An Experimental Approach to Study the Effects of Realistic Environmental Mixture of Linuron and Propamocarb on Zebrafish Synaptogenesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Zebrafish Maintenance and Egg Collection
2.3. Zebrafish Embryos Exposure
2.4. Morphometric Measurements
2.5. RT-PCR
2.6. Statistical Analysis
3. Results
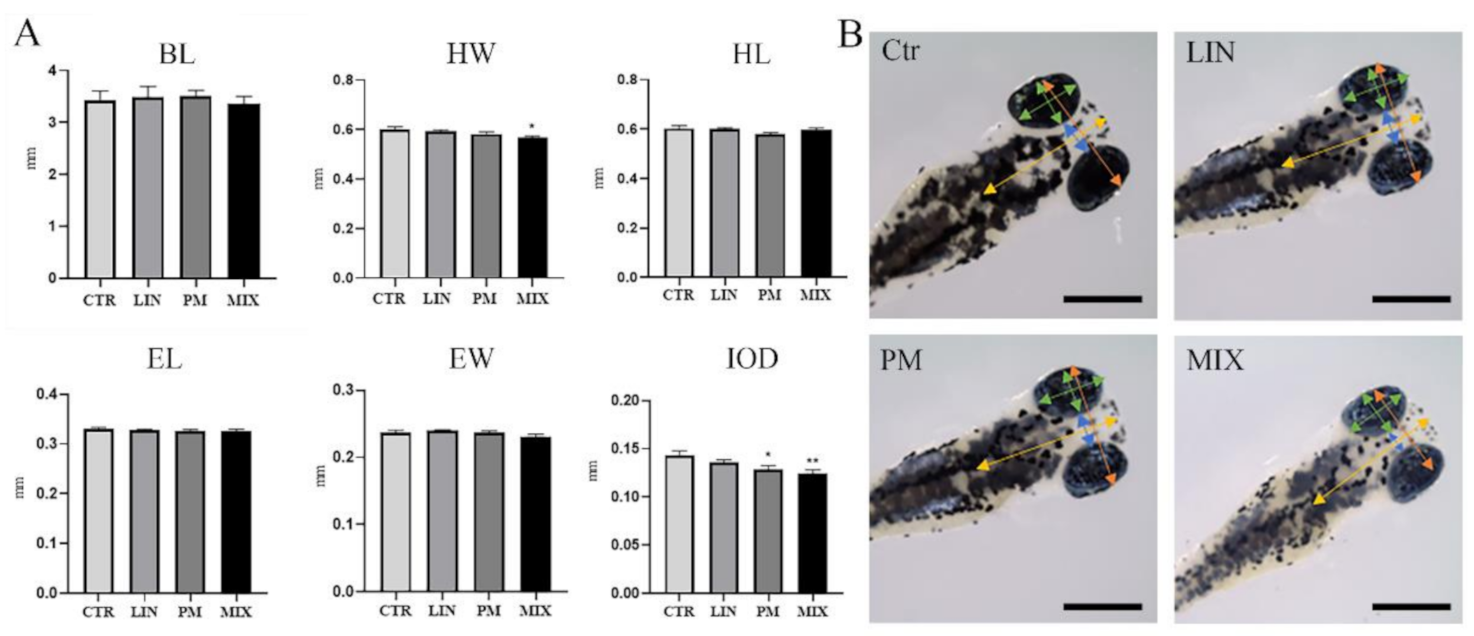
3.1. Morphometric Analyses
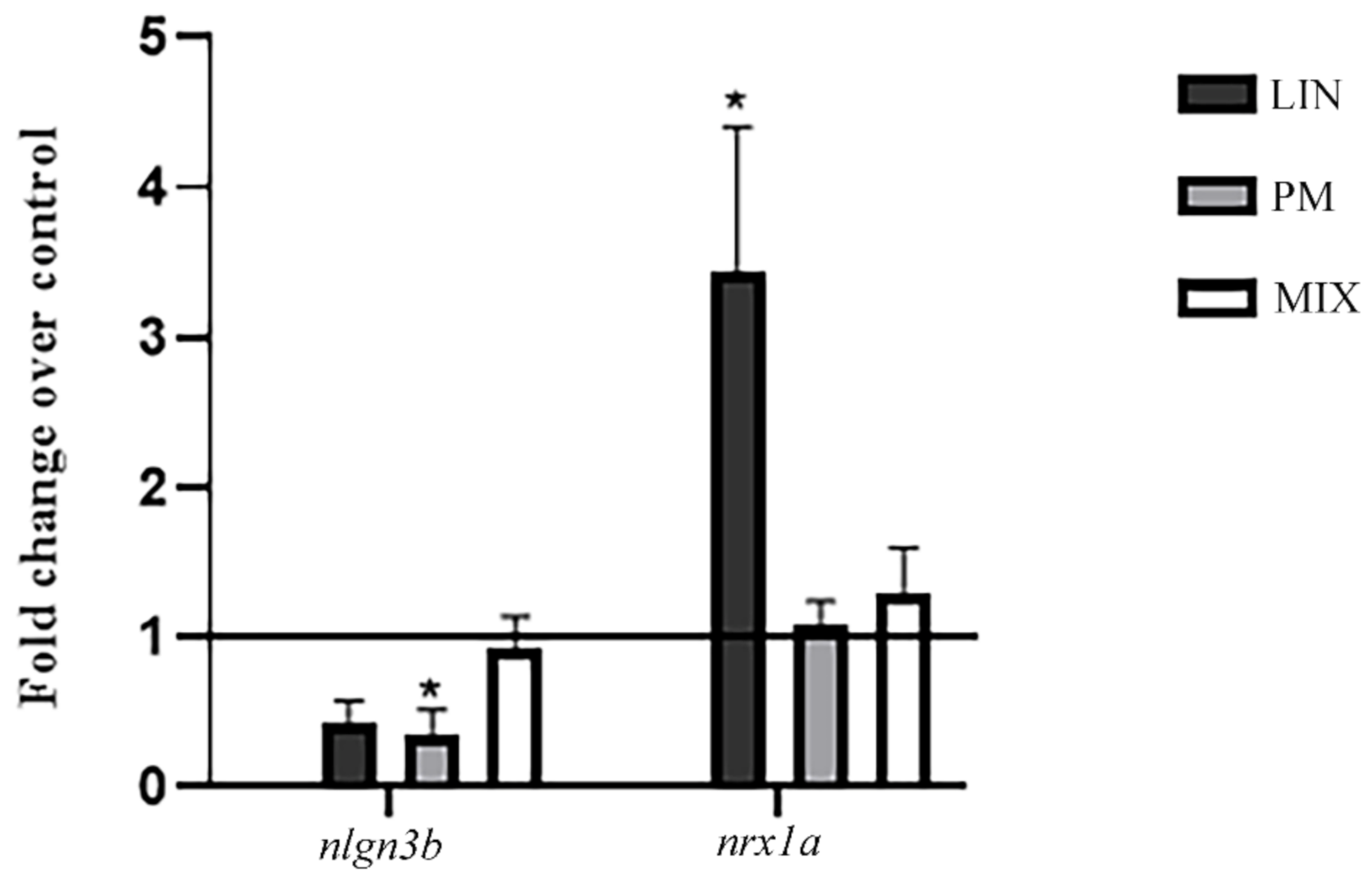
3.2. RT-PCR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Iqubal, A.; Ahmed, M.; Ahmad, S.; Sahoo, C.R.; Iqubal, M.K.; Haque, S.E. Environmental Neurotoxic Pollutants: Review. Environ. Sci. Pollut. Res. 2020, 27, 41175–41198. [Google Scholar] [CrossRef] [PubMed]
- Legradi, J.B.; Di Paolo, C.; Kraak, M.H.S.; van der Geest, H.G.; Schymanski, E.L.; Williams, A.J.; Dingemans, M.M.L.; Massei, R.; Brack, W.; Cousin, X.; et al. An Ecotoxicological View on Neurotoxicity Assessment. Environ. Sci. Eur. 2018, 30, 46. [Google Scholar] [CrossRef] [PubMed]
- Pistollato, F.; de Gyves, E.M.; Carpi, D.; Bopp, S.K.; Nunes, C.; Worth, A.; Bal-Price, A. Assessment of Developmental Neurotoxicity Induced by Chemical Mixtures Using an Adverse Outcome Pathway Concept. Environ. Health 2020, 19, 23. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Landrigan, P.J. Neurobehavioural Effects of Developmental Toxicity. Lancet Neurol. 2014, 13, 330–338. [Google Scholar] [CrossRef]
- Paparella, M.; Bennekou, S.H.; Bal-Price, A. An Analysis of the Limitations and Uncertainties of in Vivo Developmental Neurotoxicity Testing and Assessment to Identify the Potential for Alternative Approaches. Reprod. Toxicol. 2020, 96, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, Y.; Murakami, S.; Ashikawa, Y.; Sasagawa, S.; Umemoto, N.; Shimada, Y.; Tanaka, T. Zebrafish as a Systems Toxicology Model for Developmental Neurotoxicity Testing. Congenit. Anom. 2015, 55, 1–16. [Google Scholar] [CrossRef] [PubMed]
- d’Amora, M.; Giordani, S. The Utility of Zebrafish as a Model for Screening Developmental Neurotoxicity. Front. Neurosci. 2018, 12, 976. [Google Scholar] [CrossRef]
- Costa, L.G.; Giordano, G.; Guizzetti, M.; Vitalone, A. Neurotoxicity of Pesticides: A Brief Review. Front. Biosci 2008, 13, 1240–1249. [Google Scholar] [CrossRef]
- Richardson, J.R.; Fitsanakis, V.; Westerink, R.H.S.; Kanthasamy, A.G. Neurotoxicity of Pesticides. Acta Neuropathol. 2019, 138, 343–362. [Google Scholar] [CrossRef]
- Bjørling-Poulsen, M.; Andersen, H.R.; Grandjean, P. Potential Developmental Neurotoxicity of Pesticides Used in Europe. Environ. Health 2008, 7, 50. [Google Scholar] [CrossRef]
- Sellare, J.; Meemken, E.-M.; Qaim, M. Fairtrade, Agrochemical Input Use, and Effects on Human Health and the Environment. Ecol. Econ. 2020, 176, 106718. [Google Scholar] [CrossRef]
- Elahi, E.; Weijun, C.; Zhang, H.; Abid, M. Use of Artificial Neural Networks to Rescue Agrochemical-Based Health Hazards: A Resource Optimisation Method for Cleaner Crop Production. J. Clean. Prod. 2019, 238, 117900. [Google Scholar] [CrossRef]
- Maharaj, S.; El Ahmadie, N.; Rheingold, S.; El Chehouri, J.; Yang, L.; Souders, C.L.; Martyniuk, C.J. Sub-Lethal Toxicity Assessment of the Phenylurea Herbicide Linuron in Developing Zebrafish (Danio Rerio) Embryo/Larvae. Neurotoxicology Teratol. 2020, 81, 106917. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, R.; Jin, Y. Differential Responses of Larval Zebrafish to the Fungicide Propamocarb: Endpoints at Development, Locomotor Behavior and Oxidative Stress. Sci. Total Environ. 2020, 731, 139136. [Google Scholar] [CrossRef]
- Krueger, D.D.; Tuffy, L.P.; Papadopoulos, T.; Brose, N. The Role of Neurexins and Neuroligins in the Formation, Maturation, and Function of Vertebrate Synapses. Curr. Opin. Neurobiol. 2012, 22, 412–422. [Google Scholar] [CrossRef]
- Kabashi, E.; Brustein, E.; Champagne, N.; Drapeau, P. Zebrafish Models for the Functional Genomics of Neurogenetic Disorders. Biochim. Et Biophys. Acta Mol. Basis Dis. 2011, 1812, 335–345. [Google Scholar] [CrossRef]
- Tromp, A.; Mowry, B.; Giacomotto, J. Neurexins in Autism and Schizophrenia—a Review of Patient Mutations, Mouse Models and Potential Future Directions. Mol. Psychiatry 2021, 26, 747–760. [Google Scholar] [CrossRef]
- Rissone, A.; Monopoli, M.; Beltrame, M.; Bussolino, F.; Cotelli, F.; Arese, M. Comparative Genome Analysis of the Neurexin Gene Family in Danio Rerio: Insights into Their Functions and Evolution. Mol. Biol. Evol. 2007, 24, 236–252. [Google Scholar] [CrossRef][Green Version]
- Quinn, D.P.; Kolar, A.; Wigerius, M.; Gomm-Kolisko, R.N.; Atwi, H.; Fawcett, J.P.; Krueger, S.R. Pan-Neurexin Perturbation Results in Compromised Synapse Stability and a Reduction in Readily Releasable Synaptic Vesicle Pool Size. Sci. Rep. 2017, 7, 42920. [Google Scholar] [CrossRef]
- Ichtchenko, K.; Nguyen, T.; Südhof, T.C. Structures, Alternative Splicing, and Neurexin Binding of Multiple Neuroligins. J. Biol. Chem. 1996, 271, 2676–2682. [Google Scholar] [CrossRef]
- Südhof, T.C. Neuroligins and Neurexins Link Synaptic Function to Cognitive Disease. Nature 2008, 455, 903–911. [Google Scholar] [CrossRef]
- Yan, J.; Oliveira, G.; Coutinho, A.; Yang, C.; Feng, J.; Katz, C.; Sram, J.; Bockholt, A.; Jones, I.R.; Craddock, N.; et al. Analysis of the Neuroligin 3 and 4 Genes in Autism and Other Neuropsychiatric Patients. Mol. Psychiatry 2005, 10, 329–332. [Google Scholar] [CrossRef]
- Chanda, S.; Hale, W.D.; Zhang, B.; Wernig, M.; Südhof, T.C. Unique versus Redundant Functions of Neuroligin Genes in Shaping Excitatory and Inhibitory Synapse Properties. J. Neurosci. 2017, 37, 6816–6836. [Google Scholar] [CrossRef]
- Radyushkin, K.; Hammerschmidt, K.; Boretius, S.; Varoqueaux, F.; El-Kordi, A.; Ronnenberg, A.; Winter, D.; Frahm, J.; Fischer, J.; Brose, N.; et al. Neuroligin-3-Deficient Mice: Model of a Monogenic Heritable Form of Autism with an Olfactory Deficit. Genes Brain Behav. 2009, 8, 416–425. [Google Scholar] [CrossRef]
- Penn, H.E. Neurobiological Correlates of Autism: A Review of Recent Research. Child Neuropsychol. 2006, 12, 57–79. [Google Scholar] [CrossRef]
- Voineskos, A.N.; Lett, T.A.P.; Lerch, J.P.; Tiwari, A.K.; Ameis, S.H.; Rajji, T.K.; Müller, D.J.; Mulsant, B.H.; Kennedy, J.L. Neurexin-1 and Frontal Lobe White Matter: An Overlapping Intermediate Phenotype for Schizophrenia and Autism Spectrum Disorders. PLoS ONE 2011, 6, e20982. [Google Scholar] [CrossRef]
- Vester, A.; Caudle, W.M. The Synapse as a Central Target for Neurodevelopmental Susceptibility to Pesticides. Toxics 2016, 4, 18. [Google Scholar] [CrossRef]
- Quintaneiro, C.; Patrício, D.; Novais, S.C.; Soares, A.M.V.M.; Monteiro, M.S. Endocrine and Physiological Effects of Linuron and S-Metolachlor in Zebrafish Developing Embryos. Sci. Total Environ. 2017, 586, 390–400. [Google Scholar] [CrossRef]
- Webster, T.M.U.; Perry, M.H.; Santos, E.M. The Herbicide Linuron Inhibits Cholesterol Biosynthesis and Induces Cellular Stress Responses in Brown Trout. Environ. Sci. Technol. 2015, 49, 3110–3118. [Google Scholar] [CrossRef]
- Schuler, L.J.; Rand, G.M. Aquatic Risk Assessment of Herbicides in Freshwater Ecosystems of South Florida. Arch. Environ. Contam. Toxicol. 2008, 54, 571–583. [Google Scholar] [CrossRef] [PubMed]
- Palma, P.; Kuster, M.; Alvarenga, P.; Palma, V.L.; Fernandes, R.M.; Soares, A.M.V.M.; de Alda, M.J.L.; Barceló, D.; Barbosa, I.R. Risk Assessment of Representative and Priority Pesticides, in Surface Water of the Alqueva Reservoir (South of Portugal) Using on-Line Solid Phase Extraction-Liquid Chromatography-Tandem Mass Spectrometry. Environ. Int. 2009, 35, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Tinwell, H.; Friry-Santini, C.; Rouquié, D.; Belluco, S.; Elies, L.; Pallen, C.; Bars, R. Evaluation of the Antiandrogenic Effects of Flutamide, DDE, and Linuron in the Weanling Rat Assay Using Organ Weight, Histopathological, and Proteomic Approaches. Toxicol. Sci. 2007, 100, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Ornostay, A.; Cowie, A.M.; Hindle, M.; Baker, C.J.O.; Martyniuk, C.J. Classifying Chemical Mode of Action Using Gene Networks and Machine Learning: A Case Study with the Herbicide Linuron. Comp. Biochem. Physiol. Part D Genom. Proteom. 2013, 8, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Pan, Z.; Wang, X.; Shen, M.; Zhou, J.; Fu, Z.; Jin, Y. Short-Term Propamocarb Exposure Induces Hepatic Metabolism Disorder Associated with Gut Microbiota Dysbiosis in Adult Male Zebrafish. Acta Biochim. Et Biophys. Sin. 2019, 51, 88–96. [Google Scholar] [CrossRef]
- Wu, S.; Jin, C.; Wang, Y.; Fu, Z.; Jin, Y. Exposure to the Fungicide Propamocarb Causes Gut Microbiota Dysbiosis and Metabolic Disorder in Mice. Environ. Pollut. 2018, 237, 775–783. [Google Scholar] [CrossRef]
- Wu, S.; Luo, T.; Wang, S.; Zhou, J.; Ni, Y.; Fu, Z.; Jin, Y. Chronic Exposure to Fungicide Propamocarb Induces Bile Acid Metabolic Disorder and Increases Trimethylamine in C57BL/6J Mice. Sci. Total Environ. 2018, 642, 341–348. [Google Scholar] [CrossRef]
- Martínez, R.; Herrero-Nogareda, L.; Van Antro, M.; Campos, M.P.; Casado, M.; Barata, C.; Piña, B.; Navarro-Martín, L. Morphometric Signatures of Exposure to Endocrine Disrupting Chemicals in Zebrafish Eleutheroembryos. Aquat. Toxicol. 2019, 214, 105232. [Google Scholar] [CrossRef]
- Berkowitz, G.S.; Wetmur, J.G.; Birman-Deych, E.; Obel, J.; Lapinski, R.H.; Godbold, J.H.; Holzman, I.R.; Wolff, M.S. In Utero Pesticide Exposure, Maternal Paraoxonase Activity, and Head Circumference. Environ. Health Perspect. 2004, 112, 388–391. [Google Scholar] [CrossRef]
- Soesanti, F.; Idris, N.S.; Klipstein-Grobusch, K.; Hendarto, A.; Grobbee, D.E.; Uiterwaal, C.S.P.M. The Effect of Non-Organophosphate Household Pesticides Exposure during Pregnancy on Infants Birth Sizes and Growth Rate: A Cohort Study. BMC Pregnancy Childbirth 2020, 20, 476. [Google Scholar] [CrossRef]
- Zhang, C.; Atasoy, D.; Araç, D.; Yang, X.; Fucillo, M.V.; Robison, A.J.; Ko, J.; Brunger, A.T.; Südhof, T.C. Neurexins Physically and Functionally Interact with GABAA-Receptors. Neuron 2010, 66, 403–416. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caioni, G.; Merola, C.; Perugini, M.; d’Angelo, M.; Cimini, A.M.; Amorena, M.; Benedetti, E. An Experimental Approach to Study the Effects of Realistic Environmental Mixture of Linuron and Propamocarb on Zebrafish Synaptogenesis. Int. J. Environ. Res. Public Health 2021, 18, 4664. https://doi.org/10.3390/ijerph18094664
Caioni G, Merola C, Perugini M, d’Angelo M, Cimini AM, Amorena M, Benedetti E. An Experimental Approach to Study the Effects of Realistic Environmental Mixture of Linuron and Propamocarb on Zebrafish Synaptogenesis. International Journal of Environmental Research and Public Health. 2021; 18(9):4664. https://doi.org/10.3390/ijerph18094664
Chicago/Turabian StyleCaioni, Giulia, Carmine Merola, Monia Perugini, Michele d’Angelo, Anna Maria Cimini, Michele Amorena, and Elisabetta Benedetti. 2021. "An Experimental Approach to Study the Effects of Realistic Environmental Mixture of Linuron and Propamocarb on Zebrafish Synaptogenesis" International Journal of Environmental Research and Public Health 18, no. 9: 4664. https://doi.org/10.3390/ijerph18094664
APA StyleCaioni, G., Merola, C., Perugini, M., d’Angelo, M., Cimini, A. M., Amorena, M., & Benedetti, E. (2021). An Experimental Approach to Study the Effects of Realistic Environmental Mixture of Linuron and Propamocarb on Zebrafish Synaptogenesis. International Journal of Environmental Research and Public Health, 18(9), 4664. https://doi.org/10.3390/ijerph18094664







