The ADIPS Pilot National Diabetes in Pregnancy Benchmarking Programme
Abstract
1. Introduction
2. Materials and Methods
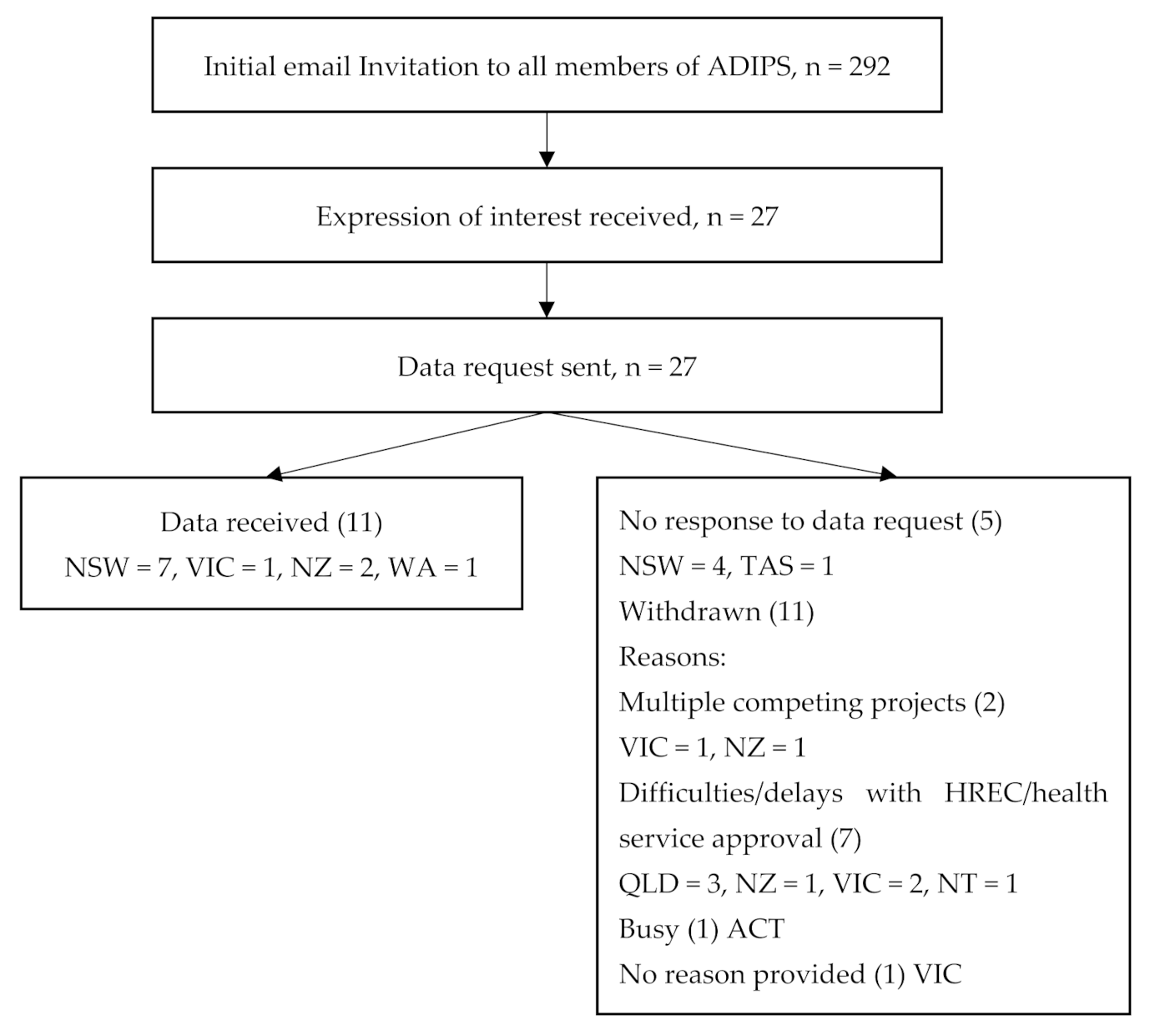
2.1. Participants
2.2. Development of the Audit Tool
2.3. Data Collection
2.4. Feedback Reports
2.5. Statistical Analysis
3. Results
3.1. Maternal Characteristics
3.2. Gestational Diabetes Mellitus
3.3. Pre-Gestational Diabetes
3.3.1. Pre-Gestational Diabetes: Pregnancy Planning
3.3.2. Pre-Gestational Diabetes: Antenatal Management
3.3.3. Pre-Gestational Diabetes: Pregnancy Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yuen, L.; Saeedi, P.; Riaz, M.; Kururanga, S.; Divakar, H.; Levitt, N.; Xilin, Y.; Simmons, D. IDF Diabetes Atlas: Projections of the prevalence of hyperglycaemia in pregnancy in 2019 and beyond: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157. [Google Scholar] [CrossRef] [PubMed]
- The HAPO Study Cooperative Research Group. Hyperglycemia and Adverse Pregnancy Outcomes. N. Engl. J. Med. 2008, 358, 1991–2002. [Google Scholar]
- Diabetes Control and Complications Trial Research Group. Pregnancy outcomes in the diabetes control and complications trial. Am. J. Obstet. Gynecol. 1996, 174, 1343–1353. [Google Scholar] [CrossRef]
- Australian Institute of Health and Welfare. Diabetes in Pregnancy 2014–2015. Bulletin no. 146. Cat. no. CDK 7; AIHW: Canberra, Australia, 2019. [Google Scholar]
- Australian Institute of Health and Welfare. Diabetes in Pregnancy: Its Impact on Australian Women and Their Babies. Diabetes Series no. 14. Cat. no. CVD 52; AIHW: Canberra, Australia, 2010. [Google Scholar]
- Kirke, A.B.; Atkinson, D.; Moore, S.; Sterry, K.; Singleton, S.; Roxburgh, C.; Parrish, K.; Porter, C.; Marley, J.V. Diabetes screening in pregnancy failing women in rural Western Australia: An audit of oral glucose tolerance test completion rates. Aust. J. Rural. Health 2019, 27, 64–69. [Google Scholar] [PubMed]
- Mackin, S.T.; Nelson, S.M.; Kerssens, J.J.; Wood, R.; Wild, S.; Colhoun, H.M.; Leese, G.P.; Philip, S.; Lindsay, R.S. SDRN Epidemiology Group. Diabetes and pregnancy: National trends over a 15 year period. Diabetologia 2018, 61, 1081–1088. [Google Scholar] [PubMed]
- Australian Atlas of Healthcare Variation 2017: 3.0 Women’s Health and Maternity—Introduction and Key Recommendations. Available online: https://www.safetyandquality.gov.au/sites/default/files/migrated/3.0-Introduction-and-key-recommendations.pdf (accessed on 20 October 2020).
- Simmons, D.; Oats, J. Diabetes in Pregnancy in New Zealand. In Textbook of Diabetes and Pregnancy; Hod, M., Ed.; Informa Healthcare: London, UK, 2008. [Google Scholar]
- Murphy, H.R.; Bell, R.; Cartwright, C.; Curnow, P.; Maresh, M.; Morgan, M.; Sylvester, C.; Young, B. Improved pregnancy outcomes in women with type 1 and type 2 diabetes but substantial clinic-to-clinic variations: A prospective nationwide study. Diabetologia 2017, 60, 1668–1677. [Google Scholar] [CrossRef] [PubMed]
- Murphy, H.R.; Howgate, C.; O’Keefe, J.; Myers, J.; Morgan, M.; Coleman, M.A.; Jolly, M.; Valabhji, J.; Scott, E.M.; Knighton, P.; et al. National Pregnancy in Diabetes (NPID) advisory group. Characteristics and outcomes of pregnant women with type 1 or type 2 diabetes: A 5-year national population-based cohort study. Lancet Diabetes Endocrinol. 2021, 9, 153–164. [Google Scholar] [CrossRef]
- Simmons, D. Adverse pregnancy outcomes in women with type 1 or type 2 diabetes. Lancet Diabetes Endocrinol. 2021, 9, 129–131. [Google Scholar] [CrossRef]
- Lee, A.S.; Colagiuri, S.; Flack, J.R. Successful implementation of diabetes audits in Australia: The Australian National Diabetes Information Audit and Benchmarking (ANDIAB) initiative. Diabet. Med. 2018, 35, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Simmons, D.; Cheung, N.W.; McIntyre, D.; Flack, J.R.; Lagstrom, J.; Bond, D.; Johnson, E.; Wolmarans, L.; Wein, P.; Sinha, A.K. The ADIPS Pilot National Diabetes in Pregnancy Audit Project. Aust. N. Z. J. Obstet. Gynae. 2007, 47, 198–206. [Google Scholar]
- Feig, D.S.; Corcoy, R.; Jensen, D.M.; Kautzky-Willer, A.; Nolan, C.J.; Oats, J.J.N.; Sacks, D.A.; Caimari, F.; McIntyre, H.D. Diabetes in pregnancy outcomes: A systematic review and proposed codification of definitions. Diabetes Metab. Res. Rev. 2015, 31, 680–690. [Google Scholar] [PubMed]
- Sinha, I.P.; Smyth, R.L.; Williamson, P.R. Using the Delphi technique to determine which outcomes to measure in clinical trials: Recommendations for the future based on a systematic review of existing studies. PLoS Med. 2011, 8, e1000393. [Google Scholar]
- Rudland, V.L.; Price, S.A.L.; Hughes, R.; Barrett, H.L.; Lagstrom, J.; Porter, C.; Britten, F.L.; Glastras, S.; Fulcher, I.; Wein, P.; et al. ADIPS 2020 guideline for pre-existing diabetes and pregnancy. Aust. N. Z. J. Obstet. Gynaecol. 2020, 60, E18–E52. [Google Scholar] [CrossRef] [PubMed]
- Australian Government. Australian Institute of Health and Welfare. Metadata Online Registry. Available online: https://meteor.aihw.gov.au/content/index.phtml/itemId/181414-title=About (accessed on 20 October 2020).
- Simmons, D. The benefits of the use of the new International Association of Diabetes in Pregnancy Study Groups guidelines for gestational diabetes mellitus. Aust. N. Z. J. Obstet. Gynaecol. 2020, 60, 486–488. [Google Scholar] [CrossRef] [PubMed]
- Sina, M.; Cade, T.J.; Flack, J.; Nolan, C.J.; Rajagopal, R.; Wong, V.; Burcher, L.; Barry, A.; Gianatti, E.; McCarthy, A.; et al. Antenatal models of care for women with gestational diabetes mellitus: Vignettes from an international meeting. Aust. N. Z. J. Obstet. Gynaecol. 2020, 60, 720–728. [Google Scholar] [CrossRef] [PubMed]
- McElduff, A.; Ross, G.P.; Lagström, J.A.; Champion, B.; Flack, J.R.; Lau, S.M.; Moses, R.G.; Seneratne, S.; McLean, M.; Cheung, N.W. Pregestational Diabetes and Pregnancy: An Australian experience. Diabetes Care 2005, 28, 1260–1261. [Google Scholar] [CrossRef] [PubMed]
- Wahabi, H.A.; Fayed, A.; Esmaeil SElmorshedy, H.; Titi, M.A.; Amer, Y.S.; Alzeidan, R.A.; Alodhayani, A.A.; Saeed, E.; Bahkali, K.H.; Kahili-Heede, M.K.; et al. Systematic review and meta-analysis of the effectiveness of pre-pregnancy care for women with diabetes for improving maternal and perinatal outcomes. PLoS ONE 2020, 15, e0237571. [Google Scholar] [CrossRef] [PubMed]
- Nankervis, A.; McIntyre, H.D.; Moses, R.; Ross, G.P.; Callaway, L.; Porter, C.; Jeffries, W.; Boorman, C.; De Vries, B.; McElduff, A. ADIPS Consensus Guidelines for the Testing and Diagnosis of Gestational Diabetes Mellitus in Australia. Available online: http://www.adips.org/downloads/ADIPSConsensusGuidelinesGDM-03.05.13VersionACCEPTEDFINAL.pdf (accessed on 30 December 2020).
- Hoffman, L.; Nolan, C.; Wilson, J.D.; Oats, J.J.; Simmons, D. The Australasian Diabetes in Pregnancy Society. Gestational diabetes mellitus—Management guidelines. Med. J. Aust. 1998, 169, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health. Screening, Diagnosis and Management of Gestational Diabetes in New Zealand: A Clinical Practice Guideline; Ministry of Health: Wellington, New Zealand, 2014. Available online: https://www.health.govt.nz/system/files/documents/publications/screening-diagnosis-management-of-gestational-diabetes-in-nz-clinical-practive-guideline-dec14-v2.pdf (accessed on 30 December 2020).
| No | Best Practice Recommendations for Women with Diabetes |
|---|---|
| 1 | All pregnancies should be intended pregnancies. |
| 2 | Pre-pregnancy counselling regarding risks should be provided. |
| 3 | Teratogenic medications should be avoided before conception and during pregnancy. |
| 4 | Blood glucose should be near normal before conception and during pregnancy. |
| 5 | Women should receive high dose (2.5–5 mg daily) folate supplementation starting 3 months prior to conception until 12 weeks gestation. |
| 6 | Oral hypoglycaemic agents should be reviewed prior to conception. |
| 7 | First antenatal visit should occur early in pregnancy, before 8 weeks of gestation. |
| 8 | Glycaemic control should be balanced against the risk of hypoglycaemia in early pregnancy. |
| 9 | Women with diabetes should be monitored for signs or progression of diabetes microvascular complications. |
| 10 | Vaginal birth should be encouraged unless there are obstetric or fetal indications for caesarean section. |
| 11 | Breastfeeding should be encouraged for all women with diabetes. |
| Category | Data Items |
|---|---|
| For all women | Patient ID, date of birth, date of referral, EDC, estimated EDC by ultrasound, ethnic group, gestation at first review, gravidity, parity, height, pre-pregnancy weight, booking weight, smoking status, chronic hypertension, gestational hypertension, preeclampsia, delivery method, baby’s date of birth, gestational age at delivery, baby sex, birth weight, APGAR at 5 mins, shoulder dystocia, other birth trauma, admission to SCN/NICU, neonatal hypoglycaemia <2.6 mmol/L, jaundice needing phototherapy, minor congenital malformation, major congenital malformation, breast feeding on discharge. |
| Women with GDM | OGTT fasting, OGTT 1 h, OGTT 2 h, HbA1c at GDM diagnosis, GDM treatment. |
| Women with type 1 or type 2 diabetes | Planned pregnancy, OHA at conception, statin at conception, ACE/ARB at conception, folate (1st trimester folate dose), aspirin use, retinopathy, nephropathy, hypoglycaemia needing assistance, number of hypoglycaemic episodes needing assistance, eGFR, urine ACR, HbA1c % trimester 1, HbA1c % trimester 2, HbA1c % trimester 3, type 1 diabetes treatment, type 2 diabetes treatment. |
| Characteristics | N | Mean ± SD or n (%) (Range) @ | Range (N, Mean or % Across All Sites) | Number of Sites Missing 100% Data (Sites Not Reporting/Collecting) † | Missing Values | |
|---|---|---|---|---|---|---|
| N | % Missing Range Across All Sites | |||||
| Number of women | 10,144 | -- | 59–2426 | 0 | -- | -- |
| GDM | 8696 | -- | 54–2397 | 0 | -- | -- |
| Type 1 diabetes | 435 | -- | 7–295 | 0 | -- | -- |
| Type 2 diabetes | 1013 | -- | 19–428 | 0 | -- | -- |
| Age (years) | 10,114 | 32.3 ± 5.5 (15–51) | 31.0–35.1 | 0 | 30 | 0–11.9 |
| Gravidity | 10,065 | 2.9 ± 1.9 (1–22) | 2.4–3.3 | 0 | 79 | 0–8.9 |
| Parity | 10,066 | 1.3 ± 1.4 (0–11) | 0.8–1.6 | 0 | 78 | 0–16.9 |
| Pre-pregnancy weight (kg) | 6471 | 74.7 ± 21.1 (34.0–185.0) | 68.5–89.8 | 3 | 3673 | 0–100 |
| Mean Pre-pregnancy BMI (kg/m2) | ||||||
| GDM | 6060 | 28.1 ± 7.0 (26.2–32.7) | 26.2–32.7 | 5 | 2636 | 30.3 |
| Type 1 diabetes | 215 | 26.0 ± 5.4 (22.8–30.7) | 22.8–30.7 | 5 | 320 | 73.6 |
| Type 2 diabetes | 244 | 31.5 ± 7.1 (29.9–38.6) | 29.1–38.6 | 4 | 769 | 75.9 |
| Booking weight (kg) | 6191 | 84.0 ± 23.5 (33.9–193.0) | 71.3–88.6 | 4 | 3953 | 0–99.9 |
| History of hypertension | 296 | 6.7 | 1.9–12.9 | 2 | 5739 | 0–100 |
| Smoking | 566 | 9.1 | 2.3–15.1 | 2 | 3919 | 0–100 |
| Variables | Mean ± SD or N/Total Available Data (%)@ | Range Across Sites | Number of Sites Missing 100% Data | Missing Across Sites Reporting N/(%) † |
|---|---|---|---|---|
| Gestation at first review (weeks) | 24.8 ± 7.2 | 22.1–26.7 *** | 4 | 6195 (71.2) |
| Diet Only | 2082/5278 (39.4) | 28.8–57.3 ** | 1 | 3418 (39.3) |
| Metformin use only | 991/5278 (18.8) | 0.4–43.7 ** | 1 | 3418 (39.3) |
| Insulin use only | 1671/5278 (31.7) | 4.1–95.6 ** | 1 | 3418 (39.3) |
Cesarean delivery:
| 1030 (12.1) 707 (9.5) 1771 (20.3) | 3.6–23.7 *** 3.5–21.2 *** 12.3–42.6 *** | 2 2 0 | 195 (2.2) 1225 (14.1) 0 |
| Gestational age at delivery (weeks) | 38.6 ± 1.5 | 38.2–38.9 *** | 0 | 174 (2.0) |
| Late preterm (34–37 weeks) | 651/8696 (7.5) | 3.7–9.4 * | 0 | 174 (2.0) |
| Very preterm/moderately preterm (<34 weeks) | 128/8696 (1.5) | 0.2–2.2 * | 0 | 174 (2.0) |
| Birth weight ≥4.0 kg ≤2.5 kg | 819/8526 (9.6) 509/8526 (6.0) | 5.9–13.9 *** 1.9–6.8 # | 0 0 | 170 (2.0) 170 (2.0) |
| LGA | 754/5759 (13.1) | 10.3–26.7 *** | 2 | 2937 (33.8) |
| SGA | 538/5759 (9.3) | 0–11.9 *** | 2 | 2937 (33.8) |
| Preeclampsia | 110/3590 (3.1) | 2.0–5.2 *** | 2 | 5106 (58.7) |
| Shoulder dystocia | 92/3321 (2.8) | 0.0–7.2 *** | 4 | 4629 (53.2) |
| Admission to SCN/NICU | 710/3347 (21.2) | 16.7–25.0 *** | 3 | 5378 (61.8) |
| Neonatal hypoglycaemia <2.6 mmol/L | 690/4079 (16.9) | 6.0–27.0 *** | 3 | 4599 (52.9) |
| Jaundice needing phototherapy | 122/3261 (4.9) | 4.7–5.3 # | 5 | 5435 (62.5) |
| Minor congenital malformations Major congenital malformations | 22/3321 (0.7) 61/3321 (1.8) | 0.0–1.2 # 0.9–3.4 ** | 4 4 | 4616 (53.1) 4629 (53.2) |
| Breast feeding on discharge | 795/1273(62.5) | 57.4–62.7 # | 6 | 7279 (83.7) |
| Variables | Type 1 Diabetes N = 435 | Type 2 Diabetes N = 1013 | ||||||
|---|---|---|---|---|---|---|---|---|
| N/(%) Mean ± SD or %@ | Range Across Sites | Number of Sites Missing 100% Data | Missing Values Across Sites Reporting N/(%) † | N/(%) Mean ± SD or %@ | Range | Number of Sites Missing 100% Data | Missing Values Across Sites Reporting N/(%) † | |
| Gestation at first review (weeks) | 9.1 ± 5.3 | 3–27 *** | 4 | 343 (78.9) | 11.1 ± 6.1 | 3.5–33 * | 5 | 862 (85.1) |
| Pregnancy planning | 65/101 (64.4) | 14.3–87.5 *** | 3 | 339 (77.9) | 97/207 (46.9) | 20.0–63.2 * | 4 | 812 (80.2) |
| OHA/GLP1RA on conception | 1/94 (1.1) | -- | 4 | 342 (78.6) | 100/210 (47.6) | 20.0–58.2 ** | 5 | 824 (81.3) |
| Statin at conception | 0/93 (0.0) | -- | 4 | 342 (78.6) | 1/152 (0.7) | -- | 5 | 862 (85.1) |
| ACEI/ARB at conception | 1/93 (1.1) | -- | 4 | 342 (78.6) | 1/152 (0.7) | -- | 5 | 863 (85.2) |
| First trimester Folate (5mg) | 92/101 (91.1) | 28.6–100 *** | 3 | 335 (77.0) | 178/207 (86.0) | 57.9–100 *** | 4 | 810 (80.0) |
| Retinopathy | 77/428 (18.0) | 6.3–39.6 ** | 1 | 49 (11.3) | 16/673 (2.4) | 0.7–7.9 * | 2 | 439 (43.3) |
| Nephropathy | 21/428 (4.9) | 3.2–6.3 # | 1 | 40 (9.2) | 95/991 (9.6) | 5.3–20.0 *** | 1 | 97 (9.6) |
| Urine ACR | 145 (33.3) | 26.4–86.8 # | 290 (66.7) | 196 (19.3) # | 9.3–94.7 | 817 (80.7) | ||
| Aspirin use in pregnancy | 215/428 (50.2) | (13.2–59.0) *** | 1 | 343 (78.9) | 178/673 (26.4) | (0–55.6) *** | 2 | 862 (85.1) |
| Hypoglycaemia needing assistance | 0/45 (0.0) | -- | 6 | 390 (89.7) | 1/24 (4.2) | -- | 7 | 989 (97.6) |
| HbA1c 1st trimester | 7.7 ± 1.6 | 5–16.5 ** | 2 | 46 (10.6) | 7.3 ± 1.7 | 4.2–14.0 * | 3 | 468 (46.2) |
| HbA1c 2nd trimester | 6.6 ± 1.1 | 4.5–11.1 ** | 1 | 321 (73.8) | 6.1 ± 1.1 | 4.3–10.4 * | 2 | 821 (81.0) |
| HbA1c 3rd trimester | 6.8 ± 1.0 | 4.8–11.0 * | 1 | 44 (10.1) | 6.3 ± 1.1 | 4.0–12.6 ** | 2 | 411 (40.6) |
| HbA1c 1st trimester >6.5% (48mmol/mol) | 78.4 | 59.6–100 * | 2 | 46 (10.6) | 54.6 | 0–58.3 ** | 3 | 469 (46.3) |
| HbA1c 2nd trimester >6.0% (42 mmol/mol) | 69.9 | 50.0–82.4 # | 1 | 322 (74.0) | 41.7 | 5.9–62.5 # | 2 | 821 (81.0) |
| HbA1c 3rd trimester >6.0% (42 mmol/mol) | 77.2 | 50.0–88.6 # | 1 | 45 (10.3) | 48.5 | 12.5–100 * | 2 | 411 (40.6 |
| Diet Only | -- | -- | -- | -- | 3/568 (0.5) | 0–0.5 | 1 | 445 (43.9) |
| Metformin use only | -- | -- | -- | -- | 158/568 (27.8) | 0–100 | 1 | 445 (43.9) |
| Insulin use only | -- | -- | -- | -- | 132/568 (23.2) | 0–100 | 1 | 445 (43.9) |
| Metformin and Insulin use | -- | -- | -- | -- | 275/568 (48.4) | 0–79.9 | 1 | 445 (43.9) |
| Insulin pump | 49/134 (36.6) | 0–68.8 | 1 | 301 (69.2) | -- | -- | - | -- |
| Cesarean delivery: (a) Elective (b) Emergency | 50/137 (36.5) 36/87 (41.4) | 12.5–45.8 # 17.4–57.7 ** | 1 1 | 298 (68.5) 348 (80.0) | 97/574 (16.9) 49/477 (10.3) | 0–73.7 *** 0–48.3 *** | 1 1 | 439 (43.3) 536 (52.9) |
| Gestational age at delivery (weeks) | 36.4 ± 1.7 | 28.1–40.2* | 0 | 1 (0.2) | 37.3 ± 1.8 | 27.3–41.3 *** | 0 | 12 (1.2) |
| Late preterm (34–37 weeks) | 208/435 (47.9) | 7.5–52.1 * | 0 | 1 (0.2) | 227/1013 (22.4) | 13.6–31.6 * | 0 | 12 (1.2) |
| Very preterm/moderately preterm ( < 34 weeks) | 36/435 (8.3) | 0.5–28.6 # | 0 | 1 (0.2) | 58/1013 (5.7) | 4.5–7.3 # | 0 | 12 (1.2) |
| Birth weight ≥ 4.0 kg ≤ 2.5 kg | 103/432 (23.8) 37/432 (8.6) | 14.3–37.5 # 2.1–14.3 # | 0 0 | 3 (0.7) 3 (0.7) | 157/1003 (15.7) 101/1003 (10.1) | 0–23.6 ** 5.3–15.8 # | 0 0 | 10 (1.0) 10 (1.0) |
| LGA | 70/112 (62.5) | 20.0–85.7 # | 1 | 323 (74.3) | 58/225 (25.8) | 15.8–33.3 # | 2 | 788 (77.8) |
| SGA | 5/112 (4.5) | 0–16.7 # | 1 | 323 (74.3) | 29/225 (12.9) | 0–20.0 # | 2 | 788 (77.8) |
| Preeclampsia | 76/425 (17.9) | 14.9–28.6# | 0 | 10 (2.3) | 74/681 (10.9) | 5.0–33.3* | 1 | 332 (32.8) |
| Shoulder dystocia | 2/102 (2.0) | 0–3.1 # | 2 | 336 (77.2) | 1/158 (0.6) | - | 3 | 860 (84.9) |
| Admission to SCN/NICU | 279/428 (65.2) | 12.5–100 *** | 1 | 14 (3.2) | 273/676 (40.4) | 21.8–50.0 * | 2 | 362 (35.7) |
| Neonatal hypoglycaemia <2.6 mmol/L | 232/435 (53.3) | 12.5–87.5 *** | 0 | 24 (5.5) | 227/695 (32.7) | 10.5–52.6 ** | 1 | 390 (38.5) |
| Jaundice needing phototherapy | 44/140 (31.4) | 12.5–51.3 ** | 1 | 301 (69.2) | 54/267 (20.2) | 5.3–40.0 # | 2 | 770 (76.0) |
| Minor congenital malformations Major congenital malformations | 5/94 (5.3) 3/94 (3.2) | 6.0–28.6 * 2.1–16.7 # | 3 3 | 344 (79.1) 343 (78.9) | 9/103 (8.7) 2/103 (1.9) | 0–15.8 # 0–5.3 # | 4 4 | 918 (90.6) 918 (90.6) |
| Breast feeding on discharge | 98/125(78.4) | 57.1–93.8 * | 3 | 313 (72.0) | 141/190 (74.2) | 20.0–89.5 * | 4 | 831 (82.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Immanuel, J.; Flack, J.; Wong, V.W.; Yuen, L.; Eagleton, C.; Graham, D.; Lagstrom, J.; Wolmarans, L.; Martin, M.; Cheung, N.W.; et al. The ADIPS Pilot National Diabetes in Pregnancy Benchmarking Programme. Int. J. Environ. Res. Public Health 2021, 18, 4899. https://doi.org/10.3390/ijerph18094899
Immanuel J, Flack J, Wong VW, Yuen L, Eagleton C, Graham D, Lagstrom J, Wolmarans L, Martin M, Cheung NW, et al. The ADIPS Pilot National Diabetes in Pregnancy Benchmarking Programme. International Journal of Environmental Research and Public Health. 2021; 18(9):4899. https://doi.org/10.3390/ijerph18094899
Chicago/Turabian StyleImmanuel, Jincy, Jeff Flack, Vincent W Wong, Lili Yuen, Carl Eagleton, Dorothy Graham, Janet Lagstrom, Louise Wolmarans, Michele Martin, Ngai Wah Cheung, and et al. 2021. "The ADIPS Pilot National Diabetes in Pregnancy Benchmarking Programme" International Journal of Environmental Research and Public Health 18, no. 9: 4899. https://doi.org/10.3390/ijerph18094899
APA StyleImmanuel, J., Flack, J., Wong, V. W., Yuen, L., Eagleton, C., Graham, D., Lagstrom, J., Wolmarans, L., Martin, M., Cheung, N. W., Padmanabhan, S., Rudland, V., Ross, G., Moses, R. G., Maple-Brown, L., Fulcher, I., Chemmanam, J., Nolan, C. J., Oats, J. J. N., ... Simmons, D. (2021). The ADIPS Pilot National Diabetes in Pregnancy Benchmarking Programme. International Journal of Environmental Research and Public Health, 18(9), 4899. https://doi.org/10.3390/ijerph18094899







