Abstract
Alcohol abuse disorder is a serious condition, implicating more than 15 million people aged 12 years and older in 2019 in the United States. Ethanol (or ethyl alcohol) is mainly oxidized in the liver, resulting in the synthesis of acetaldehyde and acetate, which are toxic and carcinogenic metabolites, as well as in the generation of a reductive cellular environment. Moreover, ethanol can interact with lipids, generating fatty acid ethyl esters and phosphatidylethanol, which interfere with physiological cellular pathways. This narrative review summarizes the impact of excessive alcohol consumption on male fertility by describing its metabolism and how ethanol consumption may induce cellular damage. Furthermore, the impact of alcohol consumption on hormonal regulation, semen quality, and genetic and epigenetic regulations is discussed based on evidence from animal and human studies, focusing on the consequences on the offspring. Finally, the limitations of the current evidence are discussed. Our review highlights the association between chronic alcohol consumption and poor semen quality, mainly due to the development of oxidative stress, as well as its genotoxic impact on hormonal regulation and DNA integrity, affecting the offspring’s health. New landscapes of investigation are proposed for the identification of molecular markers for alcohol-associated infertility, with a focus on advanced OMICS-based approaches applied to the analysis of semen samples.
1. Introduction
The consumption of alcoholic beverages is common in numerous societies, and almost 60% of the global population aged 15 years and over have been reported consuming alcoholic drinks in one year [1]. In Europe, 76% of citizens had reportedly consumed alcoholic beverages in the one year preceding a survey conducted by the European Commission [2]. Binge drinking is characterized by drinking several alcoholic beverages in a short period of time, where the blood alcohol concentration is around 0.08 g/dL [3]. According to the National Institute on Alcohol Abuse and Alcoholism, around 26% of the adult population reported binge drinking in the United States, with almost 15 million people older than 12 years showing alcohol abuse disorder [4]. This behavior is estimated to affect 4% of the adult global population [5].
Heavy alcohol consumption (defined as more than 3 and 4 drinks in a day, or more than 7 and 14 drinks weekly, for women and men, respectively) is reported to negatively affect human health, promote traffic accidents, and alter social behaviors, with severe repercussions for personal, social, and professional lives [5]. Clinically, alcohol consumption has been correlated with an increased incidence of different types of cancer [6], cardiovascular [7] and liver diseases [8], birth defects [9], and psychiatric disorders [10].
Alcohol abuse may result in alcohol dependence: this is a complex and dynamic process involving biological and socioenvironmental factors [11,12,13]. In general, alcohol acts by suppressing the activity of the nervous system. This is evident upon withdrawal after a prolonged period of abuse, where abstinence is characterized by the hyperactivation of the system itself, with consequent tachycardia, hypertension, excessive sweating, tremors, and convulsions [14]. Other common manifestations include rapid mood changes, irritability, agitation and anxiety, sleep disturbances, and the inability to experience pleasure (anhedonia), as well as a reduction in the pain threshold [15,16].
Numerous clinical studies have been conducted on alcoholic subjects to define the effects of alcohol abuse on the male reproductive capacity [17,18,19,20]. Initially, there can be alterations of the secondary sexual characteristics, such as reduction in facial and body hair, enlargement of the breasts, and accumulation of adipose tissue from the abdominal area to the hips [17]. This is followed by the establishment of a proinflammatory status, indicated by elevated levels of leukocytes in the semen [18]. From a functional point of view, alcoholics often complain of sexual dysfunction or infertility [19,20]. As a large percentage of male alcoholics are of the reproductive age and may be interested in becoming fathers [1], understanding the mechanisms leading to alcohol-related male infertility is of the utmost importance.
This narrative review aims to discuss the impact of alcohol consumption on male fertility by describing alcohol metabolism and the molecular mechanisms by which ethanol (EtOH) can induce cellular damage. Moreover, we discuss evidence from animal and human studies regarding the impact of EtOH consumption on sexual hormonal regulation, semen quality, and epigenetics, emphasizing the consequences on the offspring. Finally, we highlight the limitations of the current evidence and describe new areas of investigation for the identification of the molecular markers of alcohol-associated male infertility.
2. Metabolism of Ethanol
When an alcoholic drink is consumed, the EtOH is rapidly absorbed through the stomach (20%) and duodenum (80%), and passes directly into the bloodstream [21]. Through the blood circulation, alcohol reaches the liver, brain, heart, and kidneys within 10–15 min after consumption, while it takes about an hour to reach the muscles and adipose tissue, where it tends to concentrate [22].
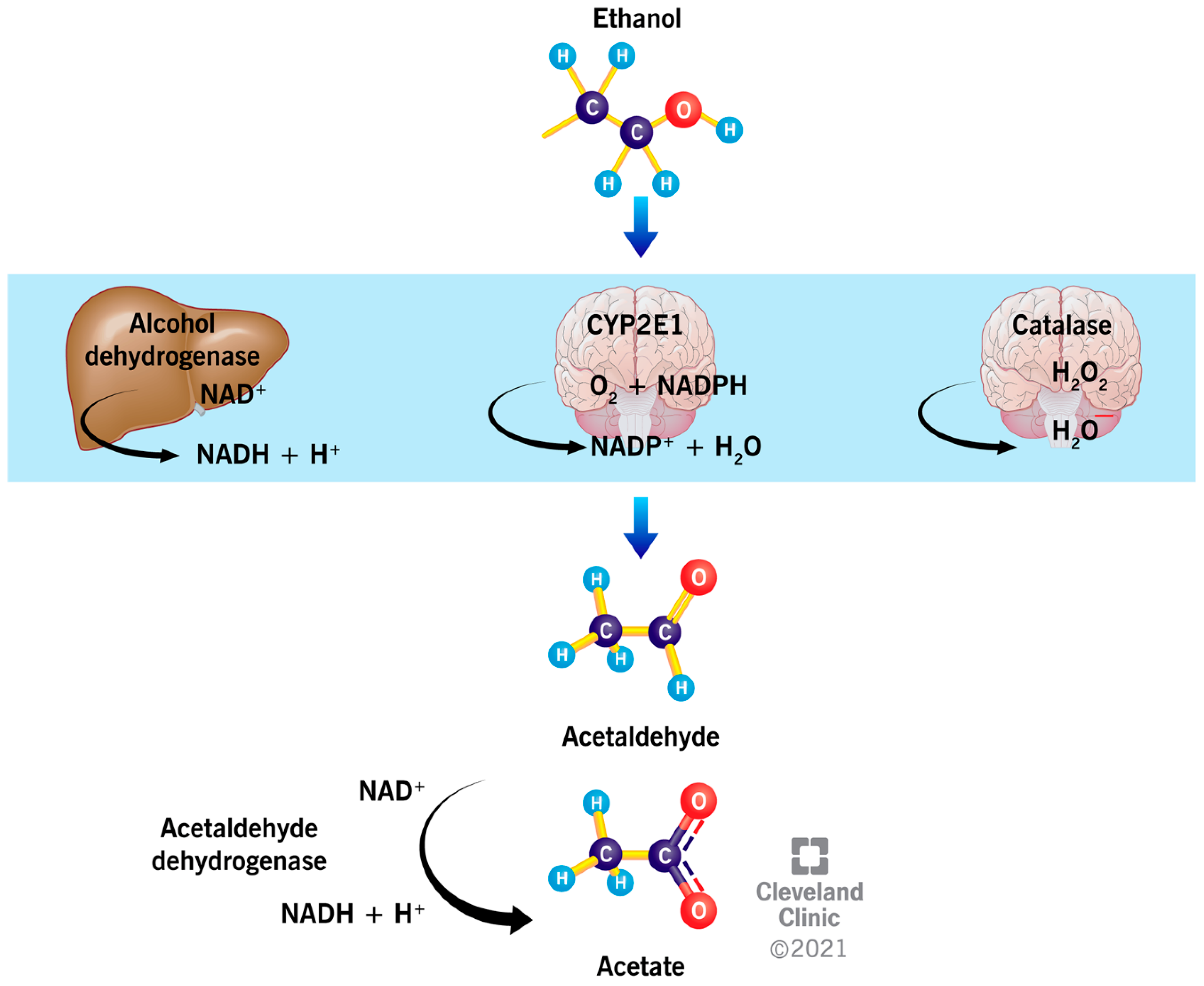
The liver metabolizes about 90% of the EtOH, while the remaining EtOH is directly eliminated through urine, feces, breast milk, sweat, and exhaled air [21]. In the liver, stomach, and intestinal tract, EtOH is oxidized primarily by alcohol dehydrogenase (ADH) into acetaldehyde, a highly toxic and reactive molecule, with a reduction in the cofactor, nicotinamide adenine dinucleotide (NAD+) [23] (Figure 1). The reduced cofactor (NADH) contributes to the cellular energy generation by means of the electronic transport chain located in the inner membrane of the mitochondria; through this pathway, the metabolism of EtOH produces energy (7 kcal/g of EtOH) [24].
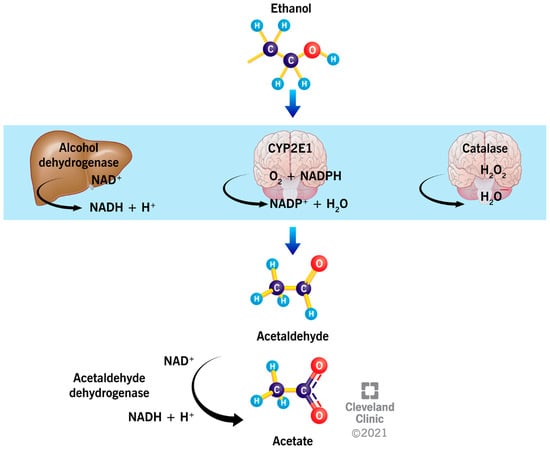
Figure 1.
The metabolism of ethanol. In the liver, ethanol is oxidized to acetaldehyde mainly by the enzyme, alcohol dehydrogenase, while, in the brain, the enzymatic activity of cytochrome P450 2E1 (CYP2E1) and catalase are more prominent. Acetaldehyde is further oxidized to acetate by the enzyme, acetaldehyde dehydrogenase.
In the brain, the enzymes, catalase and cytochrome P450 (CYP450), oxidize 65% and 20% of the EtOH into acetaldehyde (Figure 1), respectively [25]. CYP450 is a family of enzymes containing the heme group that are involved in the oxidation of steroids, fatty acids, and numerous xenobiotics of environmental origin. In particular, the cytochrome P450 2E1 (CYP2E1) activity is induced by chronic alcohol consumption, resulting in increased alcohol tolerance, oxidative stress and toxicity, with a consequently higher risk of developing cancer and liver injury [26]. Similarly, catalase, which belongs to the class of oxidoreductases, contains a heme group and is generally involved in the cellular scavenging of reactive oxygen species (ROS) [21]. The enzyme is localized in the peroxisomes, and it oxidizes EtOH in the presence of complexes that generate H2O2, such as reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase or xanthine oxidase (Figure 1). Whereas catalase plays a marginal role in EtOH oxidation in the liver, it is involved in the development of alcohol tolerance and consumption in the brain [27].
The enzyme, acetaldehyde dehydrogenase (ALDH), oxidizes acetaldehyde and produces acetate. ALDH is a zinc-dependent enzyme, which also uses NAD+ as a cofactor [21] (Figure 1). Some drugs used to treat alcoholism, such as Disulfiram, inhibit the activity of ALDH, resulting in the accumulation of acetaldehyde. This leads to unpleasant effects, such as increased sweating and tachycardia, nausea, and vomiting, thus discouraging the consumption of alcohol [28]. Acetate, which results from the oxidation of acetaldehyde, leads to increased blood flow in the liver and a depressed central nervous system, and it compromises numerous metabolic processes [29,30]. Acetate can be metabolized into acetyl-CoA, a key molecule in the synthesis of lipids and cholesterol in the mitochondria. Depending on the nutritional, energetic, and hormonal status, acetyl-CoA can be converted into CO2, fatty acids, ketones, or cholesterol [21].
The nonoxidative metabolism of alcohol is minimal, but its products can have pathological and diagnostic significance. In fact, EtOH can react with fatty acids, forming fatty acid ethyl esters [31]. These esters are synthesized in the endoplasmic reticulum and are transported to the membranes, where they bind to lipoproteins or albumin. They are potentially toxic, as they interfere with the synthesis of DNA and proteins [21]. Furthermore, EtOH can be incorporated into phospholipids as phosphatidylethanol through a reaction catalyzed by the enzyme, phospholipase D (PLD) [32]. PLD has a high Michaelis constant (Km) for ethanol, so that the enzymatic reaction occurs mainly at high concentrations of circulating alcohols. Phosphatidylethanol is poorly metabolized, and can accumulate after chronic alcohol intake, interfering with the cellular signaling pathways [32].
3. Ethanol-Induced Mechanisms of Cellular Damage
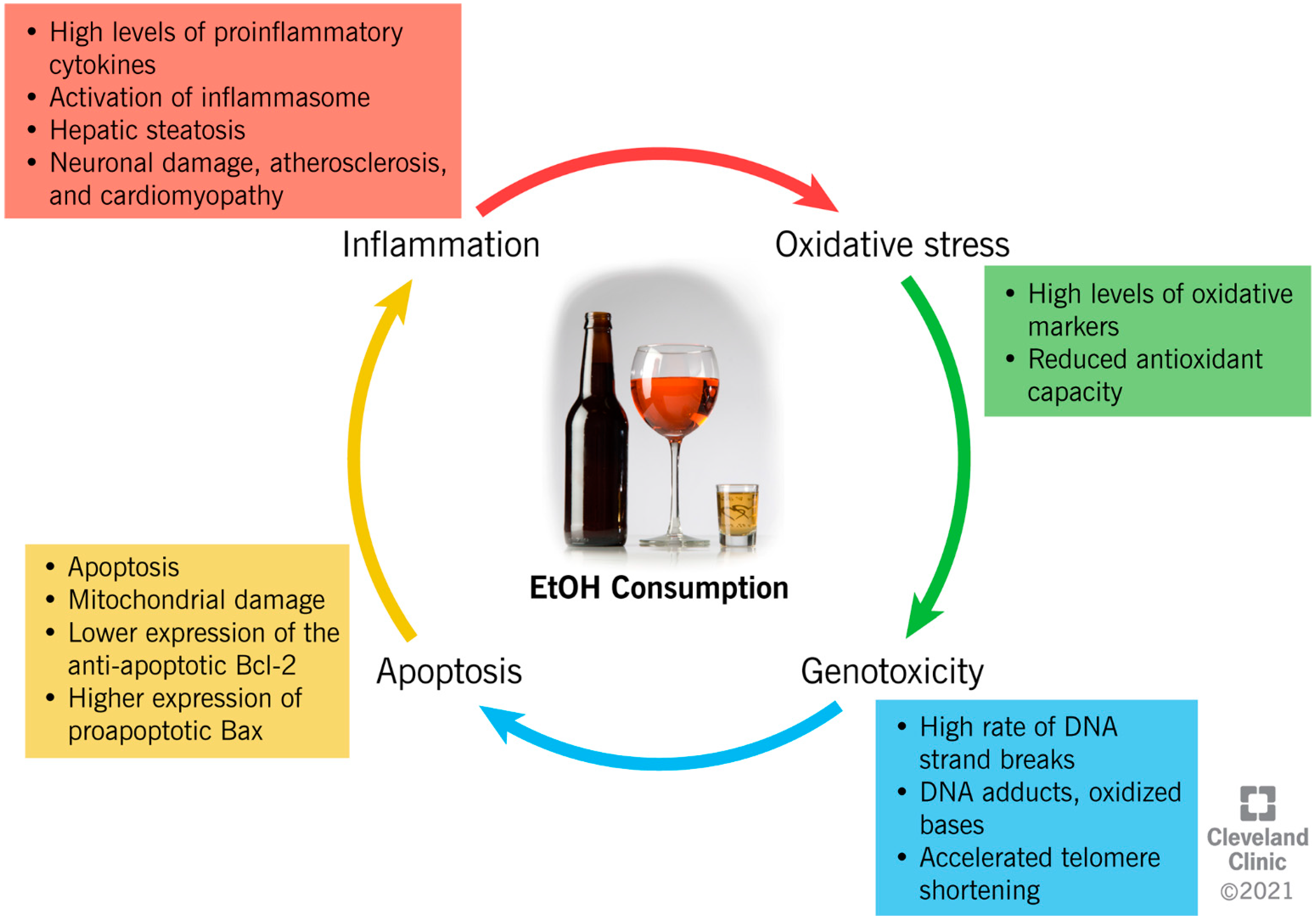
EtOH consumption has been reported to induce cellular damage through different interconnected mechanisms, which include the establishment of an inflammatory and oxidative environment, resulting in genotoxicity and enhanced apoptotic rate (Figure 2).
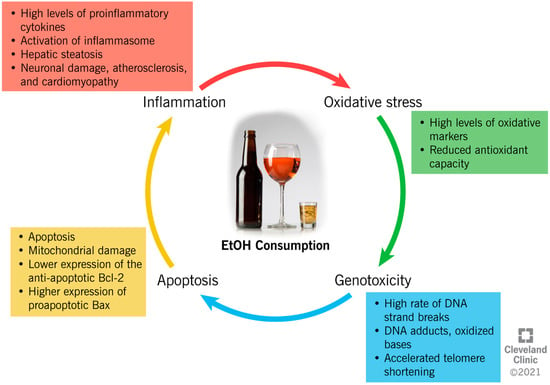
Figure 2.
Ethanol-induced mechanisms of cellular damage.
The close link between chronic alcohol consumption and the onset of inflammation has been widely investigated [33,34,35,36,37,38,39,40,41,42]. In the liver, acetaldehyde activates inflammatory signaling pathways by increasing the synthesis of circulating proinflammatory cytokines by Kupffer cells [33], such as tumor necrosis factor-alpha (TNFα) [33]. TNFα acts on the hepatic stellate cells, further exacerbating the synthesis of proinflammatory and profibrogenic molecules, impairing cell function, and contributing to alcohol-induced cirrhosis [33,34,35]. In the liver, brain, and intestine, chronic alcohol administration has been proven to activate the inflammasomes. This is an intracytoplasmic multiprotein complex that induces the activation of caspase-1 and interleukin (IL)-1β, enhancing the proinflammatory status in the same tissues [36,37]. Moreover, through a mechanism of negative feedback, inflammation enhances the negative impact of alcohol metabolism through the reduced expression of ADH1 and ALDH2, which causes systemic and hepatic increases in the EtOH and acetaldehyde levels [40]. In addition, the metabolism of acetaldehyde into acetate increases the NADH:NAD+ ratio within the cell, and consequently inhibits the fatty acid β-oxidation pathway in the liver. This results in the accumulation of triglycerides in the hepatocytes as hepatic steatosis, and the enhancement of inflammation [38].
Considering that inflammatory pathways participate in the pathophysiology of neurodegenerative disorders, an alteration of the cytokine expression in the nervous system can severely damage neuron activity [41]. EtOH exposure alters the NF-kB pathway, which is important in inflammatory and immune responses in the brain, and decreases the transcription of regulatory cyclic protein AMP-responsive element-binding protein (CREB), affecting neuronal survival and protection from apoptosis [42].
Moreover, alcohol consumption can increase oxidative stress through different mechanisms, with serious consequences for human health. ROS play a crucial role as mediators in several biological processes, including cell signaling and pathogen resistance [43]. However, ROS are highly unstable and are reactive with proteins, lipids, and any cellular structures. When ROS are overproduced, they switch the cellular redox status towards oxidative stress, which contributes to several pathological conditions [44].
EtOH metabolism enhances ROS generation, as the activity of dehydrogenases and the CYP2E1 system increases the levels of NADH, which, in turn, is oxidized by xanthine oxidase, with the generation of ROS [45]. An increased oxidative status is also due to the increased peroxisomal activity in the livers of heavy drinkers, as a result of steatosis [46].
In addition to directly increasing the production of ROS, in vitro studies have shown that acetaldehyde reportedly causes an intracellular redox imbalance by increasing the levels of oxidative stress markers (i.e., malondialdehyde (MDA)), and by reducing the concentration of antioxidant glutathione (GSH) [47,48]. Acetaldehyde also impairs the enzymatic activity of superoxide dismutase (SOD) 2, a powerful endogenous antioxidant [49]. In alveolar macrophages, chronic alcohol consumption and the subsequent onset of oxidative stress alter the expression of the NADPH oxidases (Noxes), enzymes contributing to the phagocyte-mediated host defense [50]. EtOH consumption upregulates the expression of the Noxes, as well as the regulatory proteins, p22phox, p47phox, and p67phox, both in vivo and in vitro, resulting in the higher intracellular production of O₂− and H2O2, and lower GSH levels in the lung tissue [50].
The establishment of an inflammatory and oxidative environment modifies cellular homeostasis and DNA integrity, and can have a genotoxic/mutagenic impact, eventually leading to cell death. In vitro and in vivo studies indicate that EtOH induces autophagy in neurons by inhibiting the expression of the antiapoptotic Bcl-2 family proteins, while it increases the expression of proapoptotic proteins [51]. The expression of Bcl-2 is also modulated by EtOH through epigenetic regulation. In fact, by increasing the histone deacetylase activity, EtOH affects the binding of acetyl-histone H3 to the Bcl-2 promoter, which results in a reduced Bcl-2 gene transcription [52]. Furthermore, it has been suggested that EtOH induces programmed cell death through the mitochondrial pathway, as the inhibition of caspase-9 (key molecule in this pathway) resulted in a reduced apoptotic rate [53]. The activity of caspase-3, an apoptotic marker, was also increased in the cerebral cortices of rats after EtOH treatment [54], possibly because of the depolarization of the mitochondrial membrane by acetaldehyde accumulation [55]. The role of alcohol metabolites as mediators of mitochondrial damage was also demonstrated in knock-out mice for ALDH or ADH enzymes, which showed reduced membrane potential in comparison to the controls [55].
However, whereas evidence shows a negative impact of high-dose EtOH on cellular physiology, a low dose seems to reduce the apoptotic rate. In fact, recent data suggests that a low dose of EtOH contributes to the regulation of the expression of the TNF receptor, p75NTR, and a potassium-chloride transporter (KCC2) [56]. While p75NTR is involved in the regulation of apoptosis, KCC2 regulates the cellular electrochemical gradient, and its expression is crucial to maintaining the classic hyperpolarizing GABAergic inhibition in mature adult neurons [56].
EtOH and its metabolites, including ROS, can be classified as genotoxic agents. In fact, they enhance the rate of the DNA strand breaks, leading to chromosomal rearrangements and/or the loss of genetic information, genomic instability, and micronucleus formation [57]. Studies on human peripheral blood lymphocytes show mutagenic activity in alcoholics, with an increased sister chromatid exchange frequency, indicating alterations in the DNA replication or repair processes [58]. Acetaldehyde and other toxic aldehydes produced by lipid peroxidation, such as MDA and 4-hydroxynonenal (4-HNE), damage DNA, as is described in both animal and human studies [59,60]. These molecules can increase the generation of DNA adducts, such as those with the deoxyguanosine (dG) base (N2-ethyl-2′-deoxyguanosine, 1,N2-propane-2′-dG (PDG), and propane-dG), damaging the integrity of the DNA double helix [61]. Moreover, MDA and 4-HNE form etheno-DNA adducts, with strong carcinogenic mutagenic properties [60]. The most relevant mutagenic activity is ROS-mediated, as high levels of ROS induce the oxidation of DNA and the production of various lesions, including oxidized bases (8-hydroxy-2′-deoxyguanosine), apurinic/apyrimidine sites, and single or double DNA strand breaks [62,63]. ROS-related DNA damage is also associated with accelerated telomere shortening, since shortened telomeres were reported in chronic inflammatory diseases, compared to healthy individuals (as comprehensively described in [64]). Several studies have reported a cause–effect association between early telomere shortening and EtOH consumption [65,66]. However, the mechanistic link between EtOH metabolism and telomere shortening is unclear and requires further investigation.
4. Alcohol Consumption and Male Infertility: Evidence from Animal and Human Studies
Several animal and human studies have investigated the impact of alcohol consumption on reproductive hormonal regulation, semen quality, gene transcription, genetics, and epigenetics regulation, as well as the transgenerational impact of paternal exposure on the offspring. Evidence is discussed below and is summarized in Table 1.

Table 1.
Summary of the evidence regarding the impact of alcohol consumption on male fertility in animal and human studies.
4.1. Impact of Alcohol on Reproductive Hormonal Regulation
The secretion of reproductive hormones, such as testosterone, is regulated by the hypothalamic–pituitary–gonadal (HPG) axis [114]. The hypothalamus releases gonadotropin-releasing hormone (GnRH), which reaches the adenohypophysis through the hypophyseal portal system. As a response, the GnRH triggers the release of gonadotropins (luteinizing hormone (LH), and follicle-stimulating hormone (FSH)), which act at the testicular level. LH stimulates the Leydig cells to produce testosterone [114]. LH binding triggers the internalization of cholesterol, which is converted to pregnenolone, and then to 17α-hydroxy-pregnenolone, and androgen dehydroepiandrosterone (DHEA). DHEA is converted to androstenedione via 3β-hydroxysteroid dehydrogenase (3β-HSD), and then to testosterone via 17β-hydroxysteroid dehydrogenase-3 (17β-HSD-3) [115]. FSH binds on the Sertoli cells, supporting the spermatogenesis. This process includes three phases of development: (1) The proliferation of spermatogonial cellular precursors (mitosis); (2) Maturation into spermatocytes, with recombination, reduction, and division of DNA (meiosis); and (3) Differentiation into spermatids, and, finally, into mature spermatozoa (spermiogenesis) [116].
Alcohol consumption seems to affect endocrine functions, compromising the regulation of the HPG axis; however, contradictory evidence is reported in the literature. In 2005, a study investigating fertility hormones in 66 heavy drinkers reported increased levels of FSH, LH, and estradiol [79]. Conversely, Maneesh et al. reported lower levels of the gonadotropins, FSH and LH, in 45 alcoholic men [80], while Jensen et al. did not observe any association between the levels of FSH, LH, and inhibin B and the intake of alcohol in 8344 healthy men [81]. Similar results are reported regarding testosterone concentration, with studies observing higher values in alcoholic men [81,82,83], while others observed lower testosterone [79,80]. In addition, chronic alcohol intake can increase serum prolactin (hyperprolactinemia), causing hypogonadism, reduced sperm production, impotence, and gynecomastia in men [117,118]. These differences could be due to the heterogeneity in the study designs, the populations included, and the different approaches to classifying drinking habits, whereas the studies conducted in animal models reported a reduction in the circulating levels of LH and FSH following EtOH exposure [67,68,69,70,71,72].
Studies dating back to the 1980s suggest that acute EtOH intake might directly inhibit the hypothalamic release of GnRH, and, consequently, reduce the levels of LH and testosterone [119,120], probably through the alteration of the nitric oxide (NO) pathway. NO stimulates the synthesis of PGE2 in tissues by binding the heme group of cyclooxygenase-1 [121], while EtOH inhibits the cyclooxygenase-1 activity, and eventually the synthesis of PGE2 and GnRH secretion [122]. Alcohol consumption has also been associated with a reduction in the number of Leydig cells and an altered morphology [78]. The Leydig cells of rats fed with EtOH showed reduced sizes, swollen mitochondria, and smaller amounts of cytosol, along with the reduced synthesis of testosterone [78]. Besides altering its synthesis, alcohol consumption is also associated with an enhanced rate of testosterone elimination [17,73]. In fact, EtOH stimulates the activity of aromatase, which converts testosterone into estradiol [73], resulting in elevated estrogen levels and abnormal breast enlargement, as observed in heavy alcoholics [17,83]. This further inhibits the synthesis of FSH and LH, and, consequently, the synthesis of testosterone itself [74,75]. Moreover, chronic alcohol intake has been associated with the reduced bioavailability of insulin growth factor (IGF)-1, which physiologically stimulates the synthesis of testosterone [76]. Finally, it is important to mention that NAD is the cofactor of enzymes involved in both EtOH and androgen metabolism. Therefore, high EtOH intake reduces NAD+/NADH levels, and indirectly inhibits the activity of enzymes involved in testosterone synthesis [77].
4.2. Impact of Alcohol Consumption on Semen Quality
Data from an animal study showed that an EtOH-rich diet can affect testicular function, with consequences on the semen quality. In fact, EtOH-fed mice showed compromised integrities of the testis and seminal vesicles, and altered weight of the prostate, which resulted in increased germ cell desquamation, decreased sperm concentrations, and increased abnormal sperm morphologies [123]. Besides the alterations in the semen quality (lower sperm concentration, motility, and percentage of normal forms), Rahimipour et al. also reported reduced DNA condensation and integrity in mice fed with ethanol compared to controls, along with increased apoptotic rates [85]. In addition, in vitro experiments showed an accelerated acrosomal loss occurring during the sperm capacitation of human and animal sperm incubated in ethanol, further reducing their fertilizing ability [124,125,126,127]. This is probably due to the capacity of ethanol to alter lipid fluidity and membrane permeability through the oxidation of the membranes’ lipids and proteins [127]. In rats, decreased sperm motility was observed after exposure to EtOH, as well as changes in the meiotic divisions, reduced gametes viability, and a higher rate of sperm with poorly condensed chromatin [86,87]. In humans, a case study reported severe oligoasthenoteratozoospermia in an alcoholic man, which evolved into cryptozoospermia, and then azoospermia after a few years [91]. In 2017, a meta-analysis investigated the impact of alcohol intake on semen quality by analyzing evidence from 18 cross-sectional studies [93]. The authors concluded that daily alcohol consumption results in a worsened semen quality, particularly in terms of the semen volume and the sperm morphology. However, this effect was not reported for occasional drinkers, while the authors observed even better sperm motility in occasional drinkers than never drinkers, despite all the limitations identified in their analysis [93]. In fact, the association between semen quality and the amount of alcohol consumed is still controversial. Surprisingly, Ricci et al. observed a positive correlation between semen volume and concentration, and moderate alcohol consumption (equal to 4–7 units/week), suggesting that a limited consumption of alcohol may improve semen quality [96]. This might be explained by the fact that some compounds present in alcoholics drinks (i.e., natural flavonoids, and polyphenols in red wine) have antioxidant and anti-inflammatory activities, and they reportedly have a positive influence on semen quality (particularly by improving sperm motility, concentration, and survival) at low concentrations [128,129,130,131]. However, a cross-sectional study including 8344 healthy men did not report any association between low/moderate alcohol consumption and semen quality [81]. Similarly, other studies failed to identify any coherent dose–response pattern in the semen parameters depending on the degree of alcohol consumption [83,88,132,133,134]. Boeri et al. suggested that the correlation between alcohol consumption and alterations in the semen parameters might be directly proportional to the amount of alcohol consumed. In fact, the semen parameters were reportedly worse in samples of heavy rather than moderate drinkers [89]. Several recent studies of different global geographic regions have confirmed the negative impact of heavy alcohol consumption on semen quality. In fact, in China, a cross-sectional study conducted in 2020 reported reduced sperm concentrations in 55 heavy drinkers suffering from secondary infertility [88], while in Italy, 45 heavy drinkers with primary infertility showed reduced sperm concentrations and motilities compared to moderate drinkers or abstainers [89]. Similarly, an inverse association between sperm counts and alcohol consumption was observed in a Brazilian population of 167 infertile men [90], while a large study conducted on a Danish population of 1221 men showed a direct association between worsening semen quality and increasing alcohol intake [92]. Other studies have also confirmed a higher rate of sperm DNA fragmentation and chromatin decondensation in heavy drinkers [89,90,94,95].
The differences in the study designs, and the discrepancies in the published studies, make it challenging to draw any conclusions regarding the association between the amount of alcohol consumed and the semen quality. Hence, much research is still warranted in this regard.
4.3. Impact of Alcohol Consumption on Gene Transcription, Genetics, and Epigenetics Regulation
Male infertility associated with chronic alcohol consumption might also be due to a differential regulation of gene expression, followed by an altered metabolism of the specific proteins involved in sperm maturation [97,98,101,135]. In fact, it was shown that EtOH can lead to oxidative damage in the epididymis by altering the mRNA expression of β-defensin, which has antimicrobial properties and is involved in sperm function [97]. Alcohol exposure also altered post-transcriptional RNA modifications in murine sperm, thereby influencing the expression of small mitochondrial RNA species, the mitochondrial function of spermatozoa, and the reproductive ability [101]. In addition, EtOH compromised sperm viability by reducing the expression of proliferating cell nuclear antigen (PCNA) in germ cells and by promoting apoptosis [135].
Several genetic variants of enzymes involved in EtOH metabolism have been identified, associated with different degrees of tolerability to alcohol. In fact, specific single nucleotide polymorphisms in ADH and ALDH show altered enzymatic activity, which results in the accumulation of acetaldehyde. Because of its unpleasant effects, this is associated with a lower risk of developing an alcohol-use disorder [101].
A correlation between EtOH consumption and epigenetic changes in sperm DNA has been explored in various studies [100,101,102,103,136]. In this regard, aberrant gene methylation in sperm DNA was associated with male infertility [102]. During early embryonic development, there are two loci in paternal DNA (differentially methylated region upstream of the H19 gene-H19 DMR, and intergenic differentially methylated region, IG-DMR) that are highly methylated, and that are fundamental for growth and neurobehavioral development [103]. A significant difference was observed in the demethylation rates of specific C-phosphate-G (CpG) sites between nondrinkers and moderate drinkers. Particularly, there was a direct correlation between the amount of alcohol consumed and the degree of demethylation at the H19 DMR and IG-DMR genes, supporting the hypothesis that EtOH consumption may reduce the DNA methyltransferase activity, and increase the risk of the transmission of defective imprinted genes [103]. In addition, as shown in the rat testis, EtOH exposure may affect spermatogenesis, as it increases the acetylation of lysine 9 in histone 3 (Ac-H3-lys9), which results in impaired sperm chromatin organization and embryonic development [100].
4.4. Consequences of Paternal Alcohol Consumption on the Offspring
Several studies were conducted in animal models to investigate the transgenerational effect of paternal alcohol exposure, showing low fetal and birth weights in the offspring, as well as altered organ weights and hormonal regulations (for a review on the topic, see [101]). An animal study showed that paternal alcohol exposure led to hormonal and nervous system anomalies in the offspring. Specifically, the expression of nerve growth factor (NGF), a well-characterized neurotrophin involved in the development of the nervous system in vertebrates [105], was strongly reduced in the frontal cortices of the offspring of EtOH-fed mice, as well as in the hippocampal, hypothalamic, and olfactory lobes, leading to the conclusion that paternal alcohol consumption can produce critical alterations in the brains of offspring [106]. The anogenital distance, which is a male fertility marker related to the proper functioning of the endocrine system, was also shorter in the offspring of EtOH-consuming fathers, indicating that alcohol may have an adverse effect on the reproductive development of offspring [107]. Paternal alcoholism before conception was also associated with limited offspring growth and decreased placental efficiency [104].
In a case of paternal alcohol exposure, the offspring showed reproductive dysfunctions similar to those reported for direct alcohol consumption, including alterations in the hormonal axis and the semen quality [113].
Three possible mechanisms have been proposed to explain the effect of paternal alcohol consumption on the offspring: (a) An alteration of the sperm chemical composition, leading to behavioral, biochemical, and hormonal disturbances in the offspring; (b) A failure of the elimination of EtOH-damaged sperm; and (c) An induction of genetic mutations in sperm DNA that can be transmitted to the offspring [137]. Although chemical alterations in sperm and/or seminal plasma may compromise embryonic development, this remains to be confirmed. The second hypothesis refers to the mechanisms of natural selection that may fail to remove spermatozoa damaged by EtOH during spermatogenesis, in favor of genetically intact spermatozoa. The third hypothesis emphasizes a heritable genetic change in EtOH consumers, and seems to be the most accepted [137]. However, a study published in 2017 was not able to identify any association between the phenotypes observed in mice exposed to EtOH (fetal growth restriction and altered developmental programming) and the paternal DNA methylation profiles, questioning the transgenerational effect of EtOH exposure [138]. EtOH-related epigenetic effects on the paternal germline might provide an explanation for the transgenerational influence of the father’s lifestyle habits on the development of the offspring, and it surely deserves more investigation [139].
5. Limitations of the Published Human Studies and Future Areas of Investigation
Studies on the effects of alcohol dependence in the human population suffer from significant limitations. This is due mostly to the fact that the data is self-reported and collected by using questionnaires [140]. In fact, the objective estimation of the real volume of alcohol consumed is challenging because of the possibility of an over- or underestimation [141]. Moreover, alcoholics often do not admit to having an alcohol dependence, hence leading to incorrect information about the amount of alcohol and the frequency of the drinking. The investigation is made even more challenging by the fact that alcohol consumption can be defined according to different criteria (such as frequency, or units/daily), and by the fact that the pattern of alcohol consumption can vary, from moderate/heavy alcohol consumption to binge drinking. This can influence the analyzed outcomes and can make it difficult to compare results from different publications [142].
The negative impact of EtOH on male fertility is a confirmed hypothesis; however, the molecular mechanisms that act at the sperm level remain to be clarified. In fact, studies analyzing the impact of alcohol consumption on male infertility are difficult to plan because semen quality is influenced by a multitude of other factors and varies significantly between individuals, and in the same individual as well [143]. Hence, there is a need to identify molecular markers for alcohol-related male infertility. In this context, molecular tests to assess advanced sperm characteristics (i.e., acrosome reaction, capacitation) are to be considered valid approaches for discovering biomarkers, besides the conventional semen parameters. Similarly, -OMICS sciences are to be kept in consideration. Comparative proteomic studies showed the differential expression of proteins between normozoospermic and abnormal semen samples [144], and were able to successfully identify several proteins in human sperm and seminal fluid as possible markers of male infertility and reproductive failure [144,145]. In asthenozoospermia, the altered expression of cAMP-mediated protein kinase A and actin was reported, which would explain the low motility of spermatozoa. Similarly, a proteomic screening of globozoospermic cells revealed reduced levels of α-tubulin, β-tubulin, and β-galactoside–galactoside binding protein and vimentin, indicating a weakened cytoskeletal organization, which may contribute to the pathogenesis of globozoospermia [144,146]. Furthermore, proteomic analyses of seminal plasma have allowed for the identification of the markers for obstructive and nonobstructive azoospermia (as reviewed in [147]). Hence, the high-throughput analysis of semen samples is extremely useful, both in diagnostics and for identifying new therapeutic targets, and may be successfully applied to the investigation of alcohol-induced male infertility. In association with proteomic studies, transcriptomic studies can help to identify the molecular markers of male infertility. The transcriptome is defined as the overall RNA content of a sample, including the microRNA, whereas the alteration of several microRNAs has been associated with EtOH-related damage and tolerance [148], and their expression in the human semen samples of alcoholics has not yet been investigated. On the other hand, in mice, sperm RNA sequencing showed the altered enrichment of microRNAs after EtOH exposure, suggesting that they play a role in the alcohol-related phenotypes of the offspring [149]. Furthermore, different sites can generate extracellular microvesicles (exosomes), including RNAs and proteins, which are endocytosed by sperm [150,151]. The molecular composition of exosomes is a picture of the status of their cellular origin; hence, their characterization may be helpful to identifying novel site-specific pathological biomarkers [151]. Similarly, a metabolomics analysis of seminal plasma has allowed for the identification of the metabolites involved in energy production, the maintenance of the redox status, hormonal regulation, and sperm functions [144], representing a promising approach for investigating EtOH-related male infertility.
The ability to conduct genome-wide association studies has allowed for the scanning of the entire genome for the identification of any novel genetic locus associated with specific clinical conditions. In this context, the creation of consortia has allowed for the analysis of a large population with alcohol dependence [152]. A study conducted in 2018 analyzed around 15,000 subjects with alcohol dependence, and identified functional variants of the ADH1B gene that were differentially presented in European and African populations, and that were associated with a variable rate of EtOH oxidation, and protection from EtOH-related side effects and the risk for developing alcohol dependence [153]. Similarly, a recent study investigating more than 270,000 multiethnic subjects identified different genetic loci associated with heavy drinking behavior or the risk of developing alcohol dependence [154]. Besides the genes involved in EtOH metabolism, these studies have also identified new genes (i.e., β-klotho (KLB), and fibroblast growth factor 21 (FGF21)), whose alleles are strongly associated with alcohol consumption [153,154].
In the future, the use of an integrated OMICS approach could explain the mechanisms underlying EtOH-induced infertility due to alterations in the semen parameters and the redox status, as well as disturbances in hormonal regulation, and could allow for the identification of selective markers in these subjects. Furthermore, considering the role of oxidative stress in mediating EtOH-related damage, further human studies should be planned to better understand the mechanisms involved, and to investigate the possible treatment with an adequate therapy.
6. Conclusions
Besides being an important public and social issue, alcohol consumption can also significantly impact male reproduction. The association between chronic alcohol consumption and poor semen quality has been reported in a large number of studies in both humans and animals, mainly due to excessive ROS generation following EtOH metabolism. By acting as genotoxic agents, EtOH and its metabolites alter the expression of specific genes involved in the hormonal regulation of spermatogenesis, and increase sperm DNA fragmentation, potentially with a transgenerational effect on the offspring. Despite its role in contributing to male infertility, specific markers for EtOH-related infertility have not yet been identified. In this context, high-throughput analyses of semen samples will represent the future strategies for a better understanding of the molecular mechanisms underlying alcohol-induced male reproductive disorders, as well as for identifying more effective therapeutic measures to combat the dependence.
Author Contributions
Conceptualization, R.F.; data curation, R.F. and F.M.; writing—original draft preparation, R.F. and F.M.; writing—review and editing, R.F. and F.M.; supervision, A.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors are thankful to the following reviewers: Kristian Leisegang (South Africa); Ralf Henkel (the U.K.); Pallav Sengupta (Malaysia); Rupin Shah (India); Hussein Kandil (the U.A.E.); Murat Gul (Turkey); and Taha Abo-Almagd Abdel-Meguid Hamoda (Egypt) for their constructive feedback. We are also thankful to the artists from the Cleveland Clinic’s Center for Medical Art & Photography for their help with the illustrations.
Conflicts of Interest
The authors declare no conflict of interest.
References
- World Health Organisation. Global Status Report on Alcohol and Health 2018; World Health Organization: Geneva, Switzerland, 2018.
- EU Citizens’ Attitudes towards Alcohol. Available online: https://ec.europa.eu/health/sites/default/files/alcohol/docs/ebs_331_en.pdf (accessed on 9 February 2021).
- National Institute on Alcohol Abuse and Alcoholism Drinking Levels Defined. Available online: https://www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking (accessed on 2 September 2021).
- National Institute on Alcohol Abuse and Alcoholism Alcohol Facts and Statistics. Available online: https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/alcohol-facts-and-statistics (accessed on 2 September 2021).
- Rehm, J.; Mathers, C.; Popova, S.; Thavorncharoensap, M.; Teerawattananon, Y.; Patra, J. Global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. Lancet 2009, 373, 2223–2233. [Google Scholar] [CrossRef]
- Touvier, M.; Druesne-Pecollo, N.; Kesse-Guyot, E.; Andreeva, V.A.; Galan, P.; Hercberg, S.; Latino-Martel, P. Demographic, socioeconomic, disease history, dietary and lifestyle cancer risk factors associated with alcohol consumption. Int. J. Cancer 2014, 134, 445–459. [Google Scholar] [CrossRef]
- Graff-Iversen, S.; Jansen, M.D.; Hoff, D.A.; Høiseth, G.; Knudsen, G.P.; Magnus, P.; Mørland, J.; Normann, P.T.; Næss, O.E.; Tambs, K. Divergent associations of drinking frequency and binge consumption of alcohol with mortality within the same cohort. J. Epidemiol. Community Health 2013, 67, 350–357. [Google Scholar] [CrossRef]
- Dugum, M.; McCullough, A. Diagnosis and management of alcoholic liver disease. J. Clin. Transl. Hepatol. 2015, 3, 109–116. [Google Scholar]
- Sawada Feldman, H.; Lyons Jones, K.; Lindsay, S.; Slymen, D.; Klonoff-Cohen, H.; Kao, K.; Rao, S.; Chambers, C. Prenatal alcohol exposure patterns and alcohol-related birth defects and growth deficiencies: A prospective study. Alcohol. Clin. Exp. Res. 2012, 36, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Rivas, I.; Sanvisens, A.; Bolao, F.; Fuster, D.; Tor, J.; Pujol, R.; Torrens, M.; Rey-Joly, C.; Muga, R. Impact of medical comorbidity and risk of death in 680 patients with alcohol use disorders. Alcohol. Clin. Exp. Res. 2013, 37, E221–E227. [Google Scholar] [CrossRef]
- Trudell, J.R.; Messing, R.O.; Mayfield, J.; Harris, R.A. Alcohol dependence: Molecular and behavioral evidence. Trends Pharmacol. Sci. 2014, 35, 317–323. [Google Scholar] [CrossRef]
- Vengeliene, V.; Bilbao, A.; Molander, A.; Spanagel, R. Neuropharmacology of alcohol addiction. Br. J. Pharmacol. 2008, 154, 299–315. [Google Scholar] [CrossRef]
- National Institute on Alcohol Abuse and Alcoholism Alcohol Use Disorder: A Comparison between DSM–IV and DSM–5. Available online: https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/alcohol-use-disorder-comparison-between-dsm?fbclid=IwAR0gGIMEub8BuoxUTAMbygBtNBLfJvMh0yOSN1vdLelzZMMHG4v57GjZm9g (accessed on 7 October 2021).
- Mukherjee, S. Alcoholism and its effects on the central nervous system. Curr. Neurovasc. Res. 2013, 10, 256–262. [Google Scholar] [CrossRef]
- Schuckit, M.A. Alcohol-use disorders. Lancet 2009, 373, 492–501. [Google Scholar] [CrossRef]
- Koob, G.F.; Colrain, I.M. Alcohol use disorder and sleep disturbances: A feed-forward allostatic framework. Neuropsychopharmacology 2020, 45, 141–165. [Google Scholar] [CrossRef] [PubMed]
- Adler, R.A. Clinical review 33: Clinically important effects of alcohol on endocrine function. J. Clin. Endocrinol. Metab. 1992, 74, 957–960. [Google Scholar] [PubMed]
- Close, C.E.; Roberts, P.L.; Berger, R.E. Cigarettes, alcohol and marijuana are related to pyospermia in infertile men. J. Urol. 1990, 144, 900–903. [Google Scholar] [CrossRef]
- Sansone, A.; Di Dato, C.; de Angelis, C.; Menafra, D.; Pozza, C.; Pivonello, R.; Isidori, A.; Gianfrilli, D. Smoke, alcohol and drug addiction and male fertility. Reprod. Biol. Endocrinol. 2018, 16, 3. [Google Scholar] [CrossRef]
- Grover, S.; Mattoo, S.K.; Pendharkar, S.; Kandappan, V. Sexual dysfunction in patients with alcohol and opioid dependence. Indian J. Psychol. Med. 2014, 36, 355–365. [Google Scholar] [CrossRef]
- Cederbaum, A.I. Alcohol metabolism. Clin. Liver Dis. 2012, 16, 667–685. [Google Scholar] [CrossRef]
- Wilkinson, P.K.; Sedman, A.J.; Sakmar, E.; Kay, D.R.; Wagner, J.G. Pharmacokinetics of ethanol after oral administration in the fasting state. J. Pharmacokinet. Biopharm. 1977, 5, 207–224. [Google Scholar] [CrossRef]
- Eriksson, C.J.P.; Fukunaga, T.; Sarkola, T.; Chen, W.J.; Chen, C.C.; Ju, J.M.; Cheng, A.T.; Yamamoto, H.; Kohlenberg-Muller, K.; Kimura, M.; et al. Functional relevance of human ADH polymorphism. Alcohol. Clin. Exp. Res. 2001, 25, 157S–163S. [Google Scholar] [CrossRef]
- Lieber, C.S. Perspectives: Do alcohol calories count? Am. J. Clin. Nutr. 1991, 54, 976–982. [Google Scholar] [CrossRef]
- Zimatkin, S.M.; Pronko, S.P.; Vasiliou, V.; Gonzalez, F.J.; Deitrich, R.A. Enzymatic mechanisms of ethanol oxidation in the brain. Alcohol. Clin. Exp. Res. 2006, 30, 1500–1505. [Google Scholar] [CrossRef]
- Oneta, C.M.; Lieber, C.S.; Li, J.J.; Rüttimann, S.; Schmid, B.; Lattmann, J.; Rosman, A.S.; Seitz, H.K. Dynamics of cytochrome P4502E1 activity in man: Induction by ethanol and disappearance during withdrawal phase. J. Hepatol. 2002, 36, 47–52. [Google Scholar] [CrossRef]
- Deng, X.S.; Deitrich, R.A. Putative role of brain acetaldehyde in ethanol addiction. Curr. Drug Abuse Rev. 2008, 1, 3–8. [Google Scholar] [CrossRef][Green Version]
- U.S. Department of Health and Human Services. Disulfiram. In Incorporating Alcohol Pharmacotherapies into Medical Practice: Treatment Improvement Protocol Series (TIP 49); Lulu.com: Rockville, MD, USA, 2009. [Google Scholar]
- Jiang, L.; Gulanski, B.I.; De Feyter, H.M.; Weinzimer, S.A.; Pittman, B.; Guidone, E.; Koretski, J.; Harman, S.; Petrakis, I.L.; Krystal, J.H.; et al. Increased brain uptake and oxidation of acetate in heavy drinkers. J. Clin. Investig. 2013, 123, 1605–1614. [Google Scholar] [CrossRef] [PubMed]
- Kendrick, S.F.W.; O’Boyle, G.; Mann, J.; Zeybel, M.; Palmer, J.; Jones, D.E.J.; Day, C.P. Acetate, the key modulator of inflammatory responses in acute alcoholic hepatitis. Hepatology 2010, 51, 1988–1997. [Google Scholar] [CrossRef]
- Best, C.; Laposata, M. Fatty acid ethyl esters: Toxic non-oxidative metabolites of ethanol and markers of ethanol intake. Front. Biosci. 2003, 8, e202–e217. [Google Scholar]
- Heier, C.; Xie, H.; Zimmermann, R. Nonoxidative ethanol metabolism in humans—from biomarkers to bioactive lipids. IUBMB Life 2016, 68, 916–923. [Google Scholar] [CrossRef]
- Slevin, E.; Baiocchi, L.; Wu, N.; Ekser, B.; Sato, K.; Lin, E.; Ceci, L.; Chen, L.; Lorenzo, S.R.; Xu, W.; et al. Kupffer cells: Inflammation pathways and cell-cell interactions in alcohol-associated liver disease. Am. J. Pathol. 2020, 190, 2185–2193. [Google Scholar] [CrossRef] [PubMed]
- Rocco, A.; Compare, D.; Angrisani, D.; Sanduzzi Zamparelli, M.; Nardone, G. Alcoholic disease: Liver and beyond. World J. Gastroenterol. 2014, 20, 14652–14659. [Google Scholar] [CrossRef]
- Seth, D.; Haber, P.; Syn, W.; Diehl, A.; Day, C. Pathogenesis of alcohol-induced liver disease: Classical concepts and recent advances. J. Gastroenterol. Hepatol. 2011, 26, 1089–1105. [Google Scholar] [CrossRef] [PubMed]
- Szabo, G.; Saha, B. Alcohol’s effect on host defense. Alcohol Res. 2015, 37, 159–170. [Google Scholar]
- Wang, H.J.; Zakhari, S.; Jung, M.K. Alcohol, inflammation, and gut-liver-brain interactions in tissue damage and disease development. World J. Gastroenterol. 2010, 16, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Arteel, G.E. Effect of ethanol on lipid metabolism. J. Hepatol. 2019, 70, 237–248. [Google Scholar] [CrossRef]
- Xu, G.; Li, C.; Parsiola, A.L.; Li, J.; McCarter, K.D.; Shi, R.; Mayhan, W.G.; Sun, H. Dose-dependent influences of ethanol on ischemic stroke: Role of inflammation. Front. Cell. Neurosci. 2019, 13, 6. [Google Scholar] [CrossRef]
- Ren, T.; Mackowiak, B.; Lin, Y.; Gao, Y.; Niu, J.; Gao, B. Hepatic injury and inflammation alter ethanol metabolism and drinking behavior. Food Chem. Toxicol. 2020, 136, 111070. [Google Scholar] [CrossRef]
- Roberto, M.; Patel, R.R.; Bajo, M. Ethanol and cytokines in the central nervous system. In Handbook of Experimental Pharmacology; Springer: Cham, Switzerland, 2017; Volume 248, pp. 397–431. [Google Scholar]
- Crews, F.T.; Sarkar, D.K.; Qin, L.; Zou, J.; Boyadjieva, N.; Vetreno, R.P. Neuroimmune function and the consequences of alcohol exposure. Alcohol Res. Curr. Rev. 2015, 37, 331–351. [Google Scholar]
- Yang, S.; Lian, G. ROS and diseases: Role in metabolism and energy supply. Mol. Cell. Biochem. 2020, 467, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Berndt, C.; Jones, D. Oxidative stress. Annu. Rev. Bioch 2017, 86, 715–748. [Google Scholar] [CrossRef]
- Mantle, D.; Preedy, V. Free radicals as mediators of alcohol toxicity. Advers. Druf. React. Toxicol. Rev. 1999, 18, 235–252. [Google Scholar]
- Das, S.K.; Vasudevan, D.M. Alcohol-induced oxidative stress. Life Sci. 2007, 81, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Zhao, Y.; Zhang, X. Acetaldehyde induces cytotoxicity of SH-SY5Y cells via inhibition of Akt activation and induction of oxidative stress. Oxid. Med. Cell. Longev. 2016, 2016, 4512309. [Google Scholar] [CrossRef]
- Cui, J.; Liu, Y.; Chang, X.; Gou, W.; Zhou, X.; Liu, Z.; Li, Z.; Wu, Y.; Zuo, D. Acetaldehyde induces neurotoxicity in vitro via oxidative stress- and Ca2+ imbalance-mediated endoplasmic reticulum stress. Oxid. Med. Cell. Longev. 2019, 2019, 2593742. [Google Scholar] [CrossRef] [PubMed]
- Clavijo-Cornejo, D.; Gutiérrez-Carrera, M.; Palestino-Domínguez, M.; Dominguez-Perez, M.; Nuño, N.; Souza, V.; Miranda, R.U.; Kershenobich, D.; Gutiérrez-Ruiz, M.C.; Bucio, L.; et al. Acetaldehyde targets superoxide dismutase 2 in liver cancer cells inducing transient enzyme impairment and a rapid transcriptional recovery. Food Chem. Toxicol. 2014, 69, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Yeligar, S.M.; Harris, F.L.; Hart, C.M.; Brown, L.A.S. Ethanol induces oxidative stress in alveolar macrophages via upregulation of NADPH oxidases. J. Immunol. 2012, 188, 3648–3657. [Google Scholar] [CrossRef] [PubMed]
- Luo, J. Autophagy and ethanol neurotoxicity. Autophagy 2014, 10, 2099–2108. [Google Scholar] [CrossRef]
- Yuan, F.; Chen, X.; Liu, J.; Feng, W.; Cai, L.; Wu, X.; Chen, S.Y. Sulforaphane restores acetyl-histone H3 binding to Bcl-2 promoter and prevents apoptosis in ethanol-exposed neural crest cells and mouse embryos. Exp. Neurol. 2018, 300, 60–66. [Google Scholar] [CrossRef]
- Kapasi, A.A.; Patel, G.; Goenka, A.; Nahar, N.; Modi, N.; Bhaskaran, M.; Reddy, K.; Franki, N.; Patel, J.; Singhal, P.C. Ethanol promotes T cell apoptosis through the mitochondrial pathway. Immunology 2003, 108, 313–320. [Google Scholar] [CrossRef]
- Han, J.Y.; Joo, Y.; Kim, Y.S.; Lee, Y.K.; Kim, H.J.; Cho, G.J.; Choi, W.S.; Kang, S.S. Ethanol induces cell death by activating caspase-3 in the rat cerebral cortex. Mol. Cells 2005, 20, 189–195. [Google Scholar]
- Steiner, J.L.; Lang, C.H. Etiology of alcoholic cardiomyopathy: Mitochondria, oxidative stress and apoptosis. Int. J. Biochem. Cell Biol. 2017, 89, 125–135. [Google Scholar] [CrossRef]
- Khirug, S.; Soni, S.; Saez Garcia, M.; Tessier, M.; Zhou, L.; Kulesskaya, N.; Rauvala, H.; Lindholm, D.; Ludwig, A.; Molinari, F.; et al. Protective role of low ethanol administration following ischemic stroke via recovery of KCC2 and p75NTR expression. Mol. Neurobiol. 2021, 58, 1145–1161. [Google Scholar] [CrossRef]
- Kotova, N.; Vare, D.; Schultz, N.; Meesters, D.G.; Stepnik, M.; Grawé, J.; Helleday, T.; Jenssen, D. Genotoxicity of alcohol is linked to DNA replication-associated damage and homologous recombination repair. Carcinogenesis 2013, 34, 325–330. [Google Scholar] [CrossRef]
- Santovito, A.; Cervella, P.; Delpero, M. Evidence of genotoxicity in lymphocytes of non-smoking alcoholics. Mol. Biol. Rep. 2015, 42, 53–59. [Google Scholar] [CrossRef][Green Version]
- Ceni, E.; Mello, T.; Galli, A. Pathogenesis of alcoholic liver disease: Role of oxidative metabolism. World J. Gastroenterol. 2014, 20, 17756–17772. [Google Scholar] [CrossRef] [PubMed]
- Peccerella, T.; Arslic-Schmitt, T.; Mueller, S.; Linhart, K.B.; Seth, D.; Bartsch, H.; Seitz, H.K. Chronic ethanol consumption and generation of Etheno-DNA adducts in cancer-prone tissues. Adv. Exp. Med. Biol. 2018, 1032, 81–92. [Google Scholar] [PubMed]
- Sapkota, M.; Wyatt, T.A. Alcohol, aldehydes, adducts and airways. Biomolecules 2015, 5, 2987–3008. [Google Scholar] [CrossRef] [PubMed]
- Salehi, F.; Behboudi, H.; Kavoosi, G.; Ardestani, S.K. Oxidative DNA damage induced by ROS-modulating agents with the ability to target DNA: A comparison of the biological characteristics of citrus pectin and apple pectin. Sci. Rep. 2018, 8, 13902. [Google Scholar] [CrossRef]
- Waris, G.; Ahsan, H. Reactive oxygen species: Role in the development of cancer and various chronic conditions. J. Carcinog. 2006, 5, 14. [Google Scholar] [CrossRef]
- Zhang, J.; Rane, G.; Dai, X.; Shanmugam, M.K.; Arfuso, F.; Samy, R.P.; Lai, M.K.P.; Kappei, D.; Kumar, A.P.; Sethi, G. Ageing and the telomere connection: An intimate relationship with inflammation. Ageing Res. Rev. 2016, 25, 55–69. [Google Scholar] [CrossRef]
- Yamaki, N.; Matsushita, S.; Hara, S.; Yokoyama, A.; Hishimoto, A.; Higuchi, S. Telomere shortening in alcohol dependence: Roles of alcohol and acetaldehyde. J. Psychiatr. Res. 2019, 109, 27–32. [Google Scholar] [CrossRef]
- Dixit, S.; Whooley, M.A.; Vittinghoff, E.; Roberts, J.D.; Heckbert, S.R.; Fitzpatrick, A.L.; Lin, J.; Leung, C.; Mukamal, K.J.; Marcus, G.M. Alcohol consumption and leukocyte telomere length. Sci. Rep. 2019, 9, 1404. [Google Scholar] [CrossRef]
- Salonen, I.; Pakarinen, P.; Huhtaniemi, I. Effect of chronic ethanol diet on expression of gonadotropin genes in the male rat. J. Pharmacol. Exp. Ther. 1992, 260, 463–467. [Google Scholar]
- Van Thiel, D.H.; Lester, R.; Sherins, R.J. Hypogonadism in alcoholic liver disease: Evidence for a double defect. Gastroenterology 1974, 67, 1188–1199. [Google Scholar] [CrossRef]
- Gordon, G.; Altman, K.; Southren, L.; Rupin, E.; Lieber, C. Effect of alcohol (ethanol) administration on sex-hormone metabolism in normal men. N. Engl. J. Med. 1976, 295, 793–797. [Google Scholar] [CrossRef]
- Ching, M.; Valenca, M.; Negro-Vilar, A. Acute ethanol treatment lowers hypophyseal portal plasma luteinizing hormone-releasing hormone (LH-RH) and systemic plasma LH levels in orchidectomized rats. Brain Res. 1988, 443, 325–328. [Google Scholar] [CrossRef]
- Salonen, I.; Huhtaniemi, I. Effects of chronic ethanol diet on pituitary-testicular function of the rat. Biol. Reprod. 1990, 42, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Emanuele, M.; Tentler, J.; Halloran, M.; Emanuele, N.; Wallock, L.; Kelley, M. The effect of acute in vivo ethanol exposure on follicle stimulating hormone transcription and translation. Alcohol. Clin. Exp. Res. 1992, 16, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Gordon, G.; Southren, A.L.; Vittek, J.; Lieber, C.S. The effect of alcohol ingestion on hepatic aromatase activity and plasma steroid hormones in the rat. Metabolism 1979, 28, 20–24. [Google Scholar] [CrossRef]
- Badr, F.; Bartke, A.; Dalterio, S.; Bulger, W. Suppression of testosterone production by ethyl alcohol. Possible mode of action. Steroids 1977, 30, 647–655. [Google Scholar] [CrossRef]
- Cobb, C.; Ennis, M.; Van Thiel, D.; Gavaler, J.; Lester, R. Acetaldehyde and ethanol are direct testicular toxins. Surg. Forum. 1978, 29, 641–644. [Google Scholar]
- Castilla-Cortazar, I.; Quiroga, J.; Prieto, J. Insulin-like growth factor-I, liver function, and hypogonadism in rats with experimentally induced cirrhosis. Hepatology 2000, 31, 1379. [Google Scholar] [CrossRef]
- Gordon, G.; Vittek, J.; Southren, A.L.; Munnangi, P.; Lieber, C.S. Effect of chronic alcohol ingestion on the biosynthesis of steroids in rat testicular homogenate in vitro. Endocrinology 1980, 106, 1880–1885. [Google Scholar] [CrossRef] [PubMed]
- Gavaler, J.S.; Perez, H.A.; Estes, L.; Van Thiel, D.H. Morphologic alterations of rat Leydig cells induced by ethanol. Pharmacol. Biochem. Behav. 1983, 18, 341–347. [Google Scholar] [CrossRef]
- Muthusami, K.R.; Chinnaswamy, P. Effect of chronic alcoholism on male fertility hormones and semen quality. Fertil. Steril. 2005, 84, 919–924. [Google Scholar] [CrossRef]
- Maneesh, M.; Dutta, S.; Chakrabarti, A.; Vasudevan, D. Alcohol abuse-duration dependent decrease in plasma testosterone and antioxidants in males. Indian J. Physiol. Pharmacol. 2006, 50, 291–296. [Google Scholar]
- Jensen, T.K.; Swan, S.; Jørgensen, N.; Toppari, J.; Redmon, B.; Punab, M.; Drobnis, E.Z.; Haugen, T.B.; Zilaitiene, B.; Sparks, A.E.; et al. Alcohol and male reproductive health: A cross-sectional study of 8344 healthy men from Europe and the USA. Hum. Reprod. 2014, 29, 1801–1809. [Google Scholar] [CrossRef] [PubMed]
- Shiels, M.S.; Rohrmann, S.; Menke, A.; Selvin, E.; Crespo, C.J.; Rifai, N.; Dobs, A.; Feinleib, M.; Guallar, E.; Platz, E.A. Association of cigarette smoking, alcohol consumption, and physical activity with sex steroid hormone levels in US men. Cancer Causes Control 2009, 20, 877–886. [Google Scholar] [CrossRef]
- Hansen, M.L.; Thulstrup, A.M.; Bonde, J.P.; Olsen, J.; Håkonsen, L.B.; Ramlau-Hansen, C.H. Does last week’s alcohol intake affect semen quality or reproductive hormones? A cross-sectional study among healthy young Danish men. Reprod. Toxicol. 2012, 34, 457–462. [Google Scholar] [CrossRef] [PubMed]
- La Vignera, S.; Condorelli, R.A.; Balercia, G.; Vicari, E.; Calogero, A.E. Does alcohol have any effect on male reproductive function? A review of literature. Asian J. Androl. 2013, 15, 221–225. [Google Scholar] [CrossRef]
- Rahimipour, M.; Talebi, A.R.; Anvari, M.; Sarcheshmeh, A.A.; Omidi, M. Effects of different doses of ethanol on sperm parameters, chromatin structure and apoptosis in adult mice. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 170, 423–428. [Google Scholar] [CrossRef]
- Franco Punhagui, A.P.; Rodrigues Vieira, H.; Eloisa Munhoz De Lion Siervo, G.; da Rosa, R.; Scantamburlo Alves Fernandes, G. Ethanol exposure during peripubertal period increases the mast cell number and impairs meiotic and spermatic parameters in adult male rats. Microsc. Res. Tech. 2016, 79, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Talebi, A.R.; Sarcheshmeh, A.A.; Khalili, M.A.; Tabibnejad, N. Effects of ethanol consumption on chromatin condensation and DNA integrity of epididymal spermatozoa in rat. Alcohol 2011, 45, 403–409. [Google Scholar] [CrossRef]
- Bai, S.; Wan, Y.; Zong, L.; Li, W.; Xu, X.; Zhao, Y.; Hu, X.; Zuo, Y.; Xu, B.; Tong, X.; et al. Association of alcohol intake and semen parameters in men with primary and secondary infertility: A cross-sectional study. Front. Physiol. 2020, 11, 566625. [Google Scholar] [CrossRef]
- Boeri, L.; Capogrosso, P.; Ventimiglia, E.; Pederzoli, F.; Cazzaniga, W.; Chierigo, F.; Dehò, F.; Montanari, E.; Montorsi, F.; Salonia, A. Heavy cigarette smoking and alcohol consumption are associated with impaired sperm parameters in primary infertile men. Asian J. Androl. 2019, 21, 478–485. [Google Scholar] [PubMed]
- Borges, E.; de Braga, D.P.A.F.; Provenza, R.R.; de Cassia Savio Figueira, R.; Iaconelli, A.; Setti, A.S. Paternal lifestyle factors in relation to semen quality and in vitro reproductive outcomes. Andrologia 2018, 50, e13090. [Google Scholar] [CrossRef] [PubMed]
- Sermondade, N.; Elloumi, H.; Berthaut, I.; Mathieu, E.; Delarouzire, V.; Ravel, C.; Mandelbaum, J. Progressive alcohol-induced sperm alterations leading to spermatogenic arrest, which was reversed after alcohol withdrawal. Reprod. Biomed. Online 2010, 20, 324–327. [Google Scholar] [CrossRef]
- Jensen, T.K.; Gottschau, M.; Madsen, J.O.B.; Andersson, A.M.; Lassen, T.H.; Skakkebæk, N.E.; Swan, S.H.; Priskorn, L.; Juul, A.; Jørgensen, N. Habitual alcohol consumption associated with reduced semen quality and changes in reproductive hormones; a cross-sectional study among 1221 young Danish men. BMJ Open 2014, 4, e005462. [Google Scholar] [CrossRef]
- Ricci, E.; Al Beitawi, S.; Cipriani, S.; Candiani, M.; Chiaffarino, F.; Viganò, P.; Noli, S.; Parazzini, F. Semen quality and alcohol intake: A systematic review and meta-analysis. Reprod. Biomed. Online 2017, 34, 38–47. [Google Scholar] [CrossRef]
- Anifandis, G.; Bounartzi, T.; Messini, C.I.; Dafopoulos, K.; Sotiriou, S.; Messinis, I.E. The impact of cigarette smoking and alcohol consumption on sperm parameters and sperm DNA fragmentation (SDF) measured by Halosperm. Arch. Gynecol. Obstet. 2014, 290, 777–782. [Google Scholar] [CrossRef]
- Aboulmaouahib, S.; Madkour, A.; Kaarouch, I.; Sefrioui, O.; Saadani, B.; Copin, H.; Benkhalifa, M.; Louanjli, N.; Cadi, R. Impact of alcohol and cigarette smoking consumption in male fertility potential: Looks at lipid peroxidation, enzymatic antioxidant activities and sperm DNA damage. Andrologia 2018, 50, e12926. [Google Scholar] [CrossRef]
- Ricci, E.; Noli, S.; Ferrari, S.; La Vecchia, I.; Cipriani, S.; De Cosmi, V.; Somigliana, E.; Parazzini, F. Alcohol intake and semen variables: Cross-sectional analysis of a prospective cohort study of men referring to an Italian Fertility Clinic. Andrology 2018, 6, 690–696. [Google Scholar] [CrossRef] [PubMed]
- Sadeghzadeh, M.; Shirpoor, A.; Naderi, R.; Kheradmand, F.; Gharalari, F.H.; Samadi, M.; Khalaji, N.; Gharaaghaji, R. Long-term ethanol consumption promotes changes in β-defensin isoform gene expression and induces structural changes and oxidative DNA damage to the epididymis of rats. Mol. Reprod. Dev. 2019, 86, 624–631. [Google Scholar] [CrossRef]
- Shayakhmetova, G.M.; Bondarenko, L.B.; Matvienko, A.V.; Kovalenko, V.M. Chronic alcoholism-mediated metabolic disorders in albino rat testes. Interdiscip. Toxicol. 2014, 7, 165–172. [Google Scholar] [CrossRef]
- Koh, P.O.; Won, C.K.; Cho, J.H. Ethanol decreases the expression of pituitary adenylate cyclase activating polypeptide in rat testes. J. Vet. Med. Sci. 2006, 68, 635–637. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, J.S.; Shukla, S.D. Acute in vivo effect of ethanol (binge drinking) on histone H3 modifications in rat tissues. Alcohol Alcohol. 2006, 41, 126–132. [Google Scholar] [CrossRef]
- Rompala, G.R.; Homanics, G.E. Intergenerational effects of alcohol: A review of paternal preconception ethanol exposure studies and epigenetic mechanisms in the male germline. Alcohol. Clin. Exp. Res. 2019, 43, 1032–1045. [Google Scholar] [CrossRef]
- Santi, D.; De Vincentis, S.; Magnani, E.; Spaggiari, G. Impairment of sperm DNA methylation in male infertility: A meta-analytic study. Andrology 2017, 5, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Ouko, L.A.; Shantikumar, K.; Knezovich, J.; Haycock, P.; Schnugh, D.J.; Ramsay, M. Effect of alcohol consumption on CpG methylation in the differentially methylated regions of H19 and IG-DMR in male gametes—Implications for fetal alcohol spectrum disorders. Alcohol. Clin. Exp. Res. 2009, 33, 1615–1627. [Google Scholar] [CrossRef]
- Chang, R.C.; Wang, H.; Bedi, Y.; Golding, M.C. Preconception paternal alcohol exposure exerts sex-specific effects on offspring growth and long-term metabolic programming. Epigenetics Chromatin 2019, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- Levi Montalcini, R. The nerve growth factor 35 years later. Science 1987, 237, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Ceccanti, M.; Coccurello, R.; Carito, V.; Ciafrè, S.; Ferraguti, G.; Giacovazzo, G.; Mancinelli, R.; Tirassa, P.; Chaldakov, G.N.; Pascale, E.; et al. Paternal alcohol exposure in mice alters brain NGF and BDNF and increases ethanol-elicited preference in male offspring. Addict. Biol. 2016, 21, 776–787. [Google Scholar] [CrossRef]
- Xia, R.; Jin, L.; Li, D.; Liang, H.; Yang, F.; Chen, J.; Yuan, W.; Miao, M. Association between paternal alcohol consumption before conception and anogenital distance of offspring. Alcohol. Clin. Exp. Res. 2018, 42, 735–742. [Google Scholar] [CrossRef]
- Marmorstein, N.; Iacono, W.; McGue, M. Alcohol and illicit drug dependence among parents: Associations with offspring externalizing disorders. Psychol. Med. 2009, 39, 149–155. [Google Scholar] [CrossRef]
- Cservenka, A.; Fair, D.A.; Nagel, B.J. Emotional processing and brain activity in youth at high risk for alcoholism. Alcohol. Clin. Exp. Res. 2014, 38, 1912–1923. [Google Scholar] [CrossRef]
- Monaco, A.P. An epigenetic, transgenerational model of increased mental health disorders in children, adolescents and young adults. Eur. J. Hum. Genet. 2021, 29, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Nie, Z.; Qu, Y.; Han, F.; Bell, E.M.; Zhuang, J.; Chen, J.; François, M.; Lipton, E.; Matale, R.; Cui, W.; et al. Evaluation of interactive effects between paternal alcohol consumption and paternal socioeconomic status and environmental exposures on congenital heart defects. Birth Defects Res. 2020, 112, 1273–1286. [Google Scholar] [CrossRef]
- Infante-Rivard, C.; El-Zein, M. Parental alcohol consumption and childhood cancers: A review. J. Toxicol. Environ. Health—Part B Crit. Rev. 2006, 10, 101–129. [Google Scholar] [CrossRef]
- Akison, L.K.; Moritz, K.M.; Reid, N. Adverse reproductive outcomes associated with fetal alcohol exposure: A systematic review. Reproduction 2019, 153, 329–343. [Google Scholar] [CrossRef]
- Stamatiades, G.A.; Kaiser, U.B. Gonadotropin regulation by pulsatile GnRH: Signaling and gene expression. Mol. Cell. Endocrinol. 2018, 463, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.L.; Auchus, R.J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr. Rev. 2011, 32, 81–151. [Google Scholar] [CrossRef]
- Kaur, G.; Thompson, L.A.; Dufour, J.M. Sertoli cells—Immunological sentinels of spermatogenesis. Semin. Cell Dev. Biol. 2014, 30, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Rachdaoui, N.; Sarkar, D.K. Pathophysiology of the effects of alcohol abuse on the endocrine system. Alcohol Res. 2017, 38, 255–276. [Google Scholar]
- Condorelli, R.A.; Calogero, A.E.; Vicari, E.; La Vignera, S. Chronic consumption of alcohol and sperm parameters: Our experience and the main evidences. Andrologia 2015, 47, 368–379. [Google Scholar] [CrossRef]
- Cicero, T.J.; Newman, K.S.; Gerrity, M.; Schmoeker, P.F.; Bell, R.D. Ethanol inhibits the naloxone-induced release of luteinizing hormone-releasing hormone from the hypothalamus of the male rat. Life Sci. 1982, 31, 1587–1596. [Google Scholar] [CrossRef]
- Rowe, P.; Racey, P.; Shenton, J.; Ellwood, M.; Lehane, J. Proceedings: Effects of acute administration of alcohol and barbiturates on plasma luteinizing hormone and testosterone in man. J. Endocrinol. 1974, 63, 50P–51P. [Google Scholar]
- McCann, S.M.; Mastronardi, C.; Walczewska, A.; Karanth, S.; Rettori, V.; Yu, W.H. The role of nitric oxide in reproduction. Brazilian J. Med. Biol. Res. 1999, 32, 1367–1379. [Google Scholar] [CrossRef]
- Canteros, G.; Rettori, V.; Franchi, A.; Genaro, A.; Cebral, E.; Faletti, A.; Gimeno, M.; Mccann, S.M. Ethanol inhibits luteinizing hormone-releasing hormone (LHRH) secretion by blocking the response of LHRH neuronal terminals to nitric oxide. Proc. Natl. Acad. Sci. USA 1995, 92, 3416–3420. [Google Scholar] [CrossRef] [PubMed]
- Kuller, L.H.; May, S.J.; Perper, J.A. The relationship between alcohol, liver disease, and testicular pathology. Am. J. Epidemiol. 1978, 108, 192–199. [Google Scholar] [CrossRef]
- Alvarez, J.G.; Lee, M.A.; Iozzo, R.V.; Lopez, I.; Touchstone, J.C.; Storey, B.T. Ethanol accelerates acrosomal loss in human spermatozoa. J. Androl. 1988, 9, 357–366. [Google Scholar] [CrossRef]
- Tangsrisakda, N.; Iamsaard, S. Effect of ethanol on the changes in testicular protein expression in adult male rats. Andrologia 2020, 52, e13784. [Google Scholar] [CrossRef] [PubMed]
- Rogers, B.J.; Cash, M.K.M.; Vaughn, W.K. Ethanol inhibits human and hamster sperm penetration of eggs. Gamete Res. 1987, 16, 97–107. [Google Scholar] [CrossRef]
- Sánchez, M.C.; Fontana, V.A.; Galotto, C.; Cambiasso, M.Y.; Sobarzo, C.M.A.; Calvo, L.; Calvo, J.C.; Cebral, E. Murine sperm capacitation, oocyte penetration and decondensation following moderate alcohol intake. Reproduction 2018, 155, 529–541. [Google Scholar] [CrossRef]
- Mongioì, L.M.; Perelli, S.; Condorelli, R.A.; Barbagallo, F.; Crafa, A.; Cannarella, R.; La Vignera, S.; Calogero, A.E. The role of resveratrol in human male fertility. Molecules 2021, 26, 2495. [Google Scholar] [CrossRef] [PubMed]
- Aquila, S.; Santoro, M.; De Amicis, F.; Guido, C.; Bonofiglio, D.; Lanzino, M.; Cesario, M.G.; Perrotta, I.; Sisci, D.; Morelli, C. Red wine consumption may affect sperm biology: The effects of different concentrations of the phytoestrogen Myricetin on human male gamete function. Mol. Reprod. Dev. 2013, 80, 155–165. [Google Scholar] [CrossRef]
- Lodovici, M.; Guglielmi, F.; Casalini, C.; Meoni, M.; Cheynier, V.; Dolara, P. Antioxidant and radical scavenging properties in vitro of polyphenolic extracts from red wine. Eur. J. Nutr. 2001, 40, 74–77. [Google Scholar] [CrossRef]
- Cui, X.; Jing, X.; Wu, X.; Yan, M. Protective effect of resveratrol on spermatozoa function in male infertility induced by excess weight and obesity. Mol. Med. Rep. 2016, 14, 4659–4665. [Google Scholar] [CrossRef]
- Martini, A.C.; Molina, R.I.; Estofán, D.; Senestrari, D.; Fiol De Cuneo, M.; Ruiz, R.D. Effects of alcohol and cigarette consumption on human seminal quality. Fertil. Steril. 2004, 82, 374–377. [Google Scholar] [CrossRef]
- López Teijón, M.; Garcia, F.; Serra, O.; Moragas, M.; Rabanal, A.; Olivares, R.; Alvarez, J.G. Semen quality in a population of volunteers from the province of Barcelona. Reprod. Biomed. Online 2007, 15, 434–444. [Google Scholar] [CrossRef]
- Wogatzky, J.; Wirleitner, B.; Stecher, A.; Vanderzwalmen, P.; Neyer, A.; Spitzer, D.; Schuff, M.; Schechinger, B.; Zech, N.H. The combination matters—Distinct impact of lifestyle factors on sperm quality: A study on semen analysis of 1683 patients according to MSOME criteria. Reprod. Biol. Endocrinol. 2012, 10, 115. [Google Scholar] [CrossRef] [PubMed]
- Koh, P.O.; Kim, M.O. Ethanol exposure decreases cell proliferation and increases apoptosis in rat testes. J. Vet. Med. Sci. 2006, 68, 1013–1017. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chastain, L.G.; Sarkar, D.K. Alcohol effects on the epigenome in the germline: Role in the inheritance of alcohol-related pathology. Alcohol 2017, 60, 53–66. [Google Scholar] [CrossRef]
- Cicero, T. Effects of paternal exposure. Alcohol Health Res. World 1994, 18, 37–41. [Google Scholar] [PubMed]
- Chang, R.C.; Skiles, W.M.; Chronister, S.S.; Wang, H.; Sutton, G.I.; Bedi, Y.S.; Snyder, M.; Long, C.R.; Golding, M.C. DNA methylation-independent growth restriction and altered developmental programming in a mouse model of preconception male alcohol exposure. Epigenetics 2017, 12, 841–853. [Google Scholar] [CrossRef]
- Curley, J.P.; Mashoodh, R.; Champagne, F.A. Epigenetics and the origins of paternal effects. Horm. Behav. 2011, 59, 306–314. [Google Scholar] [CrossRef]
- Rehm, J. How should prevalence of alcohol use disorders be assessed globally? Int. J. Methods Psychiatr. Res. 2016, 25, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Gilligan, C.; Anderson, K.G.; Ladd, B.O.; Yong, Y.M.; David, M. Inaccuracies in survey reporting of alcohol consumption. BMC Public Health 2019, 19, 1639. [Google Scholar] [CrossRef]
- Alcohol Research: Current Reviews Editorial Staff. Drinking patterns and their definitions. Alcohol Res. 2018, 39, 17–18. [Google Scholar]
- Wang, C.; Swerdloff, R.S. Limitations of semen analysis as a test of male fertility and anticipated needs from newer tests. Fertil. Steril. 2014, 102, 1502–1507. [Google Scholar] [CrossRef] [PubMed]
- Panner Selvam, M.K.; Finelli, R.; Agarwal, A.; Henkel, R. Proteomics and metabolomics—Current and future perspectives in clinical andrology. Andrologia 2021, 53, e13711. [Google Scholar] [CrossRef]
- Hetherington, L.; Schneider, E.K.; DeKretser, D.; Muller, C.H.; Hondermarck, H.; Velkov, T.; Baker, M.A. Deficiency in outer dense fiber 1 is a marker and potential driver of idiopathic male infertility. Mol. Cell. Proteom. 2016, 15, 3685–3693. [Google Scholar] [CrossRef]
- Holland, A.; Ohlendieck, K. Comparative profiling of the sperm proteome. Proteomics 2015, 15, 632–648. [Google Scholar] [CrossRef] [PubMed]
- Camargo, M.; Intasqui, P.; Bertolla, R.P. Understanding the seminal plasma proteome and its role in male fertility. Basic Clin. Androl. 2018, 28, 6. [Google Scholar] [CrossRef]
- Miranda, R.C.; Pietrzykowski, A.Z.; Tang, Y.; Sathyan, P.; Mayfield, D.; Keshavarzian, A.; Sampson, W.; Hereld, D. MicroRNAs: Master regulators of ethanol abuse and toxicity? Alcohol. Clin. Exp. Res. 2010, 34, 575–587. [Google Scholar] [CrossRef]
- Bedi, Y.; Chang, R.C.; Gibbs, R.; Clement, T.M.; Golding, M.C. Alterations in sperm-inherited noncoding RNAs associate with late-term fetal growth restriction induced by preconception paternal alcohol use. Reprod. Toxicol. 2019, 87, 11–20. [Google Scholar] [CrossRef]
- Crescitelli, R.; Lässer, C.; Szabó, T.G.; Kittel, A.; Eldh, M.; Dianzani, I.; Buzás, E.I.; Lötvall, J. Distinct RNA profiles in subpopulations of extracellular vesicles: Apoptotic bodies, microvesicles and exosomes. J. Extracell. Vesicles 2013, 2, 20677. [Google Scholar] [CrossRef]
- Vlassov, A.V.; Magdaleno, S.; Setterquist, R.; Conrad, R. Exosomes: Current knowledge of their composition, biological functions, and diagnostic and therapeutic potentials. Biochim. Biophys. Acta—Gen. Subj. 2012, 1820, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Schumann, G.; Liu, C.; O’Reilly, P.; Gao, H.; Song, P.; Xu, B.; Ruggeri, B.; Amin, N.; Jia, T.; Preis, S.; et al. KLB is associated with alcohol drinking, and its gene product β-Klotho is necessary for FGF21 regulation of alcohol preference. Proc. Natl. Acad. Sci. USA 2016, 113, 14372–14377. [Google Scholar] [CrossRef] [PubMed]
- Walters, R.K.; Polimanti, R.; Johnson, E.C.; McClintick, J.N.; Adams, M.J.; Adkins, A.E.; Aliev, F.; Bacanu, S.A.; Batzler, A.; Bertelsen, S.; et al. Transancestral GWAS of alcohol dependence reveals common genetic underpinnings with psychiatric disorders. Nat. Neurosci. 2018, 21, 1656–1669. [Google Scholar] [CrossRef]
- Kranzler, H.R.; Zhou, H.; Kember, R.L.; Vickers Smith, R.; Justice, A.C.; Damrauer, S.; Tsao, P.S.; Klarin, D.; Baras, A.; Reid, J.; et al. Genome-wide association study of alcohol consumption and use disorder in 274,424 individuals from multiple populations. Nat. Commun. 2019, 10, 1499. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).