The First Exposure Assessment of Mercury Levels in Hair among Pregnant Women and Its Effects on Birth Weight and Length in Semarang, Central Java, Indonesia
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Subjects
2.2. Measurement of Hair Total Mercury (T-Hg)
2.3. Estimation of Fish Consumption
2.4. Statistical Analysis
2.5. Ethical Aspects
3. Results
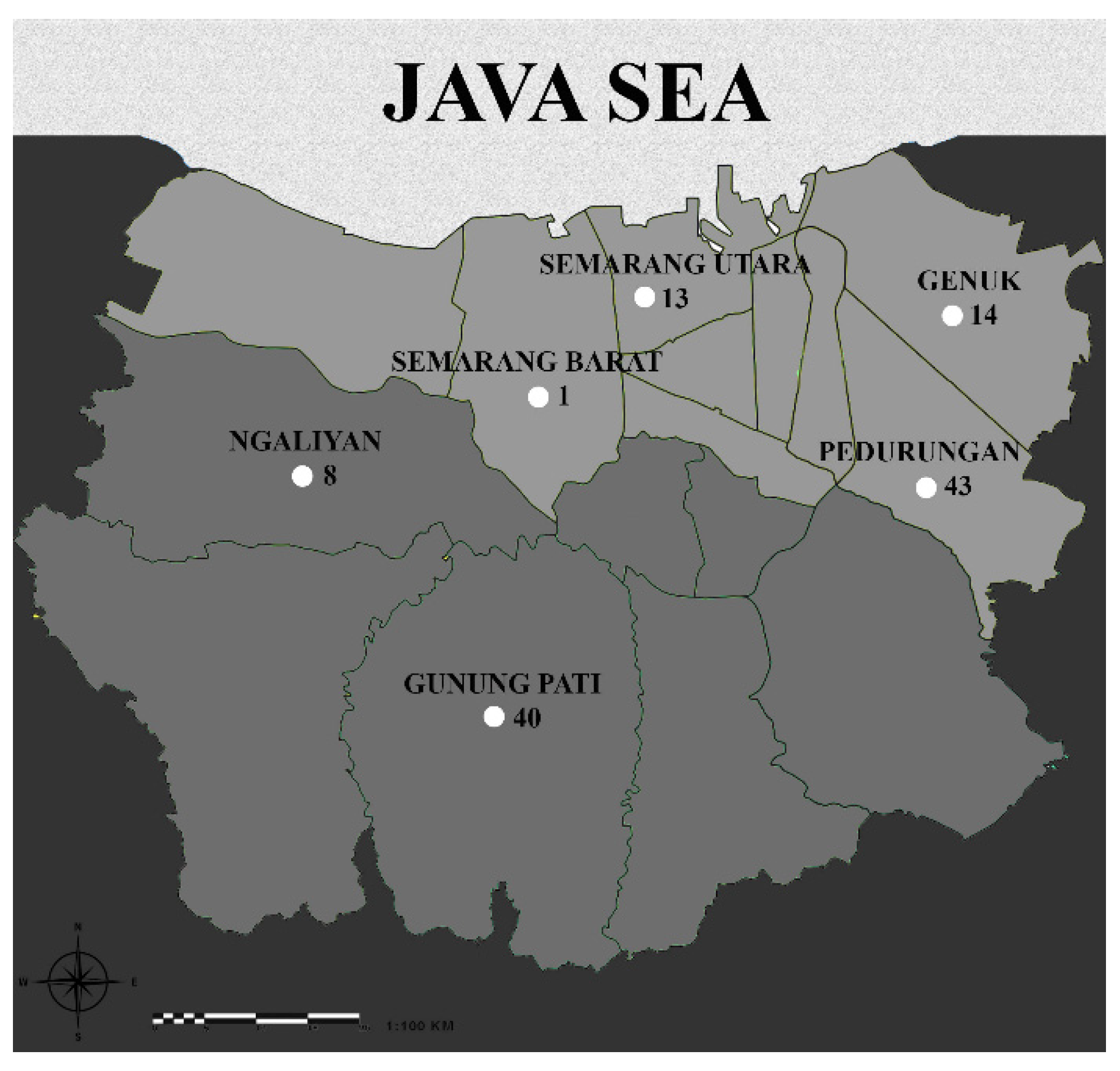
3.1. Characteristics of the Study Subjects and Area
3.2. Comparison of Hg Concentrations in Mother Conditions
3.3. Association of Maternal Hair Hg Concentrations with Birth Weight and Length of Babies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. United Nations Environment Programme Guidance for Identifying Populations at Risk from Mercury Exposure. Available online: https://wedocs.unep.org/bitstream/handle/20.500.11822/11786/IdentifyingPopnatRiskExposuretoMercury_2008Web.pdf?sequence=1&isAllowed=y (accessed on 15 June 2021).
- World Health Organization Children’s Exposure to Mercury Compounds. Available online: https://www.who.int/ceh/publications/children_exposure/en/ (accessed on 17 August 2021).
- Basu, N.; Horvat, M.; Evers, D.C.; Zastenskaya, I.; Weihe, P.; Tempowski, J. A State-of-the-Science Review of Mercury Biomarkers in Human Populations Worldwide between 2000 and 2018. Environ. Health Perspect. 2018, 126, 106001. [Google Scholar] [CrossRef]
- Bashore, C.J.; Geer, L.A.; He, X.; Puett, R.; Parsons, P.J.; Palmer, C.D.; Steuerwald, A.J.; Abulafia, O.; Dalloul, M.; Sapkota, A. Maternal mercury exposure, season of conception and adverse birth outcomes in an urban immigrant community in Brooklyn, New York, USA. Int. J. Env. Res. Public Health 2014, 11, 8414–8442. [Google Scholar] [CrossRef]
- Vigeh, M.; Nishioka, E.; Ohtani, K.; Omori, Y.; Matsukawa, T.; Koda, S.; Yokoyama, K. Prenatal mercury exposure and birth weight. Reprod. Toxicol. 2018, 76, 78–83. [Google Scholar] [CrossRef]
- Wells, E.M.; Herbstman, J.B.; Lin, Y.H.; Jarrett, J.; Verdon, C.P.; Ward, C.; Caldwell, K.L.; Hibbeln, J.R.; Witter, F.R.; Halden, R.U. Cord blood methylmercury and fetal growth outcomes in Baltimore newborns: Potential confounding and effect modification by omega-3 fatty acids, selenium, and sex. Environ. Health Perspect. 2016, 124, 373–379. [Google Scholar] [CrossRef]
- Ministry of Health Labour and Welfare Japan. Advice for Pregnant Women on Fish Consumption and Mercury. Available online: https://www.mhlw.go.jp/topics/bukyoku/iyaku/syoku-anzen/suigin/dl/051102-1en.pdf (accessed on 17 December 2021).
- Food and Drug Administration (FDA). Environmental Protection Agency (EPA) Technical Information on Development of FDA/EPA Advice about Eating Fish for Those Who Might Become or Are Pregnant or Breastfeeding and Children Ages 1–11 Years. Available online: https://www.fda.gov/food/metals-and-your-food/technical-information-development-fdaepa-advice-about-eating-fish-those-who-might-become-or-are (accessed on 20 December 2021).
- EU-Scientific Committee for Food (EU-SCF). Joint FAO/WHO Expert Committee on Food Additives (JFCFA) Reports of the Scientific Commitee for Food: First series. Available online: https://food.ec.europa.eu/system/files/2020-12/sci-com_scf_reports_01.pdf (accessed on 20 August 2022).
- United State Environmental Policy Agency. Reference Dose for Methylmercury. Available online: https://ofmpub.epa.gov/eims/eimscomm.getfile?p_download_id=36598 (accessed on 11 March 2021).
- World Health Organization. Methylmercury: In Environmental Health Criteria 101. Available online: http://www.inchem.org/documents/ehc/ehc/ehc101.htm (accessed on 27 August 2021).
- National Research Council. Toxicological Effects of Methylmercury; The National Academies Press: Washington, DC, USA, 2000; p. 364. [Google Scholar] [CrossRef]
- Bellinger, D.C.; O’Leary, K.; Rainis, H.; Gibb, H.J. Country-specific estimates of the incidence of intellectual disability associated with prenatal exposure to methylmercury. Env. Res 2016, 147, 159–163. [Google Scholar] [CrossRef]
- Sakamoto, M.; Tatsuta, N.; Izumo, K.; Phan, P.T.; Vu, L.D.; Yamamoto, M.; Nakamura, M.; Nakai, K.; Murata, K. Health Impacts and Biomarkers of Prenatal Exposure to Methylmercury: Lessons from Minamata, Japan. Toxics 2018, 6, 45. [Google Scholar] [CrossRef]
- Karagas, M.R.; Choi, A.L.; Oken, E.; Horvat, M.; Schoeny, R.; Kamai, E.; Cowell, W.; Grandjean, P.; Korrick, S. Evidence on the human health effects of low-level methylmercury exposure. Environ. Health Perspect. 2012, 120, 799–806. [Google Scholar] [CrossRef]
- Kobayashi, S.; Kishi, R.; Saijo, Y.; Ito, Y.; Oba, K.; Araki, A.; Miyashita, C.; Itoh, S.; Minatoya, M.; Yamazaki, K.; et al. Association of blood mercury levels during pregnancy with infant birth size by blood selenium levels in the Japan Environment and Children’s Study: A prospective birth cohort. Env. Int. 2019, 125, 418–429. [Google Scholar] [CrossRef]
- Oken, E.; Wright, R.O.; Kleinman, K.P.; Bellinger, D.; Amarasiriwardena, C.J.; Hu, H.; Rich-Edwards, J.W.; Gillman, M.W. Maternal fish consumption, hair mercury, and infant cognition in a U.S. Cohort. Environ. Health Perspect. 2005, 113, 1376–1380. [Google Scholar] [CrossRef]
- Jedrychowski, W.; Jankowski, J.; Flak, E.; Skarupa, A.; Mroz, E.; Sochacka-Tatara, E.; Lisowska-Miszczyk, I.; Szpanowska-Wohn, A.; Rauh, V.; Skolicki, Z.; et al. Effects of prenatal exposure to mercury on cognitive and psychomotor function in one-year-old infants: Epidemiologic cohort study in Poland. Ann. Epidemiol. 2006, 16, 439–447. [Google Scholar] [CrossRef]
- Murcia, M.; Ballester, F.; Enning, A.M.; Iñiguez, C.; Valvi, D.; Basterrechea, M.; Rebagliato, M.; Vioque, J.; Maruri, M.; Tardon, A.; et al. Prenatal mercury exposure and birth outcomes. Env. Res. 2016, 151, 11–20. [Google Scholar] [CrossRef]
- van Wijngaarden, E.; Harrington, D.; Kobrosly, R.; Thurston, S.W.; O’Hara, T.; McSorley, E.M.; Myers, G.J.; Watson, G.E.; Shamlaye, C.F.; Strain, J.J.; et al. Prenatal exposure to methylmercury and LCPUFA in relation to birth weight. Ann. Epidemiol. 2014, 24, 273–278. [Google Scholar] [CrossRef]
- Mahaffey, K.R.; Sunderland, E.M.; Chan, H.M.; Choi, A.L.; Grandjean, P.; Mariën, K.; Oken, E.; Sakamoto, M.; Schoeny, R.; Weihe, P.; et al. Balancing the benefits of n-3 polyunsaturated fatty acids and the risks of methylmercury exposure from fish consumption. Nutr. Rev. 2011, 69, 493–508. [Google Scholar] [CrossRef]
- Weihe, P.; Grandjean, P. Cohort studies of Faroese children concerning potential adverse health effects after the mothers’ exposure to marine contaminants during pregnancy. Acta Vet. Scand. 2012, 54, S7. [Google Scholar] [CrossRef]
- Food and Drug Administration. Advice about Eating Fish: For Women Who Are or Might Become Pregnant, Breastfeeding Mothers, and Young Children. Available online: https://www.fda.gov/food/consumers/advice-about-eating-fish (accessed on 9 November 2020).
- Food and Agricultural Organization. OECD-FAO Agricultural Outlook 2020–2029. Available online: https://knoema.com/HIGH_AGLINK_2020/oecd-fao-agricultural-outlook-2020-2029?commodity=1000370-Fish&origin=jp.knoema.com (accessed on 13 September 2021).
- Bappeda, B.P.S. The Guideline Book of Semarang City 2015 in Bahasa; Badan Pusat Statistik: Semarang, Indonesia, 2016; pp. 1–2. [Google Scholar]
- World Health Organization. Women of reproductive age (15–49 years) who are married or in-union who have their need for family planning satisfied with modern methods (%), UNPD. Available online: https://www.who.int/data/gho/indicator-metadata-registry/imr-details/6#:~:text=Definition%3A,who%20use%20modern%20contraceptive%20methods (accessed on 10 August 2022).
- McDowell, M.A.; Dillon, C.F.; Osterloh, J.; Bolger, P.M.; Pellizzari, E.; Fernando, R.; Montes de Oca, R.; Schober, S.E.; Sinks, T.; Jones, R.L.; et al. Hair mercury levels in U.S. children and women of childbearing age: Reference range data from NHANES 1999–2000. Environ. Health Perspect. 2004, 112, 1165–1171. [Google Scholar] [CrossRef]
- Nuttall, K.L. Interpreting Hair Mercury Levels in Individual Patients. Ann. Clin. Lab. Sci 2006, 36, 248–261. [Google Scholar]
- Hoang, V.A.T.; Do, H.T.T.; Agusa, T.; Koriyama, C.; Akiba, S.; Ishibashi, Y.; Sakamoto, M.; Yamamoto, M. Hair mercury levels in relation to fish consumption among Vietnamese in Hanoi. J. Toxicol. Sci. 2017, 42, 651–662. [Google Scholar] [CrossRef]
- David, J.; Ando, T.; Fujiyama, R.; Uwatoko, F.; Akiba, S.; Koriyama, C. Serum Docosahexaenoic Acid and Cognitive Impairment of Japanese Residents in a Remote Island. Med. J. Kagoshima Univ. 2015, 67, 1–10. [Google Scholar]
- Ververs, M.T.; Antierens, A.; Sackl, A.; Staderini, N.; Captier, V. Which anthropometric indicators identify a pregnant woman as acutely malnourished and predict adverse birth outcomes in the humanitarian context? PLoS Curr. 2013, 5. [Google Scholar] [CrossRef]
- The Major Policy of Central Java No. 560/94 about Minimum Wages in Central Java 2018 in Bahasa; Badan Keuangan Provinsi Jawa: Tengah, Indonesia, 2017.
- World Health Organization Child growth standards. Available online: https://www.who.int/tools/child-growth-standards (accessed on 11 October 2020).
- Yıldırım, E.; Derici, M.K. The Effect of Heavy Metals on Miscarriage. J Clin Obs. Gynecol 2019, 29, 31–38. [Google Scholar] [CrossRef][Green Version]
- Mosad, S.; Attalla, S.; Eldakroory, S.; Goda, H. “Chronic Mercury Exposure from Amalgam” Is it Associated with Reccurrent Miscarriage? Toxicol. Int. 2011, 14, 109–125. [Google Scholar]
- Ding, G.; Cui, C.; Chen, L.; Gao, Y.; Zhou, Y.; Shi, R.; Tian, Y. Prenatal low-level mercury exposure and neonatal anthropometry in rural northern China. Chemosphere 2013, 92, 1085–1089. [Google Scholar] [CrossRef] [PubMed]
- Baldewsingh, G.K.; Wickliffe, J.K.; van Eer, E.D.; Shankar, A.; Hindori-Mohangoo, A.D.; Harville, E.W.; Covert, H.H.; Shi, L.; Lichtveld, M.Y.; Zijlmans, W. Prenatal Mercury Exposure in Pregnant Women from Suriname’s Interior and Its Effects on Birth Outcomes. Int. J. Env. Res Public Health 2020, 17, 4032. [Google Scholar] [CrossRef]
- McCormick, M.C. The contribution of low birth weight to infant mortality and childhood morbidity. N. Engl. J. Med. 1985, 312, 82–90. [Google Scholar] [CrossRef]
- Okah, F.A.; Cai, J.; Hoff, G.L. Term-gestation low birth weight and health-compromising behaviors during pregnancy. Obs. Gynecol. 2005, 105, 543–550. [Google Scholar] [CrossRef]
- Torres-Arreola, L.P.; Constantino-Casas, P.; Flores-Hernández, S.; Villa-Barragán, J.P.; Rendón-Macías, E. Socioeconomic factors and low birth weight in Mexico. BMC Public Health 2005, 5, 20. [Google Scholar] [CrossRef]
- Verma, V.; Vishwakarma, R.K.; Nath, D.C.; Khan, H.T.A.; Prakash, R.; Abid, O. Prevalence and determinants of caesarean section in South and South-East Asian women. PLoS ONE 2020, 15, e0229906. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Betrán, A.P.; Guerrero Vela, M.; Souza, J.P.; Zhang, J. Searching for the optimal rate of medically necessary cesarean delivery. Birth 2014, 41, 237–244. [Google Scholar] [CrossRef]
- Rice, D.C. The US EPA reference dose for methylmercury: Sources of uncertainty. Env. Res. 2004, 95, 406–413. [Google Scholar] [CrossRef]
- Silbernagel, S.M.; Carpenter, D.O.; Gilbert, S.G.; Gochfeld, M.; Groth, E.; Hightower, J.M.; Schiavone, F.M. Recognizing and preventing overexposure to methylmercury from fish and seafood consumption: Information for physicians. J. Toxicol. 2011, 2011, 983072. [Google Scholar] [CrossRef]
- Sato, R.L.; Li, G.G.; Shaha, S. Antepartum seafood consumption and mercury levels in newborn cord blood. Am. J. Obs. Gynecol. 2006, 194, 1683–1688. [Google Scholar] [CrossRef]
- Yaginuma-Sakurai, K.; Murata, K.; Iwai-Shimada, M.; Nakai, K.; Kurokawa, N.; Tatsuta, N.; Satoh, H. Hair-to-blood ratio and biological half-life of mercury: Experimental study of methylmercury exposure through fish consumption in humans. J. Toxicol. Sci. 2012, 37, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Mahaffey, K.R. Mercury exposure: Medical and public health issues. Trans. Am. Clin. Clim. Assoc. 2005, 116, 127–153; discussion 153–154. [Google Scholar]
- Debes, F.; Budtz-Jørgensen, E.; Weihe, P.; White, R.F.; Grandjean, P. Impact of prenatal methylmercury exposure on neurobehavioral function at age 14 years. Neurotoxicol. Teratol. 2006, 28, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Byrd, K.A.; Thilsted, S.H.; Fiorella, K.J. Fish nutrient composition: A review of global data from poorly assessed inland and marine species. Public Health Nutr. 2021, 24, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Soccol, M.C.H.; Oetterer, M. Seafood as functional food. Braz. Arch. Biol. Technol. 2003, 46, 443–454. [Google Scholar] [CrossRef]
- Plessi, M.; Bertelli, D.; Monzani, A. Mercury and Selenium Content in Selected Seafood. J. Food Compos. Anal. 2001, 14, 461–467. [Google Scholar] [CrossRef]
| Median (Min-Max), Number (%) | p-Value | |||
|---|---|---|---|---|
| Variable | Total (n = 118) | Living Area * | ||
| Lowland (n = 70) | Highland (n = 48) | |||
| Age (year) | 29.5 (19–39) | 30.5 (19–39) | 28 (19–35) | 0.061 a |
| <30 | 59 (50.0) | 32 (45.7) | 27 (56.3) | 0.349 b |
| ≥30 | 59 (50.0) | 38 (54.3) | 21 (43.8) | |
| BMI before pregnancy (kg/m2) | 22.8 (14.4–41.1) | 23.5 (14.4–41.1) | 21.8 (15–39.7) | 0.153 a |
| <18.5 | 18 (15.3) | 9 (12.9) | 9 (18.8) | 0.828 b |
| 18.5–24.9 | 65 (55.1) | 39 (55.7) | 26 (54.2) | |
| 25–29.9 | 25 (21.2) | 16 (22.9) | 9 (18.8) | |
| ≥30 | 10 (8.5) | 6 (8.6) | 4 (8.3) | |
| Mid upper arm circumference (cm) ** | 26.5 (20.0–37.1) | 26.5 (20–37.1) | 26.5 (20–36) | 0.624 a |
| <23 | 17 (14.4) | 9 (12.9) | 8 (16.7) | 0.755 b |
| ≥23 | 101 (85.6) | 61 (87.1) | 40 (83.3) | |
| Parity before this pregnancy | 0.093 b | |||
| 0 | 36 (30.5) | 18 (25.7) | 18 (37.5) | |
| 1 | 56 (47.5) | 32 (45.7) | 24 (50) | |
| ≥2 | 26 (22.0) | 20 (28.6) | 6 (12.5) | |
| Presence of disease in previous pregnancy *** | 0.154 c | |||
| Yes | 17 (20.0) | 14 (25.5) | 3 (10.0) | |
| No | 68 (80.0) | 41 (74.6) | 27 (90.0) | |
| Education | 0.521 b | |||
| Elementary-junior | 38 (32.2) | 20 (28.6) | 18 (37.5) | |
| High school | 70 (59.3) | 43 (61.4) | 27 (56.3) | |
| Universities | 10 (8.5) | 7 (10.0) | 3 (6.3) | |
| Income (IDR.10−5 /month) **** | 23.5 (5–78) | 25 (5–78) | 23 (5–50) | 0.002 a |
| <23 | 43 (36.4) | 20 (28.6) | 23 (47.9) | 0.051 b |
| ≥23 | 75 (63.6) | 50 (71.4) | 25 (52.1) | |
| Occupation | 0.524 c | |||
| Labourer | 5 (4.2) | 3 (4.3) | 2 (4.2) | |
| Employees | 28 (23.7) | 19 (27.1) | 9 (18.8) | |
| Entrepreneur | 8 (6.8) | 6 (8.6) | 2 (4.2) | |
| Housewife | 77 (65.3) | 42 (60.0) | 35 (72.9) | |
| Median (Min-Max), Number (%) | ||||
|---|---|---|---|---|
| Birth Outcomes | Total | Living area | p-Value | |
| Lowland | Highland | |||
| Birth weight * (g) | 3100 (2260–4200) | 3100 (2260–4200) | 3120 (2260–3830) | 0.748 a |
| <2500 | 5 (4.2) | 2 (2.9) | 3 (6.3) | 0.665 b |
| ≥2500 | 113 (95.8) | 68 (97.1) | 45 (93.7) | |
| Birth length * (cm) | 49 (44–59) | 49 (45–58) | 49 (44–59) | 0.315 a |
| <50 | 73 (61.9) | 46 (65.7) | 27 (56.3) | 0.397 b |
| ≥50 | 45 (38.1) | 24 (34.3) | 21 (43.7) | |
| Gender | 0.937 b | |||
| Boy | 65 (55.1) | 35 (50.0) | 30 (62.5) | |
| Girl | 53 (44.9) | 35 (50.0) | 18 (37.5) | |
| Gestational age at birth (week) | 38.5 (34–42) | 39 (34–42) | 38 (34–42) | 0.937 a |
| <37 | 15 (12.7) | 9 (12.9) | 6 (12.5) | 1.000 b |
| ≥37 | 103 (87.3) | 61 (87.1) | 42 (87.5) | |
| Delivery process | 0.030 b | |||
| Vaginal | 67 (56.8) | 34 (48.6) | 33 (68.8) | |
| Caesarean | 51 (43.2) | 36 (51.4) | 15 (31.3) | |
| Variable | N | Hair Hg Concentration (µg/g) | p-Value | |||||
|---|---|---|---|---|---|---|---|---|
| P5 | P10 | P50 | P90 | P95 | Min-Max | |||
| All subject | 118 | 0.154 | 0.207 | 0.434 | 0.802 | 1.461 | 0.146–8.105 | |
| Age (year) | ||||||||
| <30 | 59 | 0.152 | 0.163 | 0.465 | 0.814 | 2.463 | 0.146–5.290 | 0.741 a |
| ≥30 | 59 | 0.224 | 0.236 | 0.409 | 0.776 | 1.431 | 0.154–8.105 | |
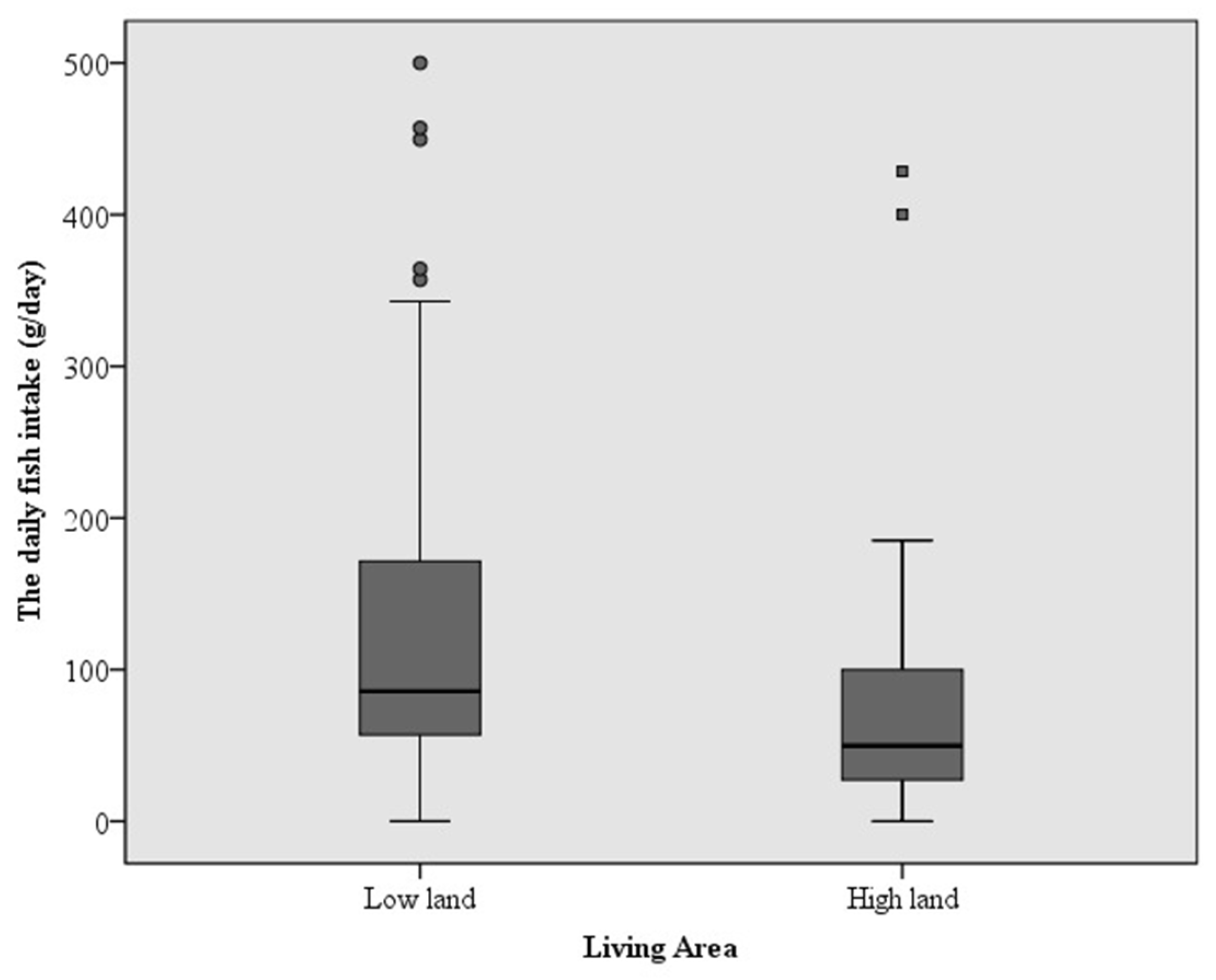
| Living area | ||||||||
| Lowland | 70 | 0.200 | 0.241 | 0.465 | 0.813 | 3.085 | 0.146–8.105 | 0.043 a |
| Highland | 48 | 0.153 | 0.154 | 0.385 | 0.747 | 1.389 | 0.150–1.956 | |
| Education | ||||||||
| Elementary–Junior | 38 | 0.180 | 0.223 | 0.450 | 1.431 | 2.604 | 0.152–5.290 | 0.269 b |
| Senior High school | 70 | 0.154 | 0.174 | 0.414 | 0.689 | 1.302 | 0.146–8.105 | |
| Universities | 10 | 0.163 | 0.175 | 0.596 | 1.847 | 1.956 | 0.163–1.956 | |
| Income (IDR.10−5/month) | ||||||||
| <23 | 43 | 0.158 | 0.192 | 0.385 | 0.792 | 1.414 | 0.152–3.845 | 0.297 a |
| ≥23 | 75 | 0.154 | 0.212 | 0.465 | 0.834 | 2.057 | 0.146–8.105 | |
| Hair treatment | ||||||||
| Yes | 14 | 0.150 | 0.152 | 0.400 | 5.975 | 8.105 | 0.150–8.105 | 0.351 a |
| No | 104 | 0.169 | 0.226 | 0.437 | 0.790 | 1.406 | 0.146–5.290 | |
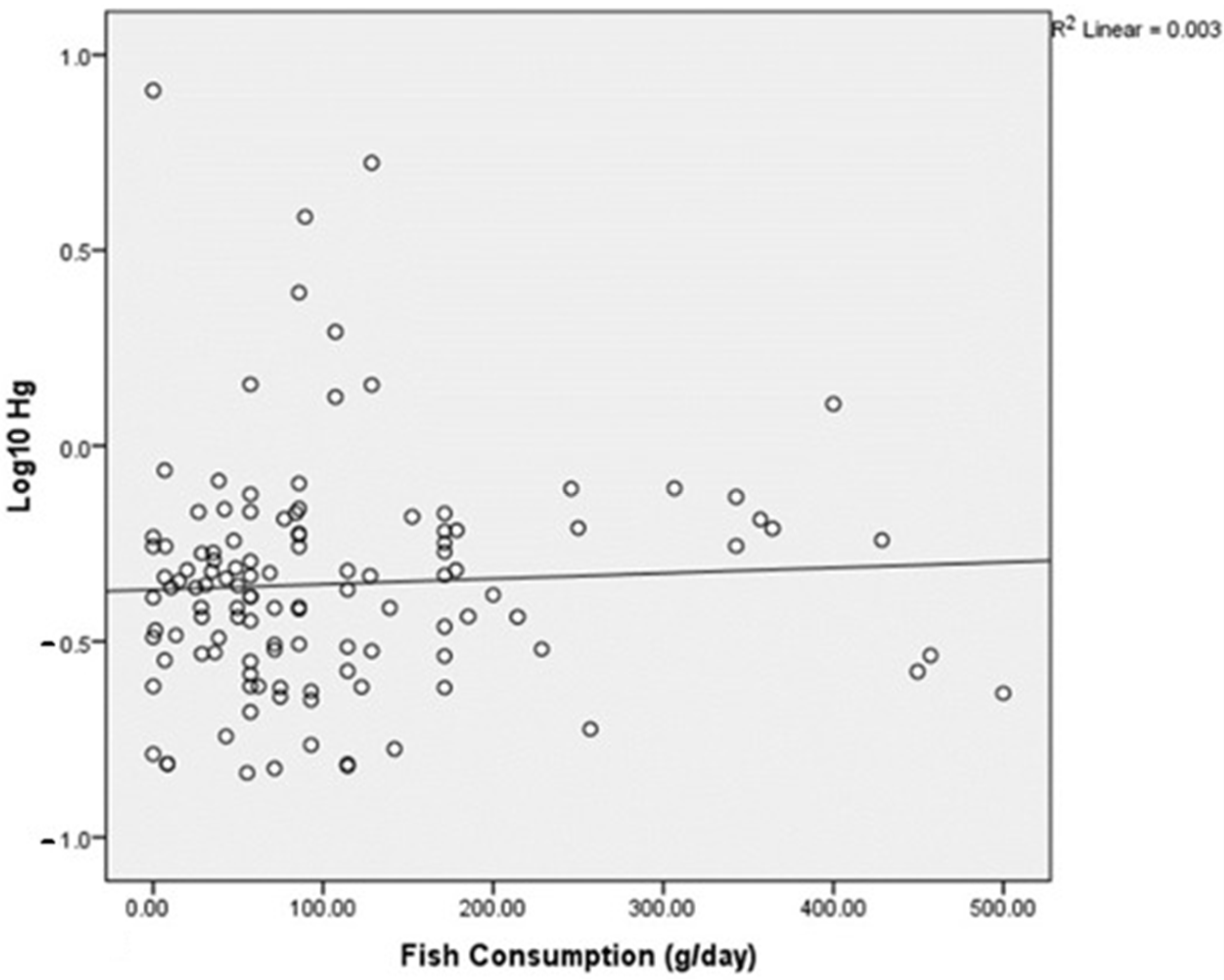
| Fish consumption (g/day) | ||||||||
| <40 | 31 | 0.154 | 0.179 | 0.434 | 0.787 | 3.761 | 0.154–8.105 | 0.314 b |
| 40–79 | 30 | 0.148 | 0.184 | 0.385 | 0.687 | 1.059 | 0.146–1.435 | |
| 80–159 | 30 | 0.153 | 0.168 | 0.447 | 2.412 | 4.495 | 0.152–5.290 | |
| ≥160 | 27 | 0.207 | 0.239 | 0.535 | 0.776 | 1.079 | 0.189–1.279 | |
| Seafood consumption | ||||||||
| Yes | 90 | 0.164 | 0.224 | 0.446 | 0.860 | 1.670 | 0.150–5.290 | 0.287 a |
| No | 21 | 0.147 | 0.157 | 0.365 | 0.579 | 0.735 | 0.146–0.751 | |
| Not eat fish | 7 | 0.163 | 0.163 | 0.409 | 8.105 | 8.105 | 0.163–8.105 | |
| Variable | Birth Length (cm) | Birth Weight (g) | ||||
|---|---|---|---|---|---|---|
| β Coefficient (CI: 95%) | SE | p-Value | β Coefficient (CI: 95%) | SE | p-Value | |
| Mothers | ||||||
| Age (year) | 0.037 (−0.046–0.119) | 0.042 | 0.383 | 6.594 (−8.249–21.44) | 7.494 | 0.381 |
| Body weight at the survey (kg) | 0.039 (0.006–0.072) | 0.017 | 0.021 | 10.72 (4.959–16.49) | 2.910 | <0.001 |
| Body weight before pregnancy (kg) | 0.034 (0.001–0.067) | 0.017 | 0.048 | 10.01 (4.205–15.81) | 2.929 | 0.001 |
| Height (cm) | 0.051 (−0.022–0.126) | 0.037 | 0.166 | 11.21 (−2.017–24.43) | 6.677 | 0.096 |
| BMI before pregnancy (kg/m2) | 0.067 (−0.018–0.152) | 0.043 | 0.119 | 22.75 (7.935–37.57) | 7.482 | 0.002 |
| Income (IDR.10−5/month) | −0.002 (−0.034–0.031) | 0.016 | 0.908 | 1.887 (−3.918–7.693) | 2.931 | 0.521 |
| Highland (vs. lowland) | 0.347 (−0.492–1.187) | 0.424 | 0.415 | −61.26 (−212.2–89.69) | 76.22 | 0.423 |
| Education (vs. senior high school) | ||||||
| Elementary-junior | −0.165 (−1.073–0.7425) | 0.458 | 0.719 | 84.92 (−73.23–243.1) | 79.84 | 0.290 |
| Universities | 0.271 (−1.252–1.795) | 0.769 | 0.725 | 370.2 (104.8–635.6) | 133.9 | 0.007 |
| Fish consumption (g/day) | −0.002 (−0.006–0.002) | 0.002 | 0.385 | −0.519 (−1.210–0.172) | 0.349 | 0.139 |
| Hair Hg, log-Hg (µg/g) | −0.043 (−1.453–1.366) | 0.712 | 0.952 | 204.96 (−45.65–455.6) | 126.5 | 0.108 |
| Babies | ||||||
| Boy (vs. girl) | 0.226 (−0.604–1.057) | 0.419 | 0.590 | 103.3 (−45.03–251.5) | 74.87 | 0.171 |
| Gestational age (week) | 0.229 (−0.037–0.495) | 0.134 | 0.090 | 90.23 (44.75–135.7) | 22.96 | <0.001 |
| Caesarian (vs. vaginal) | −0.132 (−0.966–0.703) | 0.421 | 0.755 | 29.58 (−120.4–179.6) | 75.74 | 0.697 |
| Variable | β Coefficient | CI 95% | SE | p-Value |
|---|---|---|---|---|
| Birth weight (g) | ||||
| Hair mercury, log-Hg (µg/g) | 154.6 | −73.29–382.5 | 115.0 | 0.182 |
| Boy | 72.27 | −61.74–206.2 | 67.62 | 0.288 |
| Gestational age (week) | 75.10 | 31.24–119.0 | 22.14 | 0.001 |
| BMI before pregnancy (kg/m2) | 19.99 | 6.111–33.87 | 7.01 | 0.005 |
| Education | ||||
| High school | −76.76 | −222.9–69.40 | 73.75 | 0.300 |
| Universities | 254.5 | −37.33–512.7 | 130.3 | 0.053 |
| Birth length (cm) | ||||
| Hair mercury, log-Hg (µg/g) | −0.089 | −1.518–1.340 | 0.721 | 0.902 |
| Boy | 0.115 | −0.725–0.955 | 0.424 | 0.786 |
| Gestational age (week) | 0.196 | −0.079–0.471 | 0.139 | 0.161 |
| BMI before pregnancy (kg/m2) | 0.062 | −0.026–0.149 | 0.044 | 0.164 |
| Education | ||||
| High school | 0.168 | −0.749–1.084 | 0.463 | 0.717 |
| Universities | 0.404 | −1.216–2.023 | 0.817 | 0.622 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muniroh, M.; Bakri, S.; Gumay, A.R.; Dewantiningrum, J.; Mulyono, M.; Hardian, H.; Yamamoto, M.; Koriyama, C. The First Exposure Assessment of Mercury Levels in Hair among Pregnant Women and Its Effects on Birth Weight and Length in Semarang, Central Java, Indonesia. Int. J. Environ. Res. Public Health 2022, 19, 10684. https://doi.org/10.3390/ijerph191710684
Muniroh M, Bakri S, Gumay AR, Dewantiningrum J, Mulyono M, Hardian H, Yamamoto M, Koriyama C. The First Exposure Assessment of Mercury Levels in Hair among Pregnant Women and Its Effects on Birth Weight and Length in Semarang, Central Java, Indonesia. International Journal of Environmental Research and Public Health. 2022; 19(17):10684. https://doi.org/10.3390/ijerph191710684
Chicago/Turabian StyleMuniroh, Muflihatul, Saekhol Bakri, Ainun Rahmasari Gumay, Julian Dewantiningrum, Mulyono Mulyono, Hardian Hardian, Megumi Yamamoto, and Chihaya Koriyama. 2022. "The First Exposure Assessment of Mercury Levels in Hair among Pregnant Women and Its Effects on Birth Weight and Length in Semarang, Central Java, Indonesia" International Journal of Environmental Research and Public Health 19, no. 17: 10684. https://doi.org/10.3390/ijerph191710684
APA StyleMuniroh, M., Bakri, S., Gumay, A. R., Dewantiningrum, J., Mulyono, M., Hardian, H., Yamamoto, M., & Koriyama, C. (2022). The First Exposure Assessment of Mercury Levels in Hair among Pregnant Women and Its Effects on Birth Weight and Length in Semarang, Central Java, Indonesia. International Journal of Environmental Research and Public Health, 19(17), 10684. https://doi.org/10.3390/ijerph191710684







