Walking Promotion in Pregnancy and Its Effects on Insomnia: Results of Walking_Preg Project (WPP) Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Selection Criteria for Elegibility
2.2. Intervention
2.3. Collected Variables
- -
- Change in mean steps/day after intervention in the study arms, using pedometer register, at 19th and 31st GW;
- -
- Insomnia prevalence in every trimester of pregnancy, assessed by the Athens Insomnia Scale (AIS) [16], considering insomnia from a score equal to or greater than 7 points;
- -
- Quality of sleep in every trimester of pregnancy: assessed by the Pittsburgh scale (PSQI) [17]: a PSQI score of >5 indicates poor sleep quality.
2.4. Location and Data Collection
2.5. Randomization of Participants and Concealment
2.6. Data Analysis
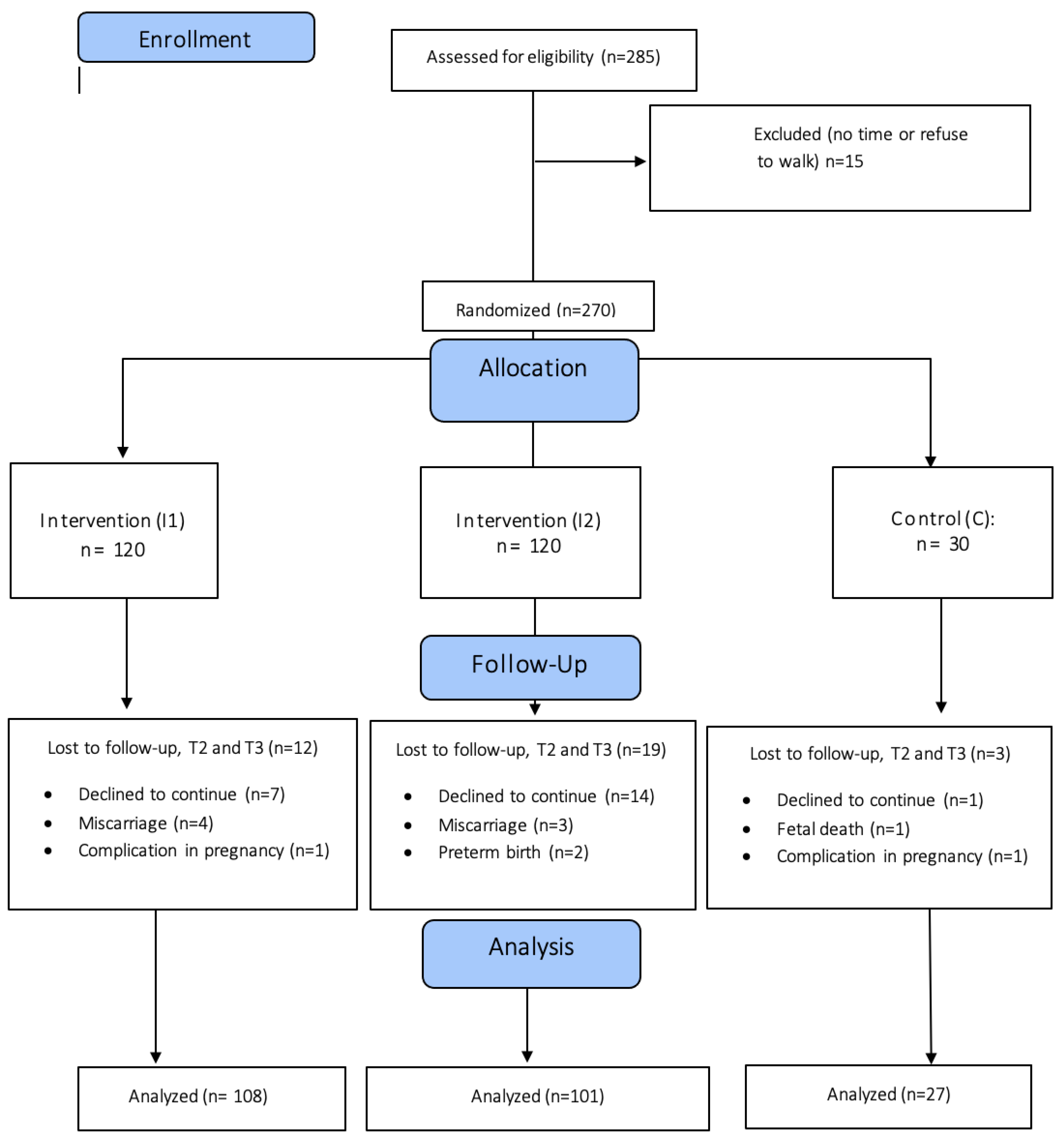
3. Results
3.1. Characteristics of the Sample
3.2. Adherence to the WPP Intervention
3.3. AIS (Insomnia) and Pitssburhg (Sleep Quality) Scores through Pregnancy in WPP Arms
3.4. Insomnia and Poor Quality Sleep Prevalence through Pregnancy in WPP Arms
3.5. Walking Intervention Effect on Insomnia and Quality of Sleep in Third Trimester of Pregnancy
4. Discussion
4.1. Main Results
4.2. Comparison with Other Studies
4.3. Limitations and Strenghs
4.4. Implication for Practice
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sateia, M.J. International Classification of Sleep Disorders-Third Edition. Chest 2014, 146, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- Amiri, S.; Hasani, J.; Satkin, M. Effect of exercise training on improving sleep disturbances: A systematic review and meta-analysis of randomized control trials. Sleep Med. 2021, 84, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Kredlow, M.A.; Capozzoli, M.C.; Hearon, B.A.; Calkins, A.W.; Otto, M.W. The effects of physical activity on sleep: A meta-analytic review. J. Behav. Med. 2015, 38, 427–449. [Google Scholar] [CrossRef]
- Nodine, P.M.; Matthews, E.E. Common sleep disorders: Management strategies and pregnancy outcomes. J. Midwifery Womens Health 2013, 58, 368–377. [Google Scholar] [CrossRef]
- Silvestri, R.; Aricò, I. Sleep disorders in pregnancy. Sleep Sci. 2019, 12, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Hashmi, A.M.; Bhatia, S.K.; Bhatia, S.K.; Khawaja, I.S. Insomnia during pregnancy: Diagnosis and Rational Interventions. Pak. J. Med. Sci. 2016, 32, 1030–1037. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.H.; Rothenberger, S.D.; Kline, C.E.; Okun, M.L. Exercise during early pregnancy is associated with greater sleep continuity. Behav. Sleep Med. 2018, 16, 482–493. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Zou, J.; Zhang, Y.; Yang, Q.; Shi, H. A Longitudinal Study of Physical Activity to Improve Sleep Quality during Pregnancy. Nat. Sci. Sleep 2020, 12, 431–442. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Blanque, R.; Sánchez-García, J.C.; Sánchez-López, A.M.; Mur-Villar, N.; Aguilar-Cordero, M.J. The influence of physical activity in water on sleep quality in pregnant women: A randomised trial. Women Birth 2018, 31, e51–e58. [Google Scholar] [CrossRef] [PubMed]
- Beddoe, A.E.; Lee, K.A.; Weiss, S.J.; Kennedy, H.P.; Yang, C.P. Effects of mindful yoga on sleep in pregnant women: A pilot study. Biol. Res. Nurs. 2010, 11, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Ba, T.; Og, S.; Of, A.; Afolabi, B. Effects of Aerobic Exercises on the Level of Insomnia and Fatigue in Pregnant Women. Internet J. Gynecol. Obstet. 2010, 15. [Google Scholar]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef]
- Doran, F.M.; Buckley, N.A. Walking patterns in pregnancy. Aust. J. Prim. Health 2013, 19, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Amezcua-Prieto, C.; Naveiro-Fuentes, M.; Arco-Jiménez, N.; Olmedo-Requena, R.; Barrios-Rodríguez, R.; Vico-Zúñiga, I.; Manzanares Galán, S.; Mozas-Moreno, J.; Jiménez-Moleón, J.J.; Gallo-Vallejo, J.L. Walking in pregnancy and prevention of insomnia in third trimester using pedometers: Study protocol of Walking_Preg project (WPP). A randomized controlled trial. BMC Pregnancy Childbirth 2020, 20, 521. [Google Scholar] [CrossRef] [PubMed]
- Opinion, A.C. Physical Activity and Exercise during Pregnancy and the Postpartum Period: ACOG Committee Opinion, Number 804. Obstet. Gynecol. 2020, 135, e178–e188. [Google Scholar]
- Gómez-Benito, J.; Ruiz, C.; Guilera, G. A Spanish version of the Athens Insomnia Scale. Qual. Life Res. 2011, 20, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F.; Monk, T.H., III; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Reichner, C.A. Insomnia and sleep deficiency in pregnancy. Obstet. Med. 2015, 8, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Roman-Galvez, R.M.; Amezcua-Prieto, C.; Salcedo-Bellido, I.; Martinez-Galiano, J.M.; Khan, K.S.; Bueno-Cavanillas, A. Factors associated with insomnia in pregnancy: A prospective Cohort Study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 221, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Borodulin, K.; Evenson, K.R.; Monda, K.; Wen, F.; Herring, A.H.; Dole, N. Physical activity and sleep among pregnant women. Paediatr. Perinat. Epidemiol. 2010, 24, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Nodine, P.M.; Leiferman, J.A.; Cook, P.F.; Matthews, E.E.; Hastings-Tolsma, M. The Impact of Physical Activity on Sleep during Pregnancy: A Secondary Analysis. Clin. Mother Child Health 2016, 13, 1–8. [Google Scholar] [CrossRef]
- Shen, W.C.; Chen, C.H. Effects of non-supervised aerobic exercise on sleep quality and maternal-fetal attachment in pregnant women: A randomized controlled trial. Complement. Ther. Med. 2021, 57, 102671. [Google Scholar] [CrossRef] [PubMed]
- Hassan Khedr, N.F.; Fadel, A.E. Effect of Practicing Walking with Deep Breathing Exercises on Insomnia among Women in Third Trimester of Pregnancy. Int. J. Nurs. Didact. 2018, 8, 19–26. [Google Scholar] [CrossRef]
- Kang, M.; Marshall, S.J.; Barreira, T.V.; Lee, J.O. Effect of pedometer-based physical activity interventions: A meta-analysis. Res. Q. Exerc. Sport 2009, 80, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Sanidad de España (Spain Ministery of Health, June 2022): Procedimiento de Actuación Para Los Servicios de Prevención de Riesgos Laborales Frente a La Exposición al Sars-Cov-2. (Action Procedure for Occupational Risk Prevention Services against Exposure to SARS-CoV-2). Available online: https://www.sanidad.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/documentos/Proteccion_Trabajadores_SARS-CoV-2.pdf (accessed on 5 July 2022).
- Gloster, A.T.; Lamnisos, D.; Lubenko, J.; Presti, G.; Squatrito, V.; Constantinou, M.; Nicolaou, C.; Papacostas, S.; Aydın, G.; Chong, Y.Y.; et al. Impact of COVID-19 pandemic on mental health: An international study. PLoS ONE 2020, 15, e0244809. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional de Estadística (Spanish National Statistical Institute, 2022). Available online: https://www.ine.es/consul/serie.do?d=true&s=EPA87 (accessed on 5 July 2022).
- Morin, C.M.; Bjorvatn, B.; Chung, F.; Holzinger, B.; Partinen, M.; Penzel, T.; Ivers, H.; Wing, Y.K.; Chan, N.Y.; Merikanto, I.; et al. Insomnia, anxiety, and depression during the COVID-19 pandemic: An international collaborative study. Sleep Med. 2021, 87, 38–45. [Google Scholar] [CrossRef]
- Drake, C.L.; Roehrs, T.; Roth, T. Insomnia causes, consequences, and therapeutics: An overview. Depress. Anxiety 2003, 18, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Conway, M.R.; Marshall, M.R.; Schlaff, R.A.; Pfeiffer, K.A.; Pivarnik, J.M. Physical Activity Device Reliability and Validity during Pregnancy and Postpartum. Med. Sci. Sports Exerc. 2018, 50, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Strycker, L.A.; Duncan, S.C.; Chaumeton, N.R.; Duncan, T.E.; Toobert, D.J. Reliability of pedometer data in samples of youth and older women. Int. J. Behav. Nutr. Phys. Act. 2007, 4, 4. [Google Scholar] [CrossRef] [PubMed]
| Variable |
Group I1 n = 120 |
Group I2 n = 120 | Group C n = 30 | |
|---|---|---|---|---|
| Age; Mean (SD) | 31.92 (4.92) | 31.91 (5.56) | 31.7 (5.36) | |
| Gestational Week at recruitment; Mean (SD) | 12.29 (0.063) | 12.21 (0.71) | 12.43 (0.73) | |
| Previous children; N (%) | 0 | 59 (49.58) | 60 (50.00) | 20 (66.67) |
| 1 | 43 (36.13) | 40 (33.33) | 8 (26.67) | |
| 2 | 14 (11.76) | 17 (14.17) | 2 (6.67) | |
| 3 | 3 (2.50) | 3 (2.50) | 0 (0.00) | |
| Children under 3 years old; N (%) | Yes | 8 (6.67) | 6 (5.00) | 1 (3.30) |
| No | 112 (93.33) | 114 (95) | 29 (96.67) | |
| Previous abortions; N (%) | 0 | 75 (62.50) | 74 (61.67) | 19 (63.33) |
| 1 | 33 (27.50) | 32 (26.67) | 9 (30.00) | |
| ≥2 | 12 (10.00) | 14 (11.67) | 2 (6.67) | |
| Nationality; N (%) | Spanish | 108 (90.76) | 109 (90.83) | 29 (96.67) |
| Other | 11 (9.24) | 11 (9.17) | 1 (3.33) | |
| Stable couple; N (%) | No | 1 (0.84) | 6 (5.00) | 2 (6.67) |
| Yes (non-cohabit) | 1 (0.84) | 2 (1.67) | 2 (6.67) | |
| Yes (cohabit) | 117 (98.32) | 112 (93.33) | 26 (86.67) | |
| Social class; N (%) | I | 14 (11.76) | 23 (19.17) | 3 (10.00) |
| II | 18 (15.13) | 16 (13.33) | 6 (20.00) | |
| III | 31 (26.05) | 20 (16.67) | 5 (16.67) | |
| IV | 39 (32.77) | 46 (38.33) | 12 (40) | |
| V | 17 (14.29) | 15 (12.50) | 4 (13.33) | |
| Pre-pregnancy BMI | Mean, SD | 25.86 (5.30) | 26.26 (5.76) | 25.46 (6.28) |
| Min–Max | 18.05–46.71 | 17.82–48.74 | 17.15–40.58 | |
| Pre-pregnancy BMI; N (%) | Underweight (<18.5) | 2 (1.68) | 2 (1.67) | 4 (13.33) |
| Normal (18.5–24.9) | 64 (53.78) | 59 (49.17) | 12 (40.00) | |
| Overweight (25–29.9) | 30 (25.21) | 30 (25.00) | 8 (26.67) | |
| Obese (30–34.9) | 14 (11.76) | 21 (17.50) | 3 (10.00) | |
| Extremely obese (>35) | 9 (7.56) | 8 (6.67) | 3 (10.00) |
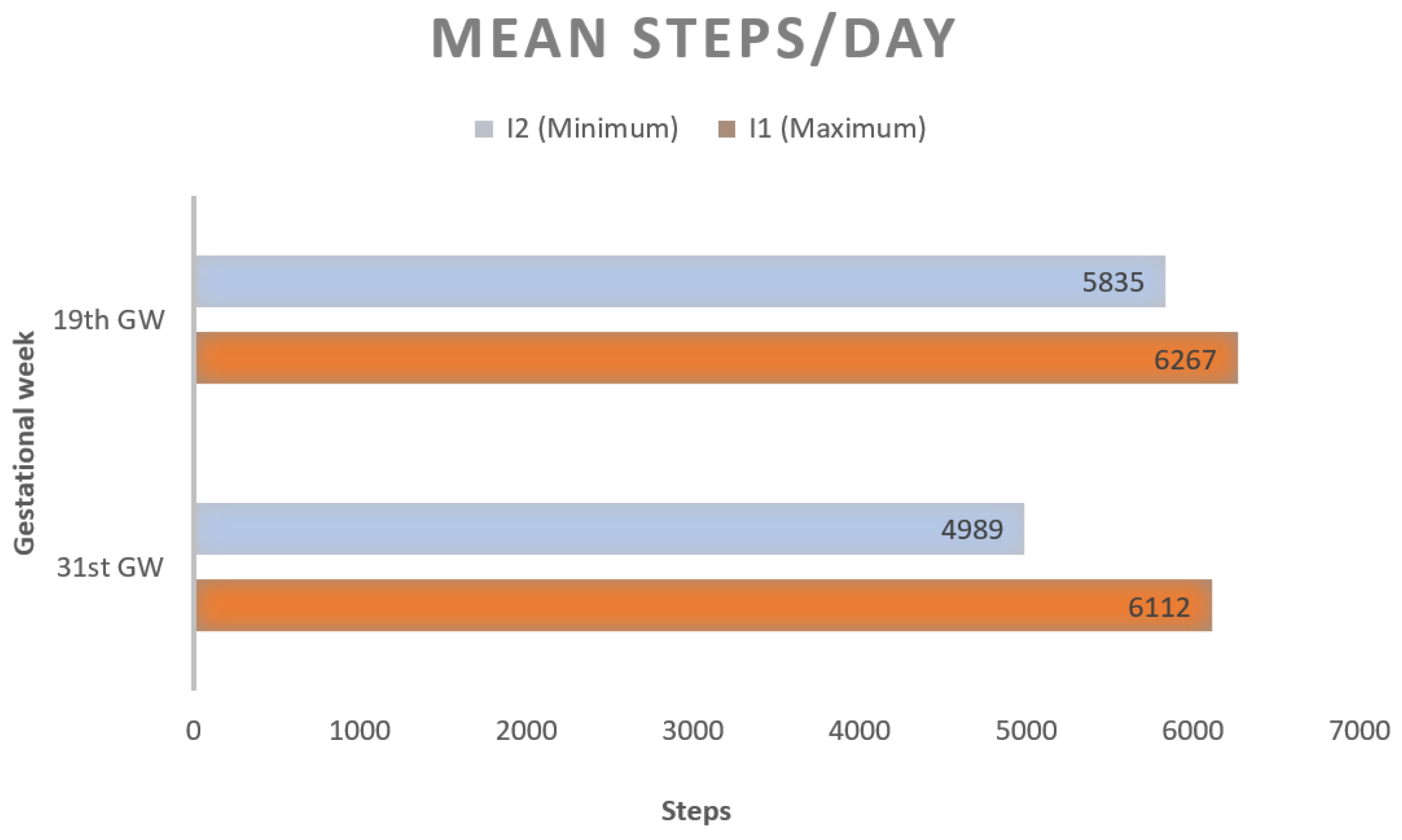
| Group I1 (Max) | Group I2 (Min) | Steps Difference | p1 | |
|---|---|---|---|---|
| Steps in 19th GW | ||||
| Mean | 6267 | 5835 | 432 | 0.347 |
| (SD) | (3854) | (2741) | ||
| Steps in 32nd GW | ||||
| Mean | 6112 | 4989 | 1123 | 0.022 |
| (SD) | (3954) | (2680) | ||
| Steps difference | −155 | −846 | ||
| p2 | 0.749 | 0.001 |
| AIS Score Mean (SD); Range | p1 | PSQI Score Mean (SD); Range | p1 | |||||
|---|---|---|---|---|---|---|---|---|
| Group I1 | Group I2 | Group C | Group I1 | Group I2 | Group C | |||
| 1st trimester of pregnancy | 3.06 (3.90) range 0–21 | 3.37 (4.53) range 0–22 | 3.40 (4.67) range 0–16 | 0.206 | 7.89 (2.98) range 3–17 | 7.80 (2.77) range 3–15 | 8.50 (3.26) range 3–15 | 0.472 |
| 2nd trimester of pregnancy | 3.22 (4.40) range 0–24 | 3.55 (5.14) range 0–23 | 4.35 (5.75) range 0–17 | 0.107 | 7.99 (2.53) range 3–15 | 8.36 (2.64) range 4–16 | 8.32 (2.65) range 4–14 | 0.893 |
| 3rd trimester of pregnancy | 5.59 (5.42) range 0–22 | 6.86 (6.06) range 0–24 | 5.93 (5.71) range 0–18 | 0.519 | 9.55 (3.02) range 4–19 | 9.90 (2.83) range 4–19.5 | 10.36 (2.85) range 5–16 | 0.795 |
| p2 | <0.001 | <0.001 | 0.211 | <0.001 | <0.001 | 0.001 | ||
| Insomnia N (%) (AIS ≥ 8) | p1 | Poor Sleep Quality N (%) (PSQI ≥ 5) | p1 | |||||
|---|---|---|---|---|---|---|---|---|
| Group I1 | Group I2 | Group C | Group I1 | Group I2 | Group C | |||
| 1st trimester of pregnancy | 16 (13.6) | 17 (14.3) | 6 (20.0) | 0.776 | 92 (76.7) | 90 (75.0) | 22 (73.3) | 0.837 |
| 2nd trimester of pregnancy | 19 (16.7) | 20 (18.5) | 7 (25.0) | 0.434 | 101 (84.2) | 104 (86.7) | 25 (83.3) | 0.823 |
| 3rd trimester of pregnancy | 41 (37.6) | 43 (42.2) | 9 (32.1) | 0.283 | 113 (94.2) | 117 (97.5) | 29 (96.7) | 0.416 |
| 1st vs. 2nd; p2 | 0.446 | 0.574 | 0.695 | 0.165 | 0.010 | 0.165 | ||
| 1st vs. 3rd; p2 | <0.001 | <0.001 | 0.239 | <0.001 | <0.001 | 0.001 | ||
| 2nd vs. 3rd; p2 | <0.001 | <0.001 | 0.433 | 0.011 | 0.006 | 0.064 | ||
| Insomnia in Third Trimester (T3) | Sleep Quality in Third Trimester (T3) | |||||||
|---|---|---|---|---|---|---|---|---|
| ß Coef. | 95% CI | p | ß Coef. | IC 95% | p | |||
| Crude models | Steps T3 | −7.02 × 10−5 | (−3.01 × 10−4–0.0) | 0.549 | Steps T3 | −9.66 × 10−5 | (−2.16 × 10−4–0.00) | 0.113 |
| Adjusted models | Steps T3 * | 0.7 × 10−4 | (−1.4 × 10−4–2.8 × 10−4) | 0.515 | Steps T3 ** | −8.93 × 10−5 | (−1.98 × 10−4–0.00) | 0.109 |
| AIS2 | 0.53 | (0.38–0.68) | <0.001 | PSQI2 | 0.53 | (0.38–0.67) | <0.001 | |
| Group 1_2 | 1.14 | (−0.31–2.59) | 0.123 | Group 1_2 | −8.73 × 10−5 | (−8.4 × 10−1–0.67) | 0.820 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benito-Villena, R.; Guerrero-Martínez, I.; Naveiro-Fuentes, M.; Cano-Ibánez, N.; Femia-Marzo, P.; Gallo-Vallejo, J.L.; Mozas-Moreno, J.; Amezcua-Prieto, C. Walking Promotion in Pregnancy and Its Effects on Insomnia: Results of Walking_Preg Project (WPP) Clinical Trial. Int. J. Environ. Res. Public Health 2022, 19, 10012. https://doi.org/10.3390/ijerph191610012
Benito-Villena R, Guerrero-Martínez I, Naveiro-Fuentes M, Cano-Ibánez N, Femia-Marzo P, Gallo-Vallejo JL, Mozas-Moreno J, Amezcua-Prieto C. Walking Promotion in Pregnancy and Its Effects on Insomnia: Results of Walking_Preg Project (WPP) Clinical Trial. International Journal of Environmental Research and Public Health. 2022; 19(16):10012. https://doi.org/10.3390/ijerph191610012
Chicago/Turabian StyleBenito-Villena, Rebeca, Ingrid Guerrero-Martínez, Mariña Naveiro-Fuentes, Naomi Cano-Ibánez, Pedro Femia-Marzo, José Luis Gallo-Vallejo, Juan Mozas-Moreno, and Carmen Amezcua-Prieto. 2022. "Walking Promotion in Pregnancy and Its Effects on Insomnia: Results of Walking_Preg Project (WPP) Clinical Trial" International Journal of Environmental Research and Public Health 19, no. 16: 10012. https://doi.org/10.3390/ijerph191610012
APA StyleBenito-Villena, R., Guerrero-Martínez, I., Naveiro-Fuentes, M., Cano-Ibánez, N., Femia-Marzo, P., Gallo-Vallejo, J. L., Mozas-Moreno, J., & Amezcua-Prieto, C. (2022). Walking Promotion in Pregnancy and Its Effects on Insomnia: Results of Walking_Preg Project (WPP) Clinical Trial. International Journal of Environmental Research and Public Health, 19(16), 10012. https://doi.org/10.3390/ijerph191610012








