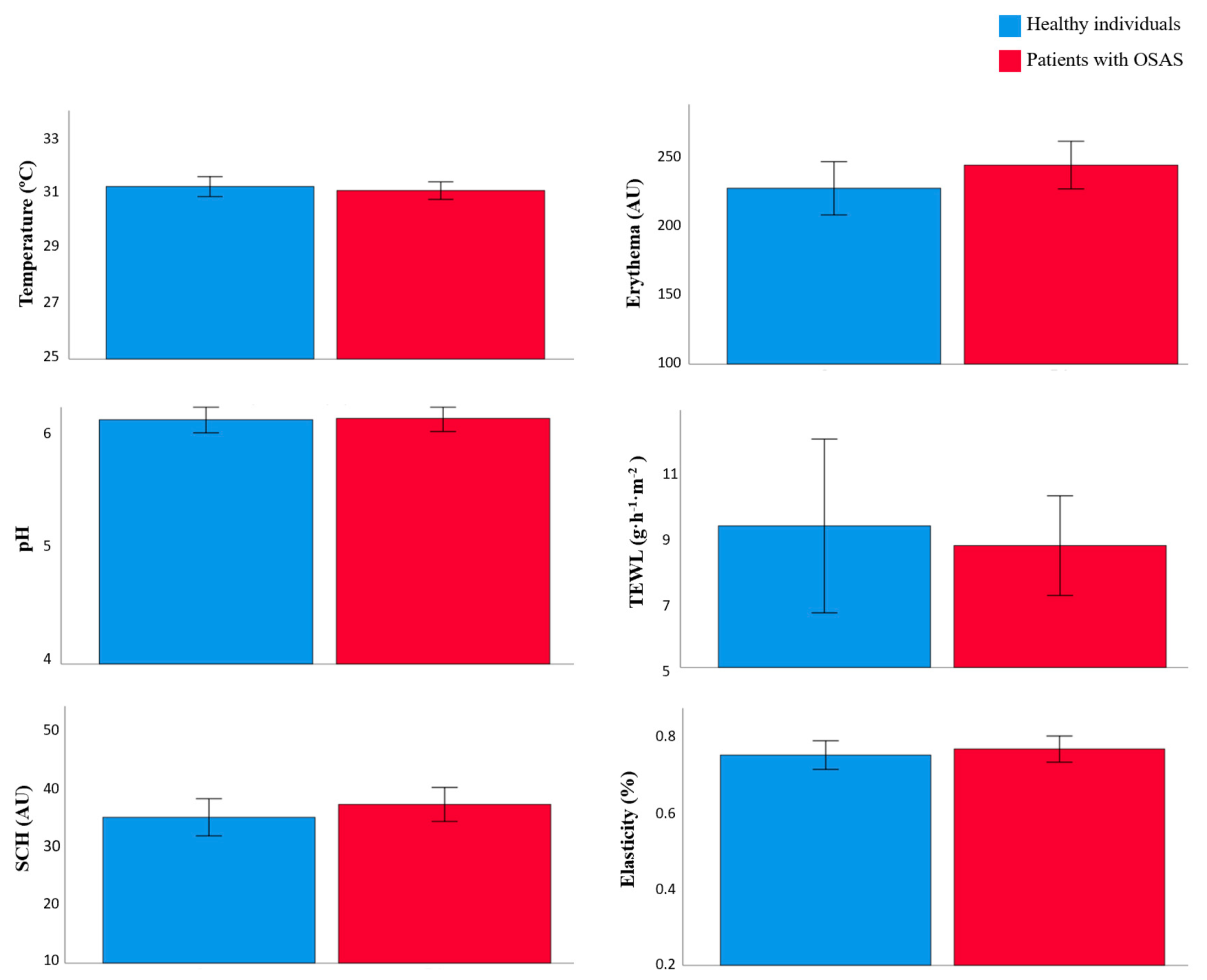
| Temperature (°C) | 31.17 (SD 0.96) | 30.99 (SD 1.16) | 0.569 |
| Erythema (AU) | 228.57 (SD 52.32) | 244.34 (SD 64.53) | 0.221 |
| pH | 6.11 (SD 0.31) | 6.12 (SD 0.42) | 0.893 |
| TEWL (g·m−2·h−1) | 8.01 (SD 1.54) | 8.68 (SD 5.63) | 0.414 |
| SCH (AU) | 35.19 (SD 8.59) | 37.42 (SD 10.96) | 0.303 |
| Elasticity | 0.74 (SD 0.09) | 0.76 (SD 0.12) | 0.532 |
| Mild OSAS (n = 8) | Moderate OSAS (n = 21) | Severe OSAS (n = 27) | p * | |
|---|---|---|---|---|
| Temperature (°C) | 30.64 (SD 2.02) | 31.09 (SD 1.09) | 31.04 (SD 0.94) | 0.621 |
| Erythema (AU) | 275.37 (SD 96.70) | 234.75 (SD 64.60) | 242.60 (SD 52.03) | 0.317 |
| pH | 6.02 (SD 0.47) | 6.09 (SD 0.44) | 6.18 (SD 0.40) | 0.617 |
| TEWL (g·m−2·h−1) | 7.08 (SD 2.50) | 8.46 (SD 4.64) | 9.31 (SD 6.89) | 0.607 |
| SCH (AU) | 35.89 (SD11.05) | 38.26 (SD 12.00) | 37.22 (SD 10.45) | 0.807 |
| Elasticity | 0.80 (SD 0.10) | 0.75 (SD 0.12) | 0.76 (SD 0.14) | 0.622 |
| Healthy (n = 30) | OSAS Patients (n = 56) | p | |
|---|---|---|---|
| PSQI | 6.47 (SD 3.92) | 11.89 (SD 4.32) | <0.001 * |
| HADS | 10.3 (SD 7.56) | 16.34 (SD 9.3) | 0.002 * |
| HADS-D | 3.83 (SD 3.62) | 7.07 (SD 4.87) | 0.001 * |
| HADS-A | 6.47 (SD 4.24) | 9.27 (SD 5.06) | 0.008 * |
| PSS | 20.7 (SD 9.94) | 25.29 (SD 10.88) | 0.058 |
| Adherence to Mediterranean Diet | 9.77 (SD 1.65) | 8.46 (SD 2.54) | 0.005 * |
| Healthy Individuals (n = 30) | OSAS Patients (n = 56) | |||||
|---|---|---|---|---|---|---|
| PSQI | Good Sleeper (<5) | Poor Sleepers (≥5) | p * | Good Sleeper (<5) | Poor Sleepers (≥5) | p ** |
| % (n/N) | 40% (12) | 60% (18) | 3.6% (2) | 96.4% (54) | ||
| TEWL (g·m−2·h−1) | 7.63 (SD 1.07) | 8.25 (SD 1.26) | 0.300 | 8.45 (SD 0.49) | 8.68 (SD 5.63) | 0.955 |
| HADS-A | Low anxiety (<8) | High anxiety (≥8) | p # | Low anxiety (<8) | High anxiety (≥8) | p ## |
| % (n/N) | 60% (18) | 40% (12) | 39.3% (22) | 60.7% (34) | ||
| TEWL (g·m−2·h−1) | 7.86 (SD 1.21) | 8.26 (SD 2.01 | 0.558 | 8.58 (6.44) | 8.74 (5.13) | 0.922 |
| HADS-D | Low depression (<8) | High depression (≥8) | p ¶ | Low depression (<8) | High depression (≥8) | p ¶¶ |
| % (n/N) | 90% (27) | 10% (3) | 62.5% (35) | 37.5% (21) | ||
| TEWL (g·m−2·h−1) | 7.91 (SD 1.30) | 9.30 (SD 4.38) | 0.228 | 8.41 (5.50) | 9.12 (6.94) | 0.648 |
| PSS | Low stress (<28) | High stress (≥28) | p ¥ | Low stress (<28) | High stress (≥28) | p ¥¥ |
| % (n/N) | 40% (12) | 60% (18) | 19.6% (11) | 80.4% (45) | ||
| TEWL (g·m−2·h−1) | 8.08 (SD 1.44) | 7.97 (SD 1.64) | 0.857 | 7.40 (3.10) | 8.99 (6.07) | 0.407 |
| Adherence to Mediterranean Diet | Low adherence (<9) | Good adherence (≥9) | p ʡ | Low adherence (<9) | Good adherence (≥9) | p ʡʡ |
| % (n) | 23.3% (7) | 76.7% (23) | 42.9% (24) | 57.1% (32) | ||
| TEWL (g·m−2·h−1) | 7.53 (SD 1.70) | 8.14 (SD 1.51) | 0.402 | 9.71 (SD 5.61) | 7.90 (SD 5.60) | 0.236 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romera-Vilchez, M.; Montero-Vilchez, T.; Herrero-Fernandez, M.; Rodriguez-Pozo, J.-A.; Jimenez-Galvez, G.; Morales-Garcia, C.; Buendia-Eisman, A.; Arias-Santiago, S. Impact of Exposome Factors on Epidermal Barrier Function in Patients with Obstructive Sleep Apnea Syndrome. Int. J. Environ. Res. Public Health 2022, 19, 659. https://doi.org/10.3390/ijerph19020659
Romera-Vilchez M, Montero-Vilchez T, Herrero-Fernandez M, Rodriguez-Pozo J-A, Jimenez-Galvez G, Morales-Garcia C, Buendia-Eisman A, Arias-Santiago S. Impact of Exposome Factors on Epidermal Barrier Function in Patients with Obstructive Sleep Apnea Syndrome. International Journal of Environmental Research and Public Health. 2022; 19(2):659. https://doi.org/10.3390/ijerph19020659
Chicago/Turabian StyleRomera-Vilchez, Maria, Trinidad Montero-Vilchez, Manuel Herrero-Fernandez, Juan-Angel Rodriguez-Pozo, Gonzalo Jimenez-Galvez, Concepcion Morales-Garcia, Agustin Buendia-Eisman, and Salvador Arias-Santiago. 2022. "Impact of Exposome Factors on Epidermal Barrier Function in Patients with Obstructive Sleep Apnea Syndrome" International Journal of Environmental Research and Public Health 19, no. 2: 659. https://doi.org/10.3390/ijerph19020659
APA StyleRomera-Vilchez, M., Montero-Vilchez, T., Herrero-Fernandez, M., Rodriguez-Pozo, J.-A., Jimenez-Galvez, G., Morales-Garcia, C., Buendia-Eisman, A., & Arias-Santiago, S. (2022). Impact of Exposome Factors on Epidermal Barrier Function in Patients with Obstructive Sleep Apnea Syndrome. International Journal of Environmental Research and Public Health, 19(2), 659. https://doi.org/10.3390/ijerph19020659








