Long-Term Consequences of COVID-19 at 6 Months and Above: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Methods
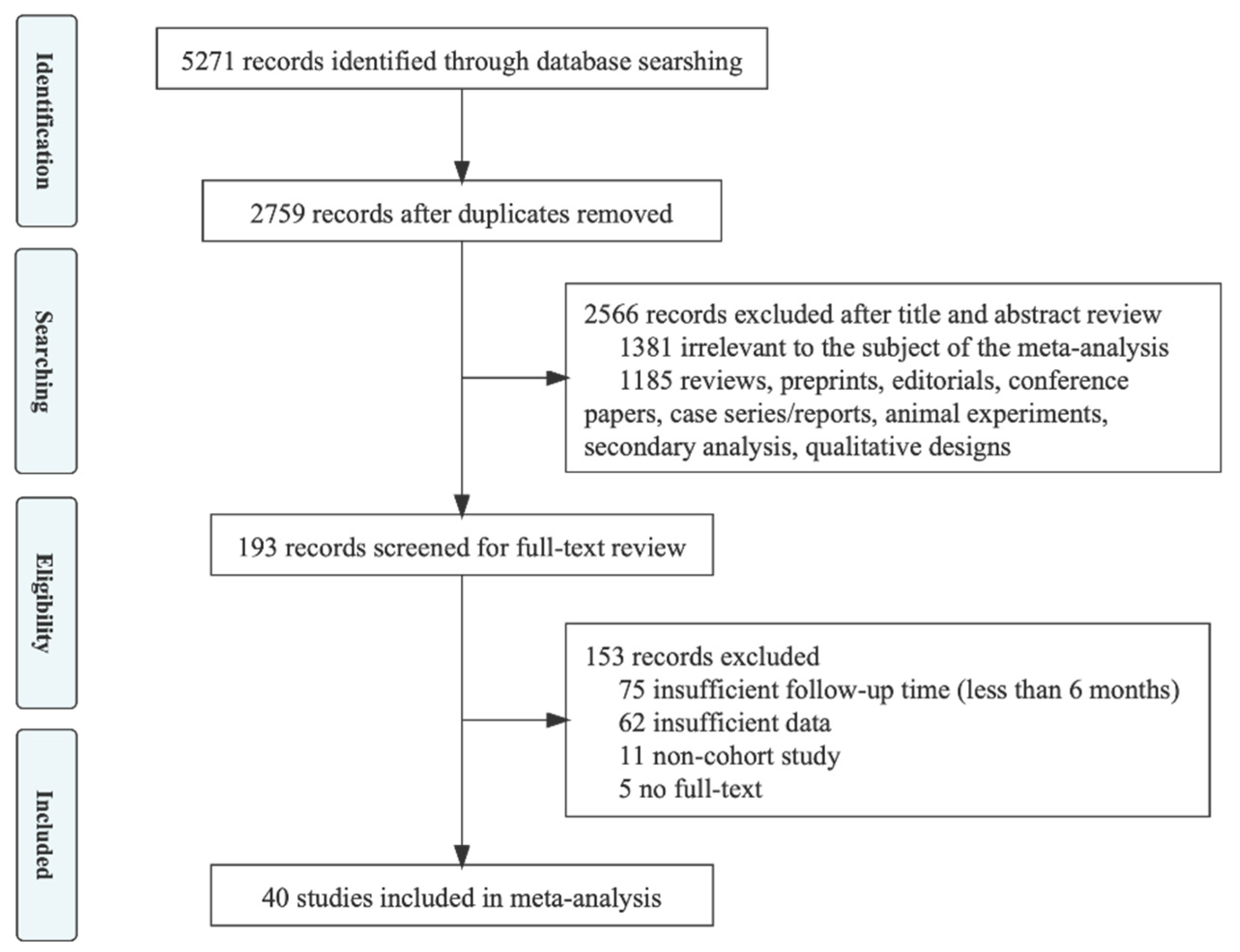
2.1. Search Strategy and Selection Criteria
2.2. Data Extraction
2.3. Quality Assessment and Risk of Bias
2.4. Data Synthesis and Statistical Analysis
3. Results
3.1. Basic Characteristics
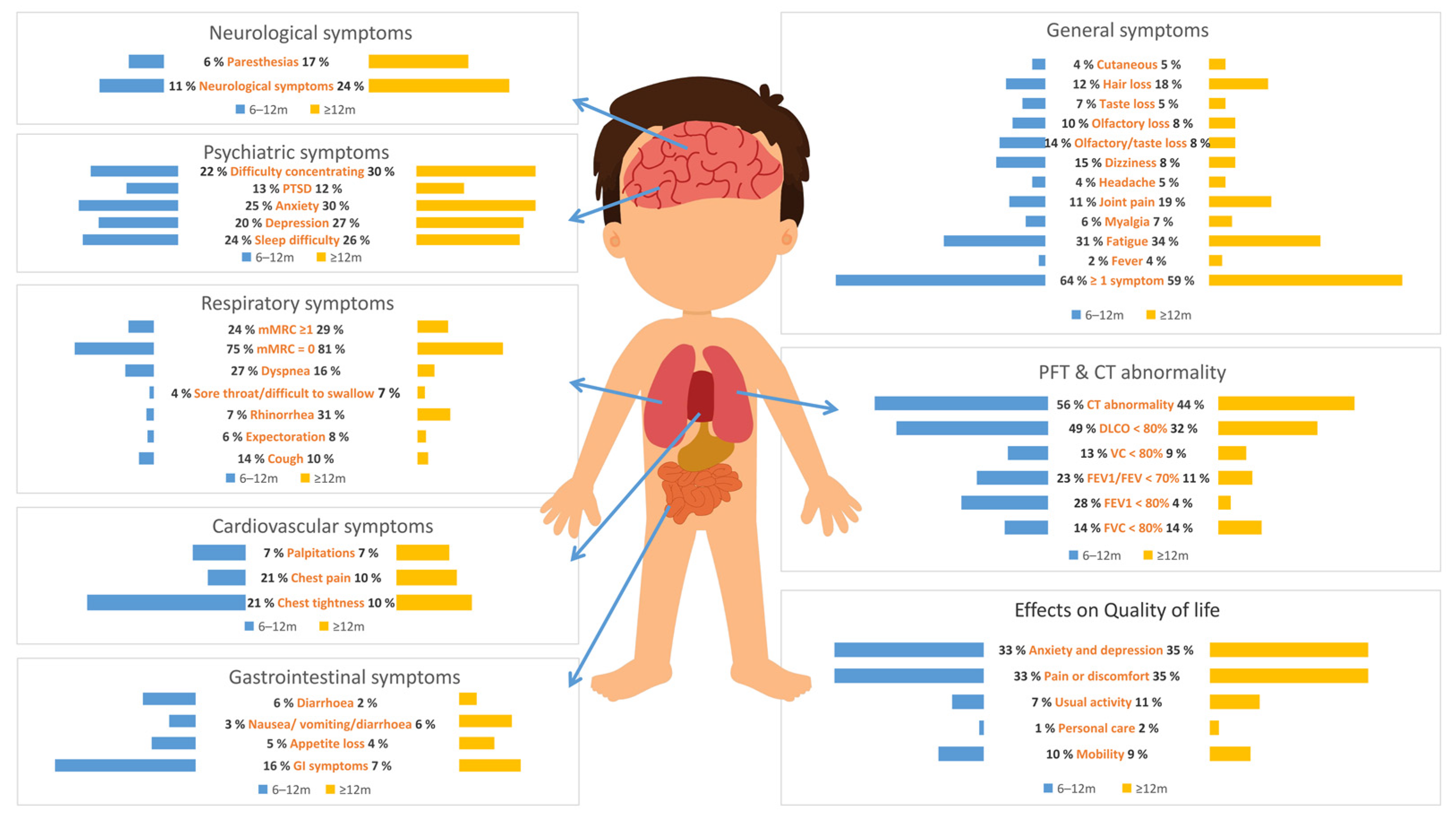
3.2. Pooled Prevalence of COVID-19 Symptoms at 6 Months and Above
3.3. Pooled Prevalence of Pulmonary Functional Test Results after COVID-19 at 6 Months and Above
3.4. Pooled Prevalence of CT Results after COVID-19 at 6 Months and Above
3.5. The Impact of COVID-19 on Quality of Life
3.6. Gender Differences in Consequences of Long-Term COVID
3.7. Quality Evaluation, Risk of Bias, and Publication Bias
3.8. GRADE Evidence Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| COVID-19 | Coronavirus disease 2019 |
| PP | Pooled prevalence |
| CI | Confidence interval |
| OR | Odds ratio |
| ICU | Intensive care unit |
| LOS | Length of stay |
| COPD | Chronic obstructive pulmonary diseases |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| PFT | Pulmonary functional test |
| WHO | World Health Organization |
| UK | United Kingdom |
| CT | Computerized tomography |
| DLCO | Carbon monoxide diffusing capacity |
| mMRC | Modified Medical Research Council Dyspnea Scale |
| GI | Gastrointestinal |
| GAD-7 | Generalized anxiety disorder-7 |
| PTSD | Post-traumatic stress disorder |
| 6 MWT | 6-min walk test |
| EQ-5D-5L | European Quality of Life Five-Dimension Five-Level Scale |
| GGO | Ground-glass opacity |
| FVC | Forced vital capacity |
| FEV1 | Forced expiratory volume in one second |
| VC | Vital capacity |
| FEV1/FEV | Forced expiratory volume in one second/forced expiratory volume |
| TLC | Total lung capacity |
| GRADE | Grading of Recommendations, Assessment, Development, and Evaluation |
| RR | Risk Ratio |
References
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard; World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- World Health Organization. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19—30 October 2020; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---30-october-2020 (accessed on 6 April 2022).
- World Health Organization. A Clinical Case Definition of Post COVID-19 Condition by a Delphi Consensus; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-Post_COVID-19_condition-Clinical_case_definition-2021.1 (accessed on 6 April 2022).
- Soriano, J.B.; Murthy, S.; Marshall, J.C.; Relan, P.; Diaz, J.V. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 2022, 22, e102–e107. [Google Scholar] [CrossRef]
- The Royal Society. Long COVID: What Is It, and What Is Needed? 2020. Available online: https://royalsociety.org/-/media/policy/projects/set-c/set-c-long-covid.pdf (accessed on 6 April 2022).
- World Health Organization. Altea: A Network for Sharing Evidence-Based Information on the Long-Term Effects of COVID-19; World Health Organization: Geneva, Switzerland, 2022; Available online: https://cdn.who.int/media/docs/default-source/science-translation/case-studies-1/cs2_altea.pdf?sfvrsn=fded8c90_4 (accessed on 6 April 2022).
- Altea Network. Symptoms Overview. 2022. Available online: https://www.altea-network.com/en/long-covid/symptoms-overview/ (accessed on 6 April 2022).
- Cabrera Martimbianco, A.L.; Pacheco, R.L.; Bagattini, Â.M.; Riera, R. Frequency, signs and symptoms, and criteria adopted for long COVID-19: A systematic review. Int. J. Clin. Pract. 2021, 75, e14357. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Leon, S.; Wegman-Ostrosky, T.; Perelman, C.; Sepulveda, R.; Rebolledo, P.A.; Cuapio, A.; Villapol, S. More than 50 long-term effects of COVID-19: A systematic review and meta-analysis. Sci. Rep. 2021, 11, 16144. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19—7 October 2021; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---7-october-2021 (accessed on 6 April 2022).
- Garg, M.; Maralakunte, M.; Garg, S.; Dhooria, S.; Sehgal, I.; Bhalla, A.S.; Vijayvergiya, R.; Grover, S.; Bhatia, V.; Jagia, P.; et al. The Conundrum of ‘Long-COVID-19’: A Narrative Review. Int. J. Gen. Med. 2021, 14, 2491–2506. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Yang, M.; Lai, C.L. Long COVID-19 Syndrome: A Comprehensive Review of Its Effect on Various Organ Systems and Recommendation on Rehabilitation Plans. Biomedicines 2021, 9, 966. [Google Scholar] [CrossRef]
- Magnúsdóttir, I.; Lovik, A.; Unnarsdóttir, A.; McCartney, D.; Ask, H.; Kõiv, K.; Christoffersen, L.; Johnson, S.; Hauksdóttir, A.; Fawns-Ritchie, C.; et al. Acute COVID-19 severity and mental health morbidity trajectories in patient populations of six nations: An observational study. Lancet Public Health 2022, 7, E406–E416. [Google Scholar] [CrossRef]
- Xie, Y.; Xu, E.; Al-Aly, Z. Risks of mental health outcomes in people with COVID-19: Cohort study. BMJ 2022, 376, e068993. [Google Scholar] [CrossRef]
- Malik, P.; Patel, K.; Pinto, C.; Jaiswal, R.; Tirupathi, R.; Pillai, S.; Patel, U. Post-acute COVID-19 syndrome (PCS) and health-related quality of life (HRQoL)-A systematic review and meta-analysis. J. Med. Virol. 2022, 94, 253–262. [Google Scholar] [CrossRef]
- Walia, N.; Walia, N.; Lat, J.O.; Tariq, R.; Tyagi, S.; Qazi, A.M.; Salari, S.W.; Jafar, A.; Kousar, T.; Bieniek, S. Post-acute sequelae of COVID-19 and the mental health implications. Discoveries 2021, 9, e140. [Google Scholar] [CrossRef]
- Douaud, G.; Lee, S.; Alfaro-Almagro, F.; Arthofer, C.; Wang, C.; McCarthy, P.; Lange, F.; Andersson, J.L.R.; Griffanti, L.; Duff, E.; et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. Nature 2022, 604, 697–707. [Google Scholar] [CrossRef]
- O’Leary, K. Brain Pathology of COVID-19. 2022. Available online: https://www.nature.com/articles/d41591-022-00043-x> (accessed on 6 April 2022).
- Du, Y.Y.; Zhao, W.; Zhou, X.L.; Zeng, M.; Yang, D.H.; Xie, X.Z.; Huang, S.H.; Jiang, Y.J.; Yang, W.H.; Guo, H.; et al. Survivors of COVID-19 exhibit altered amplitudes of low frequency fluctuation in the brain: A resting-state functional magnetic resonance imaging study at 1-year follow-up. Neural Regen. Res. 2022, 17, 1576–1581. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Xu, X.; Zhang, L.; Zheng, D.; Liu, Y.; Feng, B.; Hu, J.; Lin, Q.; Xi, X.; Wang, Q.; et al. Post-traumatic Stress Disorder Symptoms and Quality of Life of COVID-19 Survivors at 6-Month Follow-Up: A Cross-Sectional Observational Study. Front. Psychiatry 2021, 12, 782478. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, A.; Aro, M.; Reijula, J.; Mäkelä, M.J.; Ollgren, J.; Puolanne, M.; Järvinen, A.; Vasankari, T. Women report more symptoms and impaired quality of life: A survey of Finnish COVID-19 survivors. Infect. Dis. 2022, 54, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Mazza, M.G.; Palladini, M.; De Lorenzo, R.; Bravi, B.; Poletti, S.; Furlan, R.; Ciceri, F.; Vai, B.; Bollettini, I.; Melloni, E.M.T.; et al. One-year mental health outcomes in a cohort of COVID-19 survivors. J. Psychiatr. Res. 2022, 145, 118–124. [Google Scholar] [CrossRef]
- Tessitore, E.; Handgraaf, S.; Poncet, A.; Achard, M.; Höfer, S.; Carballo, S.; Marti, C.; Follonier, C.; Girardin, F.; Mach, F.; et al. Symptoms and quality of life at 1-year follow up of patients discharged after an acute COVID-19 episode. Swiss Med. Wkly. 2021, 151, w30093. [Google Scholar] [CrossRef]
- Romero-Duarte, Á.; Rivera-Izquierdo, M.; Guerrero-Fernández de Alba, I.; Pérez-Contreras, M.; Fernández-Martínez, N.F.; Ruiz-Montero, R.; Serrano-Ortiz, Á.; González-Serna, R.O.; Salcedo-Leal, I.; Jiménez-Mejías, E.; et al. Sequelae, persistent symptomatology and outcomes after COVID-19 hospitalization: The ANCOHVID multicentre 6-month follow-up study. BMC Med. 2021, 19, 129. [Google Scholar] [CrossRef]
- Xiong, L.; Li, Q.; Cao, X.; Xiong, H.; Huang, M.; Yang, F.; Liu, Q.; Meng, D.; Zhou, M.; Wang, G.; et al. Dynamic changes of functional fitness, antibodies to SARS-CoV-2 and immunological indicators within 1 year after discharge in Chinese health care workers with severe COVID-19: A cohort study. BMC Med. 2021, 19, 163. [Google Scholar] [CrossRef]
- Zhan, Y.; Zhu, Y.; Wang, S.; Jia, S.; Gao, Y.; Lu, Y.; Zhou, C.; Liang, R.; Sun, D.; Wang, X.; et al. SARS-CoV-2 immunity and functional recovery of COVID-19 patients 1-year after infection. Signal Transduct. Target. Ther. 2021, 6, 368. [Google Scholar] [CrossRef]
- Mainous, A.G.; Rooks, B.J.; Wu, V.; Orlando, F.A. COVID-19 Post-acute Sequelae Among Adults: 12 Month Mortality Risk. Front. Med. 2021, 8, 2351. [Google Scholar] [CrossRef]
- Zhou, F.; Tao, M.; Shang, L.; Liu, Y.; Pan, G.; Jin, Y.; Wang, L.; Hu, S.; Li, J.; Zhang, M.; et al. Assessment of Sequelae of COVID-19 Nearly 1 Year After Diagnosis. Front. Med. 2021, 8, 717194. [Google Scholar] [CrossRef]
- Bellan, M.; Baricich, A.; Patrucco, F.; Zeppegno, P.; Gramaglia, C.; Balbo, P.E.; Carriero, A.; Amico, C.S.; Avanzi, G.C.; Barini, M.; et al. Long-term sequelae are highly prevalent one year after hospitalization for severe COVID-19. Sci. Rep. 2021, 11, 22666. [Google Scholar] [CrossRef] [PubMed]
- Faverio, P.; Luppi, F.; Rebora, P.; Busnelli, S.; Stainer, A.; Catalano, M.; Parachini, L.; Monzani, A.; Galimberti, S.; Bini, F.; et al. Six-Month Pulmonary Impairment after Severe COVID-19: A Prospective, Multicentre Follow-Up Study. Respiration 2021, 100, 1078–1087. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yang, C.; An, X.; Xiong, Y.; Shang, Y.; He, J.; Qiu, Y.; Zhang, N.; Huang, L.; Jia, J.; et al. Follow-up study on COVID-19 survivors one year after discharge from hospital. Int. J. Infect. Dis. 2021, 112, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Eloy, P.; Tardivon, C.; Martin-Blondel, G.; Isnard, M.; Turnier, P.L.; Marechal, M.L.; CabiÉ, A.; Launay, O.; Tattevin, P.; Senneville, E.; et al. Severity of self-reported symptoms and psychological burden 6-months after hospital admission for COVID-19: A prospective cohort study. Int. J. Infect. Dis. 2021, 112, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.; Guido, G.; Zerunian, M.; Polidori, T.; Lucertini, E.; Pucciarelli, F.; Polici, M.; Rucci, C.; Bracci, B.; Nicolai, M.; et al. Post-acute sequelae of COVID-19 pneumonia: Six-month chest CT follow-up. Radiology 2021, 301, E36–E405. [Google Scholar] [CrossRef] [PubMed]
- Peghin, M.; Palese, A.; Venturini, M.; De Martino, M.; Gerussi, V.; Graziano, E.; Bontempo, G.; Marrella, F.; Tommasini, A.; Fabris, M.; et al. Post-COVID-19 symptoms 6 months after acute infection among hospitalized and non-hospitalized patients. Clin. Microbiol. Infect. 2021, 27, 1507–1513. [Google Scholar] [CrossRef]
- Maestrini, V.; Birtolo, L.I.; Francone, M.; Galardo, G.; Galea, N.; Severino, P.; Alessandri, F.; Colaiacomo, M.C.; Cundari, G.; Chimenti, C.; et al. Cardiac involvement in consecutive unselected hospitalized COVID-19 population: In-hospital evaluation and one-year follow-up. Int. J. Cardiol. 2021, 339, 235–242. [Google Scholar] [CrossRef]
- Liu, M.; Lv, F.; Zheng, Y.; Xiao, K. A prospective cohort study on radiological and physiological outcomes of recovered COVID-19 patients 6 months after discharge. Quant. Imaging Med. Surg. 2021, 11, 4181–4192. [Google Scholar] [CrossRef]
- Nehme, M.; Braillard, O.; Chappuis, F.; Courvoisier, D.S.; Guessous, I. Prevalence of symptoms more than seven months after diagnosis of symptomatic COVID-19 in an outpatient setting. Ann. Intern. Med. 2021, 174, 1252–1260. [Google Scholar] [CrossRef]
- Huang, L.; Yao, Q.; Gu, X.; Wang, Q.; Ren, L.; Wang, Y.; Hu, P.; Guo, L.; Liu, M.; Xu, J.; et al. 1-year outcomes in hospital survivors with COVID-19: A longitudinal cohort study. Lancet 2021, 398, 747–758. [Google Scholar] [CrossRef]
- Darcis, G.; Bouquegneau, A.; Maes, N.; Thys, M.; Henket, M.; Labye, F.; Rousseau, A.F.; Canivet, P.; Desir, C.; Calmes, D.; et al. Long-term clinical follow-up of patients suffering from moderate-to-severe COVID-19 infection: A monocentric prospective observational cohort study. Int. J. Infect. Dis. 2021, 109, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, M.D.M.; Foppiani, A.; Peretti, G.M.; Mangiavini, L.; Battezzati, A.; Bertoli, S.; Martinelli Boneschi, F.; Zuccotti, G.V. Long-Term Coronavirus Disease 2019 Complications in Inpatients and Outpatients: A One-Year Follow-up Cohort Study. Open Forum Infect. Dis. 2021, 8, ofab384. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Yang, H.; Liu, B.; Pang, X.; Du, J.; Liu, M.; Liu, Y.; Jing, X.; Chen, J.; Deng, S.; et al. Antibodies Can Last for More Than 1 Year After SARS-CoV-2 Infection: A Follow-Up Study From Survivors of COVID-19. Front. Med. 2021, 8, 967. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Liu, X.; Zhou, Y.; Yu, H.; Li, R.; Zhan, Q.; Ni, F.; Fang, S.; Lu, Y.; Ding, X.; et al. 3-month, 6-month, 9-month, and 12-month respiratory outcomes in patients following COVID-19-related hospitalisation: A prospective study. Lancet Respir. Med. 2021, 9, 747–754. [Google Scholar] [CrossRef]
- Menges, D.; Ballouz, T.; Anagnostopoulos, A.; Aschmann, H.E.; Domenghino, A.; Fehr, J.S.; Puhan, M.A. Burden of post-COVID-19 syndrome and implications for healthcare service planning: A population-based cohort study. PLoS ONE 2021, 16, e0254523. [Google Scholar] [CrossRef]
- Fayol, A.; Livrozet, M.; Boutouyrie, P.; Khettab, H.; Betton, M.; Tea, V.; Blanchard, A.; Bruno, R.M.; Hulot, J.S. Cardiac performance in patients hospitalized with COVID-19: A 6 month follow-up study. ESC Heart Fail. 2021, 8, 2232–2239. [Google Scholar] [CrossRef]
- Han, X.; Fan, Y.; Alwalid, O.; Li, N.; Jia, X.; Yuan, M.; Li, Y.; Cao, Y.; Gu, J.; Wu, H.; et al. Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology 2021, 299, E177–E186. [Google Scholar] [CrossRef]
- Liu, M.; Lv, F.; Huang, Y.; Xiao, K. Follow-Up Study of the Chest CT Characteristics of COVID-19 Survivors Seven Months After Recovery. Front. Med. 2021, 8, 212. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Bai, T.; Zhou, D.; Yushanjiang, F.; Wang, D.; Zhang, D.; Liu, X.; Song, J.; Zhang, J.; Hou, X.; Ma, Y. Alternation of the Autonomvous System Is ssociated with Pulmonary Sequelae in Patients With COVID-19 After Six Months of Discharge. Front. Physiol. 2021, 12, 80595. [Google Scholar] [CrossRef]
- Xianyu, Y.; Wang, M.; Yue, F.; Xu, X.; Yang, H.; Zhao, D.; Hu, K. One-year follow-up of 18 women who infected COVID-19 while pregnant. J. Med. Virol. 2022, 94, 2302–2306. [Google Scholar] [CrossRef] [PubMed]
- Augustin, M.; Schommers, P.; Stecher, M.; Dewald, F.; Gieselmann, L.; Gruell, H.; Horn, C.; Vanshylla, K.; Di Cristanziano, V.; Osebold, L.; et al. Post-COVID syndrome in non-hospitalised patient with COVID-19: A longitudinal prospective cohort study. Lancet Reg. Health-Eur. 2021, 6, 100122. [Google Scholar] [CrossRef] [PubMed]
- Van Veenendaal, N.; Van der Meulen, I.C.; Onrust, M.; Paans, W.; Dieperink, W.; Van der Voort, P.H.J. Six-Month Outcomes in COVID-19 ICU Patients and Their Family Members: A Prospective Cohort Study. Healthcare 2021, 9, 865. [Google Scholar] [CrossRef] [PubMed]
- Gamberini, L.; Mazzoli, C.A.; Prediletto, I.; Sintonen, H.; Scaramuzzo, G.; Allegri, D.; Colombo, D.; Tonetti, T.; Zani, G.; Capozzi, C.; et al. Health-related quality of life profiles, trajectories, persistent symptoms and pulmonary function one year after ICU discharge in invasively ventilated COVID-19 patients, a prospective follow-up study. Respir. Med. 2021, 189, 106665. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.; Beck, K.; Zumbrunn, S.; Memma, V.; Herzog, N.; Bissmann, B.; Gross, S.; Loretz, N.; Mueller, J.; Amacher, S.A.; et al. Long COVID 1 year after hospitalisation for COVID-19: A prospective bicentric cohort study. Swiss Med. Wkly. 2021, 151, w30091. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.J.; Kelly, J.D.; Lu, S.; Goldberg, S.A.; Davidson, M.C.; Mathur, S.; Durstenfeld, M.S.; Spinelli, M.A.; Hoh, R.; Tai, V.; et al. Persistence, Magnitude, and Patterns of Postacute Symptoms and Quality of Life Following Onset of SARS-CoV-2 Infection: Cohort Description and Approaches for Measurement. Open Forum Infect. Dis. 2022, 9, ofab640. [Google Scholar] [CrossRef]
- Erber, J.; Wiessner, J.R.; Zimmermann, G.S.; Barthel, P.; Burian, E.; Lohofer, F.; Martens, E.; Mijocevic, H.; Rasch, S.; Schmid, R.M.; et al. Longitudinal Assessment of Health and Quality of Life of COVID-19 Patients Requiring Intensive Care-An Observational Study. J. Clin. Med. 2021, 10, 5469. [Google Scholar] [CrossRef]
- Molhave, M.; Leth, S.; Gunst, J.; Jensen-Fangel, S.; Ostergaard, L.; Wejse, C.; Agergaard, J. Long-Term Symptoms among Hospitalized COVID-19 Patients 48 Weeks after Discharge-A Prospective Cohort Study. J. Clin. Med. 2021, 10, 5298. [Google Scholar] [CrossRef]
- Fortini, A.; Rosso, A.; Cecchini, P.; Torrigiani, A.; Lo Forte, A.; Carrai, P.; Alessi, C.; Fabbrizzi, F.; Lovicu, E.; Sbaragli, S.; et al. One-year evolution of DLCO changes and respiratory symptoms in patients with post COVID-19 respiratory syndrome. Infection 2022, 50, 513–517. [Google Scholar] [CrossRef]
- Kim, Y.; Bitna-Ha; Kim, S.W.; Chang, H.H.; Kwon, K.T.; Bae, S.; Hwang, S. Post-acute COVID-19 syndrome in patients after 12 months from COVID-19 infection in Korea. BMC Infect. Dis. 2022, 22, 93. [Google Scholar] [CrossRef]
- Fumagalli, C.; Zocchi, C.; Tassetti, L.; Silverii, M.V.; Amato, C.; Livi, L.; Giovannoni, L.; Verrillo, F.; Bartoloni, A.; Marcucci, R.; et al. Factors associated with persistence of symptoms 1 year after COVID-19: A longitudinal, prospective phone-based interview follow-up cohort study. Eur. J. Intern. Med. 2022, 97, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Bouteleux, B.; Henrot, P.; Ernst, R.; Grassion, L.; Raherison-Semjen, C.; Beaufils, F.; Zysman, M.; Delorme, M. Respiratory rehabilitation for COVID-19 related persistent dyspnoea: A one-year experience. Respir. Med. 2021, 189, 106648. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, J.S.; Oyelade, T.; Aldhahir, A.M.; Alghamdi, S.M.; Almehmadi, M.; Alqahtani, A.S.; Quaderi, S.; Mandal, S.; Hurst, J.R. Prevalence, Severity and Mortality associated with COPD and Smoking in patients with COVID-19: A Rapid Systematic Review and Meta-Analysis. PLoS ONE 2020, 15, e0233147. [Google Scholar] [CrossRef] [PubMed]
- George, P.M.; Wells, A.U.; Jenkins, R.G. Pulmonary fibrosis and COVID-19: The potential role for antifibrotic therapy. Lancet Respir. Med. 2020, 8, 807–815. [Google Scholar] [CrossRef]
- Testa, M.A.; Simonson, D.C. Assessment of quality-of-life outcomes. New Engl. J. Med. 1996, 334, 835–840. [Google Scholar] [CrossRef]
- Buchholz, I.; Janssen, M.F.; Kohlmann, T.; Feng, Y.S. A Systematic Review of Studies Comparing the Measurement Properties of the Three-Level and Five-Level Versions of the EQ-5D. PharmacoEconomics 2018, 36, 645–661. [Google Scholar] [CrossRef] [Green Version]
- Witteveen, D.; Velthorst, E. Economic hardship and mental health complaints during COVID-19. Proc. Natl. Acad. Sci. USA 2020, 117, 27277–27284. [Google Scholar] [CrossRef]
- McLean, S.A.; McIntosh, J.E. The mental and physical health of family mental health practitioners during COVID-19: Relationships with family violence and workplace practices. Aust. J. Psychol. 2021, 73, 395–404. [Google Scholar] [CrossRef]
| Consequences | 6–12 Months | 12 Months and Above | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number of Studies | Patients n/N | PP (%) | 95% CI (%) | p-Value | I2 | Number of Studies | Patients n/N | PP (%) | 95% CI (%) | p-Value | I2 | |
| General symptoms | ||||||||||||
| ≥1 Symptom | 13 | 4051/6477 | 63.87 | 53.64–74.09 | <0.05 | 98.70% | 8 | 1230/2290 | 58.89 | 45.87–71.91 | <0.05 | 97.20% |
| Fever | 7 | 64/3403 | 2.07 | 0.32–3.82 | <0.05 | 93.50% | 7 | 12/778 | 3.53 | −0.45–7.50 | >0.05 | 70.40% |
| Chill | 2 | 129/815 | 13.0 3 | −0.33–26.39 | >0.05 | 97.20% | ||||||
| Fatigue | 10 | 793/3000 | 30.94 | 20.21.41.66 | <0.05 | 98.20% | 14 | 822/3248 | 34.22 | 23.75–44.70 | <0.05 | 98.00% |
| Muscle weakness | 2 | 34/847 | 4.2 | 1.68–6.72 | <0.05 | 15.40% | - | - | - | - | - | - |
| Myalgia or joint pain | - | - | - | - | - | - | 2 | 187/503 | 34.52 | 9.01–60.02 | <0.05 | 97.50% |
| Fatigue or muscle weakness | 3 | 1949/3459 | 54.21 | 45.16–63.27 | <0.05 | 96.40% | - | - | - | - | - | - |
| Limited mobility | 3 | 83/943 | 21.81 | −4.17–47.78 | <0.05 | 97.90% | - | - | - | - | - | - |
| Myalgia | 9 | 271/4988 | 6.34 | 3.89–8.79 | <0.05 | 93.90% | 9 | 128/2368 | 6.59 | 4.05–9.13 | <0.05 | 79.80% |
| Joint pain | 6 | 396/3900 | 11.25 | 7.53–14.98 | <0.05 | 92.60% | 8 | 320/2058 | 18.73 | 12.24 –25.22 | <0.05 | 91.60% |
| Headache | 8 | 174/5134 | 3.68 | 2.20–5.15 | <0.05 | 89.70% | 5 | 93/1787 | 5.24 | 3.47–7.01 | <0.05 | 37.00% |
| Dizziness | 5 | 263/3289 | 14.96 | 9.72 –20.19 | <0.05 | 95.40% | 3 | 100/1607 | 8.14 | 3.82–12.46 | <0.05 | 77.70% |
| Olfactory or taste loss | 4 | 161/1556 | 14.38 | 8.40–20.36 | <0.05 | 90.20% | 2 | 21/259 | 8.21 | 2.84–13.58 | <0.05 | 54.60% |
| Olfactory loss | 8 | 491/4507 | 10.07 | 5.47–14.68 | <0.05 | 97.20% | 8 | 127/2004 | 8.22 | 5.21–11.23 | <0.05 | 70.00% |
| Taste loss | 8 | 338/4507 | 7.48 | 4.46–10.50 | <0.05 | 94.70% | 7 | 88/2308 | 4.55 | 2.45–6.65 | <0.05 | 78.00% |
| Hair loss | 7 | 712–4485 | 11.58 | 4.08–19.08 | <0.05 | 98.90% | 4 | 255–1807 | 18.42 | 9.21–27.63 | <0.05 | 94.30% |
| Cutaneous | 7 | 150/4200 | 3.87 | 2.32–5.43 | <0.05 | 84.50% | 5 | 99/2121 | 4.5 | 3.42–5.58 | <0.05 | 16.40% |
| Respiratory symptoms | ||||||||||||
| Respiratory symptoms | - | - | - | - | - | - | 2 | 79/241 | 32.7 | 3.97–61.43 | <0.05 | 96.20% |
| Cough | 12 | 381/3241 | 13.85 | 9.00–18.70 | <0.05 | 96.40% | 9 | 81/973 | 9.54 | 5.26–13.81 | <0.05 | 83.10% |
| Expectoration | 3 | 36/554 | 6.45 | 1.01–11.90 | <0.05 | 86.60% | 4 | 30/488 | 7.97 | 1.23–14.71 | <0.05 | 88.50% |
| Rhinorrhea | 2 | 13/267 | 7.44 | –5.43–20.30 | >0.05 | 89.00% | 2 | 68/210 | 30.93 | 11.60–50.26 | <0.05 | 90.10% |
| Sore throat or difficulty swallowing | 7 | 233/4885 | 4.43 | 2.49–6.37 | <0.05 | 92.60% | 6 | 78/1870 | 7.33 | 3.19–11.48 | <0.05 | 78.20% |
| Dyspnea | 12 | 717/3173 | 27.06 | 18.67–35.44 | <0.05 | 97.60% | 8 | 127/1129 | 16.43 | 9.66–23.20 | <0.05 | 91.90% |
| mMRC = 0 | 5 | 3491/3673 | 74.5 | 66.94–82.06 | <0.05 | 91.50% | 3 | 1042/1448 | 80.64 | 62.87–98.42 | <0.05 | 97.70% |
| mMRC ≥ 1 | 5 | 3491/3673 | 24.49 | 21.17–27.81 | <0.05 | 76.30% | 4 | 510/1622 | 29.1 | 10.64–47.56 | <0.05 | 98.10% |
| Cardiovascular symptoms | ||||||||||||
| Chest tightness | 2 | 200/815 | 21.18 | 4.94–37.43 | <0.05 | 97.00% | 3 | 24/278 | 10.24 | 0.77–19.71 | <0.05 | 62.30% |
| Chest pain | 9 | 265/5572 | 4.78 | 2.88–6.68 | <0.05 | 92.20% | 5 | 117–2009 | 7.76 | 2.60–12.91 | <0.05 | 93.50% |
| Back pain | 2 | 20/478 | 7.19 | −3.04–17.42 | >0.05 | 84.30% | - | - | - | - | - | - |
| Palpitations | 5 | 303/3604 | 7.19 | 3.68–10.71 | <0.05 | 93.40% | 7 | 173/2299 | 6.79 | 3.81–9.78 | < 0.05 | 86.00% |
| Gastrointestinal symptoms | ||||||||||||
| GI symptoms | 4 | 87/1049 | 15.62 | 4.91–26.34 | <0.05 | 96.80% | 5 | 71/1178 | 6.57 | 2.48–10.65 | <0.05 | 90.60% |
| Loss of appetite | 7 | 310/5106 | 4.65 | 1.98–7.32 | <0.05 | 96.70% | 5 | 58/1666 | 3.87 | 1.86–5.88 | <0.05 | 47.10% |
| Nausea, vomiting or diarrhea | 4 | 126/3699 | 3.47 | 1.41–5.52 | <0.05 | 91.80% | 4 | 34/1550 | 5.86 | 0.73–11.00 | <0.05 | 84.80% |
| Nausea | - | - | - | - | - | - | 2 | 8/63 | 10.55 | 0.75–20.35 | <0.05 | 41.70% |
| Vomiting | 2 | 16/996 | 2.01 | 1.03–2.98 | <0.05 | 0 | - | - | - | - | - | - |
| Diarrhea | 8 | 146/2272 | 6 | 2.86–9.15 | <0.05 | 94.10% | 4 | 8/397 | 2.18 | –0.56–4.91 | >0.05 | 45.90% |
| Stomachache | 2 | 50/865 | 6.52 | 2.48–10.57 | <0.05 | 40.70% | - | - | - | - | - | - |
| Constipation | 2 | 22/865 | 6.01 | −3.69–15.71 | >0.05 | 84.50% | - | - | - | - | - | - |
| Altered bowel habits | - | - | - | - | - | - | 2 | 27/165 | 16.17 | 10.56–21.78 | <0.05 | 0.00% |
| Neurological symptoms | ||||||||||||
| Neurological symptoms | 3 | 232/1803 | 10.81 | 0.40–21.21 | <0.05 | 98.60% | 4 | 167/634 | 23.85 | 11.42–36.29 | <0.05 | 92.70% |
| Polyneuropathy | 2 | 32/847 | 7.48 | −2.95–17.91 | >0.05 | 79.30% | - | - | - | - | - | - |
| Paresthesias | 4 | 68/1305 | 6.24 | 2.24–10.24 | <0.05 | 93.60% | 4 | 127/679 | 17.42 | 6.90–27.95 | <0.05 | 92.70% |
| Disorientation or confusion | 3 | 37/1237 | 2.7 | 0.31–5.09 | <0.05 | 88.10% | - | - | - | - | - | - |
| Forgetfulness | 2 | 131/815 | 18.65 | 5.23–32.08 | <0.05 | 94.80% | - | - | - | - | - | - |
| Memory loss | 3 | 89/850 | 10.65 | 1.86–19.43 | <0.05 | 96.50% | - | - | - | - | - | - |
| Visual impairment | 3 | 30/760 | 8.11 | −0.22–16.45 | >0.05 | 89.40% | - | - | - | - | - | - |
| Hearing impairment | 2 | 15/815 | 1.76 | 0.86–2.67 | <0.05 | 0.00% | - | - | - | - | - | - |
| Psychiatric symptoms | ||||||||||||
| Sleep difficulty | 9 | 1146/5121 | 24.11 | 14.67–33.56 | <0.05 | 98.90% | 5 | 476/2120 | 26.31 | 15.73–36.89 | <0.05 | 96.20% |
| GAD-7 score ≥ 10 | 2 | 62/639 | 10.8 | 8.26–13.34 | <0.05 | - | - | - | - | - | - | - |
| Depression | 6 | 301/1968 | 20.16 | 10.36–29.97 | <0.05 | 97.30% | 5 | 196/737 | 27.26 | 16.23–38.30 | <0.05 | 92.30% |
| Anxiety | 6 | 374/1970 | 25.19 | 13.88–36.49 | <0.05 | 97.60% | 5 | 213/737 | 29.78 | 16.29–43.27 | <0.05 | 94.70% |
| PTSD | 3 | 73/522 | 13.41 | 4.30–22.51 | <0.05 | 88.70% | 3 | 68/523 | 11.57 | 0.50–22.64 | <0.05 | 95.70% |
| Difficulty concentrating | 3 | 111/719 | 22.47 | 4.49–40.44 | <0.05 | 96.90% | 3 | 100/ 376 | 29.47 | 19.80–39.14 | <0.05 | 69.50% |
| PFT | ||||||||||||
| FVC < 80% | 4 | 45/441 | 13.66 | 5.23–22.09 | <0.05 | 64.90% | 5 | 43/ 374 | 12.78 | 3.99–21.56 | <0.05 | 87.10% |
| FEV1 < 80% | 2 | 11/46 | 28.03 | 1.04–55.03 | <0.05 | 66.80% | 3 | 28/216 | 13.81 | 5.57–22.05 | <0.05 | 59.50% |
| FEV1/FEV < 70% | 2 | 8/46 | 22.86 | 8.95–36.77 | <0.05 | - | 3 | 9/ 223 | 4.01 | −1.37–9.39 | >0.05 | 67.10% |
| VC < 80% | 2 | 43/323 | 13.27 | 9.57–16.97 | <0.05 | 0.00% | 2 | 22/199 | 11.05 | 6.70–15.41 | <0.05 | 0.00% |
| TLC < 80% | - | - | - | - | - | - | 4 | 28/285 | 9.28 | 4.28–14.27 | <0.05 | 49.40% |
| DLCO < 80% | 4 | 223/510 | 49.1 | 33.27–64.92 | <0.05 | 90.60% | 6 | 115/371 | 31.8 | 18.65–44.95 | <0.05 | 88.10% |
| CT results | ||||||||||||
| CT abnormality | 4 | 291/627 | 55.68 | 26.75–84.62 | <0.05 | 98.40% | 4 | 139/330 | 43.76 | 7.78–79.74 | <0.05 | 98.30% |
| GGO | 5 | 102/408 | 21.25 | 9.79–32.71 | <0.05 | 89.10% | 4 | 74/292 | 21.35 | 8.30–34.39 | <0.05 | 87.80% |
| Consolidation | 4 | 11/325 | 2.56 | 0.78–4.35 | <0.05 | 5.50% | 2 | 2/112 | 2.13 | –0.79–5.04 | >0.05 | - |
| Reticular pattern | 4 | 36/290 | 11.3 | 3.29–19.30 | <0.05 | 80.30% | 3 | 7/195 | 3.93 | 1.07–6.79 | <0.05 | 0.00% |
| Fibrosis | 3 | 108/272 | 66.28 | 52.35–80.21 | <0.05 | 63.00% | 3 | 29/209 | 13.88 | 6.04–21.72 | <0.05 | 56.40% |
| Crazy paving pattern | 3 | 0/211 | - | - | - | - | - | - | - | - | - | - |
| Air bronchogram | 3 | 0/211 | - | - | - | - | - | - | - | - | - | - |
| Bronchiectasis | 3 | 57/315 | 16.19 | −1.82–34.21 | >0.05 | 96.50% | 2 | 15/180 | 7.39 | −5.54–20.33 | >0.05 | 91.90% |
| Traction bronchiectasis | 2 | 16/93 | 17.63 | −3.45–38.70 | >0.05 | 86.20% | - | - | - | - | - | - |
| Nodules | 2 | 20/166 | 9 | −5.43–23.44 | >0.05 | 92.70% | 3 | 87/209 | 38.56 | 18.95–58.17 | <0.05 | 87.20% |
| Irregular interface | 2 | 12/93 | 12.88 | 6.07–19.68 | <0.05 | 0.00% | - | - | - | - | - | - |
| Parenchymal band | 2 | 30/93 | 32.53 | 16.45–48.60 | <0.05 | 64.90% | - | - | - | - | - | - |
| Pleural effusion | 3 | 2/284 | 1.69 | −0.63–4.02 | >0.05 | - | - | - | - | - | - | - |
| Pericardial effusion | 2 | 16/170 | 13.56 | 7.38–19.74 | <0.05 | - | - | - | - | - | - | - |
| Lymphadenopathy | 2 | 5/170 | 4.24 | 0.60–7.87 | <0.05 | - | - | - | - | - | - | - |
| Interlobular septal thickening | 4 | 63/294 | 21.75 | 6.66–36.84 | <0.05 | 91.60% | 3 | 14/195 | 7.33 | 1.68–12.98 | <0.05 | 53.80% |
| Lines and bands | - | - | - | - | - | - | 2 | 60/191 | 30.97 | −0.91–62.85 | >0.05 | 96.30% |
| Quality of life evaluation (EQ-5D-5L) | ||||||||||||
| Mobility | 4 | 278/3421 | 10.36 | 6.88–13.83 | <0.05 | 90.90% | 2 | 132/1436 | 9.18 | 7.68–10.67 | <0.05 | 0.00% |
| Personal care | 4 | 40/3422 | 0.94 | 0.11–1.77 | <0.05 | 83.20% | 2 | 23/1436 | 1.6 | 0.95–2.25 | <0.05 | 0.00% |
| Usual activity | 4 | 129/3413 | 6.68 | 3.61–9.76 | <0.05 | 96.30% | 2 | 52/1436 | 10.76 | −8.04–29.55 | >0.05 | 97.30% |
| Pain or discomfort | 4 | 989/3415 | 33.26 | 27.01–39.51 | <0.05 | 92.60% | 2 | 441/1436 | 35.31 | 22.38–48.24 | <0.05 | 90.60% |
| Anxiety and depression | 4 | 882/3418 | 33.49 | 23.87–43.12 | <0.05 | 97.20% | 2 | 406/1436 | 35.4 | 16.39–54.41 | <0.05 | 95.60% |
| 6MWT (distance lower than expected %) | 3 | 1086/3258 | 17.33 | 10.64–24.02 | <0.05 | 95.60% | - | - | - | - | - | - |
| Consequences | Study Number | Male n/N | Female n/N | OR | 95% CI | p-Value | I2 |
|---|---|---|---|---|---|---|---|
| ≥1 symptom | 5 | 977/1790 | 1113/1749 | 0.64 | 0.55–0.75 | <0.05 | 0.0% |
| General symptoms | |||||||
| Fever | 3 | 28/1435 | 32/1370 | 0.79 | 0.46–1.33 | >0.05 | 0.0% |
| Fatigue | 7 | 851/1971 | 942/1850 | 0.69 | 0.60–0.79 | <0.05 | 0.0% |
| Muscle weakness | 2 | 529/1284 | 639/1168 | 0.80 | 0.19–3.42 | >0.05 | 92.5% |
| Limited mobility | 3 | 61/619 | 71/577 | 0.76 | 0.51–1.15 | >0.05 | 0.0 |
| Myalgia | 3 | 79/1435 | 93/1370 | 0.79 | 0.49–1.27 | >0.05 | 36.9% |
| Headache | 3 | 26/1435 | 62/1370 | 0.40 | 0.25–0.65 | <0.05 | 0.0% |
| Dizziness | 2 | 70/907 | 77/843 | 0.79 | 0.55–1.14 | >0.05 | 0.0% |
| Olfactory or taste loss | 4 | 223/1487 | 251/1413 | 0.85 | 0.69–1.04 | >0.05 | 0.0% |
| Olfactory loss | 2 | 109/1007 | 119/1001 | 0.92 | 0.70–1.21 | >0.05 | 0.0% |
| Taste loss | 2 | 69/1007 | 90/1001 | 0.71 | 0.42–1.21 | >0.05 | 46.1% |
| Hair loss | 2 | 176/1007 | 192/1001 | 0.36 | 0.03–3.93 | >0.05 | 67.5% |
| Respiratory symptoms | |||||||
| Cough | 3 | 108/629 | 112/614 | 0.79 | 0.58–1.09 | >0.05 | 0.0% |
| Sore throat or difficulty swallowing | 3 | 68/1435 | 78/1370 | 0.79 | 0.57–1.11 | >0.05 | 0.0% |
| Dyspnea | 2 | 161/481 | 128/411 | 0.82 | 0.31–2.16 | >0.05 | 75.6% |
| mMRC = 0 | 2 | 786/1023 | 709/987 | 1.34 | 1.01–1.77 | <0.05 | 30.6% |
| mMRC ≥ 1 | 2 | 237/1023 | 278/955 | 0.64 | 0.36–1.11 | >0.05 | 78.6% |
| Cardiovascular symptoms | |||||||
| Chest pain | 3 | 66/1284 | 62/1168 | 0.96 | 0.67–1.37 | >0.05 | 0.0% |
| Gastrointestinal symptoms | |||||||
| GI symptoms | 3 | 101/618 | 105/576 | 0.83 | 0.61–1.13 | >0.05 | 0.0% |
| Loss of appetite | 2 | 73/1284 | 73/1168 | 0.92 | 0.66–1.30 | >0.05 | 0.0% |
| Nausea, vomiting or diarrhea | 3 | 78/1435 | 104/1370 | 0.65 | 0.41–1.03 | >0.05 | 38.7% |
| Diarrhea | 2 | 36/579 | 50/571 | 0.60 | 0.38–0.95 | <0.05 | 0.0% |
| Neurological symptoms | |||||||
| Paresthesias | 2 | 49/566 | 62/534 | 0.99 | 0.35–2.76 | >0.05 | 78.3% |
| Psychiatric symptoms | |||||||
| Sleep difficulty | 4 | 325/1474 | 365/1377 | 0.75 | 0.52–1.07 | >0.05 | 52.6% |
| Depression | 3 | 93/776 | 112/641 | 0.54 | 0.37–0.79 | <0.05 | 21.3% |
| Anxiety | 3 | 104/775 | 152/640 | 0.41 | 0.31–0.56 | <0.05 | 0.0% |
| Quality of life evaluation (EQ-5D-5L) | |||||||
| Mobility | 2 | 69/1046 | 92/1005 | 0.70 | 0.51–0.91 | <0.05 | 0.0% |
| Personal care | 2 | 6/1047 | 7/1005 | 0.82 | 0.27–2.45 | >0.05 | 0.0% |
| Usual activity | 2 | 25/1041 | 45/1000 | 0.52 | 0.31–0.85 | <0.05 | 0.0% |
| Pain or discomfort | 2 | 266/1044 | 316/1000 | 0.74 | 0.61–0.90 | <0.05 | 0.0% |
| Anxiety and depression | 2 | 200/1046 | 300/1001 | 0.55 | 0.45–0.68 | <0.05 | 0.0% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Y.; Deng, J.; Liu, Q.; Du, M.; Liu, M.; Liu, J. Long-Term Consequences of COVID-19 at 6 Months and Above: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 19, 6865. https://doi.org/10.3390/ijerph19116865
Ma Y, Deng J, Liu Q, Du M, Liu M, Liu J. Long-Term Consequences of COVID-19 at 6 Months and Above: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2022; 19(11):6865. https://doi.org/10.3390/ijerph19116865
Chicago/Turabian StyleMa, Yirui, Jie Deng, Qiao Liu, Min Du, Min Liu, and Jue Liu. 2022. "Long-Term Consequences of COVID-19 at 6 Months and Above: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 19, no. 11: 6865. https://doi.org/10.3390/ijerph19116865
APA StyleMa, Y., Deng, J., Liu, Q., Du, M., Liu, M., & Liu, J. (2022). Long-Term Consequences of COVID-19 at 6 Months and Above: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 19(11), 6865. https://doi.org/10.3390/ijerph19116865







