Higher Content but Not Activity of Stromelysin-2 (MMP-10) in Comparison to Stromelysin-1 (MMP-3) in Human Renal Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Tissue Material
2.2. Specimen Preparation
2.3. DNA Content
2.4. Contents of Stromelysines
2.5. Western Blot of Stromelysines
2.6. Activity of Stromelysines
2.7. Protein Determination
2.8. Statistical Analysis
3. Results
3.1. DNA Content
3.2. Stromelysin-1 and Stromelysin-2 Contents
3.3. Expression
3.3.1. Expression of MMP-3
3.3.2. Expression of MMP-10
3.4. Activity of Stromelysin-1 and Stromelysin-2
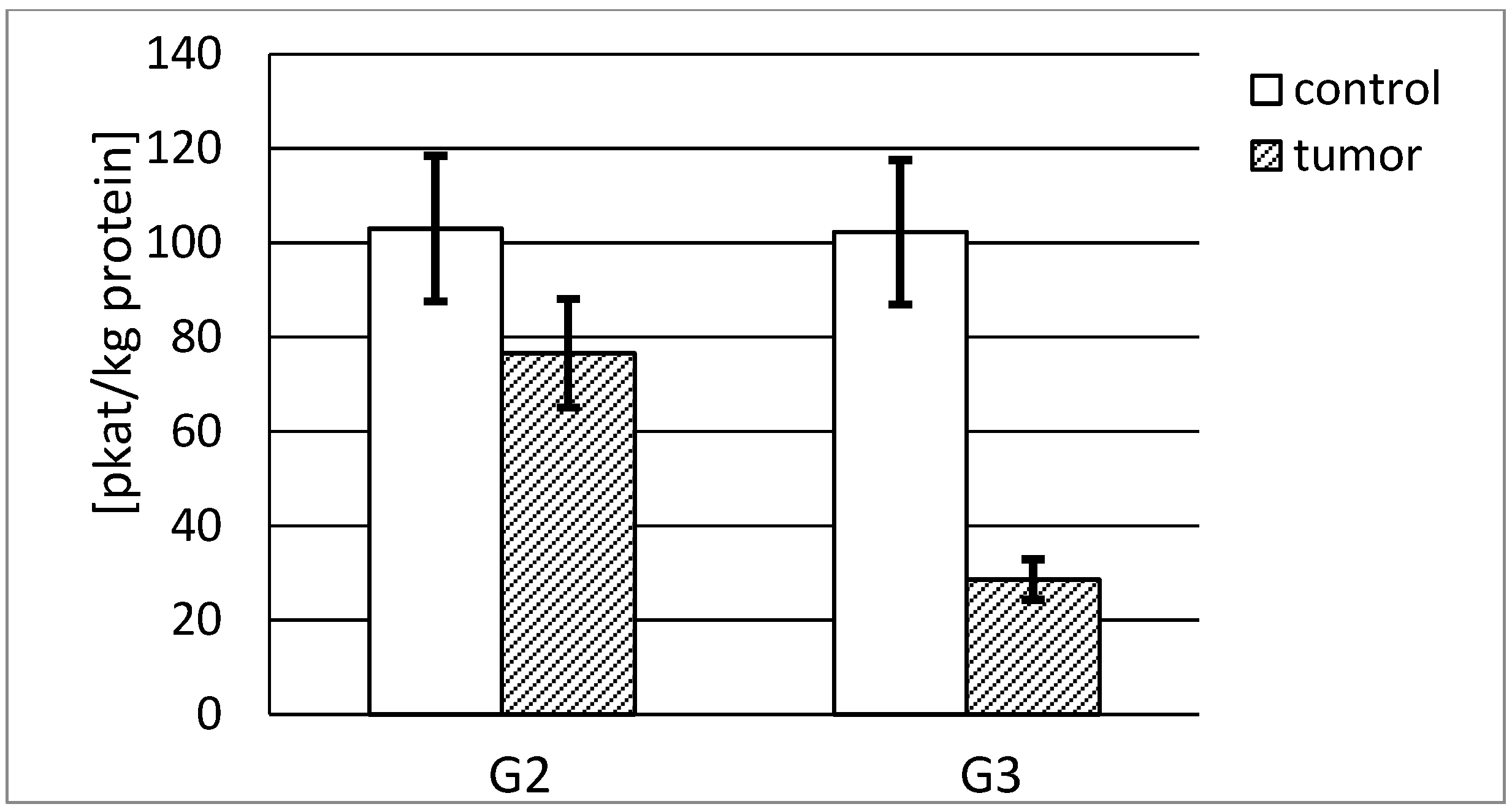
3.4.1. Actual and Specific Activity of Stromelysin-1
3.4.2. Actual and Specific Activity of Stromelysin-2
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mollica, V.; Santoni, M.; Matrana, M.R.; Basso, U.; De Giorgi, U.; Rizzo, A.; Maruzzo, M.; Marchetti, A.; Rosellini, M.; Bleve, S.; et al. Concomitant Proton Pump Inhibitors and Outcome of Patients Treated with Nivolumab Alone or Plus Ipilimumab for Advanced Renal Cell Carcinoma. Target Oncol. 2022, 17, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.; Mollica, V.; Dall’Olio, F.G.; Ricci, A.D.; Maggio, I.; Marchetti, A.; Rosellini, M.; Santoni, M.; Ardizzoni, A.; Massari, F. Quality of life assessment in renal cell carcinoma Phase II and III clinical trials published between 2010 and 2020: A systematic review. Future Oncol. 2021, 17, 2671–2681. [Google Scholar] [CrossRef] [PubMed]
- Bosman, F.T.; Stamenkovic, I. Functional structure and composition of the extracellular matrix. J. Pathol. 2003, 200, 423–428. [Google Scholar] [CrossRef]
- Boor, P.; Sebeková, K.; Ostendorf, T.; Floege, J. Treatment targets in renal fibrosis. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc.-Eur. Ren. Assoc. 2007, 22, 3391–3407. [Google Scholar] [CrossRef]
- Catania, J.M.; Chen, G.; Parrish, A.R. Role of matrix metalloproteinases in renal pathophysiologies. Am. J. Physiol. Ren. Physiol. 2007, 292, F905–F911. [Google Scholar] [CrossRef] [PubMed]
- Pardo, A.; Selman, M. Matrix metalloproteases in aberrant fibrotic tissue remodeling. Proc. Am. Thorac. Soc. 2006, 3, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Miner, J.H. Glomerular basement membrane and related glomerular disease. Transl. Res. J. Lab. Clin. Med. 2012, 160, 291–297. [Google Scholar] [CrossRef] [Green Version]
- Kudelski, J.; Młynarczyk, G.; Darewicz, B.; Bruczko-Goralewska, M.; Romanowicz, L. Dominative role of MMP-14 over MMP-15 in human urinary bladder carcinoma on the basis of its enhanced specific activity. Medicine 2020, 99, e19224. [Google Scholar] [CrossRef]
- Lochter, A.; Galosy, S.; Muschler, J.; Freedman, N.; Werb, Z.; Bissell, M.J. Matrix metalloproteinase stromelysin-1 triggers a cascade of molecular alterations that leads to stable epithelial-to-mesenchymal conversion and a premalignant phenotype in mammary epithelial cells. J. Cell Biol. 1997, 139, 1861–1872. [Google Scholar] [CrossRef]
- Kageyama, Y.; Nakamura, M.; Igari, Y.; Yamaguchi, S.; Oguchi, A.; Murakawa, Y.; Hattori, Y.; Sasano, Y. Expression of matrix metalloproteinase-3 and -10 is up-regulated in the periodontal tissues of aged mice. J. Periodontal Res. 2022, 57, 733–741. [Google Scholar] [CrossRef]
- Wan, J.; Zhang, G.; Li, X.; Qiu, X.; Ouyang, J.; Dai, J.; Min, S. Matrix Metalloproteinase 3: A Promoting and Destabilizing Factor in the Pathogenesis of Disease and Cell Differentiation. Front. Physiol. 2021, 12, 663978. [Google Scholar] [CrossRef] [PubMed]
- Kudelski, J.; Młynarczyk, G.; Gudowska-Sawczuk, M.; Mroczko, B.; Darewicz, B.; Bruczko-Goralewska, M.; Sobolewski, K.; Romanowicz, L. Enhanced Expression but Decreased Specific Activity of Matrix Metalloproteinase 10 (MMP-10) in Comparison with Matrix Metalloproteinase 3 (MMP-3) in Human Urinary Bladder Carcinoma. J. Clin. Med. 2021, 10, 3683. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.M.; Chen, C.T.; Torzilli, P.A. Increased stromelysin-1 (MMP-3), proteoglycan degradation (3B3- and 7D4) and collagen damage in cyclically load-injured articular cartilage. Osteoarthr. Cartil. 2004, 12, 485–496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rechardt, O.; Elomaa, O.; Vaalamo, M.; Pääkkönen, K.; Jahkola, T.; Höök-Nikanne, J.; Hembry, R.M.; Häkkinen, L.; Kere, J.; Saarialho-Kere, U. Stromelysin-2 is upregulated during normal wound repair and is induced by cytokines. J. Investig. Dermatol. 2000, 115, 778–787. [Google Scholar] [CrossRef] [Green Version]
- Bhuvarahamurthy, V.; Kristiansen, G.O.; Johannsen, M.; Loening, S.A.; Schnorr, D.; Jung, K.; Staack, A. In situ gene expression and localization of metalloproteinases MMP1, MMP2, MMP3, MMP9, and their inhibitors TIMP1 and TIMP2 in human renal cell carcinoma. Oncol. Rep. 2006, 15, 1379–1384. [Google Scholar] [CrossRef] [Green Version]
- Miyata, Y.; Iwata, T.; Maruta, S.; Kanda, S.; Nishikido, M.; Koga, S.; Kanetake, H. Expression of matrix metalloproteinase-10 in renal cell carcinoma and its prognostic role. Eur. Urol. 2007, 52, 791–797. [Google Scholar] [CrossRef]
- Burton, K. Determination of DNA concentration with diphenylamine. Methods Enzym. 1968, 12, 163–166. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Shi, Z.D.; Ji, X.Y.; Qazi, H.; Tarbell, J.M. Interstitial flow promotes vascular fibroblast, myofibroblast, and smooth muscle cell motility in 3-D collagen I via upregulation of MMP-1. American journal of physiology. Heart Circ. Physiol. 2009, 297, H1225–H1234. [Google Scholar] [CrossRef] [Green Version]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Amin, M.B.; Edge, S.; Greene, F.; Byrd, D.R.; Brookland, R.K.; Washington, M.K.; Gershenwald, J.E.; Compton, C.C.; Hess, K.R.; Sullivan, D.C.; et al. (Eds.) AJCC Cancer Staging Manual; Springer International Publishing: New York, NY, USA, 2017. [Google Scholar]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.J.; Affolter, M.; Kussmann, M. A nutrigenomics view of protein intake: Macronutrient, bioactive peptides, and protein turnover. Prog. Mol. Biol. Transl. Sci. 2012, 108, 51–74. [Google Scholar] [CrossRef] [PubMed]
- Frantz, C.; Stewart, K.M.; Weaver, V.M. The extracellular matrix at a glance. J. Cell Sci. 2010, 123 Pt 24, 4195–4200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zitka, O.; Kukacka, J.; Krizkova, S.; Huska, D.; Adam, V.; Masarik, M.; Prusa, R.; Kizek, R. Matrix metalloproteinases. Curr. Med. Chem. 2010, 17, 3751–3768. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.G.; Yang, K.E.; Hwang, J.W.; Kang, H.S.; Lee, S.Y.; Choi, S.; Shin, J.; Jang, I.S.; An, H.J.; Chung, H.; et al. Degradation of Kidney and Psoas Muscle Proteins as Indicators of Post-Mortem Interval in a Rat Model, with Use of Lateral Flow Technology. PLoS ONE 2016, 11, e0160557. [Google Scholar] [CrossRef] [Green Version]
- Qiao, Z.K.; Li, Y.L.; Lu, H.T.; Wang, K.L.; Xu, W.H. Expression of tissue levels of matrix metalloproteinases and tissue inhibitors of metalloproteinases in renal cell carcinoma. World J. Surg. Oncol. 2013, 11, 1. [Google Scholar] [CrossRef] [Green Version]
- Cannon, G.M., Jr.; Getzenberg, R.H. Urinary matrix metalloproteinase activity is not significantly altered in patients with renal cell carcinoma. Urology 2006, 67, 848–850. [Google Scholar] [CrossRef]
- Genovese, F.; Manresa, A.A.; Leeming, D.J.; Karsdal, M.A.; Boor, P. The extracellular matrix in the kidney: A source of novel non-invasive biomarkers of kidney fibrosis? Fibrogenesis Tissue Repair 2014, 7, 4. [Google Scholar] [CrossRef]








Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kudelski, J.; Młynarczyk, G.; Gudowska-Sawczuk, M.; Mroczko, B.; Darewicz, B.; Bruczko-Goralewska, M.; Romanowicz, L. Higher Content but Not Activity of Stromelysin-2 (MMP-10) in Comparison to Stromelysin-1 (MMP-3) in Human Renal Carcinoma. Int. J. Environ. Res. Public Health 2022, 19, 12613. https://doi.org/10.3390/ijerph191912613
Kudelski J, Młynarczyk G, Gudowska-Sawczuk M, Mroczko B, Darewicz B, Bruczko-Goralewska M, Romanowicz L. Higher Content but Not Activity of Stromelysin-2 (MMP-10) in Comparison to Stromelysin-1 (MMP-3) in Human Renal Carcinoma. International Journal of Environmental Research and Public Health. 2022; 19(19):12613. https://doi.org/10.3390/ijerph191912613
Chicago/Turabian StyleKudelski, Jacek, Grzegorz Młynarczyk, Monika Gudowska-Sawczuk, Barbara Mroczko, Barbara Darewicz, Marta Bruczko-Goralewska, and Lech Romanowicz. 2022. "Higher Content but Not Activity of Stromelysin-2 (MMP-10) in Comparison to Stromelysin-1 (MMP-3) in Human Renal Carcinoma" International Journal of Environmental Research and Public Health 19, no. 19: 12613. https://doi.org/10.3390/ijerph191912613







