Global Variation in Hand Hygiene Practices Among Adolescents: The Role of Family and School-Level Factors
Abstract
1. Introduction
2. Materials and Methods
2.1. Data
2.2. Study Population
2.3. Measurements
2.3.1. Outcome Variables
2.3.2. Independent Variables
2.4. Statistical Analyses
3. Results
3.1. Sample Characteristics
3.2. Hand Hygiene Practices
3.3. Correlate of Hand Hygiene Practices
4. Discussion
4.1. Main Findings and Contributions
4.2. Interpretation and Comparability
4.3. Strength and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Best, M.; Neuhauser, D. Ignaz Semmelweis and the birth of infection control. Qual. Saf. Health Care 2004, 13, 233–234. [Google Scholar] [CrossRef]
- Jarvis, W.R. Handwashing—The Semmelweis lesson forgotten? Lancet 1994, 344, 1311–1312. [Google Scholar] [CrossRef]
- Curtis, V.; Cairncross, S. Effect of washing hands with soap on diarrhea risk in the community: A systematic review. Lancet Infect. Dis. 2003, 3, 275–281. [Google Scholar] [CrossRef]
- UNICEF. Handwashing with Soap, Critical in the Fight against Coronavirus, Is “Out of Reach” for Billions. Available online: https://www.unicef.org.au/about-us/media/march-2020/handwashing-out-of-reach-for-billions (accessed on 5 April 2020).
- Zhan, S.; Yang, Y.Y.; Fu, C. Public’s early response to the novel coronavirus-infected pneumonia. Emerg. Microbes Infect. 2020, 9. [Google Scholar] [CrossRef]
- Aiello, A.E.; Larson, E.L. What is the evidence for a causal link between hygiene and infections? Lancet Infect. Dis. 2002, 2, 103–110. [Google Scholar] [CrossRef]
- UNICEF. Handwashing Stations and Supplies for the COVID-19 Response. Available online: https://www.unicef.org/media/68896/file/Handwashing-Facility-Factsheet.pdf (accessed on 24 May 2020).
- UNICEF. WASH—Understanding Hygiene promotion in the context of Risk Communication & Community Engagement (RCCE) and Infection Control and Prevention (IPC) for the COVID-19 Outbreak. Available online: https://www.unicef.org/media/66401/file/WASH-COVID-19-hygiene-programming-guidance-2020.pdf (accessed on 24 May 2020).
- Chisholm, R.H.; Campbell, P.T.; Wu, Y.; Tong, S.Y.C.; McVernon, J.; Geard, N. Implications of asymptomatic carriers for infectious disease transmission and control. R. Soc. Open Sci. 2018, 5. [Google Scholar] [CrossRef]
- Christie, D.; Viner, R. Adolescent development. BMJ 2005, 330. [Google Scholar] [CrossRef] [PubMed]
- Patton, G.C.; Olsson, C.A.; Skirbekk, V.; Saffery, R.; Wlodek, M.E.; Azzopardi, P.S.; Stonawski, M.; Rasmussen, B.; Spry, E.; Francis, K.; et al. Adolescence and the next generation. Nature 2018, 554, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Yalçin, S.; Yalçin, S.; Altin, S. Hand washing and adolescents. A study from seven schools in Konya, Turkey. Int. J. Adolesc. Med. Health 2004, 16, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Bolt, E.; Shordt, K.; Krukkert, I. School Sanitation and Hygiene Education: Results from the Assessment of a 6-Country Pilot Project. 2006. Available online: https://www.ircwash.org/resources/school-sanitation-and-hygiene-education-results-assessment-6-country-pilot-project (accessed on 5 May 2020).
- Ridenhour, B.J.; Braun, A.; Teyrasse, T.; Goldsman, D. Controlling the spread of disease in schools. PLoS ONE 2011, 6, 16–18. [Google Scholar] [CrossRef]
- Peltzer, K.; Pengpid, S. Health risk behaviour among in-school adolescents in the philippines: Trends between 2003, 2007 and 2011, a cross-sectional study. Int. J. Environ. Res. Public Health 2015, 13, 1–12. [Google Scholar] [CrossRef]
- Salleh, N.C.; Fuad, M.; Anuar, M.; Abdullah, N.A.; Yaw, S.L.; Wong, N.I.; Pei, T.T.; Awaluddin, S.M.; Tahir, A. Prevalence and Factors Associated with Oral and Hand Hygiene Practices among Adolescents in Malaysia: Findings from the National Health and Morbidity Survey 2017. Asia Pacific J. Public Health 2019, 31. [Google Scholar] [CrossRef] [PubMed]
- Brauer, M.; Zhao, J.T.; Bennitt, F.B.; Stanaway, J.D. Global access to Handwashing: Implications for COVID-19 control in low-income countries. Environ. Health Perspect. 2020, 128, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.C.; Stocks, M.E.; Cumming, O.; Jeandron, A.; Higgins, J.P.T.; Wolf, J.; Prüss-Ustün, A.; Bonjour, S.; Hunter, P.R.; Fewtrell, L.; et al. Hygiene and Health: Systematic Review of Handwashing Practices Worldwide and Update of Health Effects. Trop. Med. Int. Health 2014, 19, 906–916. [Google Scholar] [CrossRef] [PubMed]
- Wolf, J.; Hunter, P.R.; Freeman, M.C.; Cumming, O.; Clasen, T.; Bartram, J.; Higgins, J.P.T.; Johnston, R.; Medlicott, K.; Boisson, S.; et al. Impact of drinking water, sanitation and handwashing with soap on childhood diarrhoeal disease: Updated meta-analysis and meta-regression. Trop. Med. Int. Health 2018, 23, 508–525. [Google Scholar] [CrossRef]
- Ranasinghe, S.; Ramesh, S.; Jacobsen, K.H. Hygiene and mental health among middle school students in India and 11 other countries. J. Infect. Public Health 2016, 9, 429–435. [Google Scholar] [CrossRef]
- Peltzer, K.; Pengpid, S. Oral and hand hygiene behaviour and risk factors among in-school adolescents in four Southeast Asian countries. Int. J. Environ. Res. Public Health 2014, 11, 2780–2792. [Google Scholar] [CrossRef] [PubMed]
- Pengpid, S.; Peltzer, K. Hygiene behaviour and associated factors among in-school adolescents in nine African countries. Int. J. Behav. Med. 2011, 18, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Onyango-Ouma, W.; Aagaard-Hansen, J.; Jensen, B.B. The potential of schoolchildren as health change agents in rural western Kenya. Soc. Sci. Med. 2005, 61, 1711–1722. [Google Scholar] [CrossRef]
- Rohde, J.E.; Sadjimin, T. Elementary-school pupils as health educators: Role of school health programmes in primary health-care. Lancet 1980, 21, 1350–1352. [Google Scholar] [CrossRef]
- Kretzer, E.K.; Larson, E.L. Behavioral interventions to improve infection control practices. Am. J. Infect. Control 1998, 26, 245–253. [Google Scholar] [CrossRef]
- Ajzen, I.; Fishbein, M. Understanding the Attitudes and Predicting Social Behavior; Prentice-Hall: Englewood Cliffs, NJ, USA, 1980. [Google Scholar]
- Fishbein, M.; Guinan, M. Behavioral science and public health: A necessary partnership for HIV prevention. Public Health Rep. 1996, 111, 5–10. [Google Scholar] [PubMed]
- Lopez-Quintero, C.; Freeman, P.; Neumark, Y. Hand washing among school children in Bogotá, Colombia. Am. J. Public Health 2009, 99, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Song, I.H.; Kim, S.-A.; Park, W.-S. Family Factors Associated with Children’s Handwashing Hygiene Behavior. J. Child Health Care 2013, 17, 164–173. [Google Scholar] [CrossRef]
- Tran, D.; Phongsavan, P.; Bauman, A.; Havea, D.; Galea, G. Hygiene behaviour of adolescents in the Pacific: Associations with socio-demographic, health behaviour and school environment. Asia Pacific J. Public Health 2006, 18, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Taneja, D.K.; Badhan, S.K.; Ingle, G.K. Effect of a school-based hand washing promotion program on knowledge and hand washing behavior of girl students in a middle school of Delhi. Indian J. Public Health 2013, 57, 109–112. [Google Scholar] [CrossRef]
- Rani, M.D.M.; Mohamed, N.A.; Jamaluddin, T.Z.M.T.; Ismail, Z.; Ramli, S.; Faroque, H.; Samad, F.N.A.; Ariffien, A.R.; Farid, A.A.R.C.A.; Isahak, I. Effectiveness of comprehensive hand hygiene module on preschool children in Klang Valley, Malaysia. Clin. Experiemntal Pediatr. 2020, 63, 115–116. [Google Scholar] [CrossRef] [PubMed]
- Ramseier, C.A.; Leiggener, I.; Lang, N.P.; Bagramian, R.A.; Inglehart, M.R. Short-term effects of hygiene education for preschool (kindergarten) children: A clinical study. Oral Health Prev. Dent. 2007, 5, 19–24. [Google Scholar]
- Rosen, L.; Manor, O.; Engelhard, D.; Brody, D.; Rosen, B.; Peleg, H.; Meir, M.; Zucker, D. Can a Handwashing Intervention Make a Difference? Results from a Randomized Controlled Trial in Jerusalem Preschools. Prev. Med. 2006, 42, 27–32. [Google Scholar] [CrossRef]
- WHO. Global School-Based Student Health Survey (GSHS): Purpose and Methodology. Available online: www.who.int/chp/gshs/methodology/en/index.html (accessed on 10 September 2020).
- National Center for Immunization and Respiratory Diseases (NCIRD). How to Protect Yourself & Others. Available online: https://www.cdc.gov/coronavirus/2019-ncov/prevent-getting-sick/prevention.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Fprepare%2Fprevention.html (accessed on 31 May 2020).
- McLeroy, K.R.; Bibeau, D.; Steckler, A.; Glanz, K. An Ecological Perspective on Health Promotion Programs. Health Educ. Q 1988, 15, 351–377. [Google Scholar] [CrossRef]
- Grossi, V.; Klimschak, E.; Rechenburg, A.; Shinee, E.; Schmoll, O. The Situation of Water, Sanitation and Hygiene in Schools in the Pan-European Region. Available online: http://www.euro.who.int/en/publications/abstracts/situation-of-water,-sanitation-and-hygiene-in-schools-in-the-pan-european-region-the-2016 (accessed on 5 May 2020).
- Dreibelbis, R.; Winch, P.; Leontsini, E.; Hulland, K.R.; Ram, P.K.; Unicomb, L.; Luby, S.P. The integrated behavioural model for water, sanitation, and hygiene: A systematic review of behavioural models and a framework for designing and evaluating. BMC Public Health 2013, 13, 1–13. [Google Scholar] [CrossRef]
- Biswas, T.; Scott, J.G.; Munir, K.; Thomas, H.J.; Huda, M.M.; Hasan, M.M.; David de Vries, T.; Baxter, J.; Mamun, A.A. Global variation in the prevalence of bullying victimisation amongst adolescents: Role of peer and parental supports. EClinicalMedicine 2020, 20, 100276. [Google Scholar] [CrossRef] [PubMed]
- Pengpid, S.; Peltzer, K. Leisure-time sedentary behavior is associated with psychological distress and substance use among school-going adolescents in five southeast Asian countries: A cross-sectional study. Int. J. Environ. Res. Public Health 2019, 16, 2091. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.L.; Nakamura, K.; Seino, K.; Al-Sobaihi, S. Impact of parent-adolescent bonding on school bullying and mental health in Vietnamese cultural setting: Evidence from the global school-based health survey. BMC Psychol. 2019, 7, 1–10. [Google Scholar] [CrossRef]
- Pati, S.; Kadam, S.S.; Chauhan, A.S. Hand hygiene behavior among urban slum children and their care takers in Odisha, India. J. Prev. Med. Hyg. 2014, 55, 65–68. [Google Scholar]
- Rabbi, S.E.; Dey, N.C. Exploring the gap between hand washing knowledge and practices in Bangladesh: A cross-sectional comparative study. BMC Public Health 2013, 13, 89. [Google Scholar] [CrossRef] [PubMed]
- Luby, S.P.; Halder, A.K. Associations among handwashing indicators, wealth, and symptoms of childhood respiratory illness in urban Bangladesh. Trop. Med. Int. Health 2008, 13, 835–844. [Google Scholar] [CrossRef]
- Vessey, J.A.; Sherwood, J.J.; Warner, D.; Clark, D. Comparing Hand Washing to Hand Sanitizers in Reducing Elementary School Students’ Absenteeism. Pediatr. Nurs. 2007, 33, 368–372. [Google Scholar]
- Pengpid, S.; Peltzer, K. Hand and Oral Hygiene Practices among Adolescents in Dominican Republic, Suriname and Trinidad and Tobago: Prevalence, Health, Risk Behavior, Mental Health and Protective Factors. Int. J. Environ. Res. Public Health 2020, 17, 7860. [Google Scholar] [CrossRef]
- Sharma, M.; Batra, K.; Davis, R.E.; Wilkerson, A.H. Explaining Handwashing Behavior in a Sample of College Students during COVID-19 Pandemic Using the Multi-Theory Model (MTM) of Health Behavior Change: A Single Institutional Cross-Sectional Survey. Healthcare 2021, 9, 55. [Google Scholar] [CrossRef] [PubMed]
- World Bank. Are Your Hands Clean Enough? Baseline and Consumer Research Study findings on Handwashing with Soap Behaviour in Kenya. Available online: https://www.pseau.org/outils/ouvrages/wsp_africa_are_your_hands_clean_enough_2009.pdf (accessed on 5 May 2020).
- Steinberg, L. We know some things: Parent-adolescent relationships in retrospect and prospect. J. Res. Adolesc. 2001, 11, 1–19. [Google Scholar] [CrossRef]
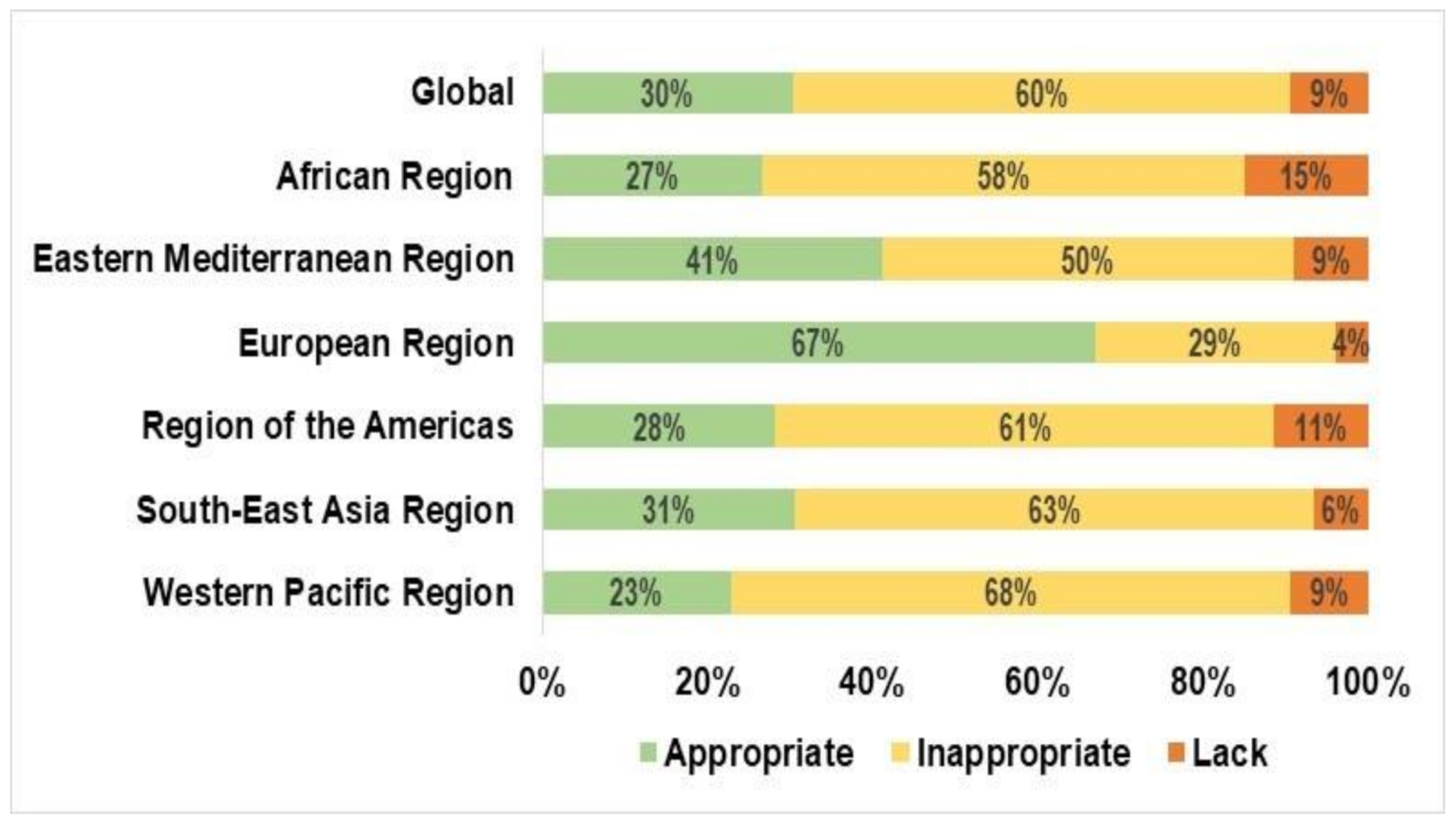
| Global | African Region | Eastern Mediterranean Region | European Region | Region of the Americas | South-East Asia Region | Western Pacific Region | |
|---|---|---|---|---|---|---|---|
| Age | |||||||
| 13 years | 65,546 (22.4) | 7767 (20.2) | 15,160 (26.9) | 455 (26.8) | 18,294 (21.7) | 9876 (27.4) | 13,994 (13.2) |
| 14 years | 83,515 (28.5) | 10,969 (24.9) | 18,728 (29.9) | 448 (26.8) | 24,125 (29.5) | 12,203 (31.6) | 17,042 (24.9) |
| 15 years | 78,710 (24.5) | 12,629 (27.1) | 16,730 (25.1) | 569 (25.1) | 21,382 (27.9) | 10,599 (21.9) | 16,801 (25) |
| 16+ years | 79,999 (24.6) | 15,822 (27.8) | 13,697 (18.1) | 539 (21.3) | 15,526 (20.9) | 11,580 (19.1) | 22,835 (36.9) |
| Sex | |||||||
| Male | 168,504 (51.4) | 27,753 (51.6) | 36,500 (53.6) | 1033 (52.3) | 41,780 (49.7) | 24,040 (52.1) | 37,398 (48.8) |
| Female | 181,673 (48.6) | 29,010 (48.4) | 37,379 (46.4) | 1053 (47.7) | 45,963 (50.3) | 27,402 (47.9) | 40,866 (51.2) |
| SES | |||||||
| Average | 240,305 (68.3) | 28,225 (61.9) | 55,412 (75.5) | 1986 (94.7) | 71,995 (83.2) | 29,840 (61.9) | 52,847 (70.1) |
| Below average | 94,731 (31.7) | 20,308 (38.1) | 18,528 (24.5) | 108 (5.3) | 16,063 (16.8) | 14,202 (38.1) | 25,522 (29.9) |
| Physically active | |||||||
| No | 215,791 (68.2) | 26,979 (65.8) | 45,045 (70.7) | 503 (23.6) | 57,038 (71.6) | 31,921 (66.9) | 54,305 (69.3) |
| Yes | 110,649 (31.8) | 18,378 (34.2) | 18,728 (29.3) | 1459 (76.4) | 29,526 (28.4) | 19,160 (33.1) | 23,398 (30.7) |
| Sedentary behavior | |||||||
| <3 h | 219,251 (70.6) | 34,015 (73.7) | 46,619 (74.1) | 922 (47.8) | 49,301 (64) | 37,870 (73.2) | 50,524 (63.9) |
| ≥3 h | 120,231 (29.4) | 15,822 (26.3) | 25,915 (25.9) | 1092 (52.2) | 36,877 (36) | 13,218 (26.8) | 27,307 (36.1) |
| Smoking | |||||||
| No | 279,620 (91) | 43,244 (92.5) | 64,261 (92.1) | 1700 (85.8) | 58,259 (83.8) | 43,099 (90.8) | 69,057 (91.1) |
| Yes | 37,220 (9) | 4014 (7.5) | 7942 (7.9) | 324 (14.2) | 11,256 (16.2) | 5187 (9.2) | 8497 (8.9) |
| Alcohol consumption | |||||||
| No | 196,182 (85.6) | 34,675 (86.3) | 7542 (90.7) | 1088 (60.6) | 48,977 (67.2) | 38,075 (93.7) | 65,825 (79.7) |
| Yes | 57,827 (14.4) | 8028 (13.7) | 1007 (9.3) | 794 (39.4) | 32,651 (32.8) | 4608 (6.3) | 10,739 (20.3) |
| Bullying victimization | |||||||
| No | 212,727 (65.1) | 26,507 (55.3) | 39,852 (54.5) | 1749 (90) | 62,339 (70.9) | 28,402 (72.1) | 53,878 (68.1) |
| Yes | 102,063 (34.9) | 22,349 (44.7) | 24,825 (45.5) | 193 (10) | 21,536 (29.1) | 12,695 (27.9) | 20,465 (31.8) |
| Loneliness | |||||||
| No | 296,365 (88.6) | 41,885 (86.9) | 57,896 (84.2) | 1954 (93.6) | 77,867 (89.7) | 46,657 (92) | 70,106 (87.8) |
| Yes | 40,834 (11.4) | 7240 (13.1) | 11,071 (15.8) | 144 (6.4) | 9763 (10.3) | 4728 (8) | 7888 (12.2) |
| Parental supervision | |||||||
| No | 171,657 (61) | 17,140 (51.9) | 34,364 (55.7) | No obs | 41,997 (59.1) | 30,110 (57.8) | 48,046 (75.3) |
| Yes | 104,788 (39) | 12,751 (48.1) | 29,069 (44.3) | No obs | 25,146 (40.9) | 20,680 (42.2) | 17,142 (24.7) |
| Parental bonding | |||||||
| No | 152,668 (58.5) | 18,686 (62.5) | 34,723 (57.6) | No obs | 32,536 (50.8) | 27,440 (55) | 39,283 (65) |
| Yes | 122,459 (41.5) | 11,020 (37.5) | 28,458 (42.4) | No obs | 34,159 (49.2) | 23,045 (45) | 25,777 (35) |
| Variable | Hand Hygiene Practices | |||
|---|---|---|---|---|
| Lacking | Inappropriate | Appropriate | p-Value | |
| Age | <0.0001 | |||
| 13 years | 8.4% | 58.1% | 33.5% | |
| 14 years | 8.6% | 59.4% | 32% | |
| 15 years | 9.5% | 59.8% | 30.6% | |
| 16+ years | 10.7% | 64.2% | 25.1% | |
| Sex | <0.0001 | |||
| Male | 10.6% | 60.6% | 28.8% | |
| Female | 7.9% | 60.2% | 31.9% | |
| SES | <0.0001 | |||
| Average | 9.2% | 59.2% | 31.6% | |
| Below average | 9.4% | 63.1% | 27.5% | |
| Physically active | <0.0001 | |||
| No | 9.2% | 62.4% | 28.6 | |
| Yes | 9.9% | 58.9% | 31.2% | |
| Sedentary behavior | <0.0001 | |||
| No | 8.7% | 58.6% | 32.7% | |
| Yes | 10.6% | 65.1% | 24.3% | |
| Smoking | <0.0001 | |||
| No | 8.6% | 60.4% | 31.1% | |
| Yes | 15.6% | 62.0% | 22.4% | |
| Alcohol consumption | <0.0001 | |||
| No | 8.3% | 62.6% | 29.1% | |
| Yes | 13.8% | 67.2% | 19.0% | |
| Bullying victimization | <0.0001 | |||
| No | 7.5% | 60.3% | 32.3% | |
| Yes | 11.3% | 62.5% | 26.3% | |
| Loneliness | <0.0001 | |||
| No | 9.0% | 61.1% | 29.9% | |
| Yes | 11.2% | 61.2% | 27.6% | |
| Parental supervision | <0.0001 | |||
| No | 11.1% | 65.8% | 23.1% | |
| Yes | 6.2% | 53.9% | 39.9% | |
| Parental bonding | <0.0001 | |||
| No | 11.2% | 63.4% | 25.4% | |
| Yes | 6.4% | 58.0% | 35.6% | |
| Inappropriate vs. Appropriate (Ref) | Lacking vs. Appropriate (Ref) | |||
|---|---|---|---|---|
| RRR (95% CI) | ARRR (95% CI) | RRR (95% CI) | ARRR (95% CI) | |
| Age | ||||
| 13 years | ||||
| 14 years | 1.01 (0.94–1.08) | 0.93 (0.84–1.03) | 1.00 (0.89–1.14) | 0.87 (0.72–1.06) |
| 15 years | 1.05 (0.96–1.14) | 0.91 (0.79–1.04) | 1.14 (0.97–1.34) | 1.03 (0.80–1.33) |
| 16+ years | 1.19 (1.10–1.29) *** | 1.00 (0.89–1.13) | 1.46 (1.26–1.68) *** | 1.20 (0.96–1.5) |
| Sex | ||||
| Male | ||||
| Female | 0.86 (0.82–0.9) *** | 0.93 (0.86–1.01) | 0.66 (0.60–0.73) *** | 0.80 (0.70–0.92) ** |
| SES | ||||
| Average | ||||
| Below average | 1.22 (1.15–1.29) *** | 0.98 (0.90–1.07) | 1.15 (1.05–1.27) ** | 0.70 (0.59–0.82) *** |
| Physically active | ||||
| No | ||||
| Yes | 0.9 (0.86–0.95) *** | 0.89 (0.83–0.96) ** | 1.00 (0.91–1.11) | 0.99 (0.84–1.16) |
| Sedentary behavior | ||||
| No | ||||
| Yes | 1.40 (1.33–1.46) *** | 1.41 (1.31–1.51) *** | 1.61 (1.49–1.74) *** | 1.57 (1.37–1.80) *** |
| Smoking | ||||
| No | ||||
| Yes | 1.44 (1.31–1.59) *** | 1.06 (0.9–1.23) | 2.67 (2.17–3.29) *** | 1.60 (1.09–2.36) * |
| Alcohol consumption | ||||
| No | ||||
| Yes | 1.49 (1.39–1.60) *** | 1.18 (1.07–1.30) ** | 2.39 (2.05–2.79) *** | 1.31 (1.06–1.61) |
| Being bullied | ||||
| No | ||||
| Yes | 1.40 (1.33–1.48) *** | 1.20 (1.10–1.30) *** | 2.07 (1.86–2.31) *** | 1.69 (1.44–2.0) *** |
| Loneliness | ||||
| No | ||||
| Yes | 1.11 (1.05–1.18) *** | 1.03 (0.92–1.15) | 1.34 (1.21–1.48) *** | 1.06 (0.86–1.30) |
| Parental supervision | ||||
| No | ||||
| Yes | 0.51 (0.48–0.54) *** | 0.55 (0.50–0.59) *** | 0.32 (0.29–0.36) *** | 0.46 (0.39–0.54) *** |
| Parental bonding | ||||
| No | ||||
| Yes | 0.67 (0.64–0.71) *** | 0.81 (0.75–0.87) *** | 0.40 (0.36–0.44) *** | 0.55 (0.48–0.63) *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jatrana, S.; Hasan, M.M.; Mamun, A.A.; Fatima, Y. Global Variation in Hand Hygiene Practices Among Adolescents: The Role of Family and School-Level Factors. Int. J. Environ. Res. Public Health 2021, 18, 4984. https://doi.org/10.3390/ijerph18094984
Jatrana S, Hasan MM, Mamun AA, Fatima Y. Global Variation in Hand Hygiene Practices Among Adolescents: The Role of Family and School-Level Factors. International Journal of Environmental Research and Public Health. 2021; 18(9):4984. https://doi.org/10.3390/ijerph18094984
Chicago/Turabian StyleJatrana, Santosh, Md. Mehedi Hasan, Abdullah A. Mamun, and Yaqoot Fatima. 2021. "Global Variation in Hand Hygiene Practices Among Adolescents: The Role of Family and School-Level Factors" International Journal of Environmental Research and Public Health 18, no. 9: 4984. https://doi.org/10.3390/ijerph18094984
APA StyleJatrana, S., Hasan, M. M., Mamun, A. A., & Fatima, Y. (2021). Global Variation in Hand Hygiene Practices Among Adolescents: The Role of Family and School-Level Factors. International Journal of Environmental Research and Public Health, 18(9), 4984. https://doi.org/10.3390/ijerph18094984






