Comparative Effectiveness of Various Eradication Regimens for Helicobacter Pylori Infection in the Northeastern Region of Poland
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Tests Used to Confirm H. pylori Infection
2.3. Treatment Regimens for Eradication
2.4. Ethics of the Study
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hooi, J.K.Y.; Lai, W.Y.; Ng, W.K.; Suen, M.M.Y.; Underwood, F.E.; Tanyingoh, D.; Malfertheiner, P.; Graham, D.Y.; Wong, V.W.S.; Wu, J.C.Y.; et al. Global Prevalence of Helicobacter pylori Infection: Systematic Review and Meta-Analysis. Gastroenterology 2017, 153, 420–429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zamani, M.; Ebrahimtabar, F.; Zamani, V.; Miller, W.H.; Alizadeh-Navaei, R.; Shokri-Shirvani, J.; Derakhshan, M.H. Systematic review with meta-analysis: The worldwide prevalence of Helicobacter pylori infection. Aliment. Pharmacol. Ther. 2018, 47, 868–876. [Google Scholar] [CrossRef] [Green Version]
- Łaszewicz, W.; Iwańczak, F.; Iwańczak, B.; Annabhani, A.; Bała, G.; Bąk-Romaniszyn, L.; Budzyńska, A.; Cader, J.; Celiński, K.; Cichy, W.; et al. Seroprevalence of Helicobacter pylori infection in Polish children and adults depending on socioeconomic status and living conditions. Adv. Med. Sci. 2014, 59, 147–150. [Google Scholar] [CrossRef]
- Kanotra, R.; Ahmed, M.; Patel, N.; Thakkar, B.; Solanki, S.; Tareen, S.; Fasullo, M.J.; Kesavan, M.; Nalluri, N.; Khan, A.; et al. Seasonal Variations and Trends in Hospitalization for Peptic Ulcer Disease in the United States: A 12-Year Analysis of the Nationwide Inpatient Sample. Cureus 2016, 8, e854. [Google Scholar] [CrossRef] [Green Version]
- Saleem, N.; Howden, C.W. Update on the Management of Helicobacter pylori Infection. Curr. Treat. Options Gastroenterol. 2020, 18, 476–487. [Google Scholar] [CrossRef] [PubMed]
- Mera, R.M.; Bravo, L.E.; Camargo, M.C.; Bravo, J.C.; Delgado, A.G.; Romero-Gallo, J.; Yepez, M.C.; Realpe, J.L.; Schneider, B.G.; Morgan, D.R.; et al. Dynamics of Helicobacter pylori infection as a determinant of progression of gastric precancerous lesions: 16-year follow-up of an eradication trial. Gut 2017, 67, 1239–1246. [Google Scholar] [CrossRef]
- De Martel, C.; Georges, D.; Bray, F.; Ferlay, J.; Clifford, G.M. Global burden of cancer attributable to infections in 2018: A worldwide incidence analysis. Lancet Glob. Health 2020, 8, e180–e190. [Google Scholar] [CrossRef] [Green Version]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef] [Green Version]
- Malfertheiner, P.; Megraud, F.; O’Morain, C.A.; Gisbert, J.P.; Kuipers, E.J.; Axon, A.T.; Bazzoli, F.; Gasbarrini, A.; Atherton, J.; Graham, D.Y.; et al. Management of Helicobacter pylori infection—The Maastricht V/Florence Consensus Report. Gut 2017, 66, 6–30. [Google Scholar] [CrossRef] [Green Version]
- Hopkins, R.J.; Girardi, L.S.; Turney, E.A. Relationship between Helicobacter pylori eradication and reduced duodenal and gastric ulcer recurrence: A review. Gastroenterology 1996, 110, 1244–1252. [Google Scholar] [CrossRef]
- Kumar, S.; Metz, D.C.; Ellenberg, S.; Kaplan, D.E.; Goldberg, D.S. Risk Factors and Incidence of Gastric Cancer After Detection of Helicobacter pylori Infection: A Large Cohort Study. Gastroenterology 2020, 158, 527–536.e7. [Google Scholar] [CrossRef] [Green Version]
- Ford, A.C.; Yuan, Y.; Moayyedi, P. Helicobacter pylori eradication therapy to prevent gastric cancer: Systematic review and meta-analysis. Gut 2020, 69, 2113–2121. [Google Scholar] [CrossRef]
- Lee, Y.-C.; Chiang, T.-H.; Chou, C.-K.; Tu, Y.-K.; Liao, W.-C.; Wu, M.-S.; Graham, D.Y. Association Between Helicobacter pylori Eradication and Gastric Cancer Incidence: A Systematic Review and Meta-analysis. Gastroenterology 2016, 150, 1113–1124.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Savoldi, A.; Carrara, E.; Graham, D.Y.; Conti, M.; Tacconelli, E. Prevalence of Antibiotic Resistance in Helicobacter pylori: A Systematic Review and Meta-analysis in World Health Organization Regions. Gastroenterology 2018, 155, 1372–1382.e17. [Google Scholar] [CrossRef] [Green Version]
- Bińkowska, A.; Biernat, M.M.; Laczmanski, L.; Gościniak, G. Molecular Patterns of Resistance among Helicobacter pylori Strains in South-Western Poland. Front. Microbiol. 2018, 9, 3154. [Google Scholar] [CrossRef] [Green Version]
- Chey, W.D.; Leontiadis, G.I.; Howden, C.W.; Moss, S.F. ACG Clinical Guideline: Treatment of Helicobacter pylori Infection. Am. J. Gastroenterol. 2017, 112, 212–239. [Google Scholar] [CrossRef]
- Fallone, C.A.; Chiba, N.; van Zanten, S.V.; Fischbach, L.; Gisbert, J.P.; Hunt, R.H.; Jones, N.L.; Render, C.; Leontiadis, G.I.; Moayyedi, P.; et al. The Toronto Consensus for the Treatment of Helicobacter pylori Infection in Adults. Gastroenterology 2016, 151, 51–69.e14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartnik, W.; Celińska-Cedro, D.; Dzieniszewski, J.; Laszewicz, W.; Mach, T.; Przytulski, K.; Skrzydlo-Radomanska, B. Guidelines fromt he Polish Society of Gastroenterology for the diagnosis and treatment of Helicobacter pylori infection. Gastroenterol. Prakt. 2014, 2, 33–41. [Google Scholar]
- Nyssen, O.P.; Bordin, D.; Tepes, B.; Pérez-Aisa, Á.; Vaira, D.; Caldas, M.; Bujanda, L.; Castro-Fernandez, M.; Lerang, F.; Leja, M.; et al. European Registry on Helicobacter pylori management (Hp-EuReg): Patterns and trends in first-line empirical eradication prescription and outcomes of 5 years and 21,533 patients. Gut 2021, 70, 40–54. [Google Scholar] [CrossRef]
- Batts, K.P.; Ketover, S.; Kakar, S.; Krasinskas, A.M.; Mitchell, K.A.; Wilcox, R.; Westerhoff, M.; Rank, J.; Gibson, J.; Mattia, A.R.; et al. Appropriate use of special stains for identifying Helicobacter pylori: Recommendations from the Rodger C. Haggitt Gastrointestinal Pathology Society. Am. J. Surg. Pathol. 2013, 37, e12–e22. [Google Scholar] [CrossRef] [Green Version]
- Liou, J.-M.; Chen, P.-Y.; Luo, J.-C.; Lee, J.-Y.; Chen, C.-C.; Fang, Y.-J.; Yang, T.-H.; Chang, C.-Y.; Bair, M.-J.; Chen, M.-J.; et al. Efficacies of Genotypic Resistance-Guided vs Empirical Therapy for Refractory Helicobacter pylori Infection. Gastroenterology 2018, 155, 1109–1119. [Google Scholar] [CrossRef]
- Graham, D.Y.; Lee, Y.; Wu, M. Rational Helicobacter pylori Therapy: Evidence-Based Medicine Rather Than Medicine-Based Evidence. Clin. Gastroenterol. Hepatol. 2014, 12, 177–186.e3. [Google Scholar] [CrossRef] [Green Version]
- Malfertheiner, P.; Mégraud, F.; Oʼmorain, C.; Bell, D.; Porro, B.G.; Deltenre, M.; Forman, D.; Gasbarrini, G.; Jaup, B.; Misiewicz, J.J.; et al. Current European concepts in the management of Helicobacter pylori infection—The Maastricht Consensus Report. Eur. J. Gastroenterol. Hepatol. 1997, 9, 1–2. [Google Scholar] [CrossRef] [Green Version]
- Thung, I.; Aramin, H.; Vavinskaya, V.; Gupta, S.; Park, J.Y.; Crowe, S.E.; Valasek, M.A. Review article: The global emergence of Helicobacter pylori antibiotic resistance. Aliment. Pharmacol. Ther. 2016, 43, 514–533. [Google Scholar] [CrossRef] [Green Version]
- Dzierżanowska-Fangrat, K.; Rożynek, E.; Celińska-Cedro, D.; Jarosz, M.; Pawłowska, J.; Szadkowski, A.; Budzyńska, A.; Nowak, J.; Romańczuk, W.; Prosiecki, R. Antimicrobial resistance of Helicobacter pylori in Poland: A multicentre study. Int. J. Antimicrob. Agents 2005, 26, 230–234. [Google Scholar] [CrossRef]
- Fallone, C.A.; Moss, S.F.; Malfertheiner, P. Reconciliation of Recent Helicobacter pylori Treatment Guidelines in a Time of Increasing Resistance to Antibiotics. Gastroenterology 2019, 157, 44–53. [Google Scholar] [CrossRef]
- Nyssen, O.P.; Vaira, D.; Tepes, B.; Kupcinskas, L.; Bordin, D.; Pérez-Aisa, Á.; Gasbarrini, A.; Castro-Fernández, M.; Bujanda, L.; Garre, A.; et al. Room for Improvement in the Treatment of Helicobacter pylori Infection: Lessons from the European Registry on H. pylori Management (Hp-EuReg). J. Clin. Gastroenterol. 2022, 56, e98–e108. [Google Scholar] [CrossRef]
- Tursi, A.; Di Mario, F.; Franceschi, M.; De Bastiani, R.; Elisei, W.; Baldassarre, G.; Ferronato, A.; Grillo, S.; Landi, S.; Zamparella, M.; et al. New bismuth-containing quadruple therapy in patients infected with Helicobacter pylori: A first Italian experience in clinical practice. Helicobacter 2017, 22, e12371. [Google Scholar] [CrossRef]
- Miehlke, S.; Frederking, D.; Günther, T.; Glocker, E.; Eisele, B.; Andresen, V.; Schröder, S.; Morgner, A. Efficacy of three-in-one capsule bismuth quadruple therapy for Helicobacter pylori eradication in clinical practice in a multinational patient population. Helicobacter 2017, 22, e12429. [Google Scholar] [CrossRef]
- Macías-García, F.; Baston-Rey, I.; de la Iglesia-García, D.; Calviño-Suárez, C.; Nieto-García, L.; Domínguez-Muñoz, J.E. Bismuth-containing quadruple therapy versus concomitant quadruple therapy as first-line treatment for Helicobacter pylori infection in an area of high resistance to clarithromycin: A prospective, cross-sectional, comparative, open trial. Helicobacter 2019, 24, e12546. [Google Scholar] [CrossRef] [Green Version]
- Delchier, J.C.; Malfertheiner, P.; Thieroff-Ekerdt, R. Use of a combination formulation of bismuth, metronidazole and tetracycline with omeprazole as a rescue therapy for eradication of Helicobacter pylori. Aliment. Pharmacol. Ther. 2014, 40, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Shiota, S.; Reddy, R.; Alsarraj, A.; El-Serag, H.B.; Graham, D.Y. Antibiotic Resistance of Helicobacter pylori Among Male United States Veterans. Clin. Gastroenterol. Hepatol. 2015, 13, 1616–1624. [Google Scholar] [CrossRef]
- Rokkas, T.; Gisbert, J.P.; Malfertheiner, P.; Niv, Y.; Gasbarrini, A.; Leja, M.; Megraud, F.; O’Morain, C.; Graham, D.Y. Comparative Effectiveness of Multiple Different First-Line Treatment Regimens for Helicobacter pylori Infection: A Network Meta-analysis. Gastroenterology 2021, 161, 495–507.e4. [Google Scholar] [CrossRef]
- Graham, D.Y.; Lee, S.-Y. How to effectively use bismuth quadruple therapy: The good, the bad, and the ugly. Gastroenterol. Clin. N. Am. 2015, 44, 537–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malfertheiner, P.; Bazzoli, F.; Delchier, J.-C.; Celiñski, K.; Giguère, M.; Rivière, M.; Mégraud, F. Helicobacter pylori eradication with a capsule containing bismuth subcitrate potassium, metronidazole, and tetracycline given with omeprazole versus clarithromycin-based triple therapy: A randomised, open-label, non-inferiority, phase 3 trial. Lancet 2011, 377, 905–913. [Google Scholar] [CrossRef]
- Laine, L.; Hunt, R.; El-Zimaity, H.; Nguyen, B.; Osato, M.; Spenard, J. Bismuth-Based Quadruple Therapy Using a Single Capsule of Bismuth Biskalcitrate, Metronidazole, and Tetracycline Given With Omeprazole Versus Omeprazole, Amoxicillin, and Clarithromycin for Eradication of Helicobacter pylori in Duodenal Ulcer Patients: A Prospective, Randomized, Multicenter, North American Trial. Am. J. Gastroenterol. 2003, 98, 562–567. [Google Scholar] [CrossRef]
- Villoria, A.; Garcia, P.; Calvet, X.; Gisbert, J.P.; Vergara, M. Meta-analysis: High-dose proton pump inhibitors versus standard dose in triple therapy for Helicobacter pylori eradication. Aliment. Pharmacol. Ther. 2008, 28, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Matsuo, K.; Sawaki, A.; Ito, H.; Hirose, K.; Wakai, K.; Sato, S.; Nakamura, T.; Yamao, K.; Ueda, R.; et al. Systematic review and meta-analysis: Importance of CagA status for successful eradication of Helicobacter pylori infection. Aliment. Pharmacol. Ther. 2006, 24, 273–280. [Google Scholar] [CrossRef]
- Wang, D.; Li, Q.; Gong, Y.; Yuan, Y. The association between vacA or cagA status and eradication outcome of Helicobacter pylori infection: A meta-analysis. PLoS ONE 2017, 12, e0177455. [Google Scholar] [CrossRef] [PubMed]
- Iwanczak, B.; Laszewicz, W.; Iwanczak, F.; Dzierzanowska-Fangrat, K.; Rozynek, M.; Dzierzanowska, D.; Gosciniak, G.; Dlugosz, J. Genotypic and clinical differences of se-ropositive Helicobacter pylori children and adults in the Polish population. J. Physiol. Pharmacol. 2014, 65, 801–807. [Google Scholar]
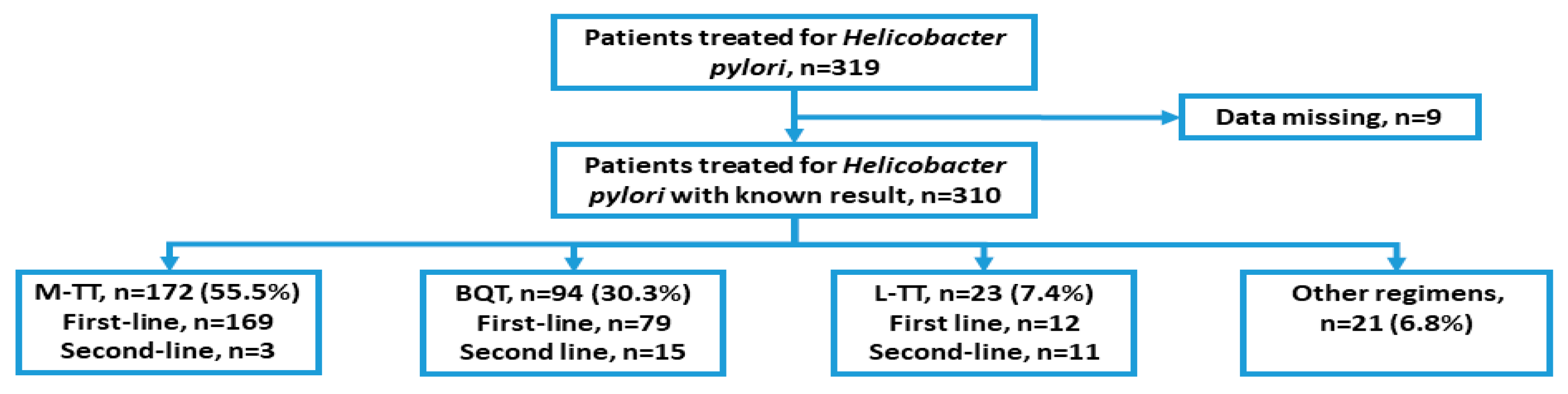
| Number of Patients | Percentage of Patients | ||
|---|---|---|---|
| Sex | Female | 187 | 64.7% |
| Male | 102 | 35.3% | |
| Indication to test H. pylori infection | Dyspepsia | 211 | 73% |
| Peptic ulcer disease | 36 | 12.5% | |
| Family history of gastric cancer | 24 | 8.3% | |
| Anaemia | 16 | 5.5% | |
| MALT lymphoma | 2 | 0.7% | |
| Test to detect H. pylori infection | Urease rapid test | 135 | 47% |
| Stool antigen test | 78 | 27% | |
| Histopathology | 54 | 19% | |
| H. pylori IgG antibody | 22 | 7% | |
| Treatment regimen | Bismuth quadruple therapy | 94 | 32.5% |
| Metronidazole-based triple therapy | |||
| 10 days | 139 | 48.1% | |
| 14 days | 33 | 11.4% | |
| Levofloxacin-based triple therapy | |||
| 10 days | 13 | 4.5% | |
| 14 days | 10 | 3.5% | |
| Proton pump inhibitor | Esomeprazole | 128 | 44.3% |
| Omeprazole | 105 | 36.3% | |
| Pantoprazole | 51 | 17.6% | |
| Lansoprazole | 5 | 1.7% | |
| H. pylori eradication attempt | First-line therapy | 260 | 90.0% |
| Second-line therapy | 29 | 10.0% |
| Variable | Successful | Unsuccessful | p | |||
|---|---|---|---|---|---|---|
| n | % | n | % | |||
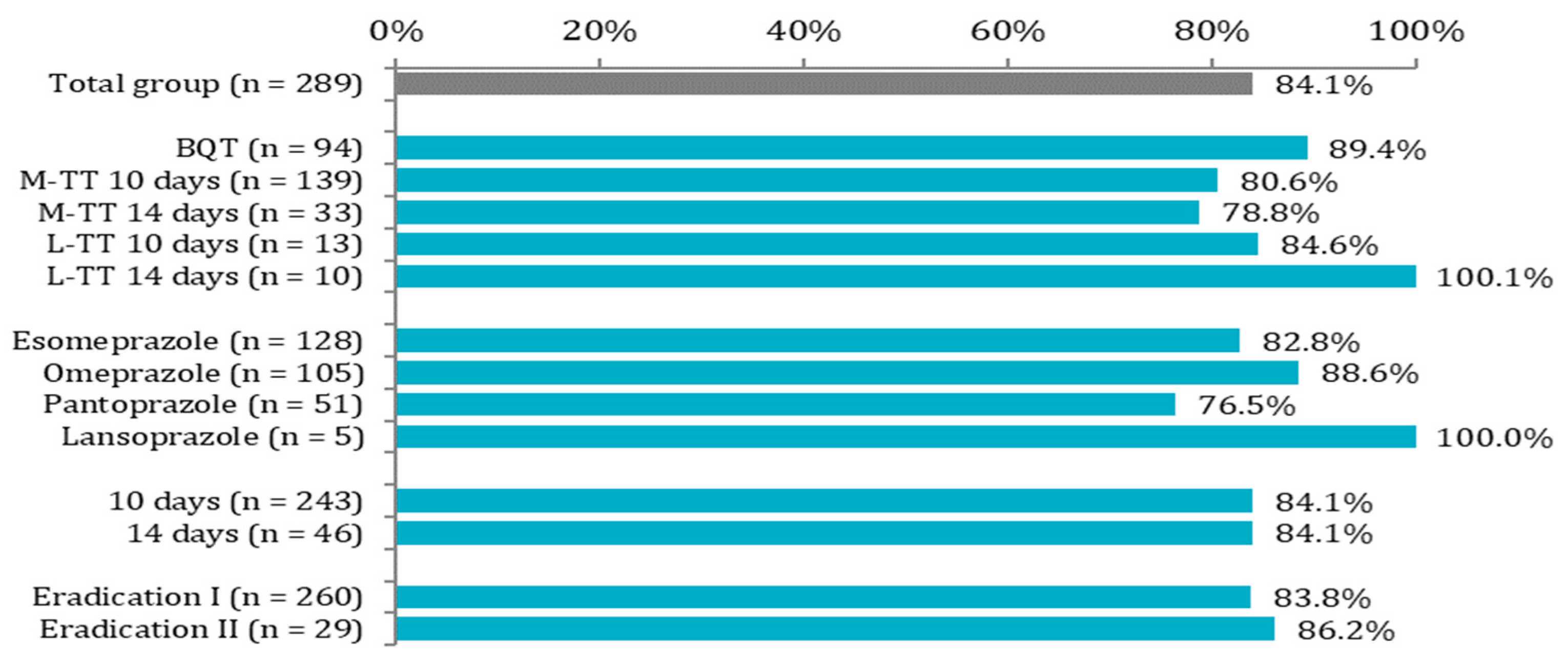
| Treatment regimen | Bismuth quadruple therapy | 84 | 34.6% | 10 | 21.7% | 0.213 |
| Metronidazole-based triple therapy 10 days | 112 | 46.1% | 27 | 58.7% | ||
| Metronidazole-based triple therapy 14 days | 26 | 10.7% | 7 | 15.2% | ||
| Levofloxacin-based triple therapy 10 days | 11 | 4.5% | 2 | 4.3% | ||
| Levofloxacin-based triple therapy 14 days | 10 | 4.1% | 0 | 0.0% | ||
| Proton pump inhibitor | Esomeprazole | 106 | 43.6% | 22 | 47.8% | 0.180 |
| Omeprazole | 93 | 38.3% | 12 | 26.1% | ||
| Pantoprazole | 39 | 16.0% | 12 | 26.1% | ||
| Lansoprazole | 5 | 2.1% | 0 | 0.0% | ||
| H. pylori eradication attempt | First-line therapy | 218 | 89.7% | 42 | 91.3% | 0.951 |
| Second-line therapy | 25 | 10.3% | 4 | 8.7% | ||
| Variables | First-Line Therapy (n = 260) | Second-Line Therapy (n = 29) | |||||
|---|---|---|---|---|---|---|---|
| Successful (n) | Unsuccessful (n) | p | Successful (n) | Unsuccessful (n) | p | ||
| Treatment regimen | BQT | 70 (88.6%) | 9 (11.4%) | 0.243 | 14 (93.3%) | 1 (6.7%) | 0.077 1 |
| M-TT | 136 (80.5%) | 33 (19.5%) | 1 (33.3%) | 2 (66.7%) | |||
| L-TT | 11 (91.7%) | 1 (8.3%) | 10 (90.9%) | 1 (9.1%) | |||
| Proton pump inhibitor | Esomeprazole | 88 (83.0%) | 18 (17.0%) | 0.222 1 | 18 (81.8%) | 4 (18.2%) | >0.999 1 |
| Omeprazole | 89 (88.1%) | 12 (11.9%) | 4 (100%) | 0 (0.0%) | |||
| Pantoprazole | 37 (75.5%) | 12 (24.5%) | 2 (100%) | 0 (0.0%) | |||
| Lansoprazole | 4 (100%) | 0 (0.0%) | 1 (100%) | 0 (0.0%) | |||
| Tratment duration | 10 | 185 (83.7) | 36 (16.3%) | >0.999 | 21 (87.5%) | 3 (12.5%) | 0.557 1 |
| 14 | 33 (84.6%) | 6 (15.4%) | 4 (80.0%) | 1 (20.0%) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wasielica-Berger, J.; Gugnacki, P.; Mlynarczyk, M.; Rogalski, P.; Swidnicka-Siergiejko, A.; Antonowicz, S.; Krzyzak, M.; Maslach, D.; Dabrowski, A.; Daniluk, J. Comparative Effectiveness of Various Eradication Regimens for Helicobacter Pylori Infection in the Northeastern Region of Poland. Int. J. Environ. Res. Public Health 2022, 19, 6921. https://doi.org/10.3390/ijerph19116921
Wasielica-Berger J, Gugnacki P, Mlynarczyk M, Rogalski P, Swidnicka-Siergiejko A, Antonowicz S, Krzyzak M, Maslach D, Dabrowski A, Daniluk J. Comparative Effectiveness of Various Eradication Regimens for Helicobacter Pylori Infection in the Northeastern Region of Poland. International Journal of Environmental Research and Public Health. 2022; 19(11):6921. https://doi.org/10.3390/ijerph19116921
Chicago/Turabian StyleWasielica-Berger, Justyna, Patryk Gugnacki, Maryla Mlynarczyk, Pawel Rogalski, Agnieszka Swidnicka-Siergiejko, Stefania Antonowicz, Michalina Krzyzak, Dominik Maslach, Andrzej Dabrowski, and Jaroslaw Daniluk. 2022. "Comparative Effectiveness of Various Eradication Regimens for Helicobacter Pylori Infection in the Northeastern Region of Poland" International Journal of Environmental Research and Public Health 19, no. 11: 6921. https://doi.org/10.3390/ijerph19116921
APA StyleWasielica-Berger, J., Gugnacki, P., Mlynarczyk, M., Rogalski, P., Swidnicka-Siergiejko, A., Antonowicz, S., Krzyzak, M., Maslach, D., Dabrowski, A., & Daniluk, J. (2022). Comparative Effectiveness of Various Eradication Regimens for Helicobacter Pylori Infection in the Northeastern Region of Poland. International Journal of Environmental Research and Public Health, 19(11), 6921. https://doi.org/10.3390/ijerph19116921






