Amateur Athlete with Sinus Arrest and Severe Bradycardia Diagnosed through a Heart Rate Monitor: A Six-Year Observation—The Necessity of Shared Decision-Making in Heart Rhythm Therapy Management
Abstract
:1. Introduction
- note the “additional” role of some contemporary sports HRMs as reliable and relevant medical devices in the diagnosis of cardiac arrhythmias, in this case, bradyarrhythmias.
- draw attention to the fact of excessive bradyarrhythmias in the amateur athlete, mainly described in professional endurance athletes.
- describe the importance of shared decision-making in cardiac electrophysiology procedures and arrhythmia management (i.e., pacing, cardioneuroablation, observation, and deconditioning) while treating SA in both professional and amateur athletes.
2. Materials and Methods
2.1. Sports Biography
2.2. Study Protocol
2.2.1. ECG Tests
2.2.2. Transthoracic Echocardiography
2.2.3. Holter ECG
2.2.4. Exercise Stress Test
2.2.5. HRM
2.2.6. TILT Test
2.3. Statistical Analysis
2.4. Ethical Approval
3. Results
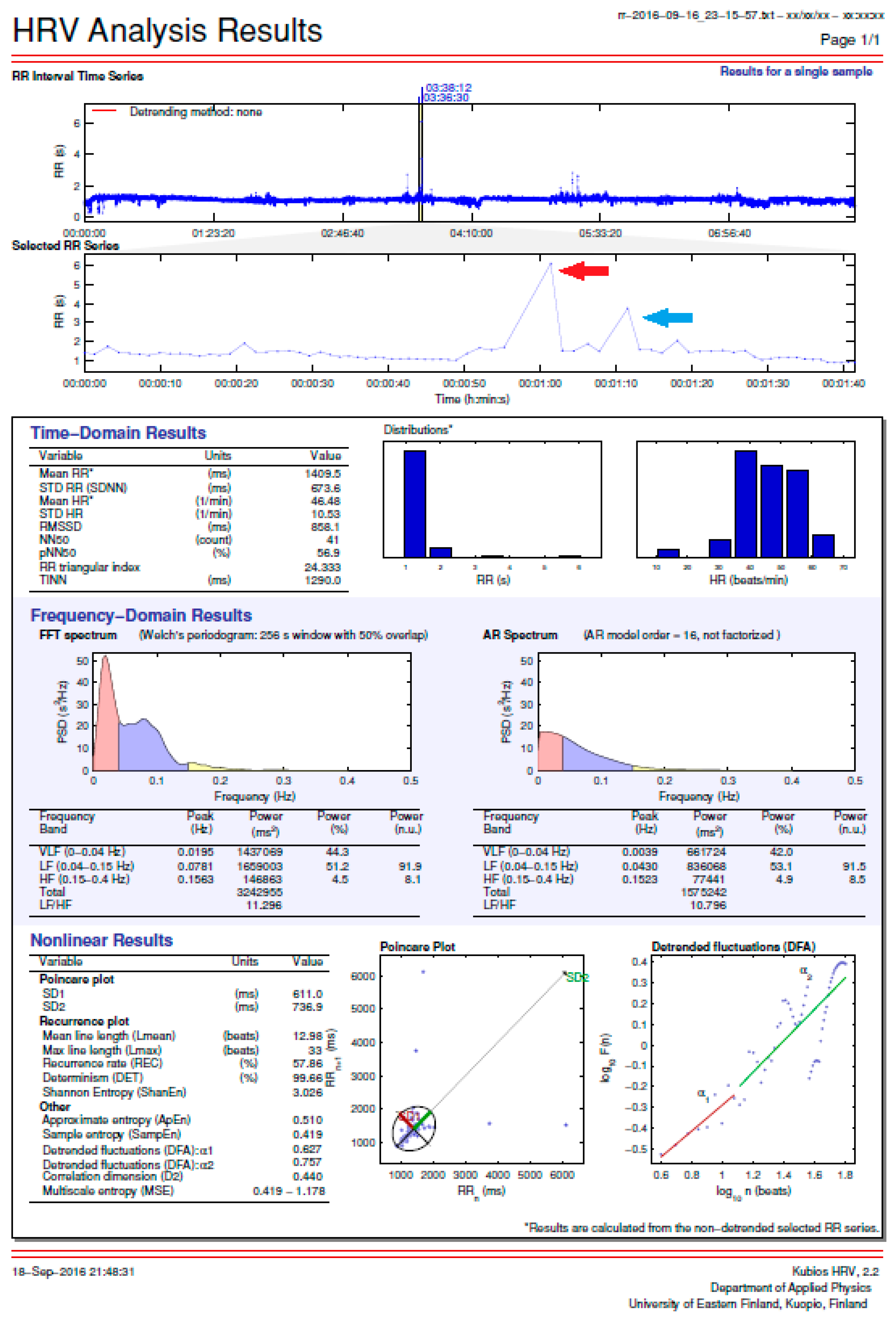
3.1. HRM Data Analysis
3.2. ECG Tests
3.3. Echocardiography
3.4. Exercise Stress Tests
3.5. TILT Tests
3.6. Holter ECG
4. Discussion
4.1. Major Findings
4.2. Recommendations for Treating SND with Cardiac Pacing
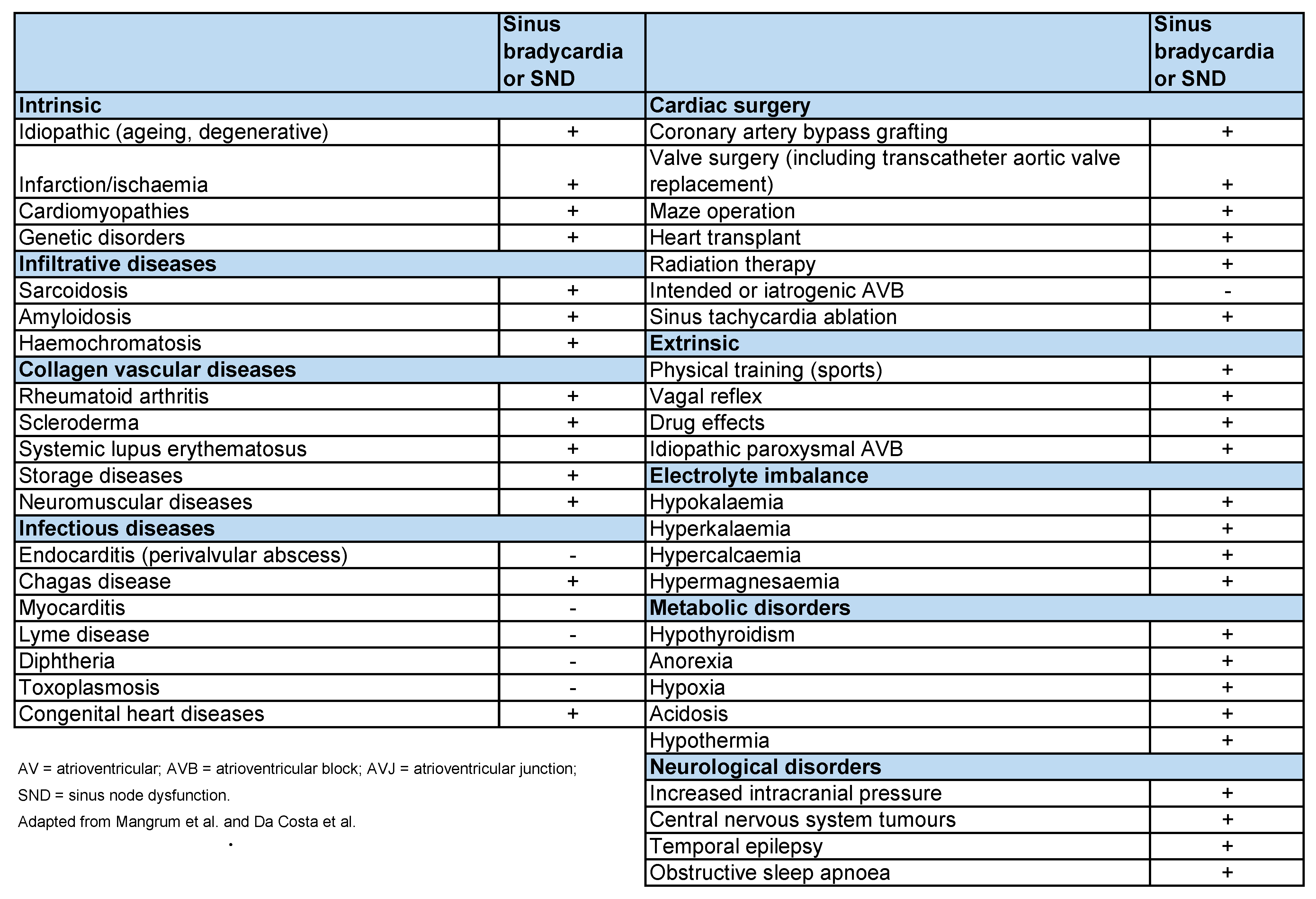
4.3. Bradycardia or SND in Trained Athletes: The Real Reason
4.4. Eligibility Criteria for Competitive Athletes with Sinus Bradycardia or SND
- Athletes with asymptomatic sinus bradycardia, exit block, pauses, and arrhythmia can participate in all competitive athletic activities unless they present other underlying structural heart diseases or arrhythmias (Class I; Level of Evidence C).
- Athletes with symptomatic sinus bradycardia should be evaluated for structural heart disease and receive treatment for the bradycardia, generally by pacemaker implantation. They should be restricted from both training and athletic competition while under evaluation. If the bradycardia treatment eliminates the symptoms, they can participate in athletic training and competition unless they present other underlying structural heart diseases or arrhythmias (Class I; Level of Evidence C).
4.5. Very Long Pauses during Sleep in Athletes
4.6. Cardioneuroablation in Athletes with SND
4.7. Shared Decision-Making in Cardiac Electrophysiology Procedures and Arrhythmia
4.8. Is a Sports HRM Just a Gadget or a Reliable Medical Device?
4.9. Limitations, Strengths, and Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Northcote, R.J.; Canning, G.P.; Ballantyne, D. Electrocardiographic findings in male veteran endurance athletes. Br. Heart J. 1989, 61, 155–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hawks, M.K.; Paul, M.L.B.; Malu, O.O. Sinus Node Dysfunction. Am. Fam. Physician 2021, 104, 179–185. [Google Scholar] [PubMed]
- Baldesberger, S.; Bauersfeld, U.; Candinas, R.; Seifert, B.; Zuber, M.; Ritter, M.; Jenni, R.; Oechslin, E.; Luthi, P.; Scharf, C.; et al. Sinus node disease and arrhythmias in the long-term follow-up of former professional cyclists. Eur. Heart J. 2008, 29, 71–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hughes, D.C.; Ellefsen, S.; Baar, K. Adaptations to Endurance and Strength Training. Cold Spring Harb. Perspect. Med. 2018, 8, a029769. [Google Scholar] [CrossRef]
- Białek, B. Statistical Description of Arrival Sequences in Amateur Long-Distance Races. Phys. Act. Rev. 2017, 5, 44–53. [Google Scholar] [CrossRef]
- Podrigalo, O.; Borisova, O.; Podrigalo, L.; Iermakov, S.; Romanenko, V.; Bodrenkova, I. The analysis of psychophysiological features of football players and water sports athletes. Phys. Act. Rev. 2020, 8, 64–73. [Google Scholar] [CrossRef]
- Levine, B.D.; Baggish, A.L.; Kovacs, R.J.; Link, M.S.; Maron, M.S.; Mitchell, J.H.; American Heart Association Electrocardiography and Arrhythmias Committee of Council on Clinical Cardiology, Council on Cardiovascular Disease in Young, Council on Cardiovascular and Stroke Nursing, Council on Functional Genomics and Translational Biology, and American College of Cardiology. Eligibility and Disqualification Recommendations for Competitive Athletes with Cardiovascular Abnormalities: Task Force 1: Classification of Sports: Dynamic, Static, and Impact. Circulation 2015, 132, e262–e266. [Google Scholar] [CrossRef]
- Pelliccia, A.; Zipes, D.P.; Maron, B.J. Bethesda Conference #36 and the European Society of Cardiology Consensus Recommendations Revisited: A Comparison of U.S. and European Criteria for Eligibility and Disqualification of Competitive Athletes with Cardiovascular Abnormalities. J. Am. Coll. Cardiol. 2008, 52, 1990–1996. [Google Scholar] [CrossRef]
- Link, M.S.; Homoud, M.K.; Wang, P.J.; Estes, N.M. Cardiac Arrhythmias in the Athlete. Cardiol. Rev. 2001, 9, 21–30. [Google Scholar] [CrossRef]
- Doyen, B.; Matelot, D.; Carré, F. Asymptomatic bradycardia amongst endurance athletes. Physician Sportsmed. 2019, 47, 249–252. [Google Scholar] [CrossRef]
- Gajda, R.; Klisiewicz, A.; Matsibora, V.; Piotrowska-Kownacka, D.; Biernacka, E.K. Heart of the World’s Top Ultramarathon Runner—Not Necessarily Much Different from Normal. Diagnostics 2020, 10, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avram, R.; Tison, G.H.; Aschbacher, K.; Kuhar, P.; Vittinghoff, E.; Butzner, M.; Runge, R.; Wu, N.; Pletcher, M.J.; Marcus, G.M.; et al. Real-world heart rate norms in the Health eHeart study. Npj Digit. Med. 2019, 2, 58. [Google Scholar] [CrossRef] [PubMed]
- Mubarik, A.; Iqbal, A.M. Holter Monitor. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK538203 (accessed on 20 February 2022).
- Gajda, R.; Gajda, J.; Czuba, M.; Gębska-Kuczorowska, A.; Knechtle, B. Heart Rate Monitors Used by Athletes-from Gadget to Medical Equipment. A Decade of Own Observations. Med. Res. J. 2021, 6, 64–70. [Google Scholar] [CrossRef]
- Jabbour, F.; Kanmanthareddy, A. Sinus Node Dysfunction. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK544253 (accessed on 20 February 2022).
- Glikson, M.; Nielsen, J.C.; Kronborg, M.B.; Michowitz, Y.; Auricchio, A.; Barbash, I.M.; Barrabés, J.A.; Boriani, G.; Braunschweig, F.; Brignole, M.; et al. 2021 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy. Eur. Heart J. 2022, 42, 3427–3520. [Google Scholar] [CrossRef] [PubMed]
- Zipes, D.P.; Ackerman, M.J.; Estes, N.A.M.; Grant, A.O.; Myerburg, R.J.; Van Hare, G. Task Force 7: Arrhythmias. J. Am. Coll. Cardiol. 2005, 45, 1354–1363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lentner, C. Physical Chemistry, Composition of Blood, Hematology, Somatometric Data 1968, 38th ed.; NJ CIBA-GEIGY Co.: West Caldwell, NJ, USA.
- Rubenstein, J.J.; Schulman, C.L.; Yurchak, P.M.; Desanctis, R.W. Clinical Spectrum of the Sick Sinus Syndrome. Circulation 1972, 46, 5–13. [Google Scholar] [CrossRef] [Green Version]
- Estes, N.M.; Link, M.S.; Cannom, D.; Naccarelli, G.V.; Prystowsky, E.N.; Maron, B.J.; Olshansky, B. Expert Consensus Conference on Arrhythmias in the Athlete of the North American Society of Pacing and Electrophysiology. Report of the NASPE policy conference on arrhythmias and the athlete. J. Cardiovasc. Electrophysiol. 2001, 12, 1208–1219. [Google Scholar] [CrossRef]
- Aubert, A.E.; Seps, B.; Beckers, F. Heart Rate Variability in Athletes. Sports Med. 2003, 33, 889–919. [Google Scholar] [CrossRef]
- Monfredi, O.; Lyashkov, A.E.; Johnsen, A.-B.; Inada, S.; Schneider, H.; Wang, R.; Nirmalan, M.; Wisloff, U.; Maltsev, V.A.; Lakatta, E.G.; et al. Biophysical Characterization of the Underappreciated and Important Relationship Between Heart Rate Variability and Heart Rate. Hypertension 2014, 64, 1334–1343. [Google Scholar] [CrossRef] [Green Version]
- D’Souza, A.; Sharma, S.; Boyett, M.R. CrossTalk opposing view: Bradycardia in the trained athlete is attributable to a downregulation of a pacemaker channel in the sinus node. J. Physiol. 2015, 593, 1749–1751. [Google Scholar] [CrossRef]
- Boyett, M.R.; D’Souza, A.; Zhang, H.; Morris, G.; Dobrzynski, H.; Monfredi, O. Viewpoint: Is the resting bradycardia in athletes the result of remodeling of the sinoatrial node rather than high vagal tone? J. Appl. Physiol. 2013, 114, 1351–1355. [Google Scholar] [CrossRef] [PubMed]
- Katona, P.G.; McLean, M.; Dighton, D.H.; Guz, A. Sympathetic and parasympathetic cardiac control in athletes and nonathletes at rest. J. Appl. Physiol. 1982, 52, 1652–1657. [Google Scholar] [CrossRef]
- Milanesi, R.; Baruscotti, M.; Gnecchi-Ruscone, T.; DiFrancesco, D. Familial Sinus Bradycardia Associated with a Mutation in the Cardiac Pacemaker Channel. N. Engl. J. Med. 2006, 354, 151–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Khoury, N.; Mathieu, S.; Marger, L.; Ross, J.; El Gebeily, G.; Ethier, N.; Fiset, C. Upregulation of the Hyperpolarization-Activated Current Increases Pacemaker Activity of the Sinoatrial Node and Heart Rate During Pregnancy in Mice. Circulation 2013, 127, 2009–2020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Epstein, A.E.; DiMarco, J.P.; Ellenbogen, K.A.; Estes, N.A.M., III; Freedman, R.A.; Gettes, L.S.; Gillinov, A.M.; Gregoratos, G.; Hammill, S.C.; Hayes, D.L.; et al. ACC/AHA/HRS 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the ACC/AHA/NASPE 2002 Guideline Update for Implantation of Cardiac Pacemakers and Antiarrhythmia Devices) developed in collaboration with the American Association for Thoracic Surgery and Society of Thoracic Surgeons. J. Am. Coll. Cardiol. 2008, 51, e1–e62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tracy, C.M.; Epstein, A.E.; Darbar, D.; DiMarco, J.P.; Dunbar, S.B.; Estes, N.M.; Ferguson, T.B.; Hammill, S.C.; Karasik, P.E.; Link, M.S.; et al. 2012 ACCF/AHA/HRS Focused Update of the 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities. J. Am. Coll. Cardiol. 2012, 60, 1297–1313. [Google Scholar] [CrossRef] [Green Version]
- Heidbuchel, H. The athlete’s heart is a proarrhythmic heart, and what that means for clinical decision making. Europace 2017, 20, 1401–1411. [Google Scholar] [CrossRef]
- Pachon, J.C.; Pachon, E.I.; Lobo, T.J.; Pachon, M.Z.; Vargas, R.N.; Jatene, A.D. Cardioneuroablation—New treatment for neurocardiogenic syncope, functional AV block and sinus dysfunction using catheter RF-ablation. Europace 2005, 7, 1–13. [Google Scholar] [CrossRef]
- Lakkireddy, D. Cardioneuroablation in the Management of Vasovagal Syncope, Sinus Node Dysfunction and Functional Atrioventricular Block: Patient Selection Based on Supporting Evidence. J. Atr. Fibrillation 2020, 13, 2396. [Google Scholar] [CrossRef]
- Chung, M.K.; Fagerlin, A.; Wang, P.J.; Ajayi, T.B.; Allen, L.A.; Baykaner, T.; Benjamin, E.J.; Branda, M.; Cavanaugh, K.L.; Chen, L.Y.; et al. Shared Decision Making in Cardiac Electrophysiology Procedures and Arrhythmia Management. Circ. Arrhythmia Electrophysiol. 2021, 14, e007958. [Google Scholar] [CrossRef]
- Stacey, D.; Légaré, F.; Lewis, K.; Barry, M.J.; Bennett, C.L.; Eden, K.B.; Holmes-Rovner, M.; Llewellyn-Thomas, H.; Lyddiatt, A.; Thomson, R.; et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst. Rev. 2017, 2017, CD001431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gajda, R. Is Continuous ECG Recording on Heart Rate Monitors the Most Expected Function by Endurance Athletes, Coaches, and Doctors? Diagnostics 2020, 10, 867. [Google Scholar] [CrossRef] [PubMed]
- Gajda, R. Heart Rate Monitor Instead of Ablation? Atrioventricular Nodal Re-Entrant Tachycardia in a Leisure-Time Triathlete: 6-Year Follow-Up. Diagnostics 2020, 10, 391. [Google Scholar] [CrossRef] [PubMed]
- Gajda, R.; Biernacka, E.K.; Drygas, W. Are heart rate monitors valuable tools for diagnosing arrhythmias in endurance athletes? Scand. J. Med. Sci. Sports 2018, 28, 496–516. [Google Scholar] [CrossRef]
- Gajda, R. Extreme Bradycardia and Bradyarrhythmias at Athletes. What will Technology Development Bring as a Help to Diagnosis Them? Res. Investig. Sports Med. 2019, 5, 704–731. [Google Scholar] [CrossRef]
- Gajda, R.; Biernacka, E.K.; Drygas, W. The Problem of Arrhythmias in Endurance Athletes: Are Heart Rate Monitors Valuable Tools for Diagnosing Arrhythmias? In Horizons in World Cardiovascular Research; Bennington, H.B., Ed.; Nova Science Publishers: New York, NY, USA, 2009; Volume 15, pp. 1–64. [Google Scholar]
- Gajda, R.; Kowalik, E.; Rybka, S.; Rębowska, E.; Śmigielski, W.; Nowak, M.; Kwaśniewska, M.; Hoffman, P.; Drygas, W. Evaluation of the Heart Function of Swimmers Subjected to Exhaustive Repetitive Endurance Efforts During a 500-km Relay. Front. Physiol. 2019, 10, 296. [Google Scholar] [CrossRef]
- Jagodzińska, M.; Szperl, M.; Ponińska, J.; Kosiec, A.; Gajda, R.; Kukla, P.; Biernacka, E.K. Coexistence of Andersen-Tawil Syndrome with Polymorphisms inhERG1Gene (K897T) andSCN5AGene (H558R) in One Family. Ann. Noninvasive Electrocardiol. 2015, 21, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Krych, M.; Biernacka, E.K.; Ponińska, J.; Kukla, P.; Filipecki, A.; Gajda, R.; Hasdemir, C.; Antzelevitch, C.; Kosiec, A.; Szperl, M.; et al. Andersen-Tawil syndrome: Clinical presentation and predictors of symptomatic arrhythmias—Possible role of polymorphisms K897T in KCNH2 and H558R in SCN5A gene. J. Cardiol. 2017, 70, 504–510. [Google Scholar] [CrossRef] [Green Version]
- Szczepańska-Gieracha, J.; Jóźwik, S.; Cieślik, B.; Mazurek, J.; Gajda, R. Immersive Virtual Reality Therapy as a Support for Cardiac Rehabilitation: A Pilot Randomized-Controlled Trial. Cyberpsycholg. Behav. Soc. Netw. 2021, 24, 543–549. [Google Scholar] [CrossRef]
- Gajda, R. Relationship Between Arrhythmias and Level Activity of Athlete’s-Role of HRMs. Examines Phys. Med. Rehabil. 2019, 2, 44–45. [Google Scholar] [CrossRef]
- Senturk, T.; Xu, H.; Puppala, K.; Krishnan, B.; Sakaguchi, S.; Chen, L.Y.; Karim, R.; Dickinson, O.; Benditt, D.G. Cardiac pauses in competitive athletes: A systematic review examining the basis of current practice recommendations. Europace 2016, 18, 1873–1879. [Google Scholar] [CrossRef] [PubMed]
- Bodin, A.; Bisson, A.; Gaborit, C.; Herbert, J.; Clementy, N.; Babuty, D.; Lip, G.Y.; Fauchier, L. Ischemic Stroke in Patients with Sinus Node Disease, Atrial Fibrillation, and Other Cardiac Conditions. Stroke 2020, 51, 1674–1681. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Alsheikhtaha, Z.; Mehra, R. A 48-Year-Old Athletic Man with Bradycardia During Sleep. Chest 2018, 154, e139–e142. [Google Scholar] [CrossRef] [PubMed]
- Svennberg, E.; Tjong, F.; Goette, A.; Akoum, N.; Di Biase, L.; Bordachar, P.; Boriani, G.; Burri, H.; Conte, G.; Deharo, J.C.; et al. How to use digital devices to detect and manage arrhythmias: An EHRA practical guide. Europace 2022, 24, euac038. [Google Scholar] [CrossRef] [PubMed]
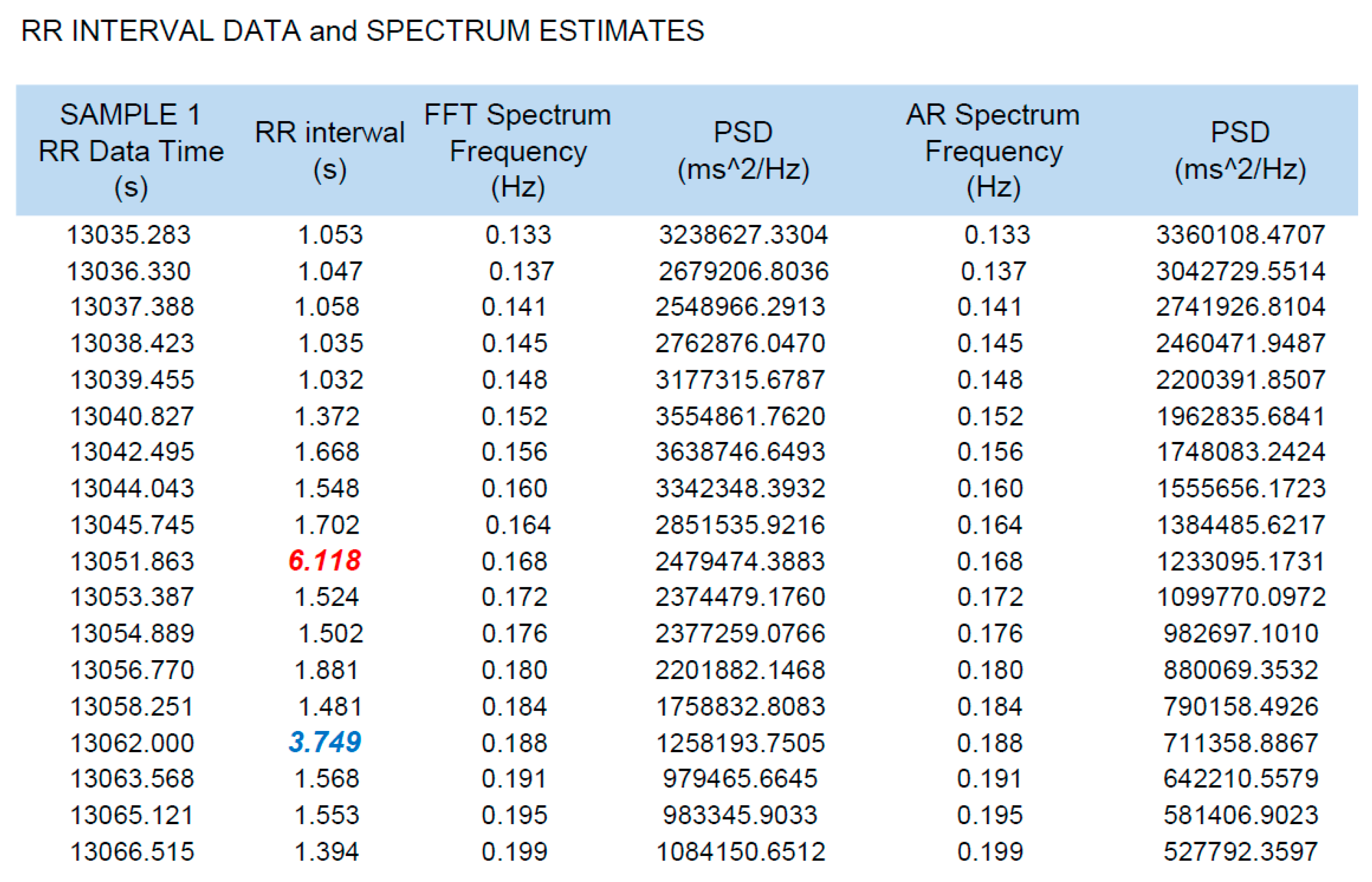
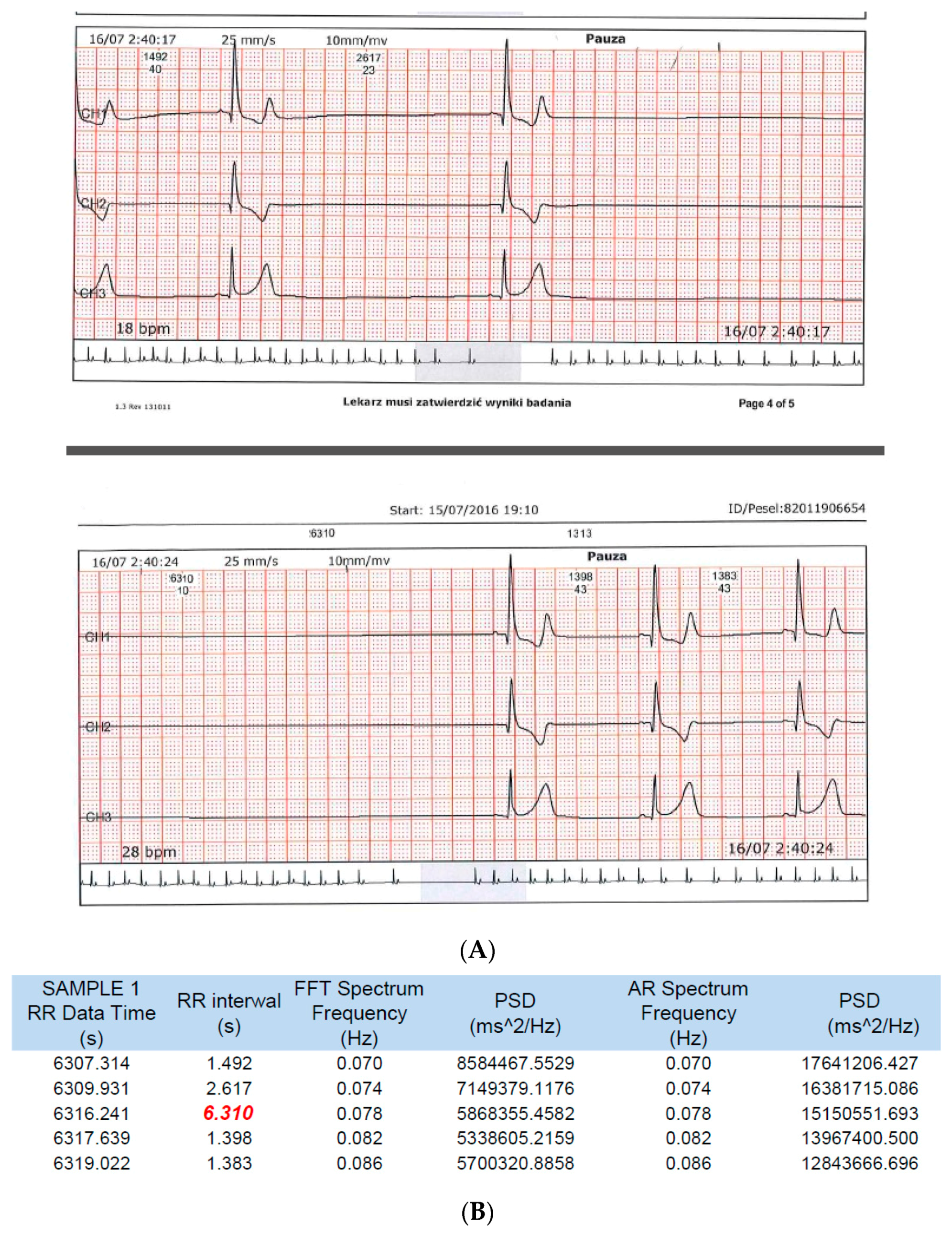
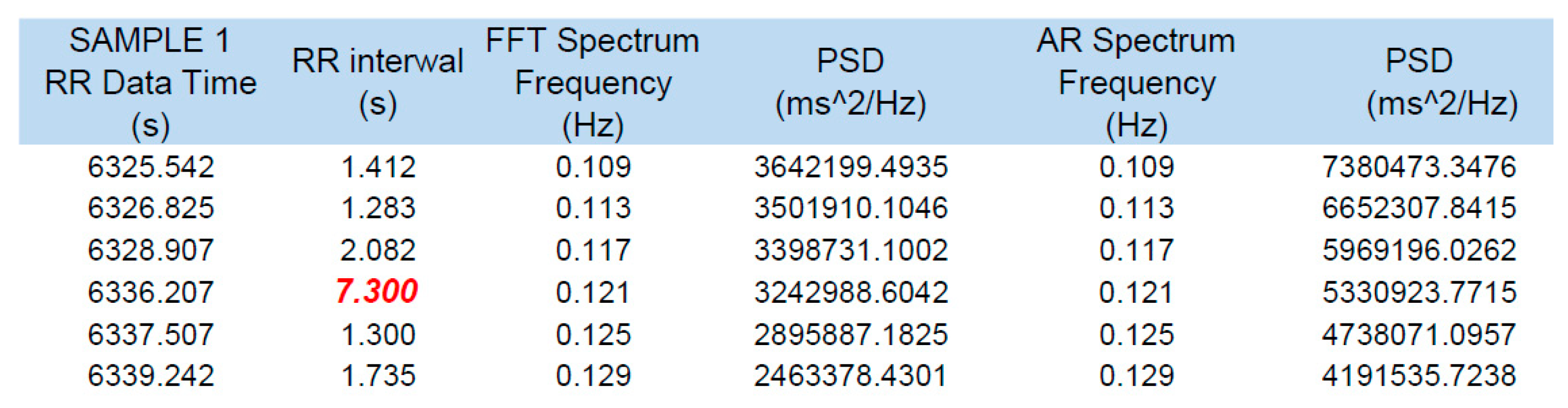
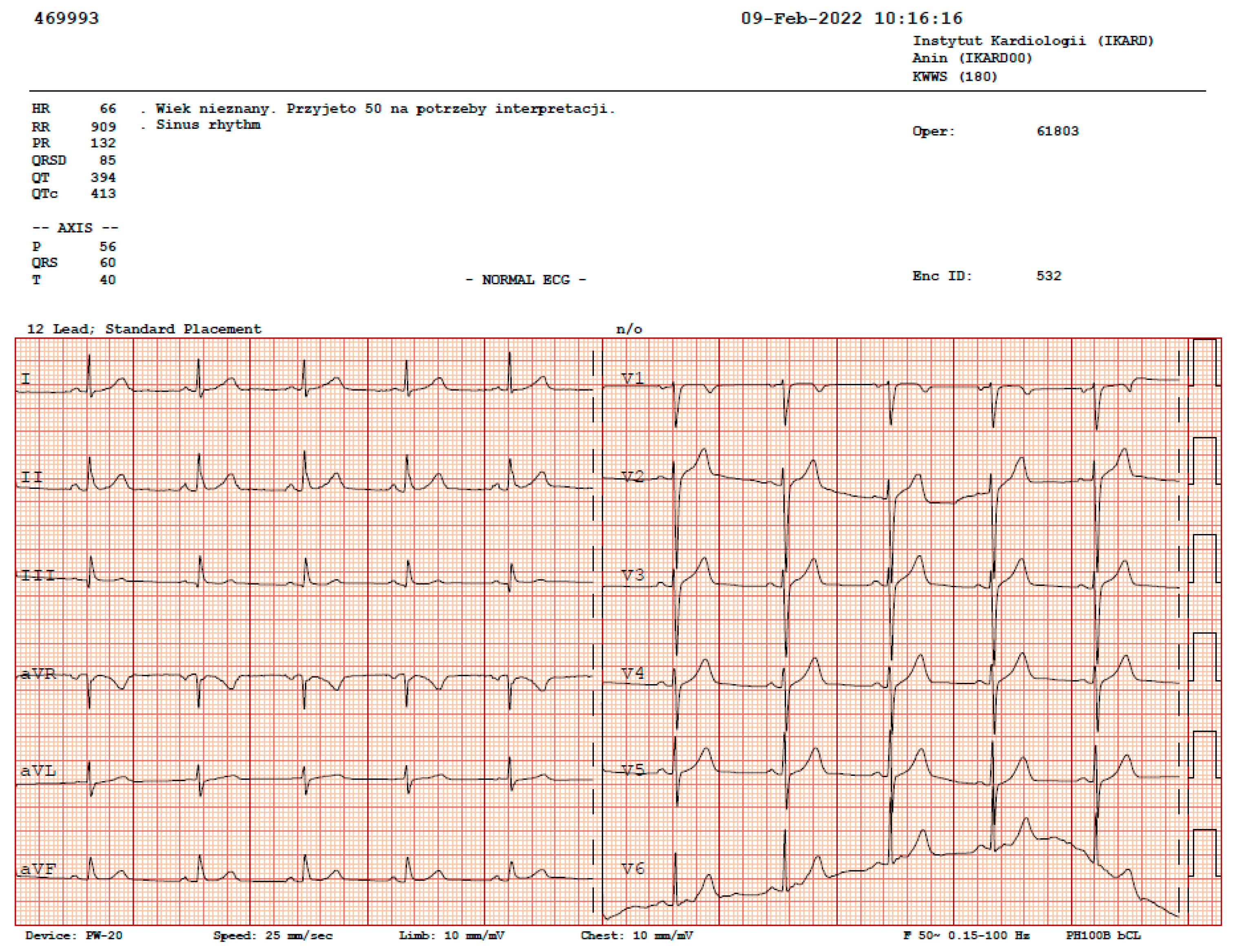
| Age (Yrs) | Discipline | Training—Number of Hours/Week | Number of Years in Training | Hours of Training in the Sports Discipline | Comments: |
|---|---|---|---|---|---|
| 9–20 | Soccer | 6 | 12 | 3700 | Probably healthy |
| 21–34 | Indoor soccer | 4 | 14 | 2900 | Probably healthy |
| 34–34 | Break–2 months | 0 | 0 | 0 | Pauses on HRM |
| 35–36 | Taekwondo | 6 | 2 | 600 | No control |
| 37–37 | Break–2 months | 0 | 0 | 0 | No control |
| 37–38 | Taekwondo | 6 | 4 | 1200 | No control |
| 39–40 | Break–2 years | 0 | 0 | 0 | No control |
| Total years spent training in different sports disciplines: 32 years | All disciplines | About 5.5 | 32 | 8400 (about) | About 8400 h in total, on average 5.5 h/week/32 years |
| Number | Hour | HR on Holter ECG | HR on HRM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Number of Recorded Beats | Mean (bpm) (s) | Min (bpm) (s) | Max (bpm) (s) | Pause > 4.0 s | Number of Recorded Beats | Mean (bpm) (s) | Min (bpm) (s) | Max (bpm) (s) | Pause > 4.0 s | ||
| 1 | 02:00 | 2966 | 49.43 | 33.01 | 77.12 | 6.310 | 2960 | 49.33 | 32.98 | 77.08 | 6.310 |
| 03:00 | 1.214 | 1.818 | 0.778 | 1.216 | 1.819 | 0.778 | |||||
| 2 | 00:00 | 3252 | 54.20 | 40.26 | 66.08 | 4.220 | 3268 | 54.47 | 40.27 | 66.04 | 4.202 |
| 01:00 | 1.107 | 1.490 | 0.908 | 1.102 | 1.490 | 0.909 | |||||
| 3 | 01:00 | 3117 | 51.95 | 46.32 | 75.11 | 5.215 | 3111 | 51.85 | 46.35 | 75.13 | 5.211 |
| 02:00 | 1.155 | 1.295 | 0.799 | 1.157 | 1.294 | 0.799 | |||||
| 4 | 05:00 | 3002 | 50.03 | 34.31 | 68.21 | 5.301 | 3010 | 50.17 | 34.29 | 68.19 | 5.310 |
| 06:00 | 1.199 | 1.749 | 0.880 | 1.196 | 1.750 | 0.880 | |||||
| 5 | 04:00 | 3089 | 51.48 | 41.07 | 86.42 | 4.190 | 3091 | 51.52 | 41.09 | 86.39 | 4.199 |
| 05:00 | 1.165 | 1.461 | 0.694 | 1.165 | 1.460 | 0.695 | |||||
| 6 | 00:00 | 3237 | 53.95 | 48.39 | 64.05 | 4.580 | 3242 | 54.03 | 48.41 | 64.04 | 4.589 |
| 01:00 | 1.112 | 1.240 | 0.937 | 1.110 | 1.239 | 0.937 | |||||
| 7 | 05:00 | 3287 | 54.78 | 48.09 | 75.23 | 4.803 | 3284 | 54.73 | 48.07 | 75.27 | 4.805 |
| 06:00 | 1.095 | 1.248 | 0.798 | 1.096 | 1.248 | 0.797 | |||||
| 8 | 02:00 | 3224 | 53.73 | 38.14 | 72.06 | 4.302 | 3215 | 53.58 | 38.16 | 72.09 | 4.309 |
| 03:00 | 1.117 | 1.573 | 0.833 | 1.120 | 1.572 | 0.832 | |||||
| 9 | 02:00 | 3279 | 54.65 | 38.32 | 89.16 | 4.009 | 3284 | 54.73 | 38.29 | 89.17 | 4.002 |
| 03:00 | 1.098 | 1.566 | 0.673 | 1.096 | 1.567 | 0.673 | |||||
| 10 | 03:00 | 3016 | 50.27 | 42.13 | 82.54 | 6.130 | 3020 | 50.33 | 42.16 | 82.52 | 6.135 |
| 04:00 | 1.194 | 1.424 | 0.727 | 1.192 | 1.423 | 0.727 | |||||
| Total: | 31,469 | Total: | 31,485 | ||||||||
| Parameters | Units (Normal Values) | 2016 Result 2022 | |
|---|---|---|---|
| Left ventricular end-diastolic volume | mL (106 ± 22) | 116 | 110 |
| Left ventricular end-systolic volume | mL (41 ± 10) | 36 | 35 |
| Ejection fraction two-dimensional (%) bi-plane | % (62 ± 5) | 65 | 65 |
| Global longitudinal strain | % (−20) | 20.6 | 20.8 |
| Interventricular septum diameter | mm (6–10) | 10 | 10 |
| Posterior wall diastolic diameter | mm (6–10) | 10 | 10 |
| Right ventricular end-diastolic diameter | mm (20–30) | 30 | 30 |
| S’ right ventricle | cm/s (14.1 ± 2.3) | 16 | 16 |
| Left atrium | mm (30–40) | 34 | 36 |
| Left atrial volume index | mL/m2 (16–34) | 28.0 | 28.4 |
| Right atrial area | cm2 (16 ± 5) | 14.0 | 14.0 |
| Mitral valve E-wave | cm/s (73 ± 19) | 78 | 80 |
| Mitral valve A-wave | cm/s (69 ± 17) | 50 | 52 |
| E’ lateral | cm/s (>10) | 20 | 18 |
| E’ septal | cm/s (>7) | 10 | 12 |
| E/e’ lateral | ratio (<15) | 4.0 | 4.2 |
| E/e’ septal | ratio (<13) | 7.0 | 7.2 |
| 2013 | 2021 | |
|---|---|---|
| Class | ||
| Cardiac Pacing for Bradycardia and Conduction System Disease | ||
| In patients with syncope, cardiac pacing may be considered to reduce recurrent syncope when asymptomatic pause(s) > 6 s due to sinus arrest are documented. | IIa | IIb |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gajda, R.; Knechtle, B.; Gębska-Kuczerowska, A.; Gajda, J.; Stec, S.; Krych, M.; Kwaśniewska, M.; Drygas, W. Amateur Athlete with Sinus Arrest and Severe Bradycardia Diagnosed through a Heart Rate Monitor: A Six-Year Observation—The Necessity of Shared Decision-Making in Heart Rhythm Therapy Management. Int. J. Environ. Res. Public Health 2022, 19, 10367. https://doi.org/10.3390/ijerph191610367
Gajda R, Knechtle B, Gębska-Kuczerowska A, Gajda J, Stec S, Krych M, Kwaśniewska M, Drygas W. Amateur Athlete with Sinus Arrest and Severe Bradycardia Diagnosed through a Heart Rate Monitor: A Six-Year Observation—The Necessity of Shared Decision-Making in Heart Rhythm Therapy Management. International Journal of Environmental Research and Public Health. 2022; 19(16):10367. https://doi.org/10.3390/ijerph191610367
Chicago/Turabian StyleGajda, Robert, Beat Knechtle, Anita Gębska-Kuczerowska, Jacek Gajda, Sebastian Stec, Michalina Krych, Magdalena Kwaśniewska, and Wojciech Drygas. 2022. "Amateur Athlete with Sinus Arrest and Severe Bradycardia Diagnosed through a Heart Rate Monitor: A Six-Year Observation—The Necessity of Shared Decision-Making in Heart Rhythm Therapy Management" International Journal of Environmental Research and Public Health 19, no. 16: 10367. https://doi.org/10.3390/ijerph191610367
APA StyleGajda, R., Knechtle, B., Gębska-Kuczerowska, A., Gajda, J., Stec, S., Krych, M., Kwaśniewska, M., & Drygas, W. (2022). Amateur Athlete with Sinus Arrest and Severe Bradycardia Diagnosed through a Heart Rate Monitor: A Six-Year Observation—The Necessity of Shared Decision-Making in Heart Rhythm Therapy Management. International Journal of Environmental Research and Public Health, 19(16), 10367. https://doi.org/10.3390/ijerph191610367








