A Narrative Review of Occupational Air Pollution and Respiratory Health in Farmworkers
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Assessment of Occupational Exposure to Air Pollution
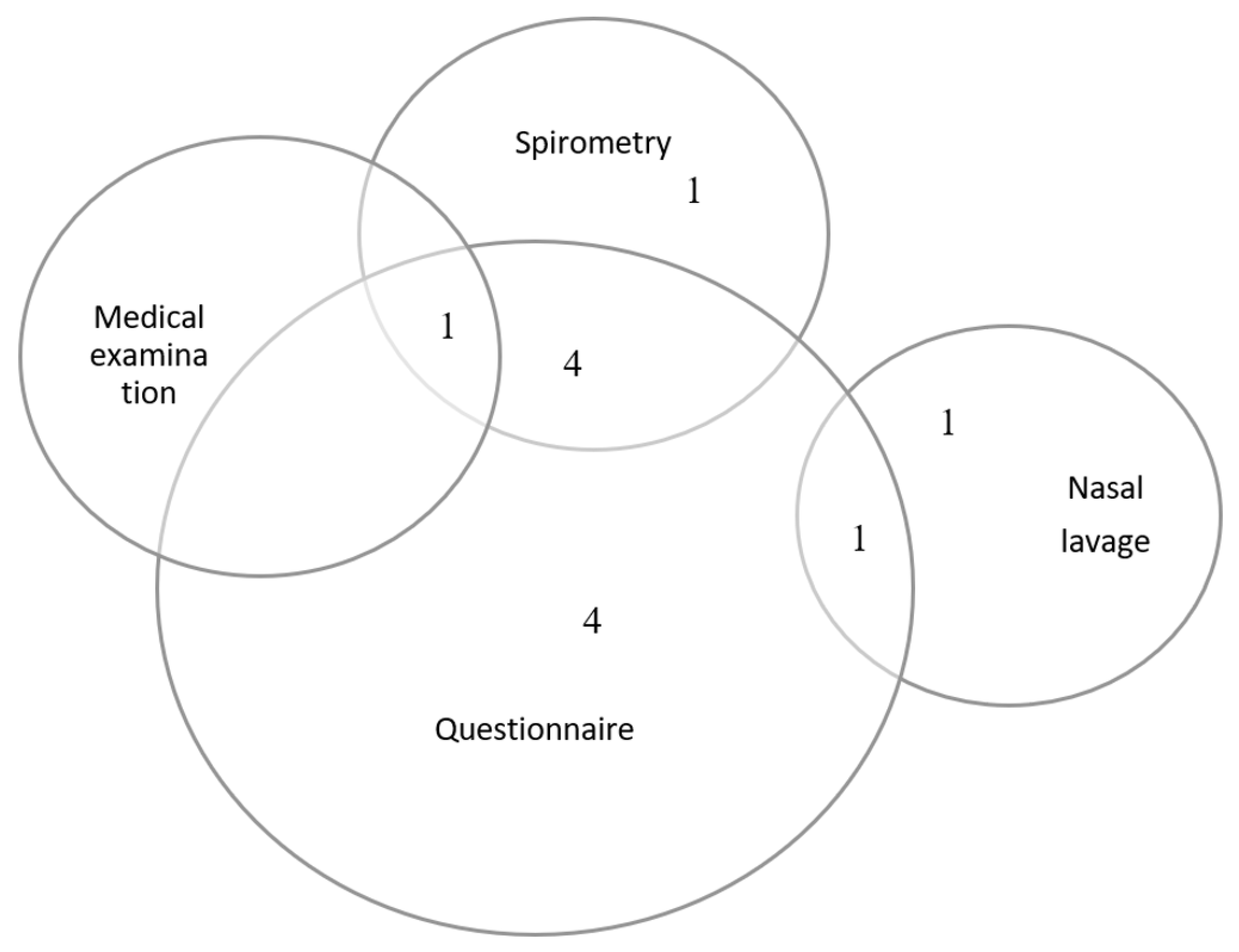
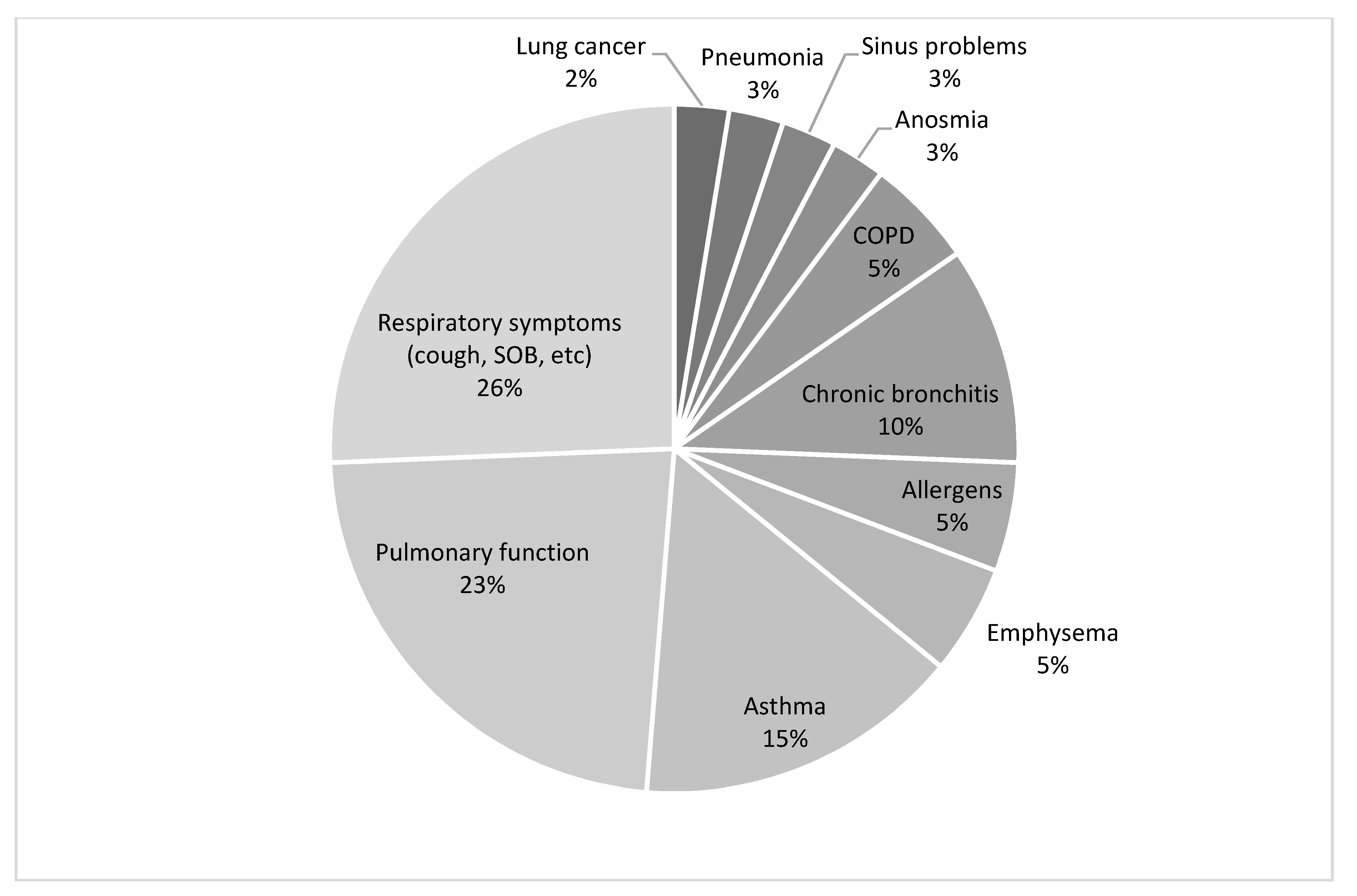
3.2. Respiratory Health Outcome Assessment
3.3. Summary of Epidemiological Findings
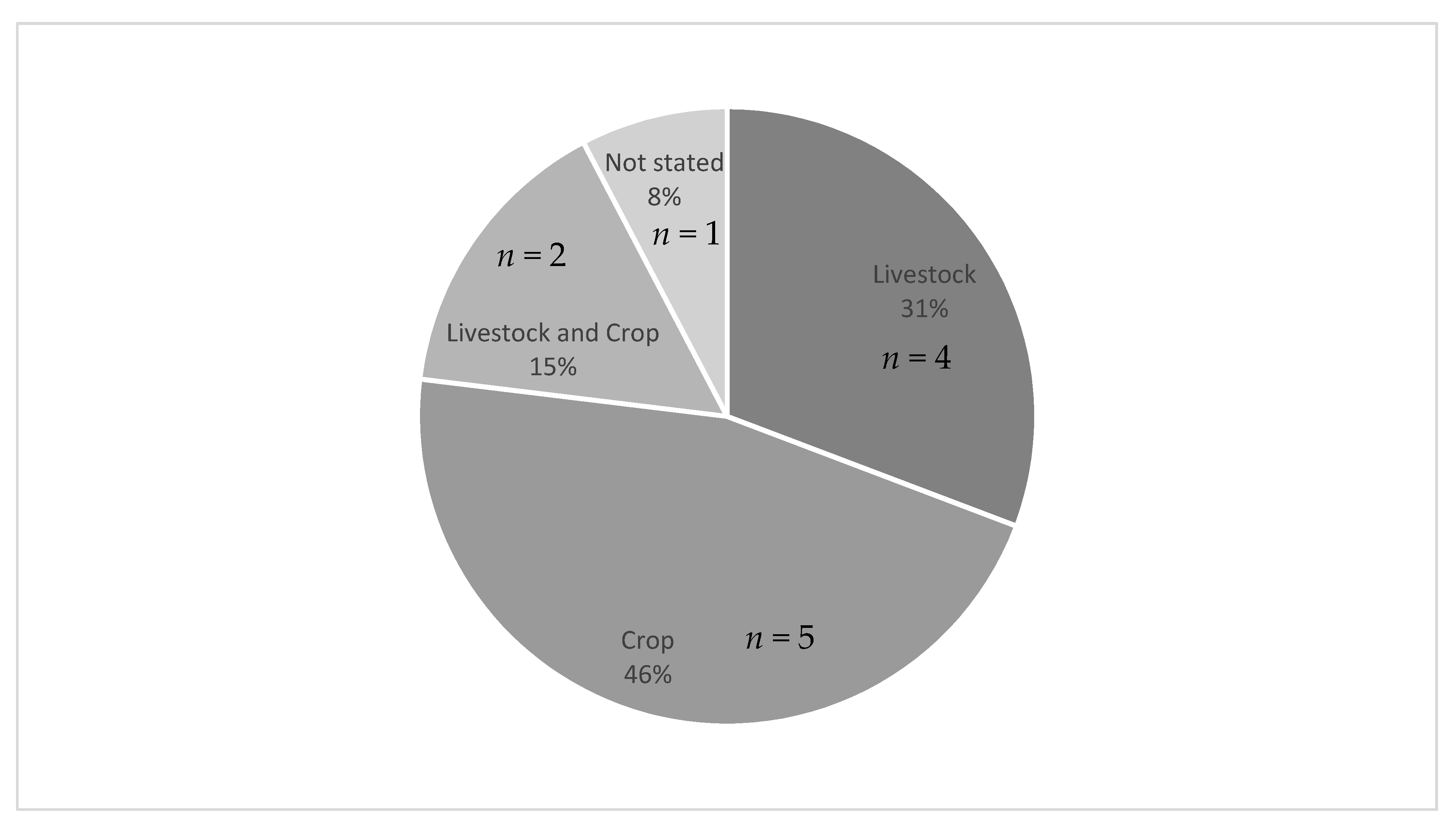
3.3.1. General (Unspecified) Farmworkers
3.3.2. Crop Farmworkers
3.3.3. Livestock Farmworkers
4. Discussion
4.1. Summary of Results and General Findings
4.2. How Have Researchers Assessed Respirable Dust Exposure among Farmworkers?
4.3. What Methods Have Been Used to Assess Respiratory Infections and What Types of Respiratory Pathogens Have Been Assessed among Farmworker Populations?
4.4. Are There Epidemiological Studies That Have Investigated the Association between Respirable Dust Exposures and Respiratory Infections in Farmworkers?
4.5. Other Gaps Identified
4.6. Limitations and Strengths
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Charania, I.; Li, X. Smart Farming: Agriculture’s Shift from a Labor Intensive to Technology Native Industry. Internet Things 2020, 9, 100142. [Google Scholar] [CrossRef]
- Schenker, M.B.; Christiani, D.; Cormier, Y.; Dimich-Ward, H.; Doekes, G.; Dosman, J.; Douwes, J.; Dowling, K.; Enarson, D.; Green, F.; et al. Respiratory Health Hazards in Agriculture. Am. J. Respir. Crit. Care Med. 1998, 158, S1–S76. [Google Scholar] [CrossRef]
- Arcury, T.A.; Marín, A.J. Latino/Hispanic Farmworkers and Farm Work in the Eastern United States: The Context for Health, Safety, and Justice. In Latino Farmworkers in the Eastern United States; Quandt, S.A., Arcury, T.A., Eds.; Springer: New York, NY, USA, 2009; pp. 15–36. ISBN 978-0-387-88346-5. [Google Scholar]
- Hu, R.; Shi, L.; Lee, D.-C.; Haile, G.P. Access to and Disparities in Care among Migrant and Seasonal Farm Workers (MSFWs) at U.S. Health Centers. J. Health Care Poor Underserved 2016, 27, 1484–1502. [Google Scholar] [CrossRef] [PubMed]
- Moran, R.E.; Bennett, D.H.; Garcia, J.; Schenker, M.B. Occupational Exposure to Particulate Matter from Three Agricultural Crops in California. Int. J. Hyg. Environ. Health 2014, 217, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Schenker, M.B.; Pinkerton, K.E.; Mitchell, D.; Vallyathan, V.; Elvine-Kreis, B.; Green, F.H.Y. Pneumoconiosis from Agricultural Dust Exposure among Young California Farmworkers. Environ. Health Perspect. 2009, 117, 988–994. [Google Scholar] [CrossRef] [PubMed]
- Liebman, A.; Roy, N.; Seda, C.; Zuroweste, E.; Roy, C. Partnerships as an Avenue to Translate Emerging Disease Ecology of SARS-CoV-2 to Agricultural Groups. J. Agromed. 2020, 1–4. [Google Scholar] [CrossRef]
- Ramos, A.; Fuentes, A.; Trinidad, N. Perception of Job-Related Risk, Training, and Use of Personal Protective Equipment (PPE) among Latino Immigrant Hog CAFO Workers in Missouri: A Pilot Study. Safety 2016, 2, 25. [Google Scholar] [CrossRef]
- Arcury, T.A.; Estrada, J.M.; Quandt, S.A. Overcoming Language and Literacy Barriers in Safety and Health Training of Agricultural Workers. J. Agromed. 2010, 15, 236–248. [Google Scholar] [CrossRef]
- Dodd, K.E.; Wood, J.; Mazurek, J.M. Mortality among Persons with Both Asthma and Chronic Obstructive Pulmonary Disease Aged ≥25 Years, by Industry and Occupation—United States, 1999–2016. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 670–679. [Google Scholar] [CrossRef]
- Sexton, K. Sociodemographic Aspects of Human Susceptibility to Toxic Chemicals. Environ. Toxicol. Pharmacol. 1997, 4, 261–269. [Google Scholar] [CrossRef]
- Milanowski, J.; Góra, A.; Skórska, C.; Mackiewicz, B.; Krysińska-Traczyk, E.; Cholewa, G.; Sitkowska, J.; Dutkiewicz, J. The Effects of Exposure to Organic Dust on the Respiratory System of Potato Processing Workers. Ann. Agric. Environ. Med. AAEM 2002, 9, 243–247. [Google Scholar]
- Fareed, M.; Pathak, M.K.; Bihari, V.; Kamal, R.; Srivastava, A.K.; Kesavachandran, C.N. Adverse Respiratory Health and Hematological Alterations among Agricultural Workers Occupationally Exposed to Organophosphate Pesticides: A Cross-Sectional Study in North India. PLoS ONE 2013, 8, e69755. [Google Scholar] [CrossRef]
- Karadžinska-Bislimovska, J.; Minov, J.; Stoleski, S.; Mijakoski, D.; Risteska-Kuc, S.; Milkovska, S. Environmental and Occupational Health Risks Among Agricultural Workers Living in a Rural Community Near Petroleum Refinery and Motorway in Skopje Region. Arch. Ind. Hyg. Toxicol. 2010, 61, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Thongtip, S.; Siviroj, P.; Deesomchok, A.; Prapamontol, T.; Wisetborisut, A.; Khacha-ananda, S. Effects of High Silica Exposure on Respiratory Disorders among Stone-Mortar Workers in Northern Thailand. Southeast. Asian J. Trop. Med. Public Health 2019, 50, 401–410. [Google Scholar]
- Bowe, B.; Xie, Y.; Yan, Y.; Al-Aly, Z. Burden of Cause-Specific Mortality Associated with PM2.5 Air Pollution in the United States. JAMA Netw. Open 2019, 2, e1915834. [Google Scholar] [CrossRef] [PubMed]
- Center of Disease Control and Prevention (CDC). Occupational Health of Hired Farmworkers in the United States National Agricultural Workers Survey Occupational Health Supplement, 1999; DHHS (NIOSH) Publication No. 2009–119, USA, 2009. Available online: https://www.cdc.gov/niosh/docs/2009-119/default.html (accessed on 14 March 2021).
- World Health Organization Chronic Respiratory Diseases; WHO: Geneva, Switzerland, 2020.
- Viegas, S.; Faísca, V.M.; Dias, H.; Clérigo, A.; Carolino, E.; Viegas, C. Occupational Exposure to Poultry Dust and Effects on the Respiratory System in Workers. J. Toxicol. Environ. Health A 2013, 76, 230–239. [Google Scholar] [CrossRef]
- Sak, Z.; Kurtuluş, Ş.; Ocakli, B.; Töreyin, Z.; Bayhan, İ.; Yeşilnacar, M.İ.; Akgün, M.; Arbak, P. Respiratory Symptoms and Pulmonary Functions before and after Pesticide Application in Cotton Farming. Ann. Agric. Environ. Med. 2018, 25, 701–707. [Google Scholar] [CrossRef]
- Madsen, A.M.; Tendal, K.; Thilsing, T.; Frederiksen, M.; Baelum, J.; Hansen, J. Fungi, β-Glucan, and Bacteria in Nasal Lavage of Greenhouse Workers and Their Relation to Occupational Exposure. Ann. Occup. Hyg. 2013. [Google Scholar] [CrossRef]
- Adhikari, A.; Gupta, J.; Wilkins, J.; Olds, R. Airborne Microorganisms, Endotoxin, and (1→3)-β-D-Glucan Exposure in Greenhouses and Assessment of Respiratory Symptoms Among Workers. Ann. Occup. Hyg. 2010. [Google Scholar] [CrossRef]
- Burch, J.B.; Svendsen, E.; Siegel, P.D.; Wagner, S.E.; von Essen, S.; Keefe, T.; Mehaffy, J.; Martinez, A.S.; Bradford, M.; Baker, L.; et al. Endotoxin Exposure and Inflammation Markers Among Agricultural Workers in Colorado and Nebraska. J. Toxicol. Environ. Health A 2009, 73, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Schenker, M.B.; Farrar, J.A.; Mitchell, D.C.; Green, R.S.; Samuels, S.J.; Lawson, R.J.; McCurdy, S.A. Agricultural Dust Exposure and Respiratory Symptoms Among California Farm Operators. J. Occup. Environ. Med. 2005, 47, 1157–1166. [Google Scholar] [CrossRef] [PubMed]
- Nonnenmann, M.W.; Gimeno Ruiz de Porras, D.; Levin, J.; Douphrate, D.; Boggaram, V.; Schaeffer, J.; Gallagher, M.; Hornick, M.; Reynolds, S. Pulmonary Function and Airway Inflammation among Dairy Parlor Workers after Exposure to Inhalable Aerosols: Pulmonary Function and Exposure to Aerosols among Dairy Workers. Am. J. Ind. Med. 2017, 60, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, D.C.; Armitage, T.L.; Schenker, M.B.; Bennett, D.H.; Tancredi, D.J.; Langer, C.E.; Reynolds, S.J.; Dooley, G.; Mehaffy, J.; Mitloehner, F.M. Particulate Matter, Endotoxin, and Worker Respiratory Health on Large Californian Dairies. J. Occup. Environ. Med. 2015, 57, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Góra, A.; Skórska, C.; Prażmo, Z.; Krysińska-Traczyk, E.; Sitkowska, J.; Dutkiewicz, J. Exposure to Bioaerosols: Allergic Reactions and Respiratory Function in Polish Hop Growers: Organic Dust Exposure and Hop Farmers Health Status. Am. J. Ind. Med. 2004, 46, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Audi, C.; Baïz, N.; Maesano, C.; Ramousse, O.; Reboulleau, D.; Magnan, A.; Caillaud, D.; Annesi-Maesano, I. Serum cytokine levels related to exposure to volatile organic compounds and PM2. 5 in dwellings and workplaces in French farmers–a mechanism to explain nonsmoking COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 1363–1374. [Google Scholar] [CrossRef]
- Guillam, M.-T.; Pédrono, G.; Le Bouquin, S.; Huneau, A.; Gaudon, J.; Leborgne, R.; Dewitte, J.-D.; Ségala, C. Chronic Respiratory Symptoms of Poultry Farmers and Model-Based Estimates of Long-Term Dust Exposure. Ann. Agric. Environ. Med. AAEM 2013, 20, 307–311. [Google Scholar]
- Rodriquez, E.J.; Stoecklin-Marois, M.T.; Bennett, D.H.; Tancredi, D.J.; Schenker, M.B. Agricultural Work Exposures and Pulmonary Function Among Hired Farm Workers in California (The MICASA Study). J. Agromed. 2014, 19, 427–436. [Google Scholar] [CrossRef]
- National Institute of Occupational Safety and Health (NIOSH) Respiratory Diseases in Agricultural Workers: Mortality and Morbidity Statistics; NIOSH Publications Dissemination 4676 Columbia Parkway Cincinnati: Cincinnati, OH, USA, 2007.
- Erb, R.; Oosting, J. Coronavirus Outbreaks Tied to Migrant Farm Workers in Michigan; Michigan Health Watch: Grand Rapids, MI, USA, 2020. [Google Scholar]
- Reiley, L. Migrant Farmworkers, Many Coronavirus Positive, Move North from Florida to Other States; Washington Post: Washington, DC, USA, 2020. [Google Scholar]
- Akbar-Khanzadeh, F.; Ames, A.; Bisesi, M.; Milz, S.; Czajkowski, K.; Kumar, A. Particulate Matter (PM) Exposure Assessment—Horizontal and Vertical PM Profiles in Relation to Agricultural Activities and Environmental Factors in Farm Fields. J. Occup. Environ. Hyg. 2012, 9, 502–516. [Google Scholar] [CrossRef]
- Fix, J.; Annesi-Maesano, I.; Baldi, I.; Boulanger, M.; Cheng, S.; Cortes, S.; Dalphin, J.-C.; Dalvie, M.A.; Degano, B.; Douwes, J.; et al. Gender Differences in Respiratory Health Outcomes among Farming Cohorts around the Globe: Findings from the AGRICOH Consortium. J. Agromed. 2020, 1–12. [Google Scholar] [CrossRef]
- Berberet, L.J.; Buchan, M.R. Beard Respirable Dust and Crystalline Silica (Quartz) Exposure Resulting from Potato Harvesting Operations. J. Agric. Saf. Health 1999, 5, 97–108. [Google Scholar] [CrossRef]
- Sedgwick, P. Cross Sectional Studies: Advantages and Disadvantages. BMJ 2014, 348, g2276. [Google Scholar] [CrossRef]
- Occupational Health and Safety Agency (OSHA). OSHA Technical Manual; Occupational Safety and Health Administration: Washington, DC, USA, 2014.
- Borghi, F.; Spinazzè, A.; Rovelli, S.; Campagnolo, D.; Del Buono, L.; Cattaneo, A.; Cavallo, D.M. Miniaturized Monitors for Assessment of Exposure to Air Pollutants: A Review. Int. J. Environ. Res. Public. Health 2017, 14, 909. [Google Scholar] [CrossRef] [PubMed]
- Volckens, J.; Quinn, C.; Leith, D.; Mehaffy, J.; Henry, C.S.; Miller-Lionberg, D. Development and Evaluation of an Ultrasonic Personal Aerosol Sampler. Indoor Air 2017, 27, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Larkin, A.; Hystad, P. Towards Personal Exposures: How Technology Is Changing Air Pollution and Health Research. Curr. Environ. Health Rep. 2017, 4, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Dangi, B.; Bhise, A. Cotton Dust Exposure: Analysis of Pulmonary Function and Respiratory Symptoms. Lung India 2017, 34, 144. [Google Scholar] [CrossRef] [PubMed]
- Bousfield, G.; Morin, S.; Jacquet, N.; Richel, A. Extraction and Refinement of Agricultural Plant Fibers for Composites Manufacturing. Comptes Rendus Chim. 2018, 21, 897–906. [Google Scholar] [CrossRef]
- Bulla, A.; Hitze, K.L. Acute Respiratory Infections: A Review. Bull. World Health Organ. 1978, 56, 481–498. [Google Scholar] [PubMed]
- Leach, R.M. Symptoms and Signs of Respiratory Disease. Medicine 2008, 36, 119–125. [Google Scholar] [CrossRef]
- Althubaiti, A. Information Bias in Health Research: Definition, Pitfalls, and Adjustment Methods. J. Multidiscip. Healthc. 2016, 211. [Google Scholar] [CrossRef] [PubMed]
- Haynes, J. Basic Spirometry Testing and Interpretation for the Primary Care Provider. Can. J. Respir. Ther. 2018, 54, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Saglani, S.; Menzie-Gow, A.N. Approaches to Asthma Diagnosis in Children and Adults. Front. Pediatr. 2019, 7, 148. [Google Scholar] [CrossRef] [PubMed]
- Myers, K.P.; Olsen, C.W.; Setterquist, S.F.; Capuano, A.W.; Donham, K.J.; Thacker, E.L.; Merchant, J.A.; Gray, G.C. Are Swine Workers in the United States at Increased Risk of Infection with Zoonotic Influenza Virus? Clin. Infect. Dis. 2006, 42, 14–20. [Google Scholar] [CrossRef]
- Krumbholz, A.; Lange, J.; Dürrwald, R.; Hoyer, H.; Bengsch, S.; Wutzler, P.; Zell, R. Prevalence of Antibodies to Swine Influenza Viruses in Humans with Occupational Exposure to Pigs, Thuringia, Germany, 2008–2009. J. Med. Virol. 2010, 82, 1617–1625. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhou, Y.; Zhao, Y.; Li, W.; Song, W.; Miao, Z. Avian Influenza H9N2 Seroprevalence among Pig Population and Pig Farm Staff in Shandong, China. Virol. J. 2015, 12, 34. [Google Scholar] [CrossRef]
- Ortiz, E.J.; Kochel, T.J.; Capuano, A.W.; Setterquist, S.F.; Gray, G.C. Avian Influenza and Poultry Workers, Peru, 2006. Influenza Other Respir. Viruses 2007, 1, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.; Lee, J.-Y.; Choi, W.; Choi, J.-H.; Chung, Y.-S.; Lee, N.-J.; Cheong, H.-M.; Katz, J.M.; Oh, H.-B.; Cho, H.; et al. Avian Influenza A (H5N1) Virus Antibodies in Poultry Cullers, South Korea, 2003–2004. Emerg. Infect. Dis. 2012, 18, 986–988. [Google Scholar] [CrossRef]
- Ameni, G.; Tadesse, K.; Hailu, E.; Deresse, Y.; Medhin, G.; Aseffa, A.; Hewinson, G.; Vordermeier, M.; Berg, S. Transmission of Mycobacterium Tuberculosis between Farmers and Cattle in Central Ethiopia. PLoS ONE 2013, 8, e76891. [Google Scholar] [CrossRef]
- Sichewo, P.R.; Vander Kelen, C.; Thys, S.; Michel, A.L. Risk Practices for Bovine Tuberculosis Transmission to Cattle and Livestock Farming Communities Living at Wildlife-Livestock-Human Interface in Northern KwaZulu Natal, South Africa. PLoS Negl. Trop. Dis. 2020, 14, e0007618. [Google Scholar] [CrossRef] [PubMed]
- Alavi, S.M.; Khoshkho, M.M. Seroprevalence Study of Leptospirosis among Rice Farmers in Khuzestan Province, South West Iran, 2012. Jundishapur J. Microbiol. 2014, 7. [Google Scholar] [CrossRef]
- Monno, R.; Fumarola, L.; Trerotoli, P.; Cavone, D.; Massaro, T.; Spinelli, L.; Rizzo, C.; Musti, M. Seroprevalence of Q-Fever, Brucellosis and Leptospirosis in Farmers and Agricultural Workers in Bari, Southern Italy. Clin. Microbiol. Infect. 2009, 15, 142–143. [Google Scholar] [CrossRef]
- van den Bogaard, A.E. Antibiotic Resistance of Faecal Escherichia Coli in Poultry, Poultry Farmers and Poultry Slaughterers. J. Antimicrob. Chemother. 2001, 47, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Bao, Z.-J.; Fan, Y.-M.; Cui, Y.-F.; Sheng, Y.-F.; Zhu, M. Effect of PM2.5 Mediated Oxidative Stress on the Innate Immune Cellular Response of Der P1 Treated Human Bronchial Epithelial Cells. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2907–2912. [Google Scholar]
- Bauer, R.N.; Diaz-Sanchez, D.; Jaspers, I. Effects of Air Pollutants on Innate Immunity: The Role of Toll-like Receptors and Nucleotide-Binding Oligomerization Domain–like Receptors. J. Allergy Clin. Immunol. 2012, 129, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.; Park, S.A.; Park, I.; Kim, P.; Cho, N.H.; Hyun, J.W.; Hyun, Y.-M. PM2.5 Exposure in the Respiratory System Induces Distinct Inflammatory Signaling in the Lung and the Liver of Mice. J. Immunol. Res. 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Miyata, R.; van Eeden, S.F. The Innate and Adaptive Immune Response Induced by Alveolar Macrophages Exposed to Ambient Particulate Matter. Toxicol. Appl. Pharmacol. 2011, 257, 209–226. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zheng, X.; Nicholas, J.; Humes, S.T.; Loeb, J.C.; Robinson, S.E.; Bisesi, J.H.; Das, D.; Saleh, N.B.; Castleman, W.L.; et al. Single-Walled Carbon Nanotubes Modulate Pulmonary Immune Responses and Increase Pandemic Influenza a Virus Titers in Mice. Virol. J. 2017, 14, 242. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Humes, S.T.; Robinson, S.E.; Loeb, J.C.; Sabaraya, I.V.; Saleh, N.B.; Khattri, R.B.; Merritt, M.E.; Martyniuk, C.J.; Lednicky, J.A.; et al. Single-Walled Carbon Nanotubes Repress Viral-Induced Defense Pathways through Oxidative Stress. Nanotoxicology 2019, 13, 1176–1196. [Google Scholar] [CrossRef] [PubMed]
- Sanpui, P.; Zheng, X.; Loeb, J.C.; Bisesi, J.H., Jr.; Khan, I.A.; Afrooz, A.R.M.N.; Liu, K.; Badireddy, A.R.; Wiesner, M.R.; Ferguson, P.L.; et al. Single-Walled Carbon Nanotubes Increase Pandemic Influenza A H1N1 Virus Infectivity of Lung Epithelial Cells. Part. Fibre Toxicol. 2014, 11, 66. [Google Scholar] [CrossRef]
- Ma, J.-H.; Song, S.-H.; Guo, M.; Zhou, J.; Liu, F.; Peng, L.; Fu, Z.-R. Long-Term Exposure to PM2.5 Lowers Influenza Virus Resistance via down-Regulating Pulmonary Macrophage Kdm6a and Mediates Histones Modification in IL-6 and IFN-β Promoter Regions. Biochem. Biophys. Res. Commun. 2017, 493, 1122–1128. [Google Scholar] [CrossRef]
- Xing, Y.-F.; Xu, Y.-H.; Shi, M.-H.; Lian, Y.-X. The Impact of PM2.5 on the Human Respiratory System. J. Thorac. Dis. 2016, 8, E69-74. [Google Scholar] [CrossRef]
- Rylander, R. Endotoxin in the Environment—Exposure and Effects. J. Endotoxin Res. 2002, 8, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, A.; Suzuki, T.; Ohara, Y.; Takahashi, K.; Sato, Y.; Ainai, A.; Nagata, N.; Tashiro, M.; Hasegawa, H. Impacts of Allergic Airway Inflammation on Lung Pathology in a Mouse Model of Influenza A Virus Infection. PLoS ONE 2017, 12, e0173008. [Google Scholar] [CrossRef] [PubMed]
- DeBord, D.G.; Carreón, T.; Lentz, T.J.; Middendorf, P.J.; Hoover, M.D.; Schulte, P.A. Use of the “Exposome” in the Practice of Epidemiology: A Primer on -Omic Technologies. Am. J. Epidemiol. 2016, 184, 302–314. [Google Scholar] [CrossRef] [PubMed]
- Castillo, M.; Simnitt, S. Farm. Labor; USDA Economic Research Service: USA, 2020. Available online: https://www.ers.usda.gov/topics/farm-economy/farm-labor/# (accessed on 14 March 2021).
- Okoye, J.O.; Eze, D.C.; Krueger, W.S.; Heil, G.L.; White, S.K.; Merrill, H.R.; Gray, G.C. Evidence for Subclinical H5N1 Avian Influenza Infections among Nigerian Poultry Workers: Avian Influenza in Nigeria. J. Med. Virol. 2014, 86, 2070–2075. [Google Scholar] [CrossRef] [PubMed]
- Pawar, S.D.; Tandale, B.V.; Raut, C.G.; Parkhi, S.S.; Barde, T.D.; Gurav, Y.K.; Kode, S.S.; Mishra, A.C. Avian Influenza H9N2 Seroprevalence among Poultry Workers in Pune, India, 2010. PLoS ONE 2012, 7, e36374. [Google Scholar] [CrossRef]
- Sanghi, A.; Mendelsohn, R. The Impacts of Global Warming on Farmers in Brazil and India. Glob. Environ. Chang. 2008, 18, 655–665. [Google Scholar] [CrossRef]

| Study | Type of Farmworker and/or Crop (N) | Type of Air Sample | Specific Instrument | Work Shift Exposure Measurement | Frequency of Measurement | Contaminant Type | PM2.5 Concentration Range |
|---|---|---|---|---|---|---|---|
| Mitchell et al. (2015) [26] | Dairy farmers (N = 205) | Personal filter sample | SKC button sampler for collecting inhalable PM (<100 μm in aerodynamic diameter) onto a Teflon 25-mm Millipore PTFE filter, with a pore size of 3.0 μm (Fisher FSLW02500). A GK2.05SH (KTL) cyclone sampler (BGI Inc., Waltham, MA) collected particles with a cut point of 2.5 μm (PM2.5) onto a Teflon filter (Fisher, FHLP03700). | Yes | Not stated | PM | Geometric mean concentration PM = 812 μg/m3 PM2.5 = 35 μg/m3 |
| Nonnenman et al. (2017) [25] | Dairy parlor workers (N = 62) A parlor is a building where cows are milked on a dairy farm. | Personal filter sample | Inhalable dust (50% cut-point at 100 μm) was sampled in the worker’s breathing zone using an inhalable sampler (Button Aerosol Sampler, SKC Inc. manufacturer, Eighty Four, Pennsylvania, USA) and personal sampling pumps (AirChek XR 5000, SKC Inc.) | Yes | Duration of a single work-shift | Inhalable dust (<Xµm) Muramic acid Endotoxin | NA |
| Burch et al. (2009) [23] | Grain elevator workers, cattle feedlot, dairy and corn farm workers (N = 125) | Personal filter sample | Personal breathing zone samples for inhalable particulate matter were collected using (IOM) sampling cassettes and 25mm PVC filters with a 5-μm pore size (SKC, Eighty Four, PA). | Yes | Duration of a single work-shift | Inhalable dust Endotoxin Endotoxin’s 3-hydroxy fatty acid (3-OHFA) | NA |
| Guillam et al. (2013) [29] | Egg production workers (N = 100) | Personal filter sampler | Personal dust sampler, 11 ozs, 10 lmp flowrate—CIP 10—ARELCO | Yes | Cold season work-shift Warm season work-shift | Respirable dust (<4 µm in diameter) | NA |
| Góra et al. (2004) [27] | Crop farmers (hop growers) (N = 69) | Personal filter sample | AP-2A personal sampler—TWOMET, Zgierz, Poland at the flow rate of 2 L/min. Glass fiber filters, with 1 µm pore size and 37 mm diameter. | Yes | Once during harvest season | Airborne microorganisms, dust and endotoxin Gram-positive bacteria (corynebacteria and bacilli) Fungi (Penicillium citrinum, Alternaria alternata, and Cladosporium epiphyllum) Thermophilic actinomycetes Gram-negative bacteria | NA |
| Madsen et al. (2013) [21] | Greenhouse vegetable crop workers (N = 33) | Personal filter sample | Gesamtstaubprobenahme (GSP) inhalable samplers—Gesamtstaubprobenahme by BGI, Inc., Waltham, MA, USA; polycarbonate filter (pore size 1 µm) | Yes | Sampling took place from 6:00 or 7:00 to 15:00 or 16:00 during the Wednesdays immediately preceding the Thursday of nasal lavage sampling | Fungi Bacteria β-glucan | NA |
| Viegas et al. (2013) [19] | Poultry farm workers (N = 47) | Personal filter sample | Portable direct reading equipment—Lighthouse, model 3016 IAQ | Yes | During performance of different tasks in pavilions | PM0.5 PM1 PM2.5 PM5 PM10 | PM0.5 = 2.8–25 µg/m3 PM1 = 4.7–32 µg/m3 PM2.5 = 20–240 µg/m3 PM5 = 220–2400 µg/m3 PM10 = 1400–15,200 µg/m3 |
| Adhikari et al. (2011) [22] | Greenhouse (flowers and plants) workers (N = 49) | Stationary monitoring | Button Inhalable Aerosol Samplers—SKC, Inc., Eighty Four, PA, USA | Yes | During winter and summer for 5 to 7 h per one work shift—four from the corners of the greenhouses and one from the center. | Fungi Bacteria Actinomycetes Endotoxin (1→3)-β-D-glucan | NA |
| Audi et al. (2017) [28] | Granary and stable workers (N = 72) | Stationary monitoring | Radiello Passive Sampler (for BTEX) to measure VOCs Aerocet 530 device to measure PM2.5 | Yes | Assessed over a 3-month period with samplers placed where participants are expected to spend the greatest number of hours, as well inside granaries and stables. | VOCs include hexane, benzene, ethylbenzene, trichloroethylene, toluene, tetrachloroethylene, decane isomers, butoxyethyl acetate and undecane isomers. Fine particles (PM2.5) | Highest mean value of PM2.5 (11 µg/m3), with the highest median value (3 µg/m3) and the highest third quartile value (8 µg/m3) |
| Sak et al. (2018) [20] | Persons living in cotton-farming villages (N = 252) | Stationary real-time with gravimetric validation | pDR 1500 Thermo Scientific Personal Data RAM pDR device and two cyclones were used to make PM10 and PM2.5 measurements | Yes | Before and after pesticide application. Fifteen-minute measurements of PM10 and PM2.5 were made with the cyclones. Measurements were made at the four village centers (in four villages spread over 65 km2) before agricultural spraying (in mid-June) and within 15 min and 48 h after agricultural spraying (in mid-August). | PM10 PM2.5 | PM10 = 11.7–334.8 µg/m3 PM2.5 = 4.7–17.2 µg/m3 |
| Schenker et al. (2005) [24] | Primary farm operator (PFO) (N = 100) | Questionnaire | Farmers were asked the following question: “In the past year, approximately what percentage of the time that you spent farming did you spend working at a dusty job?” They were also asked to report the numbers of hours they personally worked on their farm operation over the last year (by season) and the percentage of time spent in the general categories of administrative, field, and livestock tasks. Dust exposure variables were considered “none”, “low” or “high” based on percent time in dust multiplied by average yearly hours per week farming, and percent time in dusty environment. | NA | NA | Dust | NA |
| Rodriguez et al. (2014) [30] | Mexican migrant crop (melons, tomatoes, nuts, grapes, cotton, lettuce, asparagus, onion, pomegranate, etc.) farmers (N = 450) | Questionnaire | Time-weighted self-reported average (TWSRA) dust scores were calculated for dust exposure in a year by multiplying the number of weeks a participant worked in each crop type and job task combination by the average number of days worked per week. Next, the number of days worked for each crop type and job task combination was multiplied by its corresponding self-rated dust score. | NA | NA | Dust score | NA |
| Study | Study Design | Demographic Characteristics of Study Population | Statistical Analysis | Key Findings—Odds Ratio (95% CI) or β (95%CI) |
|---|---|---|---|---|
| Góra et al. (2004) [27] | Cross-sectional study | 53.6% male Median age 48 years Non-smokers 42% | Spearman test | Positive correlation between exposure to airborne endotoxin and IL-6 level in farmers’ serum r = 0.364, p < 0.01. The mean daily PEF values in farmers were significantly lower compared to controls (469.7 +/− 127.5 vs. 562.9 +/− 123.8; p < 0.001; the data were adjusted for gender, height, and smoking). PEF daily variability (amp%mean) was higher in farmers compared to controls (9.3 vs. 8.1%; p < 0.05). |
| Schenker et al. (2005) [24] | Cross-sectional study | 89.9% male Median age 54 years Ethnicity: 84.5% white Non-smoker 54% | Logistic regression | Adjusted prevalence odds ratio—persistent wheeze and current smoking status 4.7 (3.1–7.3); persistent wheeze and high dust exposure 1.8 (1.1–3.2); persistent wheeze and live on farm 1.7 (1.1–2.6); persistent wheeze and male sex 2.9 (1.4–6.4); persistent wheeze and asthma per MD 7.7 (5.1–11.8); chronic cough and age (40–59) 2.4 (1.0–5.6); chronic cough and current smoking status 7.3 (4.2–12.5); chronic bronchitis and former smoking status 1.9 (1.0–3.4); chronic bronchitis and current smoking status 5.8 (3.1–10.6); chronic bronchitis and asthma per MD 4.3 (2.4–7.8) |
| Burch et al. (2009) [23] | Cross-sectional study | 100% male Age 25–24 years—46% Race: 70% Caucasian Never tobacco use 51% | Geometric mean and least squares mean | Exposure quartiles 1 vs. 4—dust (mg/m3) and MPO (ng/mL) 57 vs. 21, p-value 0.01; endotoxin (EU/mg) vs. IL-8 (pg/mL) 145 vs. 228, p-value 0.05; sum of all 3-OH fatty acids (pmol/mg) and MPO (ng/mL) 21 vs. 53, p-value 0.01 |
| Adhikari et al. (2011) [22] | Cross-sectional study | 57.1% male Mean age 40.1 years Ethnicity: 99% white Current smokers 17.1% | Fisher’s exact test | No significant associations. Usually bringing up phlegm was higher in workers than controls with a crude PR of 4.4, p-value 0.133. |
| Guillam et al. (2013) [29] | Prospective cohort study | 60% male Mean age 45.4 Nonsmoker 63.5% | Logistic regression | Respirable dust concentration association with respiratory symptoms: day and/or night cough OR 2.65 (1.16–6.08); chronic cough OR 2.80 (1.12–7.02); chronic phlegm OR 2.07 (1.01–4.27); symptoms of chronic bronchitis OR 4.21 (1.21–14.7). |
| Madsen et al. (2013) [21] | Cross-sectional study | 60.6% male Median age 38.5 years Country of birth—Eastern and Central Europe, Denmark, the Middle East, and Southeast Asia | Pearson’s correlation coefficients (r2) | Exposure to fungi and fungi in NAL r2 = 0.62, p-value < 0.0001 Exposure to beta-glucan and glucan in NAL r2 = 0.42, p-value < 0.001 |
| Viegas et al. (2013) [19] | Cross-sectional study | 60.6% male Mean age 44.5 years Nonsmokers 56.1% | Prevalence | No significant association was found between duration of exposure, and spirometry. |
| Rodriguez et al. (2014) [30] | Prospective cohort study | 43% male Age 41–50 years—33% Country of birth—Mexico 67% Primary school education 58% Current smoker 6% | Multiple linear regression | High TWSRA dust score in past year and FEV6 estimate (SE) 0.22 (0.10), p-value 0.04; months worked in agriculture in past year and FEV1 estimate (SE) 0.08 (0.10), p-value < 0.001, FEF25%–5% estimate (SE) 0.11 (0.03), p-value < 0.001, and FEV6 estimate (SE) 0.11 (0.02), p-value < 0.001 |
| Mitchell et al. (2015) [26] | Cross-sectional study | 100% male Median age 33.7 years Ethnicity: 90.4% Hispanic Sixth grade or less education 52.8% | Logistic regression (mixed models) | Mixed models for FEV1/FVC and FEF 25–75 adjusted for age and shift time—total endotoxin and FVC, mL 24.46 (−44.65 to −4.27), p-value 0.018 |
| Nonnenmann et al. (2017) [25] | Non-randomized cross-sectional study | 92% male Mean age 32 Ethnicity: 94% Hispanic Ever smoke 70% | Beta Coefficient (Standard Error) | Relationship between endotoxin and cross-shift pulmonary health measures (FEV1): β (SE) −0.058 (0.039) p-value 0.081 |
| Audi et al. (2017) [28] | Prospective cohort study | 63.8% male Mean age 47.02 years High school education—58.33% Nonsmokers 87.5% | Mann–Whitney U test | IL cytokine concentration and woken by an attack of shortness of breath 2.3 (p-vale 0.009); IL cytokine concentration and COPD 1.1 (p-value 0.008) |
| Sak et al. (2018) [20] | Cross-sectional study | 42.9% male Nonsmoking 51.2% 38% no education | Logistic regression | PM2.5 and wheezing OR 2.153 (1.164–3.981) PM2.5 and chest tightness OR 2.211 (1.190–4.108) PM10 and chest tightness OR 1.123 (1.002–1.259) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clarke, K.; Manrique, A.; Sabo-Attwood, T.; Coker, E.S. A Narrative Review of Occupational Air Pollution and Respiratory Health in Farmworkers. Int. J. Environ. Res. Public Health 2021, 18, 4097. https://doi.org/10.3390/ijerph18084097
Clarke K, Manrique A, Sabo-Attwood T, Coker ES. A Narrative Review of Occupational Air Pollution and Respiratory Health in Farmworkers. International Journal of Environmental Research and Public Health. 2021; 18(8):4097. https://doi.org/10.3390/ijerph18084097
Chicago/Turabian StyleClarke, Kayan, Andres Manrique, Tara Sabo-Attwood, and Eric S. Coker. 2021. "A Narrative Review of Occupational Air Pollution and Respiratory Health in Farmworkers" International Journal of Environmental Research and Public Health 18, no. 8: 4097. https://doi.org/10.3390/ijerph18084097
APA StyleClarke, K., Manrique, A., Sabo-Attwood, T., & Coker, E. S. (2021). A Narrative Review of Occupational Air Pollution and Respiratory Health in Farmworkers. International Journal of Environmental Research and Public Health, 18(8), 4097. https://doi.org/10.3390/ijerph18084097







