Everyday Lives of Middle-Aged Persons with Multimorbidity: A Mixed Methods Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Eligibility Criteria
2.2.1. Types of Study
2.2.2. Participants
2.2.3. Outcomes
2.3. Information Sources and Search
2.4. Study Selection
2.5. Data Collection Process
2.6. Data Items
2.7. Risk of Bias
2.8. Summary Measures
2.9. Synthesis of Results
3. Results
3.1. Key Characteristics of the Included Studies and Participants
3.2. Risk of Bias
3.3. Everyday Life Domains of Persons Living with Multimorbidity
3.3.1. Global Functioning
3.3.2. Physical Functioning
3.3.3. Psychosocial Functioning
3.3.4. Work Functioning
3.4. Coping Skills and Resources in Persons Living with Multimorbidity
3.4.1. Coping Skills
3.4.2. Coping Resources
Financial Resources
Resources Used to Lessen Treatment (Drug) Burden
4. Discussion
4.1. Summary of Evidence
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- van den Akker, M.; Buntinx, F.; Knottnerus, J.A. Comorbidity or multimorbidity: What’s in a name? A review of literature. Eur. J. Gen. Pract. 1996, 2, 65–70. [Google Scholar] [CrossRef]
- Violán, C.; Foguet-Boreu, Q.; Flores-Mateo, G.; Salisbury, C.; Blom, J.; Freitag, M.; Glynn, L.; Muth, C.; Valderas, J.M. Prevalence, Determinants and Patterns of Multimorbidity in Primary Care: A Systematic Review of Observational Studies. PLoS ONE 2014, 9, e102149. [Google Scholar] [CrossRef] [PubMed]
- Prados-Torres, A.; Calderón-Larrañaga, A.; Hancco-Saavedra, J.; Poblador-Plou, B.; Akker, M.V.D. Multimorbidity patterns: A systematic review. J. Clin. Epidemiol. 2014, 67, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, K.; Terry, A.L.; Fortin, M.; Williamson, T.; Bauer, M.; Thind, A. Prevalence, characteristics, and patterns of patients with multimorbidity in primary care: A retrospective cohort analysis in Canada. Br. J. Gen. Pract. 2019, 69, e647–e656. [Google Scholar] [CrossRef]
- Barnett, K.; Mercer, S.; Norbury, M.; Watt, G.; Wyke, S.; Guthrie, B. Epidemiology of multimorbidity and implications for health care, research, and medical education: A cross-sectional study. Lancet 2012, 380, 37–43. [Google Scholar] [CrossRef]
- Lachman, M.E.; Teshale, S.; Agrigoroaei, S. Midlife as a pivotal period in the life course. Int. J. Behav. Dev. 2014, 39, 20–31. [Google Scholar] [CrossRef] [PubMed]
- N’Goran, A.A.; Déruaz-Luyet, A.; Haller, D.M.; Zeller, A.; Rosemann, T.; Streit, S.; Herzig, L. Comparing the self-perceived quality of life of multimorbid patients and the general population using the EQ-5D-3L. PLoS ONE 2017, 12, e0188499. [Google Scholar] [CrossRef] [PubMed]
- Hopman, P.; Heins, M.J.; Rijken, M.; Schellevis, F.G. Health care utilization of patients with multiple chronic diseases in the Netherlands: Differences and underlying factors. Eur. J. Intern. Med. 2015, 26, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Willadsen, T.G.; Siersma, V.; Nicolaisdóttir, D.R.; Køster-Rasmussen, R.; Jarbøl, D.E.; Reventlow, S.; Mercer, S.W.; Olivarius, N.D.F. Multimorbidity and mortality: A 15-year longitudinal registry-based nationwide Danish population study. J. Comorbidity 2018, 8, 2235042X1880406. [Google Scholar] [CrossRef]
- Sum, G.; Ishida, M.; Koh, G.C.-H.; Singh, A.; Oldenburg, B.; Lee, J.T. Implications of multimorbidity on healthcare utilisation and work productivity by socioeconomic groups: Cross-sectional analyses of Australia and Japan. PLoS ONE 2020, 15, e0232281. [Google Scholar] [CrossRef]
- Bratzke, L.C.; Muehrer, R.J.; Kehl, K.A.; Lee, K.S.; Ward, E.C.; Kwekkeboom, K. Self-management priority setting and decision-making in adults with multimorbidity: A narrative review of literature. Int. J. Nurs. Stud. 2015, 52, 744–755. [Google Scholar] [CrossRef]
- Spencer-Bonilla, G.; Quiñones, A.R.; Montori, V.M. Assessing the Burden of Treatment. J. Gen. Intern. Med. 2017, 32, 1141–1145. [Google Scholar] [CrossRef]
- Ko, D.; Bratzke, L.C.; Roberts, T. Self-management assessment in multiple chronic conditions: A narrative review of literature. Int. J. Nurs. Stud. 2018, 83, 83–90. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 29, n71. [Google Scholar] [CrossRef]
- Lizarondo, L.; Stern, C.; Carrier, J.; Godfrey, C.; Rieger, K.; Salmond, S.; Apostolo, J.; Kirkpatrick, P.; Loveday, H. Chapter 8: Mixed methods systematic reviews. In JBI Manual for Evidence Synthesis; Aromataris, E., Munn, Z., Eds.; JBI: Adelaide, Australia, 2020. [Google Scholar]
- Carayanni, V.; Stylianopoulou, C.; Koulierakis, G.; Babatsikou, F.; Koutis, C. Sex differences in depression among older adults: Are older women more vulnerable than men in social risk factors? The case of open care centers for older people in Greece. Eur. J. Ageing 2012, 9, 177–186. [Google Scholar] [CrossRef]
- Browne, R.H. On the use of a pilot sample for sample size determination. Stat. Med. 1995, 14, 1933–1940. [Google Scholar] [CrossRef]
- Pluye, P.; Hong, Q.N. Combining the Power of Stories and the Power of Numbers: Mixed Methods Research and Mixed Studies Reviews. Annu. Rev. Public Health 2014, 35, 29–45. [Google Scholar] [CrossRef]
- Sandelowski, M.; Voils, C.I.; Barroso, J. Defining and Designing Mixed Research Synthesis Studies. Res. Sch. 2006, 13, 29. [Google Scholar] [PubMed]
- Pearson, A.; White, H.; Bath-Hextall, F.; Salmond, S.; Apostolo, J.L.A.; Kirkpatrick, P. A mixed-methods approach to systematic reviews. Int. J. Evid.-Based Healthc. 2015, 13, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.; Noyes, J.; Flemming, K.; Gerhardus, A.; Wahlster, P.; van der Wilt, G.J.; Mozygemba, K.; Refolo, P.; Sacchini, D.; Tummers, M.; et al. Structured methodology review identified seven (RETREAT) criteria for selecting qualitative evidence synthesis approaches. J. Clin. Epidemiol. 2018, 99, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.; Harden, A. Methods for the thematic synthesis of qualitative research in systematic reviews. BMC Med. Res. Methodol. 2008, 8, 45. [Google Scholar] [CrossRef]
- Chang, Y.; Voils, C.I.; Sandelowski, M.; Hasselblad, V.; Crandell, J.L. Transforming Verbal Counts in Reports of Qualitative Descriptive Studies Into Numbers. West. J. Nurs. Res. 2009, 31, 837–852. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Beran, D.; Haghparast-Bidgoli, H.; Batura, N.; Akkazieva, B.; Abdraimova, A.; Skordis-Worrall, J. Coping with the economic burden of Diabetes, TB and co-prevalence: Evidence from Bishkek, Kyrgyzstan. BMC Health Serv. Res. 2016, 16, 118. [Google Scholar] [CrossRef]
- Bell, B.; Chalklin, L.; Mills, M.; Browne, G.; Steiner, M.; Roberts, J.; Gafni, A.; Byrne, C.; Wallik, D.; Kraemer, J.; et al. Burden of dysthymia and comorbid illness in adults in a Canadian primary care setting: High rates of psychiatric illness in the offspring. J. Affect. Disord. 2004, 78, 73–80. [Google Scholar] [CrossRef]
- Buckner, J.D.; Timpano, K.R.; Zvolensky, M.J.; Sachs-Ericsson, N.; Schmidt, N.B. Implications of comorbid alcohol dependence among individuals with social anxiety disorder. Depress. Anxiety 2008, 25, 1028–1037. [Google Scholar] [CrossRef][Green Version]
- Cheng, C.; Bai, J.; Yang, C.; Li, M.; Inder, K.; Chan, S.W. Patients’ experiences of coping with multiple chronic conditions: A qualitative descriptive study. J. Clin. Nurs. 2019, 28, 4400–4411. [Google Scholar] [CrossRef]
- Conover, C.J.; Arno, P.; Weaver, M.; Ang, A.; Ettner, S.L. Income and employment of people living with combined HIV/AIDS, chronic mental illness, and substance abuse disorders. J. Ment. Health Policy Econ. 2006, 9, 71–86. [Google Scholar]
- Dagher, R.K.; Green, K.M. Does depression and substance abuse co-morbidity affect socioeconomic status? Evidence from a prospective study of urban African Americans. Psychiatry Res. 2015, 225, 115–121. [Google Scholar] [CrossRef][Green Version]
- D’Amico, D.; Sansone, E.; Grazzi, L.; Giovannetti, A.M.; Leonardi, M.; Schiavolin, S.; Raggi, A. Multimorbidity in patients with chronic migraine and medication overuse headache. Acta Neurol. Scand. 2018, 138, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Diaz, A.; Bernardes, C.; Garvey, G.; Valery, P. Supportive care needs among Indigenous cancer patients in Queensland, Australia: Less comorbidity is associated with greater practical and cultural unmet need. Eur. J. Cancer Care 2016, 25, 242–253. [Google Scholar] [CrossRef]
- Dickson, V.V.; Buck, H.; Riegel, B. Multiple Comorbid Conditions Challenge Heart Failure Self-Care by Decreasing Self-Efficacy. Nurs. Res. 2013, 62, 2–9. [Google Scholar] [CrossRef]
- Dutton, C.E.; Adams, T.; Bujarski, S.; Badour, C.L.; Feldner, M.T. Posttraumatic stress disorder and alcohol dependence: Individual and combined associations with social network problems. J. Anxiety Disord. 2014, 28, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Egede, L.E. Major depression in individuals with chronic medical disorders: Prevalence, correlates and association with health resource utilization, lost productivity and functional disability. Gen. Hosp. Psychiatry 2007, 29, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Gehrke, A.K.; Feuerstein, M. Cancer, comorbidity and workplace discrimination: The US experience. Eur. J. Cancer Care 2017, 26, e12748. [Google Scholar] [CrossRef]
- Gulley, S.P.; Rasch, E.K.; Chan, L. The Complex Web of Health: Relationships among Chronic Conditions, Disability, and Health Services. Public Health Rep. 2011, 126, 495–507. [Google Scholar] [CrossRef]
- Hakola, R.; Kauppi, P.; Leino, T.; Ojajärvi, A.; Pentti, J.; Oksanen, T.; Haahtela, T.; Kivimaki, M.; Vahtera, J. Persistent asthma, comorbid conditions and the risk of work disability: A prospective cohort study. Allergy 2011, 66, 1598–1603. [Google Scholar] [CrossRef] [PubMed]
- Johs, N.A.; Wu, K.; Tassiopoulos, K.; Koletar, S.L.; Kalayjian, R.C.; Ellis, R.J.; Taiwo, B.; Palella, F.J.; Erlandson, K.M. Disability among Middle-Aged and Older Persons with Human Immunodeficiency Virus Infection. Clin. Infect. Dis. 2017, 65, 83–91. [Google Scholar] [CrossRef]
- Li, N.; Chan, E.; Peterson, S. The economic burden of depression among adults with rheumatoid arthritis in the United States. J. Med. Econ. 2019, 22, 372–378. [Google Scholar] [CrossRef]
- Linder, J.; Ekholm, K.S.; Jansen, G.B.; Lundh, G.; Ekholm, J. Long-term sick leavers with difficulty in resuming work: Comparisons between psychiatric–somatic comorbidity and monodiagnosis. Int. J. Rehabil. Res. 2009, 32, 20–35. [Google Scholar] [CrossRef]
- Morgan, S.A.; Eyles, C.; Roderick, P.J.; Adongo, P.B.; Hill, A.G. Women living with multi-morbidity in the Greater Accra Region of Ghana: A qualitative study guided by the Cumulative Complexity Model. J. Biosoc. Sci. 2019, 51, 562–577. [Google Scholar] [CrossRef]
- Motl, R.W.; Fernhall, B.; McAuley, E.; Cutter, G. Physical Activity and Self-Reported Cardiovascular Comorbidities in Persons with Multiple Sclerosis: Evidence from a Cross-Sectional Analysis. Neuroepidemiology 2011, 36, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Neri, L.; Rey, L.A.R.; Gallieni, M.; Brancaccio, D.; Cozzolino, M.; Colombl, A.; Burroughs, T.E. Occupational Stress is Associated with Impaired Work Ability and Reduced Quality of Life in Patients with Chronic Kidney Failure. Int. J. Artif. Organs 2009, 32, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Nikiphorou, E.; Ramiro, S.; Van Der Heijde, D.; Norton, S.; Moltó, A.; Dougados, M.; Bosch, F.V.D.; Landewé, R. Association of Comorbidities in Spondyloarthritis with Poor Function, Work Disability, and Quality of Life: Results from the Assessment of SpondyloArthritis International Society Comorbidities in Spondyloarthritis Study. Arthritis Care Res. 2018, 70, 1257–1262. [Google Scholar] [CrossRef]
- Noel, P.H.; Frueh, B.C.; Larme, A.C.; Pugh, J. Collaborative care needs and preferences of primary care patients with multimorbidity. Health Expect. 2005, 8, 54–63. [Google Scholar] [CrossRef]
- O’Brien, R.; Wyke, S.; Watt, G.G.C.M.; Guthrie, B.; Mercer, S.W. The ‘Everyday Work’ of Living with Multimorbidity in Socioeconomically Deprived Areas of Scotland. J. Comorbidity 2014, 4, 1–10. [Google Scholar] [CrossRef]
- Ørtenblad, L.; Meillier, L.; Jønsson, A.R. Multi-morbidity: A patient perspective on navigating the health care system and everyday life. Chronic Illn. 2018, 14, 271–282. [Google Scholar] [CrossRef]
- Rao, A.S.; Scher, A.I.; Vieira, R.V.; Merikangas, K.R.; Metti, A.L.; Peterlin, B.L. The Impact of Post-Traumatic Stress Disorder on the Burden of Migraine: Results From the National Comorbidity Survey-Replication. Headache J. Head Face Pain 2015, 55, 1323–1341. [Google Scholar] [CrossRef] [PubMed]
- Ridgeway, J.; Eton, D.T.; Egginton, J.S.; Tiedje, K.; Linzer, M.; Boehm, D.; Poplau, S.; de Oliveira, D.R.; Odell, L.; Montori, V.M. Factors that lessen the burden of treatment in complex patients with chronic conditions: A qualitative study. Patient Prefer. Adherence 2014, 8, 339–351. [Google Scholar] [CrossRef]
- Romera, I.; Montejo, Á.L.; Caballero, F.; Caballero, L.; Arbesú, J.; Polavieja, P.; Desaiah, D.; Gilaberte, I. Functional impairment related to painful physical symptoms in patients with generalized anxiety disorder with or without comorbid major depressive disorder: Post hoc analysis of a cross-sectional study. BMC Psychiatry 2011, 11, 69. [Google Scholar] [CrossRef]
- Sand, C.D.; Rahbek, K.; Willadsen, T.G.; Jønsson, A.R. Prioritizing social identities: Patients’ perspective on living with multimorbidity. J. Multimorb. Comorbidity 2021, 11, 26335565211009375. [Google Scholar] [CrossRef] [PubMed]
- Saris, I.M.J.; Aghajani, M.; Van Der Werff, S.J.A.; Van Der Wee, N.J.A.; Penninx, B.W.J.H. Social functioning in patients with depressive and anxiety disorders. Acta Psychiatr. Scand. 2017, 136, 352–361. [Google Scholar] [CrossRef]
- Schofield, D.; Callander, E.J.; Shrestha, R.; Passey, M.E.; Percival, R.; Kelly, S. How co-morbidities magnify the effect of arthritis on labour force participation and economic status: A costs of illness study in Australia. Rheumatol. Int. 2014, 34, 481–489. [Google Scholar] [CrossRef]
- Schonauer, K.; Achtergarde, D.; Suslow, T.; Michael, N. Comorbidity of schizophrenia and prelingual deafness: Its impact on social network structures. Soc. Psychiatry Psychiatr. Epidemiol. 1999, 34, 526–532. [Google Scholar] [CrossRef]
- Slomka, J.; Prince-Paul, M.; Webel, A.; Daly, B.J. Multimorbidity with HIV: Views of Community-Based People Living with HIV and Other Chronic Conditions. J. Assoc. Nurses AIDS Care 2017, 28, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Souĕtre, E.; Lozet, H.; Cimarosti, I.; Martin, P.; Chignon, J.; Adés, J.; Tignol, J.; Darcourt, G. Cost of anxiety disorders: Impact of comorbidity. J. Psychosom. Res. 1994, 38, 151–160. [Google Scholar] [CrossRef]
- Subramanian, L.; Quinn, M.; Zhao, J.; Lachance, L.; Zee, J.; Tentori, F. Coping with kidney disease—Qualitative findings from the Empowering Patients on Choices for Renal Replacement Therapy (EPOCH-RRT) study. BMC Nephrol. 2017, 18, 119. [Google Scholar] [CrossRef]
- Tian, H.; Robinson, R.L.; Sturm, R. Labor market, financial, insurance and disability outcomes among near elderly Americans with depression and pain. J. Ment. Health Policy Econ. 2005, 8, 219–228. [Google Scholar]
- Todd, J.; Green, G.; Harrison, M.; Ikuesan, B.A.; Self, C.; Pevalin, D.; Baldacchino, A. Social exclusion in clients with comorbid mental health and substance misuse problems. Soc. Psychiatry Psychiatr. Epidemiol. 2004, 39, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Townsend, A.; Hunt, K.; Wyke, S. Managing multiple morbidity in mid-life: A qualitative study of attitudes to drug use. BMJ 2003, 327, 837. [Google Scholar] [CrossRef]
- Warren-Jeanpiere, L.; Dillaway, H.; Hamilton, P.; Young, M.; Goparaju, L. Taking It One Day at a Time: African American Women Aging with HIV and Co-Morbidities. AIDS Patient Care STDs 2014, 28, 372–380. [Google Scholar] [CrossRef]
- Weijman, I.; Kant, I.; Swaen, G.M.; Ros, W.J.G.; Rutten, G.E.H.M.; Schaufeli, W.B.; Schabracq, M.J.; Winnubst, J.A.M. Diabetes, Employment and Fatigue-Related Complaints: A Comparison between Diabetic Employees, “Healthy” Employees, and Employees with Other Chronic Diseases. J. Occup. Environ. Med. 2004, 46, 828–836. [Google Scholar] [CrossRef]
- White, C.; Lentin, P.; Farnworth, L. Multimorbidity and the process of living with ongoing illness. Chronic Illn. 2016, 12, 83–97. [Google Scholar] [CrossRef]
- White, C.; Lentin, P.; Farnworth, L. ‘I know what I am doing’: A grounded theory investigation into the activities and occupations of adults living with chronic conditions. Scand. J. Occup. Ther. 2019, 27, 56–65. [Google Scholar] [CrossRef]
- Wittchen, H.-U.; Fuetsch, M.; Sonntag, H.; Müller, N.; Liebowitz, M. Disability and quality of life in pure and comorbid social phobia. Findings from a controlled study. Eur. Psychiatry 2000, 15, 46–58. [Google Scholar] [CrossRef]
- Wittchen, H.-U.; Carter, R.M.; Pfister, H.; Montgomery, S.A.; Kessler, R.C. Disabilities and quality of life in pure and comorbid generalized anxiety disorder and major depression in a national survey. Int. Clin. Psychopharmacol. 2000, 15, 319–328. [Google Scholar] [CrossRef]
- Deschênes, S.S.; Burns, R.J.; Schmitz, N. Associations between diabetes, major depressive disorder and generalized anxiety disorder comorbidity, and disability: Findings from the 2012 Canadian Community Health Survey—Mental Health (CCHS-MH). J. Psychosom. Res. 2015, 78, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Schneier, F.R.; Heckelman, L.R.; Garfinkel, R.; Campeas, R.; Fallon, B.A.; Gitow, A.; Street, L.; Del Bene, D.; Liebowitz, M.R. Functional impairment in social phobia. J. Clin. Psychiatry 1994, 55, 322–331. [Google Scholar]
- Cheng, C.; Inder, K.; Chan, S.W.-C. Patients’ experiences of coping with multiple chronic conditions: A meta-ethnography of qualitative work. Int. J. Ment. Health Nurs. 2019, 28, 54–70. [Google Scholar] [CrossRef]
- Leon, A.C.; Olfson, M.; Portera, L.; Farber, L.; Sheehan, D.V. Assessing Psychiatric Impairment in Primary Care with the Sheehan Disability Scale. Int. J. Psychiatry Med. 1997, 27, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Calin, A.; Garrett, S.; Whitelock, H.; Kennedy, L.G.; O’Hea, J.; Mallorie, P.; Jenkinson, T. A new approach to defining functional ability in ankylosing spondylitis: The development of the Bath Ankylosing Spondylitis Functional Index. J. Rheumatol. 1994, 21, 2281–2285. [Google Scholar] [PubMed]
- England, B.R.; Sayles, H.; Mikuls, T.R.; Johnson, D.S.; Michaud, K. Validation of the Rheumatic Disease Comorbidity Index. Arthritis Rheum. 2015, 67, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Hadjimichael, O.; Kerns, R.D.; Rizzo, M.A.; Cutter, G.; Vollmer, T. Persistent pain and uncomfortable sensations in persons with multiple sclerosis. Pain 2007, 127, 35–41. [Google Scholar] [CrossRef]
- Garvey, G.; Beesley, V.L.; Janda, M.; O’Rourke, P.K.; He, V.Y.; Hawkes, A.L.; Elston, J.K.; Green, A.C.; Cunningham, J.; Valery, P.C. Psychometric properties of an Australian supportive care needs assessment tool for Indigenous patients with cancer. Cancer 2015, 121, 3018–3026. [Google Scholar] [CrossRef]
- Weissman, M.; Prusoff, B.A.; Thompson, W.D.; Harding, P.S.; Myers, J.K. Social Adjustment by Self-Report in a Community Sample and in Psychiatric Outpatients. J. Nerv. Ment. Dis. 1978, 166, 317–326. [Google Scholar] [CrossRef]
- Epstein, N.B.; Baldwin, L.M.; Bishop, D.S. The McMaster Family Assessment Device. J. Marital Fam. Ther. 1983, 9, 171–180. [Google Scholar] [CrossRef]
- Federici, S.; Bracalenti, M.; Meloni, F.; Luciano, J.V. World Health Organization disability assessment schedule 2.0: An international systematic review. Disabil. Rehabil. 2017, 39, 2347–2380. [Google Scholar] [CrossRef]
- Brazier, J.; Roberts, J.; Deverill, M. The estimation of a preference-based measure of health from the SF-36. J. Health Econ. 2002, 21, 271–292. [Google Scholar] [CrossRef]
- Reilly, M.C.; Zbrozek, A.S.; Dukes, E.M. The Validity and Reproducibility of a Work Productivity and Activity Impairment Instrument. PharmacoEconomics 1993, 4, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Tuomi, K. Work Ability Index, 2nd ed.; Helsinki, Ed.; Finnish Institute of Occupational Health: Helsinki, Finland, 1998. [Google Scholar]
- van Vegchel, N.; de Jonge, J.; Bosma, H.; Schaufeli, W. Reviewing the effort–reward imbalance model: Drawing up the balance of 45 empirical studies. Soc. Sci. Med. 2005, 60, 1117–1131. [Google Scholar] [CrossRef] [PubMed]
- Schofield, D.J.; Callander, E.J.; Shrestha, R.N.; Passey, M.E.; Percival, R.; Kelly, S.J. Association between co-morbidities and labour force participation amongst persons with back problems. Pain 2012, 153, 2068–2072. [Google Scholar] [CrossRef]
- Moos, R.; Cronkite, R.; Billings, A.; Finney, J. Indices of coping. In Health and Daily Living Form Manual; Social Ecology Laboratory, Veterans Administration and Stanford University Medical Centre: Palo Alto, CA, USA, 1984; pp. 16–18. [Google Scholar]
- Cabral, G.G.; de Souza, A.C.D.; Barbosa, I.R.; Jerez-Roig, J.; Souza, D.L. Multimorbidity and Its Impact on Workers: A Review of Longitudinal Studies. Saf. Health Work. 2019, 10, 393–399. [Google Scholar] [CrossRef]
- Fiske, A.; Wetherell, J.L.; Gatz, M. Depression in Older Adults. Annu. Rev. Clin. Psychol. 2009, 5, 363–389. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Liu, S.; Zhang, Y.; Wu, Y.; Shen, Y.; Ke, C. Bidirectional association between depression and multimorbidity in middle-aged and elderly Chinese adults: A longitudinal cohort study. Aging Ment. Health 2021, 1–7. [Google Scholar] [CrossRef]
- Spangenberg, L.; Forkmann, T.; Brähler, E.; Glaesmer, H. The association of depression and multimorbidity in the elderly: Implications for the assessment of depression. Psychogeriatrics 2011, 11, 227–234. [Google Scholar] [CrossRef]
- Liddy, C.; Blazkho, V.; Mill, K. Challenges of self-management when living with multiple chronic conditions: Systematic review of the qualitative literature. Can. Fam. Physician 2014, 60, 1123–1133. [Google Scholar]
- Rosbach, M.; Andersen, J.S. Patient-experienced burden of treatment in patients with multimorbidity—A systematic review of qualitative data. PLoS ONE 2017, 12, e0179916. [Google Scholar] [CrossRef]
- Sum, G.; Hone, T.; Atun, R.; Millett, C.; Suhrcke, M.; Mahal, A.; Koh, G.C.H.; Lee, J.T. Multimorbidity and out-of-pocket expenditure on medicines: A systematic review. BMJ Glob. Health 2018, 3, e000505. [Google Scholar] [CrossRef] [PubMed]


| Author, Year (Reference) | Country | Setting | Design | Data Collection Methods | Response (%) | Participants n | Female Sex, % | Age Mean, SD | Condition | Everyday Life Domains/Coping Strategies under Review |
|---|---|---|---|---|---|---|---|---|---|---|
| Arnold, 2016 [24] | Kyrgyzstan | Mixed-general practice and outpatient (specialized) | Observational, quantitative | Questionnaires | nr | 32 | 38 | 54, nr | Diabetes and TB | Work functioning and coping resources (financial) |
| Bell, 2004 [25] | Canada | General practice | Observational, quantitative | Interviews | 77 | 172 | 71 | 42, 14 | Dysthymia and CM | Social functioning and coping |
| Buckner, 2008 [26] | USA | Community | Observational, quantitative | Interviews | 83 | 195 | 40 | 33, 11 | Social anxiety disorder and alcohol dependence | Social functioning and coping |
| Cheng, 2019 [27] | China | Outpatient (specialized) | Observational, qualitative | Semi-structured interviews | nr | 14 | 43 | 53, 14 | MM | Physical, psychosocial and work functioning, coping |
| Conover, 2006 [28] | USA | Community | Observational, quantitative | Interviews | nr | 1138 | 58 | 38, nr | HIV, chronic mental illness and SUD | Work functioning and coping resources (financial) |
| Dagher, 2015 [29] | USA | Community | Observational, quantitative | Survey | nr | 66 | 39 | 32, 0.5 | SUD and depression | Work functioning |
| D’Amico, 2018 [30] | Italy | Outpatient (specialized) | Observational, quantitative | Interviews | nr | 62 | 77 | 48, nr | MM (including MOH or migraines) | Physical, psychosocial and work functioning |
| Deschenes, 2015 | Canada | Community | Observational, quantitative | Interviews | nr | 145 | nr | nr | Diabetes, depression and/or GAD | Physical and social functioning |
| Díaz, 2016 [31] | Australia | Outpatient (specialized) | Observational, quantitative | Questionnaires | nr | 107 | 55 | 57, 11 | Cancer and CM | Social functioning |
| Dickson, 2013 [32] | USA and Australia | Outpatient (specialized) | Observational, mixed methods | Questionnaires | nr | 114 | 38 | 59, 15 | MM (including heart failure) | Coping |
| Dutton, 2014 [33] | USA | Community | Observational, quantitative | Interviews | nr | 56 | 57 | 41, 1 | PTSD and alcohol dependence | Social functioning |
| Egede, 2007 [34] | USA | Community | Observational, quantitative | Questionnaires | nr | 975 | 64 | 46, nr | Depression and CM | Physical and work functioning |
| Gehrke, 2017 [35] | USA | Community | Observational, quantitative | Survey | nr | 333 | 63 | 49, 9 | Cancer and CM | Work functioning |
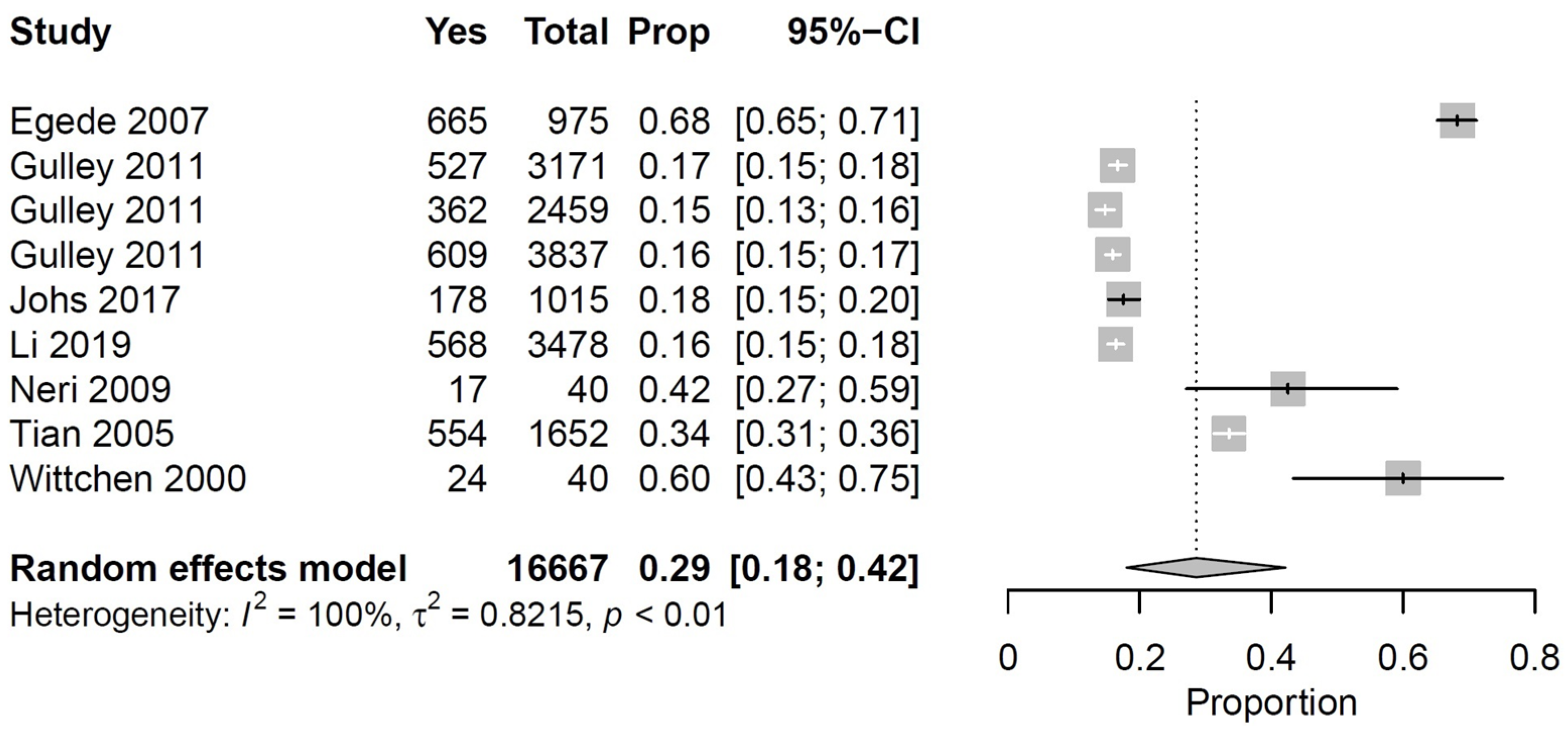
| Gulley, 2011 [36] | USA | Community | Observational, quantitative | Survey | nr | 29,171 | 57 | 45, 0.1 | Arthritis, diabetes and depression | Physical functioning |
| Hakola, 2011 [37] | Finland | Community | Observational, quantitative | Survey | 74 | 2332 | 85 | 45, 10 | Asthma and CM | Work functioning |
| Johs, 2017 [38] | USA | Outpatient (specialized) | Observational, quantitative | Questionnaire | nr | 1015 | 19 | 51, 8 | HIV and CM | Physical functioning |
| Li, 2019 [39] | USA | Community | Observational, quantitative | Survey | 100 | 3478 | 87 | 51, 10 | Rheumatoid arthritis and depression | Physical and work functioning |
| Linder, 2009 [40] | Sweden | Community | Observational, quantitative | Questionnaire | nr | 348 | 63 | 46, 8 | Psychiatric and somatic diagnosis | Work functioning |
| Morgan, 2019 [41] | Ghana | Outpatient (specialized) | Observational, qualitative | Interviews | nr | 20 | 100 | 55, 10 | MM | Physical, social and work functioning, coping |
| Motl, 2011 [42] | USA | Outpatient (specialized) | Observational, quantitative | Questionnaires | nr | 561 | 83 | 47, 10 | Multiple sclerosis and CVD | Physical functioning |
| Neri, 2009 [43] | USA | Outpatient (specialized) | Observational, quantitative | Questionnaires | 87 | 40 | 25 | 47, 8 | CKD and CM | Physical, social and work functioning |
| Nikiphorou, 2017 [44] | Multi-national | Outpatient (specialized) | Observational, quantitative | Questionnaires | nr | 3370 | 34 | 43, 14 | Spondylarthritis and CM | Physical and work functioning |
| Noël, 2005 [45] | USA | General practice | Observational, qualitative | Focus groups | 77 | 60 | 20 | 50, nr | MM | Physical, psychosocial and work functioning |
| O’Brien, 2014 [46] | UK | General practice | Observational, qualitative | Semi-structured interviews | nr | 14 | 50 | 54, 5 | MM | Coping |
| Ørtenblad, 2018 [47] | Denmark | Outpatient (specialized) | Observational, qualitative | Focus groups | nr | 10 | 50 | 51, 8 | MM | Social and work functioning, coping |
| Rao, 2015 [48] | USA | Community | Observational, quantitative | Survey | nr | 68 | 81 | 40, 1 | Migraines and PTSD | Social and work functioning |
| Ridgeway, 2014 [49] | USA | Outpatient (specialized) | Observational, qualitative | Interviews and focus groups | nr | 50 | 58 | 54, 13 | MM | Work functioning and coping |
| Romera, 2011 [50] | Spain | General practice | Observational, quantitative | Questionnaire | nr | 559 | 77 | 52, 15 | GAD and depression | Physical and work functioning |
| Sand, 2021 [51] | Denmark | General practice | Observational, qualitative | Semi-structured interviews | nr | 9 | 66 | 54, 9 | MM | Physical, psychosocial and work functioning, coping |
| Saris, 2017 [52] | Netherlands | Outpatient (specialized) | Observational, quantitative | Survey | nr | 748 | 68 | 41, 12 | Anxiety and depression | Social functioning |
| Schofield, 2014 [53] | Australia | Community | Observational, quantitative | Survey | nr | 1108 | nr | 53, nr | Arthritis and CM | Work functioning and coping resources (financial) |
| Schonauer, 1999 [54] | Germany | Outpatient (specialized) | Observational, quantitative | Semi-structured interview (sign language) | nr | 49 | 33 | 37, 11 | Prelingual deafness and Schizophrenia | Social and work functioning, coping |
| Slomka, 2017 [55] | USA | Outpatient (specialized) | Observational, qualitative | Focus groups | nr | 22 | 27 | 51, nr | HIV and CM | Social functioning and coping |
| Souêtre, 1994 [56] | France | General practice | Observational, quantitative | Questionnaires | nr | 604 | 60 | 41, 14 | GAD and CM | Work functioning |
| Subramanian, 2017 [57] | USA | Community | Observational, qualitative | Semi-structured interviews | 99 | 179 | 55 | 57, 7 | CKD and CM | Coping |
| Tian, 2005 [58] | USA | Community | Observational, quantitative | Survey | nr | 1652 | 65 | 60, 0.2 | Depression and CM | Physical and work functioning |
| Todd, 2004 [59] | UK | Outpatient (specialized) | Observational, quantitative | Survey | nr | 277 | 26 | 37, 11 | Mental health problems and SUD | Social and work functioning |
| Townsend, 2003 [60] | UK | Community | Observational, qualitative | Interviews | 56 | 23 | 57 | 50, nr | MM | Coping |
| Warren-Jeanpiere, 2014 [61] | USA | Outpatient (specialized) | Observational, qualitative | Focus groups | 46 | 23 | 100 | 57, 3 | HIV and CM | Coping |
| Weijman, 2004 [62] | Netherlands | Community | Observational, quantitative | Questionnaires | nr | 65 | 17 | 49, 7 | Diabetes and CM | Work functioning |
| White, 2016# [63] | Australia | Outpatient (specialized) | Observational, qualitative | Interviews | 100 | 16 | 69 | 47, 12 | MM | Social, work functioning and coping |
| White, 2019# [64] | Australia | Outpatient (specialized) | Observational, qualitative | Interviews | 100 | 16 | 69 | 47, 12 | MM | Coping |
| Wittchen, 2000 [65] | Germany | Community | Observational, quantitative | Interviews | nr | 51 | 61 | 38, 10 | Social phobia and CM | Psychosocial and work functioning |
| Wittchen, 2000 [66] | Germany | Community | Observational, quantitative | Interviews | 89 | 40 | 52 | 40, nr | GAD and depression | Physical and work functioning |
| Variable | Total—n (%) |
|---|---|
| Study characteristics | |
| Geographical location * | |
| - North America | 21 (48%) |
| - Europe | 17 (35%) |
| - Australia | 5 (10%) |
| - Asia | 1 (2%) |
| - Africa | 1 (2%) |
| Setting | |
| - Community | 19 (43%) |
| - Outpatient (specialized) | 17 (39%) |
| - General practice | 6 (14%) |
| - Mixed | 2 (5%) |
| Design | |
| - Quantitative | 30 (68%) |
| - Qualitative | 13 (30%) |
| - Mixed methods | 1 (2%) |
| Data collection methods * | |
| - Questionnaire/survey | 22 (50%) |
| - Interview | 13 (30%) |
| - Semi-structured interview | 5 (11%) |
| - Focus group | 5 (11%) |
| Sample size—Total (range) † | 49,371 (9–29,171) |
| - Observational | |
| ○ Quantitative † | 48,817 (32–29,171) |
| ○ Qualitative | 440 (9–179) |
| ○ Mixed methods | 114 (59) |
| Patients’ characteristics | |
| Type of condition * | |
| - Studies describing patients with multimorbidity | 12 (27%) |
| - Studies describing patients with a chronic condition associated with multimorbidity | |
| ○ Cancer | 2 (5%) |
| ○ Cardiovascular disease | 1 (2%) |
| ○ Chronic kidney disease | 2 (5%) |
| ○ Diabetes | 5 (11%) |
| ○ HIV | 4 (9%) |
| ○ Hypertension | 1 (2%) |
| ○ Mental health conditions | 19 (43%) |
| ○ Musculoskeletal disorders | 4 (9%) |
| ○ Neurological disorders | 2 (5%) |
| ○ Respiratory diseases | 1 (2%) |
| Age (range) | 32–60 |
| - Early middle age (30–44) | 13 (30) |
| - Late middle age (45–65) | 30 (68) |
| Sex (% female) | 28,477 (58%) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Scale Name | Score |
|---|---|---|---|---|---|
| D’Amico, 2018 [30] | MM | 48 (12) | 62 | WHODAS—mean (SD) * | 35 (13) |
| Wittchen, 2000 [65] | Social phobia and CM | 38 (10) | 51 | Disability Self-Rating Scale—LDSRS—mean [68] ** | 67.9 |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Scale Name | Score |
|---|---|---|---|---|---|
| Motl, 2011 [42] | Multiple sclerosis and CVD | 47 (12) | 561 | Patient-Determined Disease Steps (PDDS) scale [73]—Mean (SD) * | 3 (1) |
| Nikiphorou, 2017 [44] | Spondylarthritis and CM | 43 (14) | 3349 | Bath Ankylosing Spondylitis Functional Index (BASFI) [71]—Mean (SD) ** | 3 (3) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Score Mean (SD) * |
|---|---|---|---|---|
| Buckner, 2008 [26] | Social anxiety disorder and alcohol dependence | 33 (11) | 195 | 4 (2) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Outcome Categories | Score Mean (SD) * |
|---|---|---|---|---|---|
| Buckner, 2008 [26] | Social anxiety disorder and alcohol dependence | 33 (11) | 195 | Partner | 15 (4) |
| Relatives | 15 (4) | ||||
| Friends | 14 (4) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Scale Name | Score Mean (SD) * |
|---|---|---|---|---|---|
| Buckner, 2008 [26] | Social anxiety disorder and alcohol dependence | 33 (11) | 195 | Partner | 22 (3) |
| Relatives | 19 (4) | ||||
| Friends | 19 (5) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Scale Name | Score Mean (SD) |
|---|---|---|---|---|---|
| Saris, 2017 [52] | Anxiety and depression | 41 (12) | 748 | Network size * | 2 (1) |
| Social activities $ | 12 (4) | ||||
| Social support # | 25 (13) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Scale Name | Score Mean (SD) |
|---|---|---|---|---|---|
| Saris, 2017 [52] | Anxiety and depression | 41 (12) | 748 | Loneliness scale [73] * | 7 (4) |
| Affiliation $ | 4 (2) | ||||
| WHO-Disability Assessment Schedule, 2nd version (WHODAS 2.0) [77] # | 14 (5) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Outcome | n (%) |
|---|---|---|---|---|---|
| Neri, 2009 [43] | CKD and CM | 47 (8) | 40 | Severely limited social functioning | 22 (55) |
| Rao, 2015 [48] | Migraines and PTSD | 33 (11) | 68 | Difficulty interacting with others or maintaining a social life due to health problems | 27 (39) |
| First Author, Year | Type of Condition | Age, Mean (SD) | Participants, n | Scale Name | n (%) |
|---|---|---|---|---|---|
| Arnold, 2016 [24] | Diabetes and TB | 54 (NA) | 32 | Number of coping strategies, mean (95% CI) | 1.66 (1.32–1.99) |
| income and savings | 24 (75) | ||||
| social welfare and donations | 7 (22) | ||||
| social networks support | 18 (56) | ||||
| borrowing money | 3 (9) | ||||
| selling household assets | 1 (3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-González, A.I.; Brünn, R.; Nothacker, J.; Schwarz, C.; Nury, E.; Dinh, T.S.; Brueckle, M.-S.; Dieckelmann, M.; Müller, B.S.; van den Akker, M. Everyday Lives of Middle-Aged Persons with Multimorbidity: A Mixed Methods Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 6. https://doi.org/10.3390/ijerph19010006
González-González AI, Brünn R, Nothacker J, Schwarz C, Nury E, Dinh TS, Brueckle M-S, Dieckelmann M, Müller BS, van den Akker M. Everyday Lives of Middle-Aged Persons with Multimorbidity: A Mixed Methods Systematic Review. International Journal of Environmental Research and Public Health. 2022; 19(1):6. https://doi.org/10.3390/ijerph19010006
Chicago/Turabian StyleGonzález-González, Ana Isabel, Robin Brünn, Julia Nothacker, Christine Schwarz, Edris Nury, Truc Sophia Dinh, Maria-Sophie Brueckle, Mirjam Dieckelmann, Beate Sigrid Müller, and Marjan van den Akker. 2022. "Everyday Lives of Middle-Aged Persons with Multimorbidity: A Mixed Methods Systematic Review" International Journal of Environmental Research and Public Health 19, no. 1: 6. https://doi.org/10.3390/ijerph19010006
APA StyleGonzález-González, A. I., Brünn, R., Nothacker, J., Schwarz, C., Nury, E., Dinh, T. S., Brueckle, M.-S., Dieckelmann, M., Müller, B. S., & van den Akker, M. (2022). Everyday Lives of Middle-Aged Persons with Multimorbidity: A Mixed Methods Systematic Review. International Journal of Environmental Research and Public Health, 19(1), 6. https://doi.org/10.3390/ijerph19010006







