The Interaction of Vitamin D and Corticosteroids: A Mortality Analysis of 26,508 Veterans Who Tested Positive for SARS-CoV-2
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Data Source
2.2. Cohort Definition
2.3. Case Definition
2.4. Study Variables
2.5. Statistical Analysis
3. Results
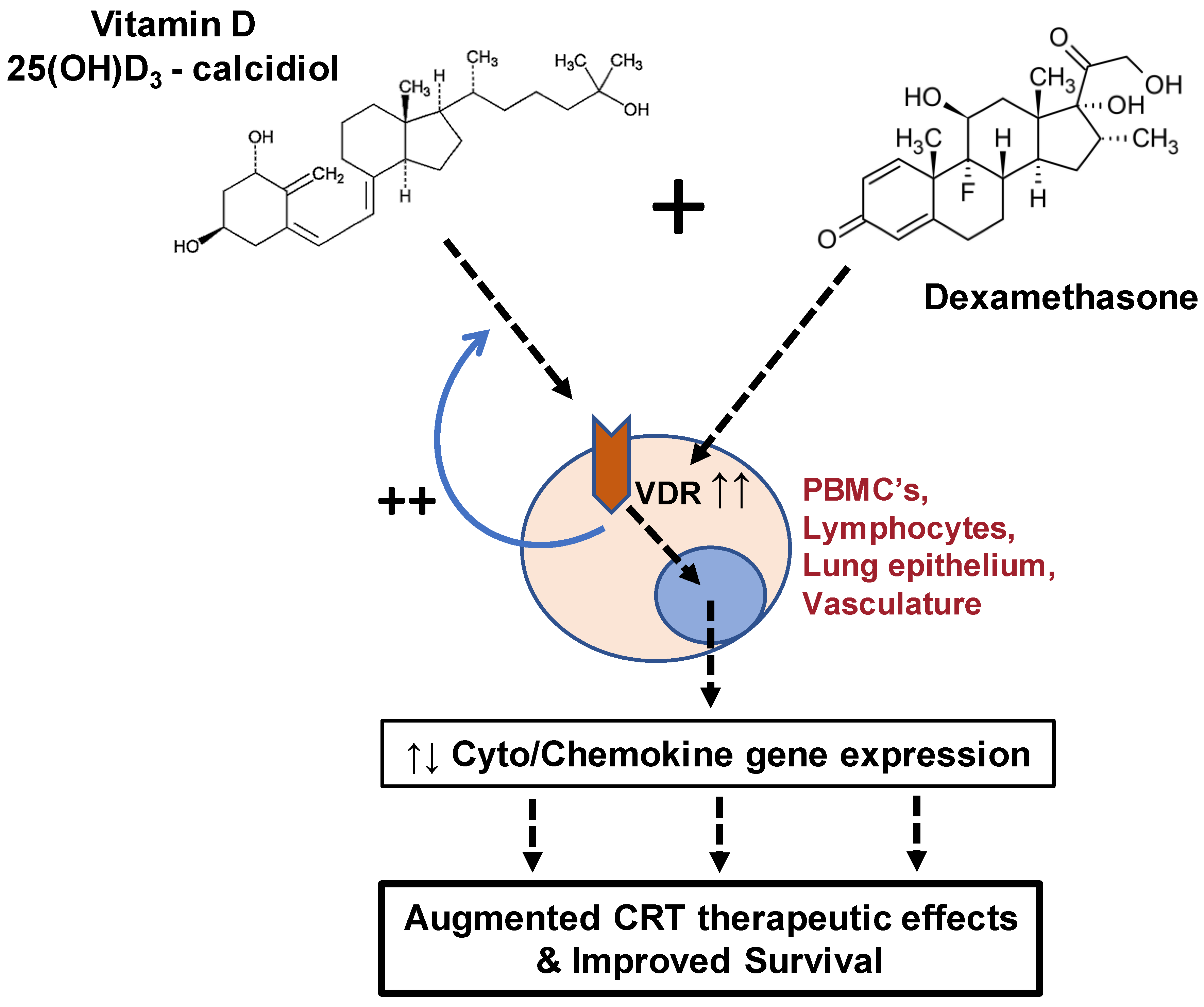
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Korf, H.; Decallonne, B.; Mathieu, C. Vitamin D for infections. Curr. Opin. Endocrinol. Diabetes Obes. 2014, 21, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; Agrawal, D.K. Vitamin D and inflammatory diseases. J. Inflamm. Res. 2014, 7, 69–87. [Google Scholar] [CrossRef]
- Ismailova, A.; White, J.H. Vitamin D, infections and immunity. Rev. Endocr. Metab. Disord. 2021, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D Deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Reijven, P.L.M.; Soeters, P.B. Vitamin D: A magic bullet or a myth? Clin. Nutr. 2020, 39, 2663–2674. [Google Scholar] [CrossRef] [PubMed]
- Brenner, H. Vitamin D Supplementation to Prevent COVID-19 Infections and Deaths-Accumulating Evidence from Epidemiological and Intervention Studies Calls for Immediate Action. Nutrients 2021, 13, 411. [Google Scholar] [CrossRef] [PubMed]
- Raisi-Estabragh, Z.; Martineau, A.R.; Curtis, E.M.; Moon, R.J.; Darling, A.; Lanham-New, S.; Ward, K.A.; Cooper, C.; Munroe, P.B.; Petersen, S.E.; et al. Vitamin D and coronavirus disease 2019 (COVID-19): Rapid evidence review. Aging Clin. Exp. Res. 2021, 33, 2031–2041. [Google Scholar] [CrossRef]
- Bilezikian, J.P.; Bikle, D.; Hewison, M.; Lazaretti-Castro, M.; Formenti, A.M.; Gupta, A.; Madhavan, M.V.; Nair, N.; Babalyan, V.; Hutchings, N.; et al. Mechanisma in endocrinology: Vitamin D and COVID-19. Eur. J. Endocrinol. 2020, 183, R133–R147. [Google Scholar] [CrossRef]
- Arthur, P. Vitamine D et COVID-19. Bull. Acad. Natl. Med. 2020, 204, 721–722. [Google Scholar]
- Vaughan, M.; Trott, M.; Sapkota, R.; Premi, G.; Roberts, J.; Ubhi, J.; Smith, L.; Pardhan, S. Changes in 25-hydroxyvitamin D levels post-vitamin D supplementation in people of Black and Asian ethnicities and its implications during COVID-19 pandemic: A systematic review. J. Hum. Nutr. Diet. 2021, 12949. [Google Scholar] [CrossRef] [PubMed]
- Iacobucci, G. COVID-19: NHS bosses told to assess risk to ethnic minority staff who may be at greater risk. BMJ 2020, 369, m1820. [Google Scholar] [CrossRef]
- Turrubiates-Hernández, F.J.; Sánchez-Zuno, G.A.; González-Estevez, G.; Hernández-Bello, J.; Macedo-Ojeda, G.; Muñoz-Valle, J.F. Potential immunomodulatory effects of vitamin D in the prevention of severe coronavirus disease 2019: An ally for Latin America (Review). Int. J. Mol. Med. 2021, 47, 1. [Google Scholar] [CrossRef]
- Kishore, J.; Grimes, D.S.; Anderson, D.C.; Bharti, O.K.; Joseph, B.; Ghai, G.; Sahoo, M. Recommendations for High Vitamin D Supplementation to help Fight the COVID-19 Pandemic and its Inclusion in National COVID-19 Management Guidelines. Epidem. Int. 2021, 6, 6–9. [Google Scholar]
- Munshi, R.; Hussein, M.H.; Toraih, E.A.; Elshazli, R.M.; Jardak, C.; Sultana, N.; Youssef, M.R.; Omar, M.; Attia, A.S.; Fawzy, M.S.; et al. Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J. Med. Virol. 2021, 93, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Liao, Q.; Shen, Y.; Li, H.; Cheng, L. Vitamin D Deficiency Is Associated with COVID-19 Incidence and Disease Severity in Chinese People. J. Nutr. 2021, 151, 98–103. [Google Scholar] [CrossRef]
- Tehrani, S.; Khabiri, N.; Moradi, H.; Mosavat, M.S.; Khabiri, S.S. Evaluation of vitamin D levels in COVID-19 patients referred to Labafinejad hospital in Tehran and its relationship with disease severity and mortality. Clin. Nutr. ESPEN 2021, 42, 313–317. [Google Scholar] [CrossRef]
- Reis, B.Z.; Fernandes, A.L.; Sales, L.P.; Santos, M.D.; Dos Santos, C.C.; Pinto, A.J.; Goessler, K.F.; Franco, A.S.; Duran, C.S.C.; Silva, C.B.R.; et al. Influence of vitamin D status on hospital length of stay and prognosis in hospitalized patients with moderate to severe COVID-19: A multicenter prospective cohort study. Am. J. Clin. Nutr. 2021, 114, 598–604. [Google Scholar] [CrossRef]
- Borsche, L.; Glauner, B.; von Mendel, J. COVID-19 mortality risk correlates inversely with vitamin D3 status, and a mortality rate close to zero could theoretically be achieved at 50 ng/ml 25(OH)D3: Results of a systematic review and meta-analysis. medRxiv 2021, 13, 3596. [Google Scholar] [CrossRef]
- Benskin, L.L. The influence of vitamin D on COVID-19 outcomes. In COVID-19 and Nutraceuticals: A Guidebook; Bohr Publishers and New Century Health Publishers: Tamil Nadu, India, 2021. [Google Scholar]
- Dramé, M.; Cofais, C.; Hentzien, M.; Proye, E.; Coulibaly, P.S.; Demoustier-Tampère, D.; Destailleur, M.H.; Lotin, M.; Cantagrit, E.; Cebille, A.; et al. Relation between Vitamin D and COVID-19 in Aged People: A Systematic Review. Nutrients 2021, 13, 1339. [Google Scholar] [CrossRef]
- Pal, R.; Banerjee, M.; Bhadada, S.K.; Shetty, A.J.; Singh, B.; Vyas, A. Vitamin D supplementation and clinical outcomes in COVID-19: A systematic review and meta-analysis. J. Endocrinol. Investig. 2021, 1–16. [Google Scholar] [CrossRef]
- Hariyanto, T.I.; Intan, D.; Hananto, J.E.; Harapan, H.; Kurniawan, A. Vitamin D supplementation and Covid-19 outcomes: A systematic review, meta-analysis and meta-regression. Rev. Med. Virol. 2021, 27, e2269. [Google Scholar] [CrossRef]
- Han, J.E.; Jones, J.L.; Tangpricha, V.; Brown, M.A.; Brown, L.A.S.; Hao, L.; Hebbar, G.; Lee, M.J.; Liu, S.; Ziegler, T.R.; et al. High Dose Vitamin D Administration in Ventilated Intensive Care Unit Patients: A Pilot Double Blind Randomized Controlled Trial. J. Clin. Transl. Endocrinol. 2016, 4, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Da Rocha, A.P.; Atallah, A.N.; Aldrighi, J.M.; Pires, A.L.R.; Dos Santos Puga, M.E.; Pinto, A. Insufficient evidence for vitamin D use in COVID-19: A rapid systematic review. Int. J. Clin. Pract. 2021, 75, e14649. [Google Scholar] [CrossRef]
- Boaz, M. Vitamin D and COVID-19: Partial Evidence. BCHD 2021, 4, 40–44. [Google Scholar] [CrossRef]
- Cereda, E.; Bogliolo, L.; Klersy, C.; Lobascio, F.; Masi, S.; Crotti, S.; De Stefano, L.; Bruno, R.; Corsico, A.G.; Di Sabatino, A.; et al. Vitamin D 25OH deficiency in COVID-19 patients admitted to a tertiary referral hospital. Clin. Nutr. 2021, 40, 2469–2472. [Google Scholar] [CrossRef]
- Cereda, E.; Bogliolo, L.; Lobascio, F.; Barichella, M.; Zecchinelli, A.L.; Pezzoli, G.; Caccialanza, R. Vitamin D supplementation and outcomes in coronavirus disease 2019 (COVID-19) patients from the outbreak area of Lombardy, Italy. Nutrition 2021, 82, 111055. [Google Scholar] [CrossRef] [PubMed]
- Campi, I.; Gennari, L.; Merlotti, D.; Mingiano, C.; Frosali, A.; Giovanelli, L.; Torlasco, C.; Pengo, M.F.; Heilbron, F.; Soranna, D.; et al. Vitamin D and COVID-19 severity and related mortality: A prospective study in Italy. BMC Infect. Dis. 2021, 21, 566. [Google Scholar] [CrossRef]
- Mariani, J.; Giménez, V.M.M.; Bergam, I.; Tajer, C.; Antonietti, L.; Inserra, F.; Ferder, L.; Manucha, W. Association Between Vitamin D Deficiency and COVID-19 Incidence, Complications, and Mortality in 46 Countries: An Ecological Study. Health Secur. 2021, 19, 302–308. [Google Scholar] [CrossRef]
- Baktash, V.; Hosack, T.; Patel, N.; Shah, S.; Kandiah, P.; Van den Abbeele, K.; Mandal, A.K.J.; Missouris, C.G. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad. Med. J. 2021, 97, 442–447. [Google Scholar] [CrossRef]
- Radujkovic, A.; Hippchen, T.; Tiwari-Heckler, S.; Dreher, S.; Boxberger, M.; Merle, U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 2020, 12, 2757. [Google Scholar] [CrossRef] [PubMed]
- Bianconi, V.; Mannarino, M.R.; Figorilli, F.; Cosentini, E.; Batori, G.; Marini, E.; Lombardini, R.; Gargaro, M.; Fallarino, F.; Scarponi, A.M.; et al. Prevalence of vitamin D deficiency and its prognostic impact on patients hospitalized with COVID-19. Nutrition 2021, 91–92, 111408. [Google Scholar] [CrossRef]
- Pecina, J.L.; Merry, S.P.; Park, J.G.; Thacher, T.D. Vitamin D Status and Severe COVID-19 Disease Outcomes in Hospitalized Patients. J. Prim. Care Community Health 2021, 12, 21501327211041206. [Google Scholar] [CrossRef]
- Butler-Laporte, G.; Nakanishi, T.; Mooser, V.; Morrison, D.R.; Abdullah, T.; Adeleye, O.; Mamlouk, N.; Kimchi, N.; Afrasiabi, Z.; Rezk, N.; et al. Vitamin D and COVID-19 susceptibility and severity in the COVID-19 Host Genetics Initiative: A Mendelian randomization study. PLoS Med. 2021, 18, e1003605. [Google Scholar] [CrossRef] [PubMed]
- Soliman, A.R.; Abdelaziz, T.S.; Fathy, A. Impact of Vitamin D Therapy on the Progress COVID-19: Six Weeks Follow-Up Study of Vitamin D Deficient Elderly Diabetes Patients. Proc. Singap. Health 2021, 20101058211041405. [Google Scholar] [CrossRef]
- Murai, I.H.; Fernandes, A.L.; Sales, L.P.; Pinto, A.J.; Goessler, K.F.; Duran, C.S.C.; Silva, C.B.R.; Franco, A.S.; Macedo, M.B.; Dalmolin, H.H.H.; et al. Effect of a Single High Dose of Vitamin D3 on Hospital Length of Stay in Patients with Moderate to Severe COVID-19: A Randomized Clinical Trial. JAMA 2021, 325, 1053–1060. [Google Scholar] [CrossRef]
- Güven, M.; Gültekin, H. The effect of high-dose parenteral vitamin D(3) on COVID-19-related inhospital mortality in critical COVID-19 patients during intensive care unit admission: An observational cohort study. Eur. J. Clin. Nutr. 2021, 75, 1383–1388. [Google Scholar] [CrossRef]
- Bassatne, A.; Basbous, M.; Chakhtoura, M.; El Zein, O.; Rahme, M.; El-Hajj Fuleihan, G. The link between COVID-19 and VItamin D (VIVID): A systematic review and meta-analysis. Metabolism 2021, 119, 154753. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, A.; Mohammadi, V.; Aghababaee, S.K.; Golzarand, M.; Clark, C.C.T.; Babajafari, S. Association of Vitamin D Status with SARS-CoV-2 Infection or COVID-19 Severity: A Systematic Review and Meta-analysis. Adv. Nutr. 2021, 12, 1636–1658. [Google Scholar] [CrossRef] [PubMed]
- Santaolalla, A.; Beckmann, K.; Kibaru, J.; Josephs, D.; Van Hemelrijck, M.; Irshad, S. Association Between Vitamin D and Novel SARS-CoV-2 Respiratory Dysfunction-A Scoping Review of Current Evidence and Its Implication for COVID-19 Pandemic. Front. Physiol. 2020, 11, 564387. [Google Scholar] [CrossRef]
- Sánchez-Zuno, G.A.; González-Estevez, G.; Matuz-Flores, M.G.; Macedo-Ojeda, G.; Hernández-Bello, J.; Mora-Mora, J.C.; Pérez-Guerrero, E.E.; García-Chagollán, M.; Vega-Magaña, N.; Turrubiates-Hernández, F.J.; et al. Vitamin D Levels in COVID-19 Outpatients from Western Mexico: Clinical Correlation and Effect of Its Supplementation. J. Clin. Med. 2021, 10, 2378. [Google Scholar] [CrossRef]
- Gibson-Moore, H. Vitamin D: What’s new a year on from the COVID-19 outbreak? Nutr. Bull. 2021, 46, 195–205. [Google Scholar] [CrossRef]
- Craft, J.F.; Travassos, M.A.; Foppiano Palacios, C.; Openshaw, J.J. Inadequate Minority Representation within SARS-CoV-2 Vaccine Trials. Am. J. Trop. Med. Hyg. 2021, 104, 32–34. [Google Scholar] [CrossRef] [PubMed]
- Szeto, B.; Zucker, J.E.; LaSota, E.D.; Rubin, M.R.; Walker, M.D.; Yin, M.T.; Cohen, A. Vitamin D Status and COVID-19 Clinical Outcomes in Hospitalized Patients. Endocr. Res. 2021, 46, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Carpagnano, G.E.; Di Lecce, V.; Quaranta, V.N.; Zito, A.; Buonamico, E.; Capozza, E.; Palumbo, A.; Di Gioia, G.; Valerio, V.N.; Resta, O. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J. Endocrinol. Investig. 2021, 44, 765–771. [Google Scholar] [CrossRef]
- Arvinte, C.; Singh, M.; Marik, P.E. Serum Levels of Vitamin C and Vitamin D in a Cohort of Critically Ill COVID-19 Patients of a North American Community Hospital Intensive Care Unit in May 2020: A Pilot Study. Med. Drug Discov. 2020, 8, 100064. [Google Scholar] [CrossRef]
- Abrishami, A.; Dalili, N.; Mohammadi Torbati, P.; Asgari, R.; Arab-Ahmadi, M.; Behnam, B.; Sanei-Taheri, M. Possible association of vitamin D status with lung involvement and outcome in patients with COVID-19: A retrospective study. Eur. J. Nutr. 2021, 60, 2249–2257. [Google Scholar] [CrossRef]
- Pizzini, A.; Aichner, M.; Sahanic, S.; Böhm, A.; Egger, A.; Hoermann, G.; Kurz, K.; Widmann, G.; Bellmann-Weiler, R.; Weiss, G.; et al. Impact of Vitamin D Deficiency on COVID-19-A Prospective Analysis from the CovILD Registry. Nutrients 2020, 12, 2775. [Google Scholar] [CrossRef]
- Kerget, B.; Kerget, F.; Kızıltunç, A.; Koçak, A.O.; Araz, Ö.; Yılmazel Uçar, E.; Akgün, M. Evaluation of the relationship of serum vitamin D levels in COVID-19 patients with clinical course and prognosis. Tuberk. Torak 2020, 68, 227–235. [Google Scholar] [CrossRef]
- Karonova, T.L.; Andreeva, A.T.; Vashukova, M.A. Serum 25 (OH) D level in COVID-19 patients. J. Infectol. 2020, 12, 21–27. [Google Scholar] [CrossRef]
- Faul, J.L.; Kerley, C.P.; Love, B.; O’Neill, E.; Cody, C.; Tormey, W.; Hutchinson, K.; Cormican, L.J.; Burke, C.M. Vitamin D Deficiency and ARDS after SARS-CoV-2 Infection. Ir. Med. J. 2020, 113, 84. [Google Scholar]
- De Smet, D.; De Smet, K.; Herroelen, P.; Gryspeerdt, S.; Martens, G.A. Serum 25(OH)D Level on Hospital Admission Associated with COVID-19 Stage and Mortality. Am. J. Clin. Pathol. 2021, 155, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Ye, K.; Tang, F.; Liao, X.; Shaw, B.A.; Deng, M.; Huang, G.; Qin, Z.; Peng, X.; Xiao, H.; Chen, C.; et al. Does Serum Vitamin D Level Affect COVID-19 Infection and Its Severity? A Case-Control Study. J. Am. Coll. Nutr. 2020, 40, 724–731. [Google Scholar] [CrossRef]
- Mardani, R.; Alamdary, A.; Mousavi Nasab, S.D.; Gholami, R.; Ahmadi, N.; Gholami, A. Association of vitamin D with the modulation of the disease severity in COVID-19. Virus Res. 2020, 289, 198148. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.K.; Wenban, C.; Heer, R.S.; Baktash, V.; Missouris, C.G. Does Vitamin D have a role to play in COVID-19 in the dexamethasone era? Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 102237. [Google Scholar] [CrossRef] [PubMed]
- Wenban, C.; Heer, R.S.; Baktash, V.; Kandiah, P.; Katsanouli, T.; Pandey, A.; Goindoo, R.; Ajaz, A.; Van den Abbeele, K.; Mandal, A.K.J.; et al. Dexamethasone treatment may mitigate adverse effects of vitamin D deficiency in hospitalized COVID-19 patients. J. Med. Virol. 2021, 93, 6605–6610. [Google Scholar] [CrossRef] [PubMed]
- Hahn, D.L. Glucocorticosteroids are potential confounders in studies of vitamin D and asthma. Am. J. Respir. Crit. Care Med. 2012, 185, 1245; author reply 1245–1246. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.A.; Agrawal, A.D.; Appanna, V.; Chaudagar, K.K. Vitamin D improves corticosteroid efficacy and attenuates its side-effects in an animal model of asthma. Can. J. Physiol. Pharmacol. 2015, 93, 53–61. [Google Scholar] [CrossRef]
- Agrawal, A.D. Evaluation of Corticosteroids-Vitamin D Combination in Asthma. PhD Thesis, Gujarat Technological University, Ahmedabad, India, 2016. [Google Scholar]
- Dhawan, P.; Christakos, S. Novel regulation of 25-hydroxyvitamin D3 24-hydroxylase (24(OH)ase) transcription by glucocorticoids:Ccooperative effects of the glucocorticoid receptor, C/EBP beta, and the Vitamin D receptor in 24(OH)ase transcription. J. Cell. Biochem. 2010, 110, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Skversky, A.L.; Kumar, J.; Abramowitz, M.K.; Kaskel, F.J.; Melamed, M.L. Association of glucocorticoid use and low 25-hydroxyvitamin D levels: Results from the National Health and Nutrition Examination Survey (NHANES): 2001–2006. J. Clin. Endocrinol. Metab. 2011, 96, 3838–3845. [Google Scholar] [CrossRef]
- Davidson, Z.E.; Walker, K.Z.; Truby, H. Do glucocorticosteroids alter vitamin D status? A systematic review with meta-analyses of observational studies. J. Clin. Endocrinol. Metab. 2012, 97, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Zoorob, R.J.; Cender, D. A different look at corticosteroids. Am. Fam. Physician 1998, 58, 443–450. [Google Scholar]
- Hidalgo, A.A.; Deeb, K.K.; Pike, J.W.; Johnson, C.S.; Trump, D.L. Dexamethasone enhances 1alpha,25-dihydroxyvitamin D3 effects by increasing vitamin D receptor transcription. J. Biol. Chem. 2011, 286, 36228–36237. [Google Scholar] [CrossRef]
- Zella, L.A.; Meyer, M.B.; Nerenz, R.D.; Lee, S.M.; Martowicz, M.L.; Pike, J.W. Multifunctional enhancers regulate mouse and human vitamin D receptor gene transcription. Mol. Endocrinol. 2010, 24, 128–147. [Google Scholar] [CrossRef]
- Kurahashi, I.; Matsunuma, A.; Kawane, T.; Abe, M.; Horiuchi, N. Dexamethasone enhances vitamin D-24-hydroxylase expression in osteoblastic (UMR-106) and renal (LLC-PK1) cells treated with 1alpha,25-dihydroxyvitamin D3. Endocrine 2002, 17, 109–118. [Google Scholar] [CrossRef]
- Xystrakis, E.; Kusumakar, S.; Boswell, S.; Peek, E.; Urry, Z.; Richards, D.F.; Adikibi, T.; Pridgeon, C.; Dallman, M.; Loke, T.K.; et al. Reversing the defective induction of IL-10-secreting regulatory T cells in glucocorticoid-resistant asthma patients. J. Clin. Investig. 2006, 116, 146–155. [Google Scholar] [CrossRef]
- Searing, D.A.; Zhang, Y.; Murphy, J.R.; Hauk, P.J.; Goleva, E.; Leung, D.Y. Decreased serum vitamin D levels in children with asthma are associated with increased corticosteroid use. J. Allergy Clin. Immunol. 2010, 125, 995–1000. [Google Scholar] [CrossRef]
- Zhang, Y.G.; Wu, S.; Sun, J. Vitamin D, Vitamin D Receptor, and Tissue Barriers. Tissue Barriers 2013, 1, e23118. [Google Scholar] [CrossRef]
- Ferreira, G.B.; Kleijwegt, F.S.; Waelkens, E.; Lage, K.; Nikolic, T.; Hansen, D.A.; Workman, C.T.; Roep, B.O.; Overbergh, L.; Mathieu, C. Differential protein pathways in 1,25-dihydroxyvitamin d(3) and dexamethasone modulated tolerogenic human dendritic cells. J. Proteome Res. 2012, 11, 941–971. [Google Scholar] [CrossRef] [PubMed]
- Penna, G.; Amuchastegui, S.; Giarratana, N.; Daniel, K.C.; Vulcano, M.; Sozzani, S.; Adorini, L. 1,25-Dihydroxyvitamin D3 selectively modulates tolerogenic properties in myeloid but not plasmacytoid dendritic cells. J. Immunol. 2007, 178, 145–153. [Google Scholar] [CrossRef]
- Garg, S.; Patel, K.; Pham, H.; Whitaker, M.; O’Halloran, A.; Milucky, J.; Anglin, O.; Kirley, P.D.; Reingold, A.; Kawasaki, B.; et al. Clinical Trends Among U.S. Adults Hospitalized With COVID-19, March to December 2020: A Cross-Sectional Study. Ann. Intern. Med. 2021, 174, 1409–1419. [Google Scholar] [CrossRef]
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef]
- Suzuki, A.; Efird, J.T.; Redding, T.S.t.; Thompson, A.D., Jr.; Press, A.M.; Williams, C.D.; Hostler, C.J.; Hunt, C.M. COVID-19-Associated Mortality in US Veterans with and without SARS-CoV-2 Infection. Int. J. Environ. Res. Public Health 2021, 18, 8486. [Google Scholar] [CrossRef]
- DuVall, S.; Scehnet, J. VA Informatics and Computing Infrastructure: Introduction to the VA COVID-19 Shared Data Resource and Its Use for Research NW Washington DC 204202020. Available online: https://www.hsrd.research.va.gov/cyberseminars/catalog-upcoming-session.cfm?UID=3810 (accessed on 29 November 2021).
- Chapman, A.; Peterson, K.; Turano, A.; Box, T.; Wallace, K.; Jones, M. A Natural Language Processing System for National COVID-19 Surveillance in the US Department of Veterans Affairs. Available online: https://openreview.net/forum?id=ZQ_HvBxcdCv (accessed on 4 December 2020).
- Basheer, M.; Saad, E.; Shlezinger, D.; Assy, N. Convalescent Plasma Reduces Mortality and Decreases Hospitalization Stay in Patients with Moderate COVID-19 Pneumonia. Metabolites 2021, 11, 761. [Google Scholar] [CrossRef]
- Breslow, N.E.; Day, N.E. The Analysis of Case-control Studies. In Statistical Methods in Cancer Research; IARC Scientific Publications No. 32; International Agency for Research on Cancer: Lyon, France, 1980; Volume 1. [Google Scholar]
- Bland, M. Introduction to Medical Statistics; Oxford University Press: Oxford, UK, 2015; p. 464. [Google Scholar]
- Efird, J.T. Goldilocks Rounding: Achieving Balance between Accuracy and Parsimony in the Reporting of Relative Effect Estimates. Cancer Inform. 2021, 20, 1176935120985132. [Google Scholar] [CrossRef]
- Benjamini, Y.; Yekutieli, D. False Discovery Rate–Adjusted Multiple Confidence Intervals for Selected Parameters. J. Am. Stat. Assoc. 2005, 100, 71–81. [Google Scholar] [CrossRef]
- Hochberg, Y. A sharper Bonferroni procedure for multiple tests of significance. Biometrika 1988, 75, 800–802. [Google Scholar] [CrossRef]
- Sundberg, R. An iterative method for solution of the likelihood equations for incomplete data from exponential families. Commun. Stat.-Simul. Comput. 1976, 5, 55–64. [Google Scholar] [CrossRef]
- Maghbooli, Z.; Sahraian, M.A.; Jamalimoghadamsiahkali, S.; Asadi, A.; Zarei, A.; Zendehdel, A.; Varzandi, T.; Mohammadnabi, S.; Alijani, N.; Karimi, M.; et al. Treatment with 25-Hydroxyvitamin D(3) (Calcifediol) Is Associated with a Reduction in the Blood Neutrophil-to-Lymphocyte Ratio Marker of Disease Severity in Hospitalized Patients with COVID-19: A Pilot Multicenter, Randomized, Placebo-Controlled, Double-Blinded Clinical Trial. Endocr. Pract. 2021, 27, 1242–1251. [Google Scholar] [CrossRef]
- Ohaegbulam, K.C.; Swalih, M.; Patel, P.; Smith, M.A.; Perrin, R. Vitamin D Supplementation in COVID-19 Patients: A Clinical Case Series. Am. J. Ther. 2020, 27, e485–e490. [Google Scholar] [CrossRef]
- Lakkireddy, M.; Gadiga, S.G.; Malathi, R.D.; Karra, M.L.; Raju, I.; Ragini; Chinapaka, S.; Baba, K.; Kandakatla, M. Impact of daily high dose oral vitamin D therapy on the inflammatory markers in patients with COVID 19 disease. Sci. Rep. 2021, 11, 10641. [Google Scholar] [CrossRef]
- Annweiler, C.; Hanotte, B.; Grandin de l’Eprevier, C.; Sabatier, J.M.; Lafaie, L.; Célarier, T. Vitamin D and survival in COVID-19 patients: A quasi-experimental study. J. Steroid Biochem. Mol. Biol. 2020, 204, 105771. [Google Scholar] [CrossRef]
- Annweiler, G.; Corvaisier, M.; Gautier, J.; Dubée, V.; Legrand, E.; Sacco, G.; Annweiler, C. Vitamin D Supplementation Associated to Better Survival in Hospitalized Frail Elderly COVID-19 Patients: The GERIA-COVID Quasi-Experimental Study. Nutrients 2020, 12, 3377. [Google Scholar] [CrossRef]
- Ling, S.F.; Broad, E.; Murphy, R.; Pappachan, J.M.; Pardesi-Newton, S.; Kong, M.F.; Jude, E.B. High-Dose Cholecalciferol Booster Therapy is Associated with a Reduced Risk of Mortality in Patients with COVID-19: A Cross-Sectional Multi-Centre Observational Study. Nutrients 2020, 12, 3799. [Google Scholar] [CrossRef] [PubMed]
- Giannini, S.; Passeri, G.; Tripepi, G.; Sella, S.; Fusaro, M.; Arcidiacono, G.; Torres, M.O.; Michielin, A.; Prandini, T.; Baffa, V.; et al. Effectiveness of In-Hospital Cholecalciferol Use on Clinical Outcomes in Comorbid COVID-19 Patients: A Hypothesis-Generating Study. Nutrients 2021, 13, 219. [Google Scholar] [CrossRef]
- Entrenas Castillo, M.; Entrenas Costa, L.M.; Vaquero Barrios, J.M.; Alcalá Díaz, J.F.; López Miranda, J.; Bouillon, R.; Quesada Gomez, J.M. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef] [PubMed]
- Alcala-Diaz, J.F.; Limia-Perez, L.; Gomez-Huelgas, R.; Martin-Escalante, M.D.; Cortes-Rodriguez, B.; Zambrana-Garcia, J.L.; Entrenas-Castillo, M.; Perez-Caballero, A.I.; López-Carmona, M.D.; Garcia-Alegria, J.; et al. Calcifediol Treatment and Hospital Mortality Due to COVID-19: A Cohort Study. Nutrients 2021, 13, 1760. [Google Scholar] [CrossRef]
- Cangiano, B.; Fatti, L.M.; Danesi, L.; Gazzano, G.; Croci, M.; Vitale, G.; Gilardini, L.; Bonadonna, S.; Chiodini, I.; Caparello, C.F.; et al. Mortality in an Italian nursing home during COVID-19 pandemic: Correlation with gender, age, ADL, vitamin D supplementation, and limitations of the diagnostic tests. Aging 2020, 12, 24522–24534. [Google Scholar] [CrossRef]
- Vasheghani, M.; Jannati, N.; Baghaei, P.; Rezaei, M.; Aliyari, R.; Marjani, M. The relationship between serum 25-hydroxyvitamin D levels and the severity of COVID-19 disease and its mortality. Sci. Rep. 2021, 11, 17594. [Google Scholar] [CrossRef]
- Tan, C.W.; Ho, L.P.; Kalimuddin, S.; Cherng, B.P.Z.; Teh, Y.E.; Thien, S.Y.; Wong, H.M.; Tern, P.J.W.; Chandran, M.; Chay, J.W.M.; et al. Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B(12) in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrition 2020, 79–80, 111017. [Google Scholar] [CrossRef]
- Rastogi, A.; Bhansali, A.; Khare, N.; Suri, V.; Yaddanapudi, N.; Sachdeva, N.; Puri, G.D.; Malhotra, P. Short term, high-dose vitamin D supplementation for COVID-19 disease: A randomised, placebo-controlled, study (SHADE study). Postgrad. Med. J. 2020, 1–4. [Google Scholar] [CrossRef]
- Jungreis, I.; Kellis, M. Mathematical analysis of Córdoba calcifediol trial suggests strong role for Vitamin D in reducing ICU admissions of hospitalized COVID-19 patients. medRxiv 2020. [Google Scholar] [CrossRef]
- Alguwaihes, A.M.; Sabico, S.; Hasanato, R.; Al-Sofiani, M.E.; Megdad, M.; Albader, S.S.; Alsari, M.H.; Alelayan, A.; Alyusuf, E.Y.; Alzahrani, S.H.; et al. Severe vitamin D deficiency is not related to SARS-CoV-2 infection but may increase mortality risk in hospitalized adults: A retrospective case-control study in an Arab Gulf country. Aging Clin. Exp. Res. 2021, 33, 1415–1422. [Google Scholar] [CrossRef]
- Jain, A.; Chaurasia, R.; Sengar, N.S.; Singh, M.; Mahor, S.; Narain, S. Analysis of vitamin D level among asymptomatic and critically ill COVID-19 patients and its correlation with inflammatory markers. Sci. Rep. 2020, 10, 20191. [Google Scholar] [CrossRef]
- Macaya, F.; Espejo Paeres, C.; Valls, A.; Fernández-Ortiz, A.; González Del Castillo, J.; Martín-Sánchez, F.J.; Runkle, I.; Rubio Herrera, M. Interaction between age and vitamin D deficiency in severe COVID-19 infection. Nutr. Hosp. 2020, 37, 1039–1042. [Google Scholar] [CrossRef]
- Mendy, A.; Apewokin, S.; Wells, A.A.; Morrow, A.L. Factors Associated with Hospitalization and Disease Severity in a Racially and Ethnically Diverse Population of COVID-19 Patients. medRxiv 2020. [Google Scholar] [CrossRef]
- Panagiotou, G.; Tee, S.A.; Ihsan, Y.; Athar, W.; Marchitelli, G.; Kelly, D.; Boot, C.S.; Stock, N.; Macfarlane, J.; Martineau, A.R.; et al. Low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID-19 are associated with greater disease severity. Clin. Endocrinol. 2020, 93, 508–511. [Google Scholar] [CrossRef]
- Barassi, A.; Pezzilli, R.; Mondoni, M.; Rinaldo, R.F.; DavÌ, M.; Cozzolino, M.; Melzi D’Eril, G.V.; Centanni, S. Vitamin D in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) patients with non-invasive ventilation support. Panminerva Med. 2021. [Google Scholar] [CrossRef]
- Seal, K.; Bertenthal, D.; Carey, E. Impact of Low Vitamin D Levels on COVID19-Related Hospitalization and Mortality: Results From a National Cohort of Veterans Affairs Patients. Glob. Adv. Health Med. 2021, 10, 1–64. [Google Scholar] [CrossRef]
- Gönen, M.S.; Alaylıoğlu, M.; Durcan, E.; Özdemir, Y.; Şahin, S.; Konukoğlu, D.; Nohut, O.K.; Ürkmez, S.; Küçükece, B.; Balkan, İ.; et al. Rapid and Effective Vitamin D Supplementation May Present Better Clinical Outcomes in COVID-19 (SARS-CoV-2) Patients by Altering Serum INOS1, IL1B, IFNg, Cathelicidin-LL37, and ICAM1. Nutrients 2021, 13, 4047. [Google Scholar] [CrossRef]
- Loucera, C.; Peña-Chilet, M.; Esteban-Medina, M.; Muñoyerro-Muñiz, D.; Villegas, R.; Lopez-Miranda, J.; Rodriguez-Baño, J.; Túnez, I.; Bouillon, R.; Dopazo, J.; et al. Real world evidence of calcifediol or vitamin D prescription and mortality rate of COVID-19 in a retrospective cohort of hospitalized Andalusian patients. Sci. Rep. 2021, 11, 23380. [Google Scholar] [CrossRef]
- Jevalikar, G.; Mithal, A.; Singh, A.; Sharma, R.; Farooqui, K.J.; Mahendru, S.; Dewan, A.; Budhiraja, S. Lack of association of baseline 25-hydroxyvitamin D levels with disease severity and mortality in Indian patients hospitalized for COVID-19. Sci. Rep. 2021, 11, 6258. [Google Scholar] [CrossRef] [PubMed]
- Hernández, J.L.; Nan, D.; Fernandez-Ayala, M.; García-Unzueta, M.; Hernández-Hernández, M.A.; López-Hoyos, M.; Muñoz-Cacho, P.; Olmos, J.M.; Gutiérrez-Cuadra, M.; Ruiz-Cubillán, J.J.; et al. Vitamin D Status in Hospitalized Patients with SARS-CoV-2 Infection. J. Clin. Endocrinol. Metab. 2021, 106, e1343–e1353. [Google Scholar] [CrossRef]
- Lohia, P.; Nguyen, P.; Patel, N.; Kapur, S. Exploring the link between vitamin D and clinical outcomes in COVID-19. Am. J. Physiol. Metab. 2021, 320, E520–E526. [Google Scholar] [CrossRef] [PubMed]
- Pouramini, A.; Kafi, F.; Hassanzadeh, S. Vitamin D and COVID-19 infection; recent findings. J. Ren. Endocrinol. 2021, 7, e19. [Google Scholar] [CrossRef]
- Dayal, D.; Gupta, B.M.; Surulinathi, M.; Nanda, P. COVID-19 and Vitamin D Deficiency: A Scientometric Assessment of Global Publications during 2020–21. J. Young Pharm. 2021, 13, s89–s94. [Google Scholar] [CrossRef]
- Filep, J.G.; Delalandre, A.; Payette, Y.; Földes-Filep, E. Glucocorticoid receptor regulates expression of L-selectin and CD11/CD18 on human neutrophils. Circulation 1997, 96, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Burton, J.L.; Kehrli, M.E., Jr.; Kapil, S.; Horst, R.L. Regulation of L-selectin and CD18 on bovine neutrophils by glucocorticoids: Effects of cortisol and dexamethasone. J. Leukoc. Biol. 1995, 57, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Weber, P.S.; Toelboell, T.; Chang, L.C.; Tirrell, J.D.; Saama, P.M.; Smith, G.W.; Burton, J.L. Mechanisms of glucocorticoid-induced down-regulation of neutrophil L-selectin in cattle: Evidence for effects at the gene-expression level and primarily on blood neutrophils. J. Leukoc. Biol. 2004, 75, 815–827. [Google Scholar] [CrossRef]
- Smith, C.W. Endothelial adhesion molecules and their role in inflammation. Can. J. Physiol. Pharmacol. 1993, 71, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Van Eeden, S.; Miyagashima, R.; Haley, L.; Hogg, J.C. L-selectin expression increases on peripheral blood polymorphonuclear leukocytes during active marrow release. Am. J. Respir. Crit. Care Med. 1995, 151, 500–507. [Google Scholar] [CrossRef]
- Török, C.; Lundahl, J.; Hed, J.; Lagercrantz, H. Diversity in regulation of adhesion molecules (Mac-1 and L-selectin) in monocytes and neutrophils from neonates and adults. Arch. Dis. Child. 1993, 68, 561–565. [Google Scholar] [CrossRef]
- Cronstein, B.N.; Kimmel, S.C.; Levin, R.I.; Martiniuk, F.; Weissmann, G. A mechanism for the antiinflammatory effects of corticosteroids: The glucocorticoid receptor regulates leukocyte adhesion to endothelial cells and expression of endothelial-leukocyte adhesion molecule 1 and intercellular adhesion molecule 1. Proc. Natl. Acad. Sci. USA 1992, 89, 9991–9995. [Google Scholar] [CrossRef] [PubMed]
- Tahir, N.; Zahra, F. Neutrophilia. StatPearls Publishing Copyright © 2021. In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2021. [Google Scholar]
- Sterne, J.A.C.; Murthy, S.; Diaz, J.V.; Slutsky, A.S.; Villar, J.; Angus, D.C.; Annane, D.; Azevedo, L.C.P.; Berwanger, O.; Cavalcanti, A.B.; et al. Association between Administration of Systemic Corticosteroids and Mortality among Critically Ill Patients with COVID-19: A Meta-analysis. JAMA 2020, 324, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- Cohn, B.A.; Cirillo, P.M.; Murphy, C.C.; Krigbaum, N.Y.; Wallace, A.W. SARS-CoV-2 vaccine protection and deaths among US veterans during 2021. Science 2021, eabm0620. [Google Scholar] [CrossRef]
- Mazess, R.B.; Bischoff-Ferrari, H.A.; Dawson-Hughes, B. Vitamin D: Bolus Is Bogus—A Narrative Review. JBMR Plus 2021, e10567. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; Goodall, E.C.; et al. Vitamin D supplementation to prevent acute respiratory infections: Individual participant data meta-analysis. Health Technol. Assess. 2019, 23, 1–44. [Google Scholar] [CrossRef] [PubMed]
- Griffin, G.; Hewison, M.; Hopkin, J.; Kenny, R.A.; Quinton, R.; Rhodes, J.; Subramanian, S.; Thickett, D. Perspective: Vitamin D supplementation prevents rickets and acute respiratory infections when given as daily maintenance but not as intermittent bolus: Implications for COVID-19. Clin. Med. 2021, 21, e144–e149. [Google Scholar] [CrossRef]
- Ketha, H.; Thacher, T.D.; Oberhelman, S.S.; Fischer, P.R.; Singh, R.J.; Kumar, R. Comparison of the effect of daily versus bolus dose maternal vitamin D(3) supplementation on the 24,25-dihydroxyvitamin D(3) to 25-hydroxyvitamin D(3) ratio. Bone 2018, 110, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Burt, L.A.; Billington, E.O.; Rose, M.S.; Raymond, D.A.; Hanley, D.A.; Boyd, S.K. Effect of High-Dose Vitamin D Supplementation on Volumetric Bone Density and Bone Strength: A Randomized Clinical Trial. JAMA 2019, 322, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Klinger, M.; Alexiewicz, J.M.; Linker-Israeli, M.; Pitts, T.O.; Gaciong, Z.; Fadda, G.Z.; Massry, S.G. Effect of parathyroid hormone on human T cell activation. Kidney Int. 1990, 37, 1543–1551. [Google Scholar] [CrossRef]
- Shurtz-Swirski, R.; Shkolnik, T.; Shasha, S.M. Parathyroid hormone and the cellular immune system. Nephron 1995, 70, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Lewin, E.; Ladefoged, J.; Brandi, L.; Olgaard, K. Parathyroid hormone dependent T cell proliferation in uremic rats. Kidney Int. 1993, 44, 379–384. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Geara, A.S.; Castellanos, M.R.; Bassil, C.; Schuller-Levis, G.; Park, E.; Smith, M.; Goldman, M.; Elsayegh, S. Effects of parathyroid hormone on immune function. Clin. Dev. Immunol. 2010, 2010, 418695. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.; Prince, D.K.; Williams, K.; Allen, N.B.; Burke, G.L.; Hoofnagle, A.N.; Li, X.; Liu, K.J.; McClelland, R.L.; Michos, E.D.; et al. Clinical and biomarker modifiers of vitamin D treatment response: The multi-ethnic study of atherosclerosis. Am. J. Clin. Nutr. 2021. [Google Scholar] [CrossRef]
- Leaf, D.E.; Ginde, A.A. Vitamin D3 to Treat COVID-19: Different Disease, Same Answer. JAMA 2021, 325, 1047–1048. [Google Scholar] [CrossRef] [PubMed]
- Hung, A.; Lee, K.M.; Lynch, J.A.; Li, Y.; Poonnen, P.; Efimova, O.V.; Hintze, B.J.; Buckingham, T.; Yong, C.; Seal, B.; et al. Chemoradiation treatment patterns among United States Veteran Health Administration patients with unresectable stage III non-small cell lung cancer. BMC Cancer 2021, 21, 824. [Google Scholar] [CrossRef] [PubMed]
- Razjouyan, J.; Helmer, D.A.; Lynch, K.E.; Hanania, N.A.; Klotman, P.E.; Sharafkhaneh, A.; Amos, C.I. Smoking Status and Factors associated with COVID-19 In-hospital Mortality among U.S. Veterans. Nicotine Tob. Res. 2021. [Google Scholar] [CrossRef]
- Griffith, G.J.; Morris, T.T.; Tudball, M.J.; Herbert, A.; Mancano, G.; Pike, L.; Sharp, G.C.; Sterne, J.; Palmer, T.M.; Davey Smith, G.; et al. Collider bias undermines our understanding of COVID-19 disease risk and severity. Nat. Commun. 2020, 11, 5749. [Google Scholar] [CrossRef]
- Song, R.J.; Ho, Y.L.; Schubert, P.; Park, Y.; Posner, D.; Lord, E.M.; Costa, L.; Gerlovin, H.; Kurgansky, K.E.; Anglin-Foote, T.; et al. Phenome-wide association of 1809 phenotypes and COVID-19 disease progression in the Veterans Health Administration Million Veteran Program. PLoS ONE 2021, 16, e0251651. [Google Scholar] [CrossRef]
- Escobar, G.J.; Adams, A.S.; Liu, V.X.; Soltesz, L.; Chen, Y.I.; Parodi, S.M.; Ray, G.T.; Myers, L.C.; Ramaprasad, C.M.; Dlott, R.; et al. Racial Disparities in COVID-19 Testing and Outcomes: Retrospective Cohort Study in an Integrated Health System. Ann. Intern. Med. 2021, 174, 786–793. [Google Scholar] [CrossRef]
- Pearce, N.; Rhodes, S.; Stocking, K.; Pembrey, L.; van Veldhoven, K.; Brickley, E.B.; Robertson, S.; Davoren, D.; Nafilyan, V.; Windsor-Shellard, B.; et al. Occupational differences in COVID-19 incidence, severity, and mortality in the United Kingdom: Available data and framework for analyses. Wellcome Open Res. 2021, 6, 102. [Google Scholar] [CrossRef] [PubMed]
- Vitamin D: Fact Sheet for Consumers. Available online: https://ods.od.nih.gov/factsheets/VitaminD-Consumer/ (accessed on 29 November 2021).
- Aboumrad, M.; Shiner, B.; Riblet, N.; Huizenga, H.; Neupane, N.; Young-Xu, Y. Trends in COVID-19 cases and clinical management in Veterans Health Administration medical facilities: A national cohort study. PLoS ONE 2021, 16, e0246217. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, G.N.; O’Hare, A.M.; Berry, K.; Fan, V.S.; Crothers, K.; Eastment, M.C.; Locke, E.; Green, P.; Shah, J.A.; Dominitz, J.A. Trends over time in the risk of adverse outcomes among patients with SARS-CoV-2 infection. Clin. Infect. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Tran, B.X.; Vu, G.T.; Le, H.T.; Pham, H.Q.; Phan, H.T.; Latkin, C.A.; Ho, R.C. Understanding health seeking behaviors to inform COVID-19 surveillance and detection in resource-scarce settings. J. Glob. Health 2020, 10, 0203106. [Google Scholar] [CrossRef]
- Hoerster, K.D.; Lehavot, K.; Simpson, T.; McFall, M.; Reiber, G.; Nelson, K.M. Health and health behavior differences: U.S. Military, veteran, and civilian men. Am. J. Prev. Med. 2012, 43, 483–489. [Google Scholar] [CrossRef]
- Maloney, S.R.; Goolkasian, P. Low Vitamin D States Observed in U.S. Marines and Navy Sailors with Early Multi-Symptom Illness. Biomolecules 2020, 10, 1032. [Google Scholar] [CrossRef] [PubMed]
- Devoto, C.; Arcurio, L.; Fetta, J.; Ley, M.; Rodney, T.; Kanefsky, R.; Gill, J. Inflammation Relates to Chronic Behavioral and Neurological Symptoms in Military Personnel with Traumatic Brain Injuries. Cell Transplant. 2017, 26, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Fogleman, S.A.; Janney, C.; Cialdella-Kam, L.; Flint, J.H. Vitamin D Deficiency in the Military: It’s Time to Act! Mil. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kojima, G.; Tamai, A.; Masaki, K.; Gatchell, G.; Epure, J.; China, C.; Ross, G.W.; Petrovitch, H.; Tanabe, M. Prevalence of vitamin D deficiency and association with functional status in newly admitted male veteran nursing home residents. J. Am. Geriatr. Soc. 2013, 61, 1953–1957. [Google Scholar] [CrossRef] [PubMed]
- Al-Anouti, F.; Mousa, M.; Karras, S.N.; Grant, W.B.; Alhalwachi, Z.; Abdel-Wareth, L.; Uddin, M.; Alkaabi, N.; Tay, G.K.; Mahboub, B.; et al. Associations between Genetic Variants in the Vitamin D Metabolism Pathway and Severity of COVID-19 among UAE Residents. Nutrients 2021, 13, 3680. [Google Scholar] [CrossRef]
- Speeckaert, M.M.; Speeckaert, R.; Delanghe, J.R. Vitamin D Sufficiency and COVID-19: Is Vitamin D Binding Protein (and Its Polymorphism) the Missing Link? Endocr. Pract. 2021, 27, 645. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Papinutti, A.; Mathew, E.; Vila, G.; Parekh, D. Vitamin D and critical illness: What endocrinology can learn from intensive care and vice versa. Endocr. Connect. 2018, 7, R304–R315. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Post-Index Medication Usage * (Vitamin D|Corticosteroids) n (%), Median (IQR) | 30-Day Mortality n (%), Median (IQR) | |||
|---|---|---|---|---|---|
| (−|+) | (−|−) | (+|+) | (+|−) | ||
| N (% of total sample size) | 5355 (20) | 20,134 (76) | 283 (1) | 736 (3) | 1612 (6) |
| Age (y) | 64 (21) | 58 (27) | 66 (16) | 63 (20) | 76 (16) |
| ≤30 | 154 (3) | 1425 (7) | 5 (2) | 14 (2) | 0 (0) |
| 31–40 | 455 (9) | 3058 (15) | 12 (4) | 59 (8) | 3 (<1) |
| 41–50 | 598 (11) | 2725 (14) | 26 (9) | 84 (11) | 25 (2) |
| 51–60 | 976 (18) | 3864 (19) | 49 (17) | 165 (22) | 80 (5) |
| 61–70 | 1362 (25) | 4101 (20) | 88 (31) | 179 (24) | 328 (20) |
| 71–80 | 1301 (24) | 3410 (17) | 76 (27) | 177 (24) | 569 (35) |
| 81–90 | 401 (7) | 1151 (6) | 19 (7) | 37 (5) | 401 (25) |
| >90 | 108 (2) | 400 (2) | 8 (3) | 21 (3) | 206 (13) |
| Male ^ | 4831 (90) | 17,970 (89) | 240 (85) | 618 (84) | 1575 (98) |
| Race | |||||
| White | 3047 (57) | 12,070 (60) | 147 (52) | 392 (53) | 1009 (63) |
| Black | 2101 (39) | 7257 (36) | 129 (46) | 328 (45) | 547 (34) |
| Asian | 59 (1) | 226 (1) | 1 (<1) | 3 (<1) | 15 (1) |
| AIAN | 50 (1) | 166 (1) | 0 (0) | 2 (<1) | 16 (1) |
| NHOPI | 51 (1) | 207 (1) | 2 (1) | 7 (1) | 13 (1) |
| Multiracial | 47 (1) | 208 (1) | 4 (1) | 4 (1) | 12 (1) |
| Latinx ^ | 790 (15) | 3536 (18) | 25 (9) | 102 (14) | 154 (10) |
| BMI (kg/m2) | 31 [8.3] | 30 [7.7] | 30 [8.5] | 30 [8.6] | 27 [9] |
| Underweight (<18.5) | 100 (2) | 243 (1) | 7 (2) | 10 (1) | 73 (5) |
| Normal (18.5–24.9) | 883 (16) | 3403 (17) | 45 (16) | 127 (17) | 489 (30) |
| Overweight (25–29.9) | 1655 (31) | 6678 (33) | 83 (29) | 249 (34) | 490 (30) |
| Class-I Obese (30–34.9) | 1462 (27) | 5748 (29) | 90 (32) | 178 (24) | 304 (19) |
| Class-II Obese (35–39.9) | 777 (15) | 2614 (13) | 34 (12) | 104 (14) | 159 (10) |
| Class-III Obese (40–44.9) | 323 (6) | 984 (5) | 18 (6) | 35 (5) | 55 (3) |
| Super Obese (≥45) | 155 (3) | 464 (2) | 6 (2) | 33 (4) | 42 (3) |
| Alcohol Use Disorder ^ | 671 (13) | 2683 (13) | 37 (13) | 118 (16) | 163 (10) |
| Smoker § | |||||
| Never | 2413 (45) | 10,167 (51) | 137 (48) | 381 (52) | 644 (40) |
| Former | 2406 (45) | 7623 (38) | 118 (42) | 297 (40) | 858 (53) |
| Current | 536 (10) | 2344 (12) | 28 (10) | 58 (8) | 110 (7) |
| Hospitalization ^ | 3149 (59) | 4340 (22) | 164 (58) | 192 (26) | 1113 (69) |
| Length of Stay (days) | 8 [11] | 6 [10] | 6 [12] | 5 [11] | 9 [9] |
| ≤7 ~ | 3751 (70) | 18,398 (91) | 209 (74) | 669 (91) | 946 (59) |
| >7–14 | 790 (15) | 854 (4) | 29 (10) | 28 (4) | 388 (24) |
| >14 | 814 (15) | 882 (4) | 45 (16) | 39 (5) | 278 (17) |
| Mechanical Ventilation ^ | 673 (13) | 465 (2) | 30 (11) | 14 (2) | 598 (37) |
| Location (USA) | |||||
| Pacific-West/Mountain | 971 (18) | 3996 (20) | 42 (15) | 97 (13) | 231 (14) |
| Mid-West/Continental | 1053 (20) | 4175 (21) | 42 (15) | 93 (13) | 251 (16) |
| Southeast | 2598 (49) | 8111 (40) | 180 (64) | 442 (60) | 592 (37) |
| Northeast | 733 (14) | 3852 (19) | 19 (7) | 104 (14) | 538 (33) |
| Time (Index, 3/1–9/10) | |||||
| March | 362 (7) | 1668 (8) | 17 (6) | 54 (7) | 253 (16) |
| April | 512 (10) | 2916 (14) | 26 (9) | 97 (13) | 445 (28) |
| May | 297 (6) | 1730 (9) | 12 (4) | 61 (8) | 190 (12) |
| June | 953 (18) | 3592 (18) | 42 (15) | 122 (17) | 205 (13) |
| July | 2155 (40) | 7136 (35) | 114 (40) | 277 (38) | 315 (20) |
| August | 998 (19) | 2950 (15) | 68 (24) | 121 (16) | 192 (12) |
| September | 78 (1) | 142 (1) | 4 (1) | 4 (1) | 12 (<1) |
| CCI | |||||
| 0 | 2198 (41) | 11,712 (58) | 93 (33) | 329 (45) | 407 (25) |
| 1–2 | 1978 (37) | 6032 (30) | 115 (41) | 291 (40) | 572 (35) |
| 3–4 | 766 (14) | 1640 (8) | 48 (17) | 77 (10) | 391 (24) |
| 5+ | 413 (8) | 750 (4) | 27 (10) | 39 (5) | 242 (15) |
| Comorbidity ^ | |||||
| Asthma | 591 (11) | 1039 (5) | 29 (10) | 56 (8) | 74 (5) |
| Atherosclerosis | 1830 (34) | 4551 (23) | 112 (40) | 207 (28) | 872 (54) |
| Cancer | 885 (17) | 2095 (10) | 80 (28) | 137 (19) | 391 (24) |
| Chronic Kidney Disease | 1173 (22) | 2664 (13) | 77 (27) | 121 (16) | 612 (38) |
| Chronic Liver Disease | 145 (3) | 408 (2) | 13 (5) | 22 (3) | 68 (4) |
| CHF | 900 (17) | 1930 (10) | 52 (18) | 89 (12) | 452 (28) |
| COPD | 1307 (24) | 2188 (11) | 79 (28) | 108 (15) | 472 (29) |
| Diabetes (Type II) | 2108 (39) | 6116 (30) | 122 (43) | 307 (42) | 813 (50) |
| Hyperlipidemia | 3227 (60) | 10,032 (50) | 183 (65) | 466 (63) | 1047 (65) |
| Hypertension | 3523 (66) | 10,461 (52) | 209 (74) | 501 (68) | 1242 (77) |
| Mental Illness | 2716 (51) | 9662 (48) | 148 (52) | 409 (56) | 709 (44) |
| Sleep Disorder | 1828 (34) | 5111 (25) | 99 (35) | 214 (29) | 416 (26) |
| Substance Abuse | 1194 (22) | 4222 (21) | 62 (22) | 173 (24) | 307 (19) |
| Post-Index | Non Survivors ^ | Survivors ^ | Multiplicity Corrected ‡ | ||
|---|---|---|---|---|---|
| Medication * | n (%) | n (%) | aRR (95% CI) † | p-Value | |
| Hospitalized (N = 7845) | |||||
| Vitamin D | Corticosteroids | ||||
| − | + | 534 (48) | 2615 (39) | 1.0 Referent | --- |
| − | − | 553 (50) | 3787 (56) | 0.66 (0.58–0.74) | <0.0001 |
| + | + | 15 (1) | 149 (2) | 0.51 (0.27–0.94) | 0.031 |
| + | ̶ | 11 (1) | 181 (3) | 0.30 (0.16–0.58) | 0.0004 |
| Non-hospitalized (N = 18,663) | |||||
| Vitamin D | Corticosteroids | ||||
| − | + | 69 (14) | 2137 (12) | 1.0 Referent | --- |
| − | − | 413 (83) | 15,381 (85) | 0.94 (0.71–1.2) | 0.66 |
| + | + | 6 (1) | 113 (1) | 2.5 (0.90–7.1) | 0.078 |
| + | ̶ | 11 (2) | 533 (3) | 0.48 (0.22–1.1) | 0.078 |
| Post-Index | Non Survivors ^ | Survivors ^ | Multiplicity Corrected ‡ | ||
|---|---|---|---|---|---|
| Medication * | n (%) | n (%) | aRR (95% CI) † | p-Value | |
| Black (N = 3281) | |||||
| Vitamin D | Corticosteroids | ||||
| − | + | 185 (45) | 1089 (38) | 1.0 Referent | --- |
| − | − | 215 (52) | 1633 (57) | 0.69 (0.55–0.86) | 0.0009 |
| + | + | 5 (1) | 70 (2) | 0.44 (0.09–0.2.0) | 0.29 |
| + | − | 5 (1) | 79 (3) | 0.34 (0.12–0.98) | 0.047 |
| White (N = 4291) | |||||
| Vitamin D | Corticosteroids | ||||
| − | + | 329 (50) | 1434 (40) | 1.0 Referent | --- |
| − | − | 318 (48) | 2020 (56) | 0.65 (0.55–0.76) | <0.0001 |
| + | + | 9 (1) | 76 (2) | 0.50 (0.26–0.96) | 0.036 |
| + | − | 6 (1) | 99 (3) | 0.29 (0.12–0.66) | 0.0032 |
| Post-Index | Non Survivors ^ | Survivors ^ | Multiplicity Corrected ‡ | ||
|---|---|---|---|---|---|
| Medication * | n (%) | n (%) | aRR (95%CI) † | p-Value | |
| Black (N = 6534) | |||||
| Vitamin D | Corticosteroids | ||||
| − | + | 16 (12) | 811 (13) | 1.0 Referent | --- |
| − | − | 116 (85) | 5293 (83) | 1.2 (0.68–2.0) | 0.56 |
| + | + | 0 (0) | 54 (<1) | ∞ | ∞ |
| + | − | 5 (4) | 239 (4) | 0.93 (0.24–3.6) | 0.92 |
| White (N = 11,365) | |||||
| Vitamin D | Corticosteroids | ||||
| − | + | 51 (15) | 1233 (11) | 1.0 Referent | --- |
| − | − | 284 (82) | 9448 (86) | 0.86 (0.42–1.7) | 0.67 |
| + | + | 6 (2) | 56 (1) | 2.7 (0.77–9.2) | 0.12 |
| + | − | 6 (2) | 281 (3) | 0.35 (0.10–1.2) | 0.093 |
| Stratum | Non- | Survivors ^ | aRR | Non | Survivors ^ | aRR | Effect (+|−) |  Stratum Stratum | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| (Col/Row) | Survivors ^ | n (%) | (95%CI) † | Survivors ^ | n (%) | (95%CI) † | ΔaRR% ♦ | PInt †,¥ | PΔV †,⅄ | ||
| n (%) | n (%) | MC’ ‡ | MC ‡ | MC’ ‡ | MC ‡ | ||||||
| Post Index → | Vitamin D (Yes versus No) | ||||||||||
| ↓ | Hospitalized + | Hospitalized − | |||||||||
| Corticosteroid + | 15 (3) | 149 (5) | 0.51 (0.29–0.88) | 6 (8) | 113 (5) | 2.5 (0.90–7.1) | −400 | 0.0071 | 0.028 | 0.057 | 0.12 |
| Corticosteroid − | 11 (2) | 181 (5) | 0.47 (0.26–0.84) | 11 (3) | 533 (3) | 0.51 (0.25–1.03) | −8 | 0.88 | 0.88 | ||
| Effect (+|−) | ΔaRR% ♦ | 8 | 400 | ||||||||
| PInt †,¥ | MC’ ‡ | 0.86 | 0.012 | ||||||||
| MC ‡ | 1 | 0.036 | |||||||||
 Stratum Stratum | PΔH †,⅄ | MC’ ‡ | 0.062 | ||||||||
| MC ‡ | 0.062 | ||||||||||
| Post Index → | Corticosteroid (Yes versus No) | ||||||||||
| ↓ | Hospitalized + | Hospitalized − | |||||||||
| Vitamin D + | 15 (58) | 149 (45) | 3.0 (0.51–18) | 6 (35) | 113 (17) | 6.2 (0.72–53) | −103 | 0.62 | 0.62 | 0.81 | 0.81 |
| Vitamin D - | 534 (49) | 2615 (41) | 1.5 (1.3–1.7) | 69 (14) | 2137 (12) | 1.1 (0.81–1.4) | 43 | 0.018 | 0.071 | ||
| Effect (+|−) | ΔaRR% ♦ | 98 | 479 | ||||||||
| PInt†,¥ | MC’ ‡ | 0.45 | 0.11 | ||||||||
| MC ‡ | 0.91 | 0.34 | |||||||||
 Stratum Stratum | PΔH †,⅄ | MC’ | 0.53 | ||||||||
| MC ‡ | 1.0 | ||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Efird, J.T.; Anderson, E.J.; Jindal, C.; Redding, T.S.; Thompson, A.D.; Press, A.M.; Upchurch, J.; Williams, C.D.; Choi, Y.M.; Suzuki, A. The Interaction of Vitamin D and Corticosteroids: A Mortality Analysis of 26,508 Veterans Who Tested Positive for SARS-CoV-2. Int. J. Environ. Res. Public Health 2022, 19, 447. https://doi.org/10.3390/ijerph19010447
Efird JT, Anderson EJ, Jindal C, Redding TS, Thompson AD, Press AM, Upchurch J, Williams CD, Choi YM, Suzuki A. The Interaction of Vitamin D and Corticosteroids: A Mortality Analysis of 26,508 Veterans Who Tested Positive for SARS-CoV-2. International Journal of Environmental Research and Public Health. 2022; 19(1):447. https://doi.org/10.3390/ijerph19010447
Chicago/Turabian StyleEfird, Jimmy T., Ethan J. Anderson, Charulata Jindal, Thomas S. Redding, Andrew D. Thompson, Ashlyn M. Press, Julie Upchurch, Christina D. Williams, Yuk Ming Choi, and Ayako Suzuki. 2022. "The Interaction of Vitamin D and Corticosteroids: A Mortality Analysis of 26,508 Veterans Who Tested Positive for SARS-CoV-2" International Journal of Environmental Research and Public Health 19, no. 1: 447. https://doi.org/10.3390/ijerph19010447
APA StyleEfird, J. T., Anderson, E. J., Jindal, C., Redding, T. S., Thompson, A. D., Press, A. M., Upchurch, J., Williams, C. D., Choi, Y. M., & Suzuki, A. (2022). The Interaction of Vitamin D and Corticosteroids: A Mortality Analysis of 26,508 Veterans Who Tested Positive for SARS-CoV-2. International Journal of Environmental Research and Public Health, 19(1), 447. https://doi.org/10.3390/ijerph19010447







