Association between Diabetes Mellitus and Oral Health Status in Patients with Cardiovascular Diseases: A Nationwide Population-Based Study
Abstract
1. Introduction
2. Materials and Methods
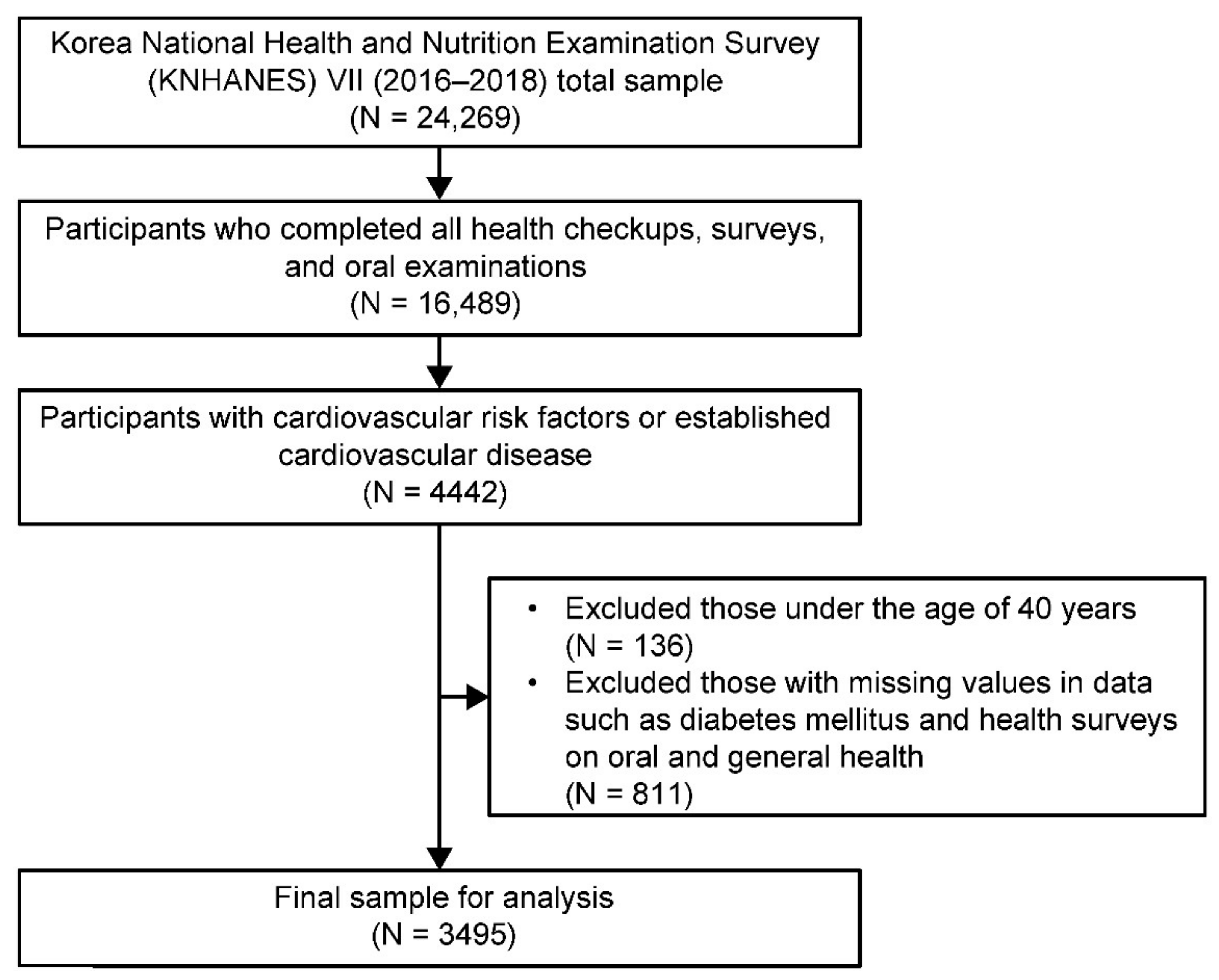
2.1. Design, Sample, and Setting
2.2. Measurement
2.2.1. Diabetes Mellitus
2.2.2. Oral Health Status
2.2.3. Other Potential Confounding Variables
2.3. Data Analysis
2.4. Ethical Considerations and Data Collection
3. Results
3.1. General Characteristics of People with and without DM Combined with CVD
3.2. Clinical Characteristics of People with and without DM Combined with CVD
3.3. Oral Health Status of People with and without DM with CVD
3.4. Logistic Regression Analysis of the Association between DM and Oral Health Status in CVD
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- GBD 2017 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1859–1922. [Google Scholar] [CrossRef]
- Mensah, G.A.; Roth, G.A.; Fuster, V. The global burden of cardiovascular diseases and risk factors: 2020 and beyond. J. Am. Coll. Cardiol. 2019, 74, 2529–2532. [Google Scholar] [CrossRef] [PubMed]
- Einarson, T.R.; Acs, A.; Ludwig, C.; Panton, U.H. Prevalence of cardiovascular disease in type 2 diabetes: A systematic literature review of scientific evidence from across the world in 2007–2017. Cardiovasc. Diabetol. 2018, 17, 83. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Simon, B.; Shi, J.; Mallhi, A.K.; Eisen, H.J. Impact of diabetes mellitus on risk of cardiovascular disease and all-cause mortality: Evidence on health outcomes and antidiabetic treatment in United States adults. World J. Diabetes 2016, 7, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Majumder, M.A.A.; Kabir, R.; Haque, M.; Gupta, S.; Arafat, S.M.Y.; Ojeh, N.; Dalvi, P. Cardiovascular disease and diabetes: Two sides of the same coin! In Recent Trends in Cardiovascular Risks; Kumar, A., Ed.; InTech Open: Croatia, Balkans, 2017; pp. 71–95. [Google Scholar]
- Liccardo, D.; Cannavo, A.; Spagnuolo, G.; Ferrara, N.; Cittadini, A.; Rengo, C.; Rengo, G. Periodontal disease: A risk factor for diabetes and cardiovascular disease. Int. J. Mol. Sci. 2019, 20, 1414. [Google Scholar] [CrossRef] [PubMed]
- Pedroso, J.F.; Lotfollahi, Z.; Albattarni, G.; Schulz, M.A.; Monteiro, A.; Sehnem, A.L.; Gidlund, M.A.; Neto, A.M.F.; Jardini, M.A.N. Influence of periodontal disease on cardiovascular markers in diabetes mellitus patients. Sci. Rep. 2019, 9, 16138. [Google Scholar] [CrossRef] [PubMed]
- Kozarov, E.; Sweier, D.; Shelburne, C.; Proguske-Fox, A.; Lopatin, D. Detection of bacterial DNA in atheromatous plaques by quantitative PCR. Microbes Infect. 2006, 8, 687–693. [Google Scholar] [CrossRef]
- Louhelainen, A.M.; Aho, J.; Tuomisto, S.; Aittoniemi, J.; Vuento, R.; Karhunen, P.J.; Pessi, T. Oral bacterial DNA findings in pericardial fluid. J. Oral Microbiol. 2014, 6, 25835. [Google Scholar] [CrossRef] [PubMed]
- Pessi, T.; Karhunen, V.; Karjalainen, P.P.; Ylitalo, A.; Airaksinen, J.K.; Niemi, M.; Pietila, M.; Lounatmaa, K.; Haapaniemi, T.; Lehtimäki, T.; et al. Bacterial signatures in thrombus aspirates of patients with myocardial infarction. Circulation 2013, 127, 1219–1228. [Google Scholar] [CrossRef]
- Carrizales-Sepulveda, E.F.; Ordaz-Farias, A.; Vera-Pineda, R.; Flores-Ramirez, R. Periodontal disease, systemic inflammation and the risk of cardiovascular disease. Heart Lung Circ. 2018, 27, 1327–1334. [Google Scholar] [CrossRef]
- Parakh, M.K.; Kasi, A.; Ayyappan, V.; Subramani, P. Knowledge and awareness of oral manifestations of diabetes mellitus and oral health assessment among diabetes mellitus patients-a cross sectional study. Curr. Diabetes Rev. 2020, 16, 156–164. [Google Scholar] [CrossRef]
- Poudel, P.; Griffiths, R.; Wong, V.W.; Arora, A.; Flack, J.R.; Khoo, C.L.; George, A. Oral health knowledge, attitudes and care practices of people with diabetes: A systematic review. BMC Public Health 2018, 18, 577. [Google Scholar] [CrossRef]
- Lee, C.Y.; Kuan, Y.H.; Tsai, Y.F.; Tai, C.J.; Tsai, T.H.; Huang, K.H. Correlation between diabetes mellitus and periodontitis in Taiwan: A nationwide cohort study. Diabetes Res. Clin. Pract. 2019, 150, 245–252. [Google Scholar] [CrossRef]
- Petrie, J.R.; Guzik, T.J.; Touyz, R.M. Diabetes, hypertension, and cardiovascular disease: Clinical insights and vascular mechanisms. Can. J. Cardiol. 2018, 34, 575–584. [Google Scholar] [CrossRef]
- Loe, H. Periodontal disease. The sixth complication of diabetes mellitus. Diabetes Care 1993, 16, 329–334. [Google Scholar] [CrossRef]
- Rawal, I.; Ghosh, S.; Hameed, S.S.; Shivashankar, R.; Ajay, V.S.; Patel, S.A.; Goodman, M.; Ali, M.K.; Narayan, K.M.V.; Tandon, N.; et al. Association between poor oral health and diabetes among Indian adult population: Potential for integration with NCDs. BMC Oral Health 2019, 19, 191. [Google Scholar] [CrossRef]
- Stewart, R.; West, M. Increasing evidence for an association between periodontitis and cardiovascular disease. Circulation 2016, 133, 549–551. [Google Scholar] [CrossRef]
- Hong, M.; Kim, H.Y.; Seok, H.; Yeo, C.D.; Kim, Y.S.; Song, J.Y.; Lee, Y.B.; Lee, D.H.; Lee, J.I.; Lee, T.K.; et al. Prevalence and risk factors of periodontitis among adults with or without diabetes mellitus. Korean J. Intern. Med. 2016, 31, 910–919. [Google Scholar] [CrossRef]
- Schenkein, H.A.; Loos, B.G. Inflammatory mechanisms linking periodontal diseases to cardiovascular diseases. J. Clin. Periodontol. 2013, 40 (Suppl. 14), S51–S69. [Google Scholar] [CrossRef]
- Rydén, L.; Buhlin, K.; Ekstrand, E.; de Faire, U.; Gustafsson, A.; Holmer, J.; Kjellström, B.; Lindahl, B.; Norhammar, A.; Nygren, Å.; et al. Periodontitis increases the risk of a first myocardial infarction: A report from the PAROKRANK study. Circulation 2016, 133, 576–583. [Google Scholar] [CrossRef]
- Aoyama, N.; Suzuki, J.I.; Kobayashi, N.; Hanatani, T.; Ashigaki, N.; Yoshida, A.; Shiheido, Y.; Sato, H.; Kumagai, H.; Ikeda, Y.; et al. Japanese cardiovascular disease patients with diabetes mellitus suffer increased tooth loss in comparison to those without diabetes mellitus -a cross-sectional study. Intern. Med. 2018, 57, 777–782. [Google Scholar] [CrossRef]
- Han, S.J.; Yi, Y.J.; Bae, K.H. The association between periodontitis and dyslipidemia according to smoking and harmful alcohol use in a representative sample of Korean adults. Clin. Oral Investig. 2020, 24, 2937–2944. [Google Scholar] [CrossRef]
- Kim, Y.T.; Choi, J.K.; Kim, D.H.; Jeong, S.N.; Lee, J.H. Association between health status and tooth loss in Korean adults: Longitudinal results from the National Health Insurance Service-Health Examinee Cohort, 2002–2015. J. Periodontal Implant Sci. 2019, 49, 158–170. [Google Scholar] [CrossRef]
- Korea Centers for Disease Control and Prevention (KCDC). The Seventh Korea National Health and Nutrition Examination Survey (KNHANES VII). 2020. Available online: http://knhanes.cdc.go.kr (accessed on 8 January 2020).
- Korea Centers for Disease Control and Prevention (KCDC). Guidelines of the Seventh Korea National Health and Nutrition Examination Survey VII (2016–2018). 2016. Available online: http://knhanes.cdc.go.kr (accessed on 8 January 2020).
- Dalal, J.J.; Padmanabhan, T.N.; Jain, P.; Patil, S.; Vasnawala, H.; Gulati, A. LIPITENSION: Interplay between dyslipidemia and hypertension. Indian J. Endocrinol. Metabol. 2012, 16, 240–245. [Google Scholar] [CrossRef]
- Salehidoost, R.; Mansouri, A.; Amini, M.; Aminorroaya, Y.S.; Aminorroaya, A. Diabetes and all-cause mortality, a 18-year follow-up study. Sci. Rep. 2020, 10, 3183. [Google Scholar] [CrossRef]
- Rhee, E.J. Prevalence and current management of cardiovascular risk factors in Korean adults based on fact sheets. Endocrinol. Metab. 2020, 35, 85–94. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Oral Health Surveys: Basic Methods, 4th ed.; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Buchwald, S.; Kocher, T.; Biffar, R.; Harb, A.; Holtfreter, B.; Meisel, P. Tooth loss and periodontitis by socio-economic status and inflammation in a longitudinal population-based study. J. Clin. Periodontol. 2013, 40, 203–211. [Google Scholar] [CrossRef]
- Kim, S.; Jung, A. Optimum cutoff value of urinary cotinine distinguishing South Korean adult smokers from nonsmokers using data from the KNHANES (2008–2010). Nicotine Tob. Res. 2013, 15, 1608–1616. [Google Scholar] [CrossRef]
- Martinez-Herrera, M.; Silvestre-Rangil, J.; Silvestre, F.J. Association between obesity and periodontal disease. A systematic review of epidemiological studies and controlled clinical trials. Med. Oral Patol. Oral Cir. Bucal. 2017, 22, e708–e715. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Regional Office for the Western Pacific. In The Asia-Pacific Perspective: Redefining Obesity and its Treatment; Health Communications Australia: Sydney, Australia, 2000. [Google Scholar]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.G. Statistical power analyses using G*Power 3.1: Tests for correlation and regression analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, Y.; Nickel, J.C.; Iwasaki, L.R.; Duan, P.; Simmer-Beck, M.; Brown, L. Gender differences in the association of periodontitis and type 2 diabetes. Int. Dent. J. 2018, 68, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, T.; Scarbro, A.; Bryant, L.L.; Puma, J. Factors associated with tooth loss in older adults in rural Colorado. J. Community Health 2016, 41, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Natto, Z.S.; Aladmawy, M.; Alasqah, M.; Papas, A. Factors contributing to tooth loss among the elderly: A cross sectional study. Singapore Dent. J. 2014, 35, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Kingman, A.; Albandar, J.M. Methodological aspects of epidemiological studies of periodontal diseases. Periodontology 2002, 29, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, T.; Kitamura, M.; Kawashita, Y.; Saito, T. Periodontal disease and percentage of calories from fat using national data. J. Periodontal. Res. 2017, 52, 114–121. [Google Scholar] [CrossRef]
- Han, S.J.; Bae, K.H.; Lee, H.J.; Kim, S.J.; Cho, H.J. Association between regular walking and periodontitis according to socioeconomic status: A cross-sectional study. Sci. Rep. 2019, 9, 12969. [Google Scholar] [CrossRef]
- Lee, S.W.; Lim, H.J.; Lee, E. Association between asthma and periodontitis: Results from the Korean National Health and Nutrition Examination Survey. J. Periodontol. 2017, 88, 575–581. [Google Scholar] [CrossRef] [PubMed]
| Variables | Total | Non-DM Group | DM Group | p-Value |
|---|---|---|---|---|
| n = 3495 | n = 2648 | n = 847 | ||
| Socio-demographics | ||||
| Age (years), n (%) | <0.001 # | |||
| 40–49 | 320 (8.9) | 268 (9.7) | 52 (6.1) | |
| 50–59 | 851 (25.1) | 694 (26.8) | 157 (19.3) | |
| 60–69 | 1127 (32.2) | 850 (31.9) | 277 (33.1) | |
| 70–79 | 937 (26.6) | 644 (24.4) | 293 (33.5) | |
| ≥80 | 260 (7.3) | 192 (7.1) | 68 (8.1) | |
| Sex, n (%) | 0.026 # | |||
| Male | 1556 (42.9) | 1148 (41.7) | 408 (47.0) | |
| Female | 1939 (57.1) | 1500 (58.3) | 439 (53.0) | |
| Living status, n (%) | <0.001 # | |||
| Alone | 647 (15.4) | 456 (14.1) | 191 (20.0) | |
| Together | 2848 (84.6) | 2192 (85.9) | 656 (80.0) | |
| Education, n (%) | <0.001 # | |||
| ≤Elementary | 1375 (38.8) | 997 (37.1) | 378 (44.6) | |
| Middle-high school | 1457 (41.6) | 1099 (41.6) | 358 (41.4) | |
| ≥College | 663 (19.6) | 552 (21.3) | 111 (14.1) | |
| Job, n (%) | 0.014 # | |||
| Yes | 1773 (49.1) | 1383 (50.5) | 390 (44.8) | |
| No | 1722 (50.9) | 1265 (49.5) | 457 (55.2) | |
| Household income a, n (%) | <0.001 # | |||
| <25% | 1113 (31.1) | 778 (28.7) | 335 (38.8) | |
| 25–49% | 913 (25.9) | 703 (26.2) | 210 (25.0) | |
| 50–74% | 732 (21.2) | 558 (21.5) | 174 (20.4) | |
| ≥75% | 737 (21.7) | 609 (23.6) | 128 (15.8) | |
| General health behaviors | ||||
| Smoking status, n (%) | 0.067 # | |||
| Non-smoker/past smoker | 2973 (85.6) | 2268 (86.3) | 705 (83.4) | |
| Current smoker | 522 (14.4) | 380 (13.7) | 142 (16.6) | |
| Drinking status, n (%) | 0.019 # | |||
| Nondrinker | 604 (16.8) | 432 (15.9) | 172 (19.8) | |
| Drinker | 2891 (83.2) | 2216 (84.1) | 675 (80.2) | |
| Subjective health status, n (%) | <0.001 # | |||
| Good | 635 (18.3) | 517 (19.7) | 118 (13.7) | |
| Moderate | 1795 (52.2) | 1396 (53.5) | 399 (48.2) | |
| Bad | 1065 (29.4) | 735 (26.8) | 330 (38.1) | |
| Oral health care | ||||
| Tooth-brushing b, n (%) | <0.001 # | |||
| ≤1 | 499 (14.0) | 349 (12.8) | 150 (17.7) | |
| 2 | 1503 (42.4) | 1113 (41.1) | 390 (46.5) | |
| ≥3 | 1493 (43.6) | 1186 (46.0) | 307 (35.8) | |
| Usage of floss or interdental brush, n (%) | 0.068 # | |||
| No | 2639 (74.9) | 1966 (74.0) | 673 (77.8) | |
| Yes | 856 (25.1) | 682 (26.0) | 174 (22.2) | |
| Dental clinic visit, n (%) | 0.037 # | |||
| No | 1329 (38.3) | 986 (37.3) | 343 (41.6) | |
| Yes | 2166 (61.7) | 1662 (62.7) | 504 (58.4) | |
| Dental checkup, n (%) | <0.001 # | |||
| No | 2325 (66.3) | 1713 (64.3) | 612 (72.9) | |
| Yes | 1170 (33.7) | 935 (35.7) | 235 (27.1) | |
| Variables | Total | Non-DM Group | DM Group | p -Value |
|---|---|---|---|---|
| n = 3495 | n = 2648 | n = 847 | ||
| Hypertension, n(%) | 0.001 # | |||
| No | 958 (28.8) | 777 (30.5) | 181 (23.1) | |
| Yes | 2537 (71.2) | 1871 (69.5) | 666 (76.9) | |
| Dyslipidemia, n (%) | <0.001 # | |||
| No | 1558 (43.1) | 1225 (45.1) | 333 (36.4) | |
| Yes | 1937 (56.9) | 1423 (54.9) | 514 (63.6) | |
| Stroke, n (%) | 0.023 # | |||
| No | 3260 (93.4) | 2487 (94.0) | 773 (91.4) | |
| Yes | 235 (6.6) | 161 (6.0) | 74 (8.6) | |
| Myocardial infarction, n (%) | 0.012 # | |||
| No | 3381 (97.0) | 2571 (97.4) | 810 (95.5) | |
| Yes | 114 (3.0) | 77 (2.6) | 37 (4.5) | |
| Renal failure, n (%) | 0.016 # | |||
| No | 3473 (99.5) | 2636 (99.7) | 837 (99.1) | |
| Yes | 22 (0.5) | 12 (0.3) | 10 (0.9) | |
| BMI (kg/m2), n (%) | 0.017 # | |||
| <18.5 | 45 (1.4) | 37 (1.4) | 8 (1.2) | |
| 18.5–22.9 | 926 (28.0) | 734 (29.2) | 192 (23.9) | |
| 23.0–24.9 | 918 (26.0) | 702 (26.4) | 216 (24.6) | |
| 25.0–29.9 | 1362 (38.7) | 996 (37.1) | 366 (43.9) | |
| ≥30.0 | 244 (6.0) | 179 (5.9) | 65 (6.4) | |
| Waist circumference (cm) | 85.97 ± 0.21 | 85.30 ± 0.23 | 88.19 ± 0.37 | <0.001 † |
| Systolic blood pressure (mmHg) | 126.53 ± 0.39 | 126.75 ± 0.43 | 125.84 ± 0.72 | 0.245 † |
| Diastolic blood pressure (mmHg) | 75.76 ± 0.23 | 76.78 ± 0.26 | 72.41 ± 0.40 | <0.001 † |
| Total cholesterol (mg/dL) | 184.80 ± 0.80 | 190.55 ± 0.89 | 165.93 ± 1.50 | <0.001 † |
| Triglyceride (mg/dL) | 151.38 ± 2.56 | 149.66 ± 2.93 | 157.02 ± 4.20 | 0.130 † |
| hs-CRP (mg/dL) | 1.29 ± 0.04 | 1.29 ± 0.05 | 1.29 ± 0.08 | 0.981 † |
| Blood urea nitrogen (mg/dL) | 16.44 ± 0.12 | 16.27 ± 0.13 | 16.99 ± 0.21 | 0.002 † |
| Serum creatinine (mg/dL) | 0.85 ± 0.01 | 0.84 ± 0.01 | 0.89 ± 0.02 | 0.001 † |
| Hemoglobin (g/dL) | 13.87 ± 0.03 | 13.92 ± 0.04 | 13.72 ± 0.07 | 0.006 † |
| Variables | Total | Non-DM Group | DM Group | p -Value |
|---|---|---|---|---|
| n = 3495 | n = 2648 | n = 847 | ||
| Periodontitis | <0.001 # | |||
| No | 1877 (54.2) | 1491 (56.8) | 386 (45.7) | |
| Yes | 1618 (45.8) | 1157 (43.2) | 461 (54.3) | |
| Number of remaining teeth | <0.001 # | |||
| ≥20 | 2636 (75.3) | 2054 (77.2) | 582 (69.1) | |
| <20 | 859 (24.7) | 594 (22.8) | 265 (30.9) | |
| Active caries | 0.221 # | |||
| No | 2574 (74.8) | 1961 (75.3) | 613 (72.9) | |
| Yes | 921 (25.2) | 687 (24.7) | 234 (27.1) |
| DM | Periodontitis | Number of Remaining Teeth | Active Caries | |||
|---|---|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | OR (95% CI) | ||||
| Model 1 | Model 2 | Model 1 | Model 2 | Model 1 | Model 2 | |
| Yes | 1.561 ** (1.300–1.874) | 1.406 * (1.160–1.705) | 1.511 ** (1.220–1.858) | 1.184 (0.934–1.510) | 1.136 (0.926–1.393) | 1.082 (0.874–1.341) |
| No | 1 | 1 | 1 | 1 | 1 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, S.-J.; Son, Y.-J.; Kim, B.-H. Association between Diabetes Mellitus and Oral Health Status in Patients with Cardiovascular Diseases: A Nationwide Population-Based Study. Int. J. Environ. Res. Public Health 2021, 18, 4889. https://doi.org/10.3390/ijerph18094889
Han S-J, Son Y-J, Kim B-H. Association between Diabetes Mellitus and Oral Health Status in Patients with Cardiovascular Diseases: A Nationwide Population-Based Study. International Journal of Environmental Research and Public Health. 2021; 18(9):4889. https://doi.org/10.3390/ijerph18094889
Chicago/Turabian StyleHan, Su-Jin, Youn-Jung Son, and Bo-Hwan Kim. 2021. "Association between Diabetes Mellitus and Oral Health Status in Patients with Cardiovascular Diseases: A Nationwide Population-Based Study" International Journal of Environmental Research and Public Health 18, no. 9: 4889. https://doi.org/10.3390/ijerph18094889
APA StyleHan, S.-J., Son, Y.-J., & Kim, B.-H. (2021). Association between Diabetes Mellitus and Oral Health Status in Patients with Cardiovascular Diseases: A Nationwide Population-Based Study. International Journal of Environmental Research and Public Health, 18(9), 4889. https://doi.org/10.3390/ijerph18094889






