Seawater Hydration Modulates IL-6 and Apelin Production during Triathlon Events: A Crossover Randomized Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects and Ethics
2.2. Experimental Design
2.3. Experimental Trial
2.4. Anthropometric and Physiological Variables
2.5. Sample Collection
2.6. Hematological Measurements
2.7. Cytokine and Myokine Proteins Multiplex Analysis
2.8. Statistical Analysis
3. Results
3.1. Participants’ Anthropometric and Physiological Characteristics
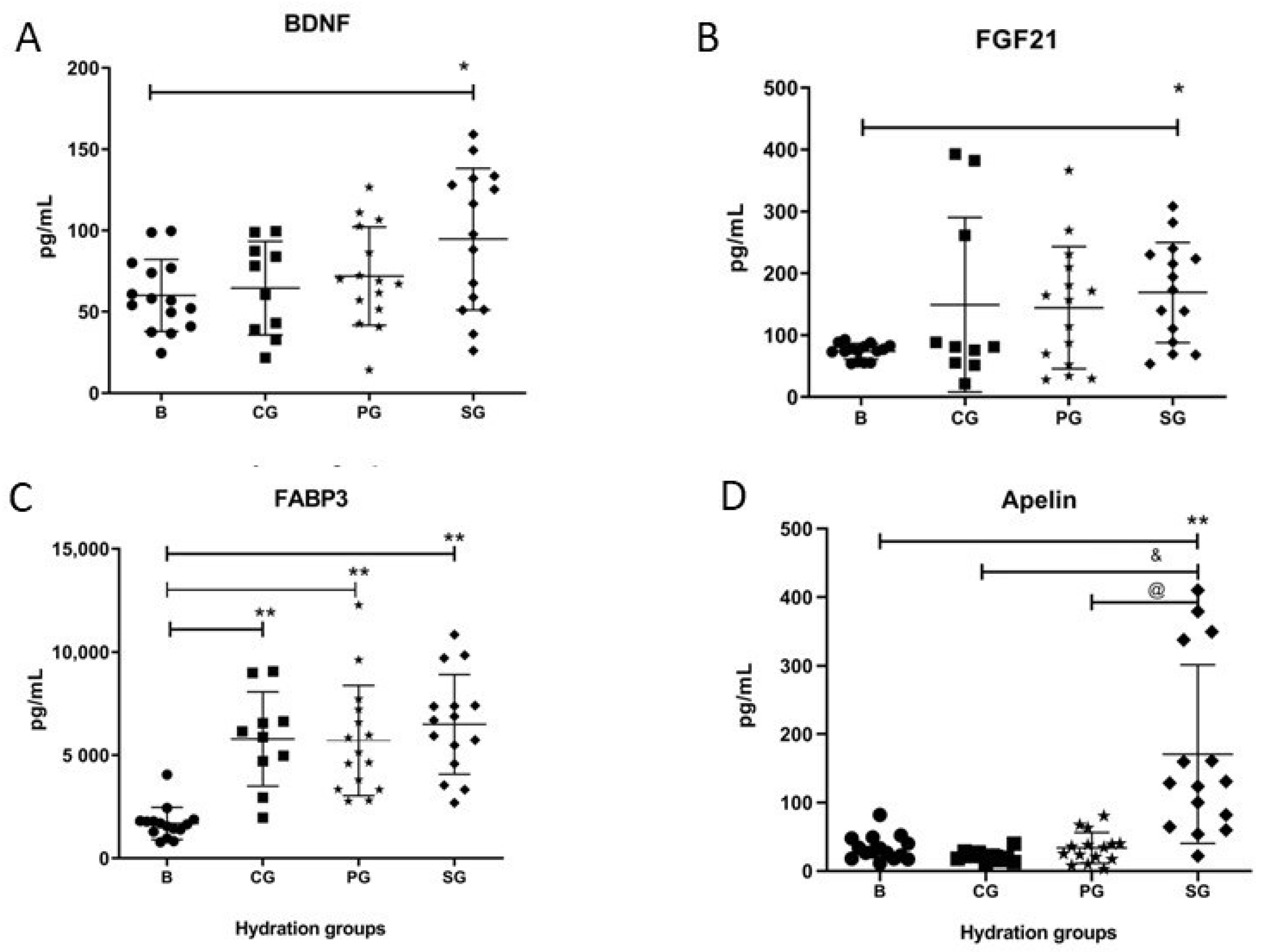
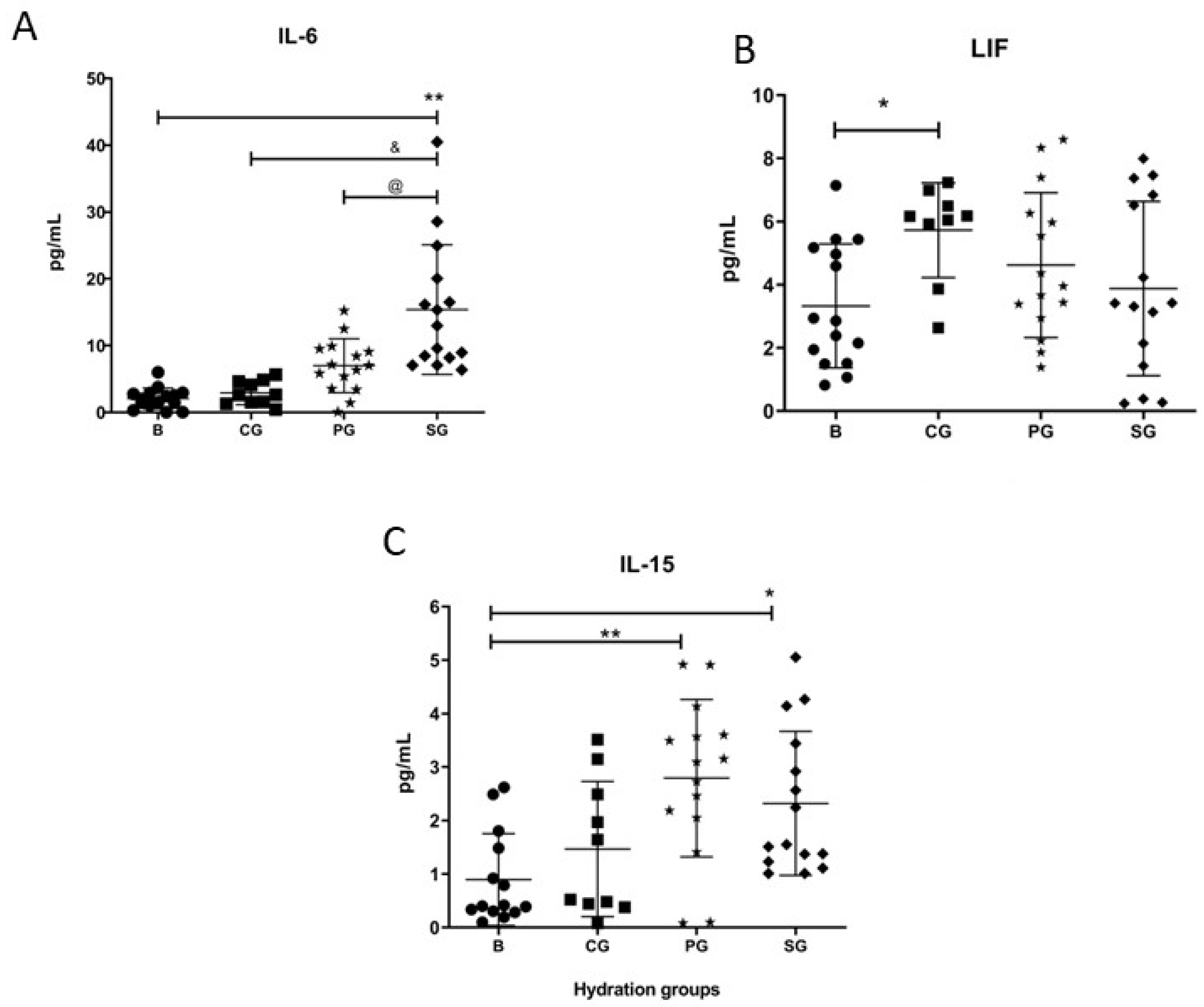
3.2. Alterations in Blood Cytokine and Myokine Protein Levels
4. Discussion
4.1. Anthropometric, Performance, and Effort Parameters
4.2. Cytokines, Myokines, and Hormones Values
4.3. Limitations and Strength
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Sawka, M.N.; Timothy, D. Noakes Does Dehydration Impair Exercise Performance? Med. Sci. Sports Exerc. 2007, 39, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Goulet, E.D.B.; Hoffman, M.D. Impact of Ad Libitum versus Programmed Drinking on Endurance Performance: A Systematic Review with Meta-Analysis. Sports Med. 2019, 49, 221–232. [Google Scholar] [CrossRef]
- José, G.-A.; Mora-Rodríguez, R.; Below, P.R.; Coyle, E.F. Dehydration markedly impairs cardiovascular function in hyperthermic endurance athletes during exercise. J. Appl. Physiol. 1997, 82, 1229–1236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hew-Butler, T.; Rosner, M.H.; Fowkes-Godek, S.; Dugas, J.P.; Hoffman, M.D.; Lewis, D.P.; Maughan, R.J.; Miller, K.C.; Montain, S.J.; Rehrer, N.J.; et al. Statement of the Third International Exercise-Associated Hyponatremia Consensus Development Conference, Carlsbad, California, 2015. Clin. J. Sports Med. 2015, 25, 303–320. [Google Scholar] [CrossRef]
- Lau, W.Y.; Kato, H.; Nosaka, K. Effect of oral rehydration solution versus spring water intake during exercise in the heat on muscle cramp susceptibility of young men. J. Int. Soc. Sports Nutr. 2021, 18, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Keen, D.A.; Constantopoulos, E.; Konhilas, J.P. The impact of post-exercise hydration with deep-ocean mineral water on rehydration and exercise performance. J. Int. Soc. Sports Nutr. 2016, 13, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, C.-W.; Tsai, Y.-S.; Jean, W.-H.; Chen, C.-Y.; Ivy, J.L.; Huang, C.-Y.; Kuo, C.-H. Deep Ocean mineral water accelerates recovery from physical fatigue. J. Int. Soc. Sports Nutr. 2013, 10, 7. [Google Scholar] [CrossRef] [Green Version]
- Murray, B. Hydration and Physical Performance. J. Am. Coll. Nutr. 2007, 26, 542S–548S. [Google Scholar] [CrossRef]
- Stasiule, L.; Capkauskiene, S.; Vizbaraite, D.; Stasiulis, A. Deep mineral water accelerates recovery after dehydrating aerobic exercise: A randomized, double-blind, placebo-controlled crossover study. J. Int. Soc. Sports Nutr. 2014, 11, 34. [Google Scholar] [CrossRef] [Green Version]
- Nieman, D.C.; Pedersen, B.K. Minerals and Exercise Immunology. In Nutrition and Exercise Immunology; London, B.R., Ed.; CRC Press: New York, NY, USA, 2000; p. 208. ISBN 9780367398798. [Google Scholar]
- Mueller, S.M.; Anliker, E.; Knechtle, P.; Knechtle, B.; Toigo, M. Changes in body composition in triathletes during an Ironman race. Eur. J. Appl. Physiol. 2013, 113, 2343–2352. [Google Scholar] [CrossRef]
- Castizo-Olier, J.; Carrasco-Marginet, M.; Roy, A.; Chaverri, D.; Iglesias, X.; Pérez-Chirinos, C.; Rodríguez, F.; Irurtia, A. Bioelectrical Impedance Vector Analysis (BIVA) and Body Mass Changes in an Ultra-Endurance Triathlon Event. J. Sports Sci. Med. 2018, 17, 571–579. [Google Scholar] [PubMed]
- Maughan, R.J.; Merson, S.J.; Broad, N.P.; Shirreffs, S.M. Fluid and Electrolyte Intake and Loss in Elite Soccer Players during Training. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 333–346. [Google Scholar] [CrossRef] [PubMed]
- Cheuvront, S.N.; Kenefick, R.W. Dehydration: Physiology, Assessment, and Performance Effects. In Comprehensive Physiology; Wiley: Hoboken, NJ, USA, 2014; pp. 257–285. [Google Scholar]
- Luk, H.-Y.; Levitt, D.E.; Lee, E.C.; Ganio, M.S.; McDermott, B.P.; Kupchak, B.R.; McFarlin, B.K.; Hill, D.W.; Armstrong, L.E.; Vingren, J.L. Pro- and anti-inflammatory cytokine responses to a 164-km road cycle ride in a hot environment. Eur. J. Appl. Physiol. 2016, 116, 2007–2015. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, K.; Rohde, T.; Asp, S.; Schjerling, P.; Pedersen, B.K. Pro- and anti-inflammatory cytokine balance in strenuous exercise in humans. J. Physiol. 1999, 515, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Besse-Patin, A.; Montastier, E.; Vinel, C.; Castan-Laurell, I.; Louche, K.; Dray, C.; Daviaud, D.; Mir, L.; Marques, M.-A.; Thalamas, C.; et al. Effect of endurance training on skeletal muscle myokine expression in obese men: Identification of apelin as a novel myokine. Int. J. Obes. 2014, 38, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Severinsen, M.C.K.; Pedersen, B.K. Muscle–Organ Crosstalk: The Emerging Roles of Myokines. Endocr. Rev. 2020, 41, 594–609. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.P. Interleukin-6 in acute exercise and training: What is the biological relevance? Exerc. Immunol. Rev. 2006, 12, 6–33. [Google Scholar]
- Petersen, A.M.W.; Pedersen, B.K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 2005, 98, 1154–1162. [Google Scholar] [CrossRef] [Green Version]
- Lu, C.-H.; Shen, C.-Y.; Hsieh, D.J.-Y.; Lee, C.-Y.; Chang, R.-L.; Ju, D.-T.; Pai, P.-Y.; Viswanadha, V.P.; Ou, H.-C.; Huang, C.-Y. Deep ocean minerals inhibit IL-6 and IGFIIR hypertrophic signaling pathways to attenuate diabetes-induced hypertrophy in rat hearts. J. Appl. Physiol. 2019, 127, 356–364. [Google Scholar] [CrossRef]
- Pérez-Turpin, J.A.; Trottini, M.; Chinchilla-Mira, J.J.; Cyganik, W. Effects of seawater ingestion on lactate response to exercise in runners. Biol. Sport 2017, 34, 407–412. [Google Scholar] [CrossRef] [Green Version]
- Rowlands, D.S.; Houltham, S.D. Multiple-Transportable Carbohydrate Effect on Long-Distance Triathlon Performance. Med. Sci. Sports Exerc. 2017, 49, 1734–1744. [Google Scholar] [CrossRef] [PubMed]
- Bejder, J.; Andersen, A.B.; Buchardt, R.; Larsson, T.H.; Olsen, N.V.; Nordsborg, N.B. Endurance, aerobic high-intensity, and repeated sprint cycling performance is unaffected by normobaric “Live High-Train Low”: A double-blind placebo-controlled cross-over study. Eur. J. Appl. Physiol. 2017, 117, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Dill, D.B.; Costill, D.L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 1974, 37, 247–248. [Google Scholar] [CrossRef] [Green Version]
- Kenefick, R.W. Drinking Strategies: Planned Drinking Versus Drinking to Thirst. Sports Med. 2018, 48, 31–37. [Google Scholar] [CrossRef] [Green Version]
- Higgins, M.F.; Rudkin, B.; Kuo, C.-H. Oral Ingestion of Deep Ocean Minerals Increases High-Intensity Intermittent Running Capacity in Soccer Players after Short-Term Post-Exercise Recovery: A Double-Blind, Placebo-Controlled Crossover Trial. Mar. Drugs 2019, 17, 309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pedersen, B.K. Muscles and their myokines. J. Exp. Biol. 2011, 214, 337–346. [Google Scholar] [CrossRef] [Green Version]
- Jorgensen, S.B. How is AMPK activity regulated in skeletal muscles during exercise? Front. Biosci. 2008, 13, 5589. [Google Scholar] [CrossRef] [Green Version]
- Wojtaszewski, J.F.P.; MacDonald, C.; Nielsen, J.N.; Hellsten, Y.; Hardie, D.G.; Kemp, B.E.; Kiens, B.; Richter, E.A. Regulation of 5′AMP-activated protein kinase activity and substrate utilization in exercising human skeletal muscle. Am. J. Physiol. Metab. 2003, 284, E813–E822. [Google Scholar] [CrossRef] [Green Version]
- MacDonald, C.; Wojtaszewski, J.F.P.; Pedersen, B.K.; Kiens, B.; Richter, E.A. Interleukin-6 release from human skeletal muscle during exercise: Relation to AMPK activity. J. Appl. Physiol. 2003, 95, 2273–2277. [Google Scholar] [CrossRef] [Green Version]
- Ha, B.G.; Park, J.-E.; Cho, H.-J.; Shon, Y.H. Stimulatory Effects of Balanced Deep Sea Water on Mitochondrial Biogenesis and Function. PLoS ONE 2015, 10, e0129972. [Google Scholar] [CrossRef] [Green Version]
- Ha, B.G.; Moon, D.-S.; Kim, H.J.; Shon, Y.H. Magnesium and calcium-enriched deep-sea water promotes mitochondrial biogenesis by AMPK-activated signals pathway in 3T3-L1 preadipocytes. Biomed. Pharmacother. 2016, 83, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, L.; Patten, D.A.; Caron, A.; Garneau, L.; Pinault-Masson, E.; Foretz, M.; Haddad, P.; Anderson, B.G.; Quinn, L.S.; Jardine, K.; et al. IL-15 improves skeletal muscle oxidative metabolism and glucose uptake in association with increased respiratory chain supercomplex formation and AMPK pathway activation. Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 395–407. [Google Scholar] [CrossRef] [PubMed]
- Matthews, V.B.; Åström, M.-B.; Chan, M.H.S.; Bruce, C.R.; Krabbe, K.S.; Prelovsek, O.; Åkerström, T.; Yfanti, C.; Broholm, C.; Mortensen, O.H.; et al. Brain-derived neurotrophic factor is produced by skeletal muscle cells in response to contraction and enhances fat oxidation via activation of AMP-activated protein kinase. Diabetologia 2009, 52, 1409–1418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinel, C.; Lukjanenko, L.; Batut, A.; Deleruyelle, S.; Pradère, J.-P.; Le Gonidec, S.; Dortignac, A.; Geoffre, N.; Pereira, O.; Karaz, S.; et al. The exerkine apelin reverses age-associated sarcopenia. Nat. Med. 2018, 24, 1360–1371. [Google Scholar] [CrossRef] [PubMed]
- Sterbova, S.; Karlsson, T.; Persson, E. Oncostatin M induces tumorigenic properties in non-transformed human prostate epithelial cells, in part through activation of signal transducer and activator of transcription 3 (STAT3). Biochem. Biophys. Res. Commun. 2018, 498, 769–774. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-S.; Galvão, D.A.; Newton, R.U.; Gray, E.; Taaffe, D.R. Exercise-induced myokines and their effect on prostate cancer. Nat. Rev. Urol. 2021, 18, 519–542. [Google Scholar] [CrossRef]
- Farrash, W.F.; Phillips, B.E.; Britton, S.L.; Qi, N.; Koch, L.G.; Wilkinson, D.J.; Smith, K.; Atherton, P.J. Myokine Responses to Exercise in a Rat Model of Low/High Adaptive Potential. Front. Endocrinol. 2021, 12, 645881. [Google Scholar] [CrossRef]
- Timmons, J.A.; Baar, K.; Davidsen, P.K.; Atherton, P.J. Is irisin a human exercise gene? Nature 2012, 488, E9–E10. [Google Scholar] [CrossRef]
- Broholm, C.; Mortensen, O.H.; Nielsen, S.; Akerstrom, T.; Zankari, A.; Dahl, B.; Pedersen, B.K. Exercise induces expression of 460 leukaemia inhibitory factor in human skeletal muscle. J. Physiol. 2008, 586, 2195–2201. [Google Scholar] [CrossRef]
| Control (n = 10) | Experimental—Placebo (n = 15) | |
|---|---|---|
| Age (years) | 40.70 ± 5.89 | 38.80 ± 5.62 |
| Body height (cm) | 176.60 ± 8.04 | 176.00 ± 6.43 |
| Body mass (kg) | 70.74 ± 12.28 | 73.26 ± 10.68 |
| Total Body Water (kg) | 46.54 ± 6.58 | 46.17 ± 5.16 |
| Protein (kg) | 12.72 ± 1.84 | 12.55 ± 1.39 |
| Body Fat Mass (kg) | 7.19 ± 3.92 | 10.22 ± 5.29 |
| Body Mass Index (kg/m2) | 22.58 ± 2.51 | 23.54 ± 2.34 |
| VO2max (mL/kg/min) | 45.31 ± 9.52 | 43.46 ± 10.43 |
| Swim | Bike | Run | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variables | Basal | PG | CG | SG | PG | CG | SG | PG | CG | SG | ||||||||||||||||||||
| Haematocrit | 46.6 | ± | 3.41 | 45.80 | ± | 3.55 | 47.50 | ± | 3.06 | 45.93 | ± | 3.71 | 46.40 | ± | 3.48 | 47.5 * | ± | 2.95 | 45.47 | ± | 3.07 | 45.20 | ± | 3.03 | 47.88 # | ± | 2.09 | 46.33 | ± | 3.11 |
| Haemoglobin | 12.2 | ± | 3.10 | 11.73 | ± | 3.45 | 14.23 | ± | 1.36 | 11.79 | ± | 1.74 | 12.28 | ± | 3.75 | 14.69 | ± | 1.42 | 11.09 | ± | 1.94 | 11.55 | ± | 1.28 | 15.16 | ± | 2.71 | 12.04 | ± | 1.81 |
| Hrmax (bpm) | xxx | ± | xxx | 143.00 | ± | 7.74 | 165.00 | ± | 8.19 | 145.40 | ± | 6.02 | 181.67 | ± | 5.29 | 160.80 | ± | 11.7 | 176.93 | ± | 4.70 | 173.93 | ± | 6.24 | 170.40 | ± | 9.61 | 177.87 | ± | 8.45 |
| Hrmean (bpm) | 61.9 | ± | 10.5 | 130.49 | ± | 34.7 | 139.10 | ± | 10.3 | 135.06 | ± | 7.13 | 162.98 | ± | 42.9 | 137.70 | ± | 10.5 | 135.06 | ± | 7.13 | 162.98 | ± | 42.91 | 155.10 | ± | 9.93 | 149.30 | ± | 7.19 |
| Hrpost (bpm) | xxx | ± | xxx | 105.93 | ± | 14 | 99.40 | ± | 19.2 | 108.73 | ± | 15.5 | 181.67 | ± | 5.29 | 149.90 | ± | 7.26 | 150.33 | ± | 8.64 | 169.67 | ± | 5.74 | 171.10 | ± | 3.21 | 175.27 | ± | 9.16 |
| SBP (mmHg) | 127 | ± | 12.6 | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | 117 | ± | 15.30 | 110 * | ± | 11.3 | 122 | ± | 15.4 |
| DBP (mmHg) | 74 | ± | 8.44 | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | 68 | ± | 7.93 | 66 | ± | 4.45 | 70 | ± | 11.5 |
| Lactate (mmol/L) | 1.68 | ± | 0.36 | 5.85 | ± | 1.68 | 7.10 | ± | 2.66 | 5.86 | ± | 2.22 | 2.47 | ± | 1.57 | 2.46 | ± | 1.16 | 3.19 | ± | 1.85 | 2.83 | ± | 1.59 | 2.92 | ± | 0.95 | 2.71 | ± | 0.62 |
| RPE | xxx | ± | xxx | 7.05 | ± | 0.68 | 7.94 | ± | 0.93 | 6.70 | ± | 0.67 | 7.80 | ± | 0.42 | 8.40 | ± | 0.54 | 8.00 | ± | 1.30 | 9.00 | ± | 0.81 | 8.87 | ± | 0.54 | 9.15 | ± | 0.88 |
| Perfor. (min) | xxx | ± | xxx | 14.68 | ± | 1.45 | 14.55 | ± | 1.39 | 14.55 | ± | 1.24 | 165.70 | ± | 12.9 | 157.20 | ± | 8.02 | 165.60 | ± | 12.8 | 51.13 | ± | 9.59 | 48.61 | ± | 6.46 | 51.95 | ± | 7.95 |
| Weight (kg) | 71.8 | ± | 12.0 | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | xxx | ± | xxx | 70.86 | ± | 11.63 | 69.15 | ± | 11.6 | 70.98 | ± | 11.5 |
| Myokines | Rest | Control Group | Placebo Group | Experimental Group | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fractalkine | 419.70 | ± | 194.30 | 435.3 | ± | 239.00 | 405.30 | ± | 169.40 | 341.10 | ± | 145.70 |
| EPO | 354.00 | ± | 206.00 | 146.00 | ± | 84.4 | 229.00 | ± | 251.00 | 256.00 | ± | 242.00 |
| Osteonectin | 153.00 | ± | 54.30 | 175.00 | ± | 44.00 | 171.00 | ± | 60.00 | 199.00 | ± | 78.90 |
| Myostatin | 5.999 | ± | 14.156 | 8.824 | ± | 12.982 | 11.580 | ± | 18.253 | 11.599 | ± | 21.667 |
| Oncostatin | 2.4 | ± | 3.8 | 1.29 | ± | 1.1 | 3.0 | ± | 6.0 | 3.8 | ± | 7.7 |
| FSTL1 | 491 | ± | 297 | 198 | ± | 117 | 464 | ± | 299 | 624 | ± | 579 |
| Irisin | 746 | ± | 601 | 367 | ± | 295 | 353 | ± | 445 | 341 * | ± | 343 |
| Osteocrin | 7.93 | ± | 3.44 | 5.48 | ± | 3.89 | 16.40 | ± | 17.40 | 17.00 | ± | 13.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acevedo, O.G.; Aragón-Vela, J.; De la Cruz Márquez, J.C.; Marín, M.M.; Casuso, R.A.; Huertas, J.R. Seawater Hydration Modulates IL-6 and Apelin Production during Triathlon Events: A Crossover Randomized Study. Int. J. Environ. Res. Public Health 2022, 19, 9581. https://doi.org/10.3390/ijerph19159581
Acevedo OG, Aragón-Vela J, De la Cruz Márquez JC, Marín MM, Casuso RA, Huertas JR. Seawater Hydration Modulates IL-6 and Apelin Production during Triathlon Events: A Crossover Randomized Study. International Journal of Environmental Research and Public Health. 2022; 19(15):9581. https://doi.org/10.3390/ijerph19159581
Chicago/Turabian StyleAcevedo, Olivia González, Jerónimo Aragón-Vela, Juan Carlos De la Cruz Márquez, Manuel Martínez Marín, Rafael A. Casuso, and Jesús R. Huertas. 2022. "Seawater Hydration Modulates IL-6 and Apelin Production during Triathlon Events: A Crossover Randomized Study" International Journal of Environmental Research and Public Health 19, no. 15: 9581. https://doi.org/10.3390/ijerph19159581
APA StyleAcevedo, O. G., Aragón-Vela, J., De la Cruz Márquez, J. C., Marín, M. M., Casuso, R. A., & Huertas, J. R. (2022). Seawater Hydration Modulates IL-6 and Apelin Production during Triathlon Events: A Crossover Randomized Study. International Journal of Environmental Research and Public Health, 19(15), 9581. https://doi.org/10.3390/ijerph19159581










