Executive Function-Related Improvements on a Commercial CBT-Based Weight Management Intervention: Pilot Randomized Controlled Trial
Abstract
:1. Introduction
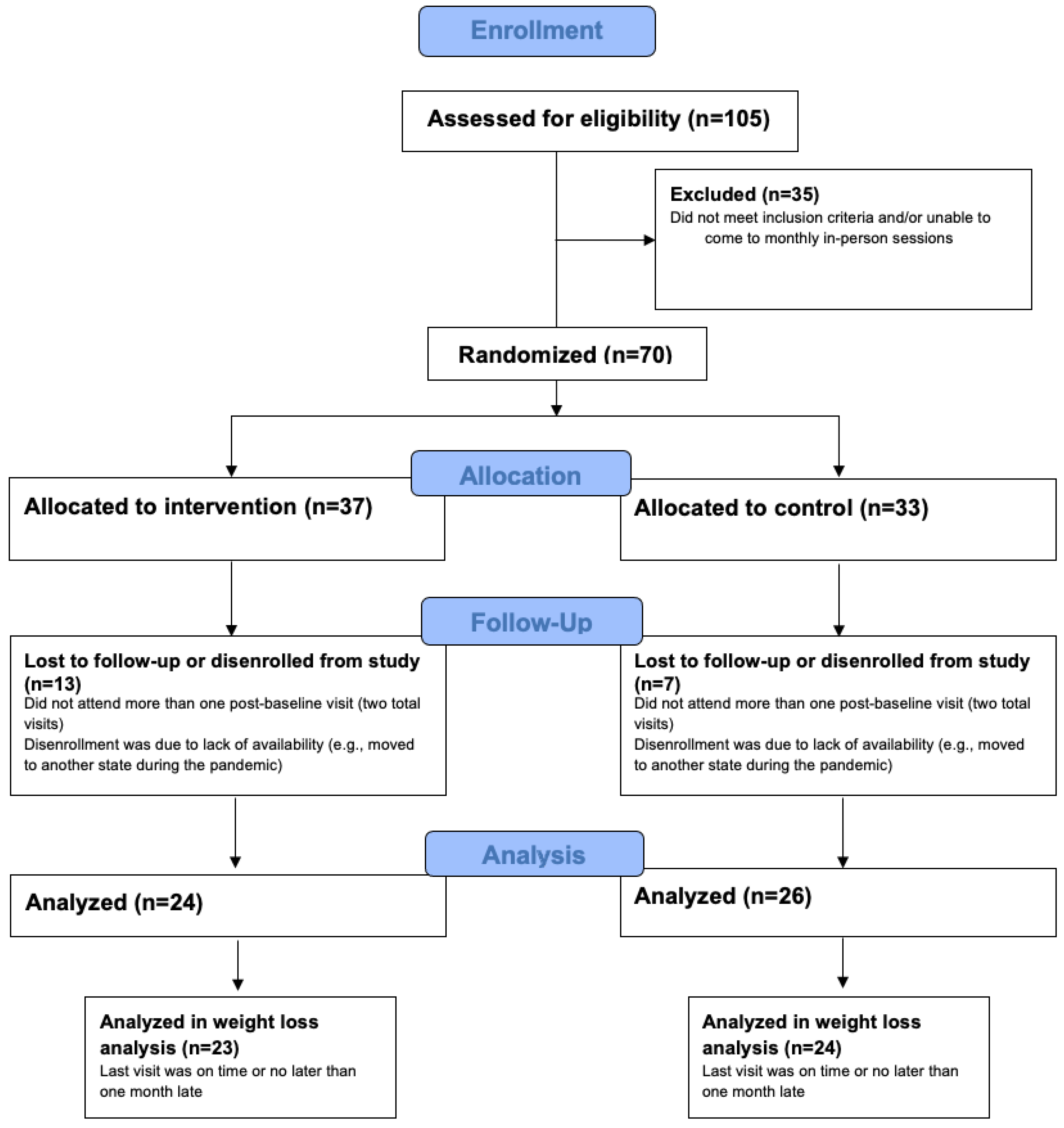
2. Materials and Methods
2.1. Study Procedure
2.2. Intervention
2.3. Measures
2.3.1. Cognitive Tasks
2.3.2. Brain Activity
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics and Weight Loss
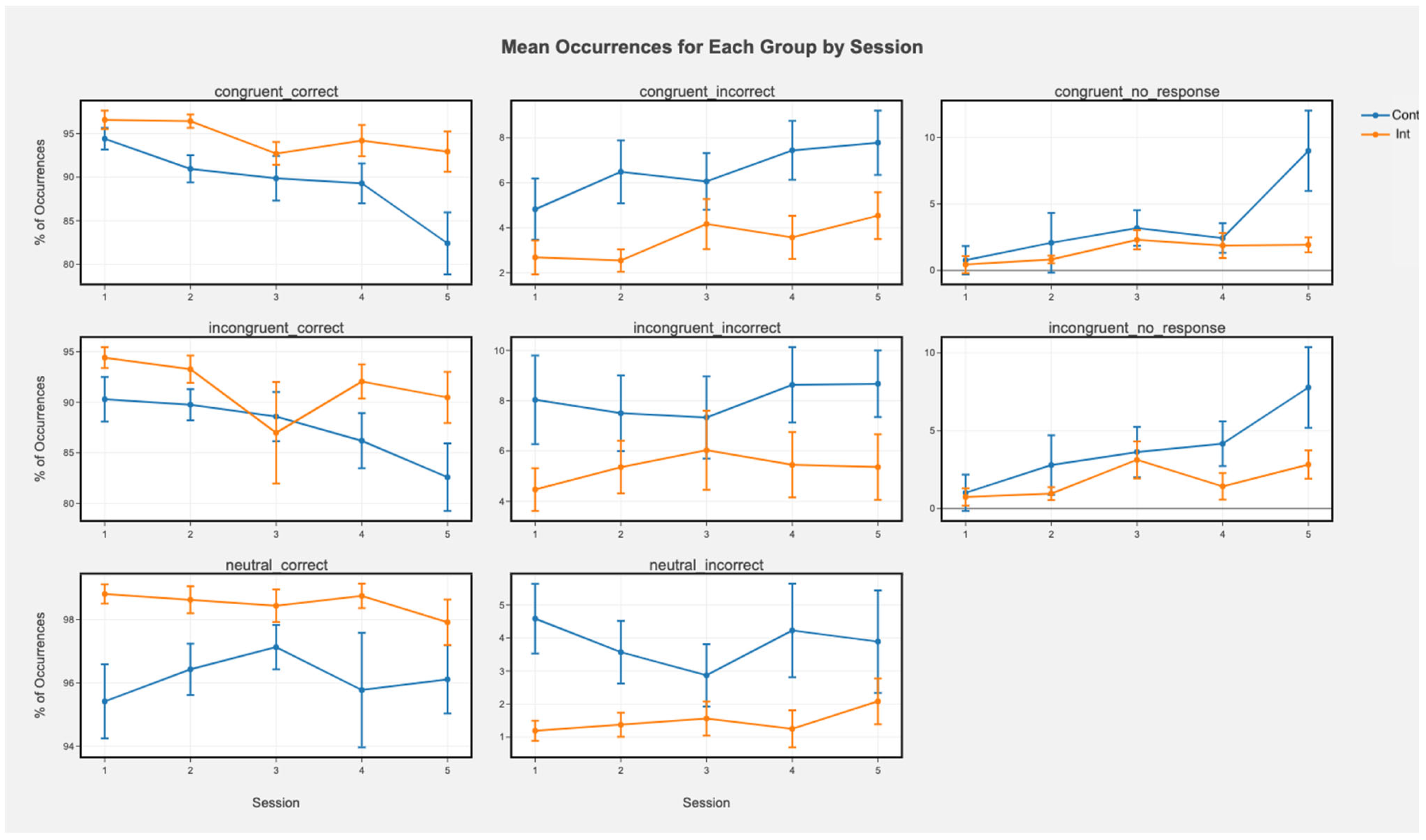
3.2. Cognitive Tasks
3.2.1. 2-Back
3.2.2. Stroop
3.2.3. Flanker
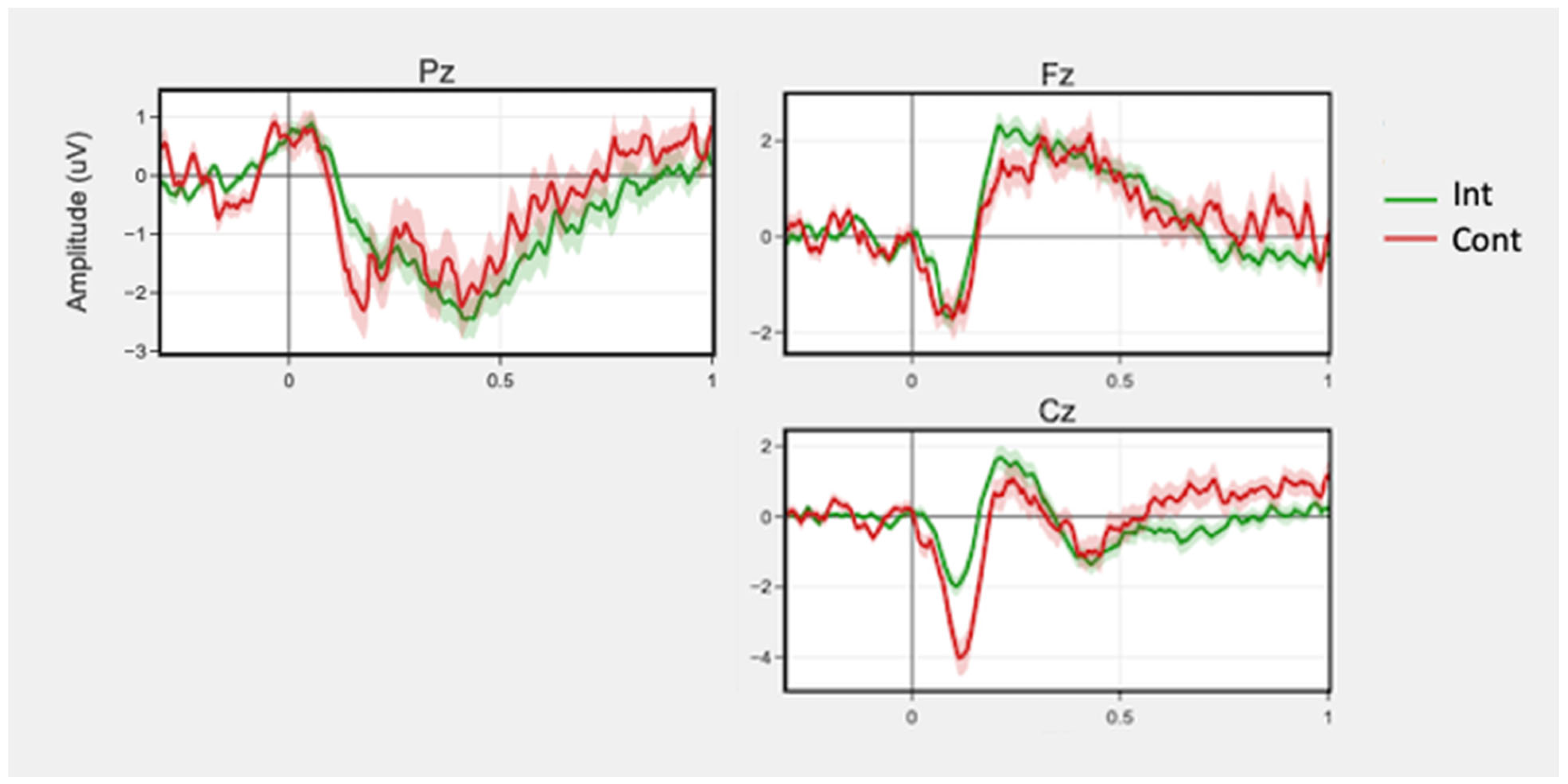
3.3. Brain Activity
3.3.1. 2-Back
3.3.2. Stroop
3.3.3. Flanker
4. Discussion
4.1. Cognitive Tasks and Brain Activity
4.2. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Blüher, M. Obesity: Global Epidemiology and Pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Diabetes Prevention Program (DPP) Research Group. The Diabetes Prevention Program (DPP): Description of Lifestyle Intervention. Diabetes Care 2002, 25, 2165–2171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Favieri, F.; Forte, G.; Casagrande, M. The Executive Functions in Overweight and Obesity: A Systematic Review of Neuropsychological Cross-Sectional and Longitudinal Studies. Front. Psychol. 2019, 10, 2126. [Google Scholar] [CrossRef] [PubMed]
- Elliott, R. Executive Functions and Their Disorders: Imaging in Clinical Neuroscience. Br. Med. Bull. 2003, 65, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Banich, M.T. Executive Function: The Search for an Integrated Account. Curr. Dir. Psychol. Sci. 2009, 18, 89–94. [Google Scholar] [CrossRef]
- Gettens, K.M.; Gorin, A.A. Executive Function in Weight Loss and Weight Loss Maintenance: A Conceptual Review and Novel Neuropsychological Model of Weight Control. J. Behav. Med. 2017, 40, 687–701. [Google Scholar] [CrossRef]
- Spitznagel, M.B.; Alosco, M.; Strain, G.; Devlin, M.; Cohen, R.; Paul, R.; Crosby, R.D.; Mitchell, J.E.; Gunstad, J. Cognitive Function Predicts 24-Month Weight Loss Success after Bariatric Surgery. Surg. Obes. Relat. Dis. 2013, 9, 765–770. [Google Scholar] [CrossRef] [Green Version]
- Butryn, M.L.; Martinelli, M.K.; Remmert, J.E.; Roberts, S.R.; Zhang, F.; Forman, E.M.; Manasse, S.M. Executive Functioning as a Predictor of Weight Loss and Physical Activity Outcomes. Ann. Behav. Med. 2019, 53, 909–917. [Google Scholar] [CrossRef]
- Best, J.R.; Nagamatsu, L.S.; Liu-Ambrose, T. Improvements to Executive Function during Exercise Training Predict Maintenance of Physical Activity over the Following Year. Front. Hum. Neurosci. 2014, 8, 353. [Google Scholar] [CrossRef] [Green Version]
- Daly, M.; McMinn, D.; Allan, J.L. A Bidirectional Relationship between Physical Activity and Executive Function in Older Adults. Front. Hum. Neurosci. 2014, 8, 1044. [Google Scholar] [CrossRef] [Green Version]
- Raman, J.; Hay, P.; Tchanturia, K.; Smith, E. A Randomised Controlled Trial of Manualized Cognitive Remediation Therapy in Adult Obesity. Appetite 2018, 123, 269–279. [Google Scholar] [CrossRef] [Green Version]
- Veronese, N.; Facchini, S.; Stubbs, B.; Luchini, C.; Solmi, M.; Manzato, E.; Sergi, G.; Maggi, S.; Cosco, T.; Fontana, L. Weight Loss Is Associated with Improvements in Cognitive Function among Overweight and Obese People: A Systematic Review and Meta-Analysis. Neurosci. Biobehav. Rev. 2017, 72, 87–94. [Google Scholar] [CrossRef]
- Smith, E.; Hay, P.; Campbell, L.; Trollor, J.N. A Review of the Association between Obesity and Cognitive Function across the Lifespan: Implications for Novel Approaches to Prevention and Treatment. Obes. Rev. 2011, 12, 740–755. [Google Scholar] [CrossRef] [PubMed]
- Allom, V.; Mullan, B.; Smith, E.; Hay, P.; Raman, J. Breaking Bad Habits by Improving Executive Function in Individuals with Obesity. BMC Public Health 2018, 18, 505. [Google Scholar] [CrossRef] [PubMed]
- Castelnuovo, G.; Pietrabissa, G.; Manzoni, G.M.; Cattivelli, R.; Rossi, A.; Novelli, M.; Varallo, G.; Molinari, E. Cognitive Behavioral Therapy to Aid Weight Loss in Obese Patients: Current Perspectives. Pseudo Rigid Body Model 2017, 10, 165–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Comsa, L.; David, O.; David, D. Outcomes and Mechanisms of Change in Cognitive-Behavioral Interventions for Weight Loss: A Meta-Analysis of Randomized Clinical Trials. Behav. Res. Ther. 2020, 132, 103654. [Google Scholar] [CrossRef] [PubMed]
- Cuijpers, P.; van Straten, A.; Andersson, G. Internet-Administered Cognitive Behavior Therapy for Health Problems: A Systematic Review. J. Behav. Med. 2008, 31, 169–177. [Google Scholar] [CrossRef] [Green Version]
- Chiesa, A.; Calati, R.; Serretti, A. Does Mindfulness Training Improve Cognitive Abilities? A Systematic Review of Neuropsychological Findings. Clin. Psychol. Rev. 2011, 31, 449–464. [Google Scholar] [CrossRef]
- Chow, G.; Gan, J.K.E.; Chan, J.K.Y.; Wu, X.V.; Klainin-Yobas, P. Effectiveness of Psychosocial Interventions among Older Adults with Mild Cognitive Impairment: A Systematic Review and Meta-Analysis. Aging Ment. Health 2021, 25, 1986–1997. [Google Scholar] [CrossRef]
- Leyland, A.; Rowse, G.; Emerson, L.-M. Experimental Effects of Mindfulness Inductions on Self-Regulation: Systematic Review and Meta-Analysis. Emotion 2019, 19, 108–122. [Google Scholar] [CrossRef]
- Pozuelos, J.; Mead, B.; Rueda, M.; Malinowski, P. Short-Term Mindful Breath Awareness Training Improves Inhibitory Control and Response Monitoring. Prog. Brain Res. 2019, 244, 137–163. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, A.-M.; Hogervorst, M.A.; van Erp, J.B.F.; Heffelaar, T.; Zimmerman, P.H.; Oostenveld, R. Estimating Workload Using EEG Spectral Power and ERPs in the N-Back Task. J. Neural. Eng. 2012, 9, 045008. [Google Scholar] [CrossRef]
- Larson, M.J.; Clayson, P.E. The Relationship between Cognitive Performance and Electrophysiological Indices of Performance Monitoring. Cogn. Affect. Behav. Neurosci. 2011, 11, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.Y.; Wittenberg, E.; Nam, C.S. Behavioral and Neural Correlates of Executive Function: Interplay between Inhibition and Updating Processes. Front. Neurosci. 2017, 11, 378. [Google Scholar] [CrossRef]
- Fissler, M.; Winnebeck, E.; Schroeter, T.A.; Gummbersbach, M.; Huntenburg, J.M.; Gärtner, M.; Barnhofer, T. Brief Training in Mindfulness May Normalize a Blunted Error-Related Negativity in Chronically Depressed Patients. Cogn. Affect. Behav. Neurosci. 2017, 17, 1164–1175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lomas, T.; Ivtzan, I.; Fu, C.H.Y. A Systematic Review of the Neurophysiology of Mindfulness on EEG Oscillations. Neurosci. Biobehav. Rev. 2015, 57, 401–410. [Google Scholar] [CrossRef]
- Crook, T.H.; Lebowitz, B.D.; Pirozzolo, F.J.; Zappalà, G.; Cavarzeran, F.; Measso, G.; Massari, D.C. Recalling Names after Introduction: Changes across the Adult Life Span in Two Cultures. Dev. Neuropsychol. 1993, 9, 103–113. [Google Scholar] [CrossRef]
- Lande, M.B.; Kaczorowski, J.M.; Auinger, P.; Schwartz, G.J.; Weitzman, M. Elevated Blood Pressure and Decreased Cognitive Function among School-Age Children and Adolescents in the United States. J. Pediatr. 2003, 143, 720–724. [Google Scholar] [CrossRef]
- Stubbs, J.; Whybrow, S.; Teixeira, P.; Blundell, J.; Lawton, C.; Westenhoefer, J.; Engel, D.; Shepherd, R.; Mcconnon, Á.; Gilbert, P.; et al. Problems in Identifying Predictors and Correlates of Weight Loss and Maintenance: Implications for Weight Control Therapies Based on Behaviour Change: Predicting Weight Outcomes. Obes. Rev. 2011, 12, 688–708. [Google Scholar] [CrossRef] [Green Version]
- Hartmann-Boyce, J.; Boylan, A.-M.; Jebb, S.A.; Fletcher, B.; Aveyard, P. Cognitive and Behavioural Strategies for Self-Directed Weight Loss: Systematic Review of Qualitative Studies. Obes. Rev. 2017, 18, 335–349. [Google Scholar] [CrossRef] [Green Version]
- Krueger, C.; Tian, L. A Comparison of the General Linear Mixed Model and Repeated Measures ANOVA Using a Dataset with Multiple Missing Data Points. Biol. Res. Nurs. 2004, 6, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, W.K. Age Differences in Short-Term Retention of Rapidly Changing Information. J. Exp. Psychol. 1958, 55, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Lamichhane, B.; Westbrook, A.; Cole, M.W.; Braver, T.S. Exploring Brain-Behavior Relationships in the N-Back Task. NeuroImage 2020, 212, 116683. [Google Scholar] [CrossRef]
- Stroop, J.R. Studies of Interference in Serial Verbal Reactions. J. Exp. Psychol. 1935, 18, 643. [Google Scholar] [CrossRef]
- Eriksen, C.W.; Schultz, D.W. Information Processing in Visual Search: A Continuous Flow Conception and Experimental Results. Percept. Psychophys. 1979, 25, 249–263. [Google Scholar] [CrossRef]
- McDermott, J.M.; Pérez-Edgar, K.; Fox, N.A. Variations of the Flanker Paradigm: Assessing Selective Attention in Young Children. Behav. Res. 2007, 39, 62–70. [Google Scholar] [CrossRef] [Green Version]
- Bugg, J.M.; Jacoby, L.L.; Toth, J.P. Multiple Levels of Control in the Stroop Task. Mem. Cogn. 2008, 36, 1484–1494. [Google Scholar] [CrossRef] [Green Version]
- Gevins, A.S.; Zeitlin, G.M.; Yingling, C.D.; Doyle, J.C.; Dedon, M.F.; Schaffer, R.E.; Roumasset, J.T.; Yeager, C.L. EEG Patterns during ‘Cognitive’ Tasks. I. Methodology and Analysis of Complex Behaviors. Electroencephalogr. Clin. Neurophysiol. 1979, 47, 693–703. [Google Scholar] [CrossRef]
- Slater, J.D.; Kalamangalam, G.P.; Hope, O. Quality Assessment of Electroencephalography Obtained from a “Dry Electrode” System. J. Neurosci. Methods 2012, 208, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Halford, J.J.; Schalkoff, R.J.; Satterfield, K.E.; Martz, G.U.; Kutluay, E.; Waters, C.G.; Dean, B.C. Comparison of a Novel Dry Electrode Headset to Standard Routine EEG in Veterans. J. Clin. Neurophysiol. 2016, 33, 530–537. [Google Scholar] [CrossRef]
- Debener, S.; Ullsperger, M.; Siegel, M.; Fiehler, K.; Von Cramon, D.Y.; Engel, A.K. Trial-by-Trial Coupling of Concurrent Electroencephalogram and Functional Magnetic Resonance Imaging Identifies the Dynamics of Performance Monitoring. J. Neurosci. 2005, 25, 11730–11737. [Google Scholar] [CrossRef] [PubMed]
- Maier, M.E.; di Pellegrino, G.; Steinhauser, M. Enhanced Error-Related Negativity on Flanker Errors: Error Expectancy or Error Significance? Psychophysiology 2012, 49, 899–908. [Google Scholar] [CrossRef]
- Yasuda, A.; Sato, A.; Miyawaki, K.; Kumano, H.; Kuboki, T. Error-Related Negativity Reflects Detection of Negative Reward Prediction Error. NeuroReport 2004, 15, 2561–2565. [Google Scholar] [CrossRef] [PubMed]
- Folstein, J.R.; Van Petten, C. Influence of Cognitive Control and Mismatch on the N2 Component of the ERP: A Review. Psychophysiology 2008, 45, 152–170. [Google Scholar] [CrossRef] [Green Version]
- Tafuro, A.; Ambrosini, E.; Puccioni, O.; Vallesi, A. Brain Oscillations in Cognitive Control: A Cross-Sectional Study with a Spatial Stroop Task. Neuropsychologia 2019, 133, 107190. [Google Scholar] [CrossRef]
- Mullen, T.R.; Kothe, C.A.; Chi, Y.M.; Ojeda, A.; Kerth, T.; Makeig, S.; Jung, T.-P.; Cauwenberghs, G. Real-Time Neuroimaging and Cognitive Monitoring Using Wearable Dry EEG. IEEE Trans. Biomed. Eng. 2015, 62, 2553–2567. [Google Scholar] [CrossRef] [Green Version]
- Gehring, W.J.; Goss, B.; Coles, M.G.; Meyer, D.E.; Donchin, E. A Neural System for Error Detection and Compensation. Psychol. Sci. 1993, 4, 385–390. [Google Scholar] [CrossRef]
- Řondík, T.; Ciniburk, J.; Mouček, R.; Mautner, P. ERP Components Detection Using Wavelet Transform and Matching Pursuit Algorithm. In Proceedings of the International Conference on Applied Electronics, Pilsen, Czech Republic, 6–7 September 2022. [Google Scholar]
- Ambrosini, E.; Vallesi, A. Asymmetry in Prefrontal Resting-State EEG Spectral Power Underlies Individual Differences in Phasic and Sustained Cognitive Control. Neuroimage 2016, 124, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Compton, R.J.; Arnstein, D.; Freedman, G.; Dainer-Best, J.; Liss, A. Cognitive Control in the Intertrial Interval: Evidence from EEG Alpha Power: Cognitive Control in the Intertrial Interval. Psychophysiology 2011, 48, 583–590. [Google Scholar] [CrossRef]
- Friese, U.; Daume, J.; Göschl, F.; König, P.; Wang, P.; Engel, A.K. Oscillatory Brain Activity during Multisensory Attention Reflects Activation, Disinhibition, and Cognitive Control. Sci. Rep. 2016, 6, 32775. [Google Scholar] [CrossRef] [Green Version]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B. Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Schielzeth, H.; Dingemanse, N.J.; Nakagawa, S.; Westneat, D.F.; Allegue, H.; Teplitsky, C.; Réale, D.; Dochtermann, N.A.; Garamszegi, L.Z.; Araya-Ajoy, Y.G. Robustness of Linear Mixed-Effects Models to Violations of Distributional Assumptions. Methods Ecol. Evol. 2020, 11, 1141–1152. [Google Scholar] [CrossRef]
- Verbeken, S.; Braet, C.; Goossens, L.; van der Oord, S. Executive Function Training with Game Elements for Obese Children: A Novel Treatment to Enhance Self-Regulatory Abilities for Weight-Control. Behav. Res. Ther. 2013, 51, 290–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verbeken, S.; Braet, C.; Naets, T.; Houben, K.; Boendermaker, W.; Zeepreventorium, V.Z.W. Computer Training of Attention and Inhibition for Youngsters with Obesity: A Pilot Study. Appetite 2018, 123, 439–447. [Google Scholar] [CrossRef]
- Im, S.; Stavas, J.; Lee, J.; Mir, Z.; Hazlett-Stevens, H.; Caplovitz, G. Does Mindfulness-Based Intervention Improve Cognitive Function?: A Meta-Analysis of Controlled Studies. Clin. Psychol. Rev. 2021, 84, 101972. [Google Scholar] [CrossRef]
- Gilbert, M.; Raman, J.; Sui, Z. Cognitive Remediation-Enabled Cognitive Behaviour Therapy for Obesity: A Case Series. Eat. Weight Disord. 2021, 26, 103–114. [Google Scholar] [CrossRef]
- Duan, X.; Rhee, J.; Mehta, R.K.; Srinivasan, D. Neuromuscular Control and Performance Differences Associated With Gender and Obesity in Fatiguing Tasks Performed by Older Adults. Front. Physiol. 2018, 9, 800. [Google Scholar] [CrossRef] [Green Version]
- Gholami, Z.; Akhlaghi, M. The Effect of Flaxseed on Physical and Mental Fatigue in Children and Adolescents with Overweight/Obesity: A Randomised Controlled Trial. Br. J. Nutr. 2021, 126, 151–159. [Google Scholar] [CrossRef]
- Gletsu-Miller, N.; Shevni, N.; Manatunga, A.; Lin, E.; Musselman, D. A Multidimensional Analysis of the Longitudinal Effects of Roux En y Gastric Bypass on Fatigue: An Association with Visceral Obesity. Physiol. Behav. 2019, 209, 112612. [Google Scholar] [CrossRef]
- Cserjési, R.; Luminet, O.; Poncelet, A.-S.; Lénárd, L. Altered Executive Function in Obesity. Exploration of the Role of Affective States on Cognitive Abilities. Appetite 2009, 52, 535–539. [Google Scholar] [CrossRef]
- Mamrot, P.; Hanć, T. The Association of the Executive Functions with Overweight and Obesity Indicators in Children and Adolescents: A Literature Review. Neurosci. Biobehav. Rev. 2019, 107, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Shigihara, Y.; Ishii, A.; Funakura, M.; Kanai, E.; Watanabe, Y. Effect of Mental Fatigue on the Central Nervous System: An Electroencephalography Study. Behav. Brain Funct. 2012, 8, 48. [Google Scholar] [CrossRef] [Green Version]
- Spitznagel, M.B.; Hawkins, M.; Alosco, M.; Galioto, R.; Garcia, S.; Miller, L.; Gunstad, J. Neurocognitive Effects of Obesity and Bariatric Surgery. Eur. Eat. Disord. Rev. 2015, 23, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Rico, E.; Río-Valle, J.S.; Albein-Urios, N.; Caracuel, A.; González-Jiménez, E.; Piqueras, M.J.; Brandi, P.; Ruiz-López, I.M.; García-Rodríguez, I.; Martín-Matillas, M.; et al. Effects of a Multicomponent Behavioral Intervention on Impulsivity and Cognitive Deficits in Adolescents with Excess Weight. Behav. Pharmacol. 2012, 23, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Norris, C.J.; Creem, D.; Hendler, R.; Kober, H. Brief Mindfulness Meditation Improves Attention in Novices: Evidence From ERPs and Moderation by Neuroticism. Front. Hum. Neurosci. 2018, 12, 315. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Xin, T.-T.; Liu, X.-H.; Zhang, Y.; Lu, H.-H.; Zhai, Y.-B. Mindfulness Can Reduce Automatic Responding: Evidences from Stroop Task and Prospective Memory Task. Acta Psychol. Sin. 2012, 44, 1180–1188. [Google Scholar] [CrossRef]
- Stoll, F.; Wilson, C.; Faraut, M.; Vezoli, J.; Knoblauch, K.; Procyk, E. The Effects of Cognitive Control and Time on Frontal Beta Oscillations. Cereb. Cortex 2016, 26, 1715–1732. [Google Scholar] [CrossRef] [Green Version]
- Herrmann, C.S.; Strüber, D.; Helfrich, R.F.; Engel, A.K. EEG Oscillations: From Correlation to Causality. Int. J. Psychophysiol. 2016, 103, 12–21. [Google Scholar] [CrossRef]
- Riesel, A.; Kathmann, N.; Klawohn, J. Flexibility of Error-Monitoring in Obsessive-Compulsive Disorder under Speed and Accuracy Instructions. J. Abnorm. Psychol. 2019, 128, 671–677. [Google Scholar] [CrossRef]
- Holroyd, C.B.; Coles, M.G.H. The Neural Basis of Human Error Processing: Reinforcement Learning, Dopamine, and the Error-Related Negativity. Psychol. Rev. 2002, 109, 679–709. [Google Scholar] [CrossRef]
- Cavanagh, J.F.; Figueroa, C.M.; Cohen, M.X.; Frank, M.J. Frontal Theta Reflects Uncertainty and Unexpectedness during Exploration and Exploitation. Cereb. Cortex 2012, 22, 2575–2586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viviani, G.; Vallesi, A. EEG-Neurofeedback and Executive Function Enhancement in Healthy Adults: A Systematic Review. Psychophysiology 2021, 58, e13874. [Google Scholar] [CrossRef] [PubMed]
- Santarossa, S.; Kane, D.; Senn, C.Y.; Woodruff, S.J. Exploring the Role of In-Person Components for Online Health Behavior Change Interventions: Can a Digital Person-to-Person Component Suffice? J. Med. Internet Res. 2018, 20, e8480. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, R.F.; Leykin, Y.; Barrera, A.Z.; Brown, C.H.; Bunge, E.L. The Impact of Phone Calls on Follow-up Rates in an Online Depression Prevention Study. Internet Interv. 2017, 8, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Gowey, M.A.; Neumeier, W.H.; Henry, S.; Wadley, V.G.; Phillips, J.; Hayden, K.M.; Espeland, M.A.; Coday, M.; Lewis, C.E.; Dutton, G.R. Executive Function in Individuals with Clinically Significant Weight Loss via Behavioral Intervention. Obes. Sci. Pract. 2021, 7, 25–34. [Google Scholar] [CrossRef]

| Intervention | Control | p-Value | |
|---|---|---|---|
| Gender | 0.99 | ||
| Female | 22 (88%) | 20 (83.3%) | |
| Male | 3 (12%) | 4 (16.7%) | |
| Age | 46.3 | 42.1 | 0.09 |
| Baseline BMI | 33.0 | 36.1 | 0.20 |
| Ethnicity | 0.30 | ||
| Asian or Pacific Islander | 3 (12%) | 0 | |
| Black/African | 3 (12%) | 1 (4.2%) | |
| Caucasian | 15 (60%) | 19 (79.2%) | |
| Hispanic/Latino | 2 (8%) | 3 (12.5%) | |
| Other | 2 (8%) | 1 (4.2%) | |
| Education | 0.50 | ||
| High school degree | 1 (4%) | 2 (8.3%) | |
| Some college | 4 (16%) | 3 (12.5%) | |
| 2-year college degree or vocational training | 1 (4%) | 4 (16.7%) | |
| 4-year college degree | 9 (36%) | 5 (20.8%) | |
| Graduate degree (Master’s, PhD, MD, JD) | 10 (40%) | 10 (41.7%) | |
| Employment status | 0.20 | ||
| Full-time employed | 17 (68%) | 21 (87.5%) | |
| Part-time employed | 5 (20%) | 0 (0%) | |
| Self-employed | 1 (4%) | 1 (4.2%) | |
| Unemployed | 0 (0%) | 1 (4.2%) | |
| Looking after family | 2 (8%) | 1 (4.2%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michaelides, A.; Mitchell, E.S.; Behr, H.; Ho, A.S.; Hanada, G.; Lee, J.; McPartland, S. Executive Function-Related Improvements on a Commercial CBT-Based Weight Management Intervention: Pilot Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2022, 19, 8763. https://doi.org/10.3390/ijerph19148763
Michaelides A, Mitchell ES, Behr H, Ho AS, Hanada G, Lee J, McPartland S. Executive Function-Related Improvements on a Commercial CBT-Based Weight Management Intervention: Pilot Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2022; 19(14):8763. https://doi.org/10.3390/ijerph19148763
Chicago/Turabian StyleMichaelides, Andreas, Ellen Siobhan Mitchell, Heather Behr, Annabell Suh Ho, Grant Hanada, Jihye Lee, and Sue McPartland. 2022. "Executive Function-Related Improvements on a Commercial CBT-Based Weight Management Intervention: Pilot Randomized Controlled Trial" International Journal of Environmental Research and Public Health 19, no. 14: 8763. https://doi.org/10.3390/ijerph19148763
APA StyleMichaelides, A., Mitchell, E. S., Behr, H., Ho, A. S., Hanada, G., Lee, J., & McPartland, S. (2022). Executive Function-Related Improvements on a Commercial CBT-Based Weight Management Intervention: Pilot Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 19(14), 8763. https://doi.org/10.3390/ijerph19148763






