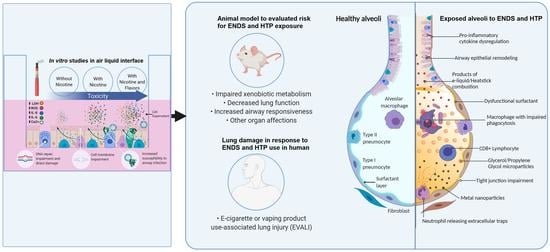
Lung Damage Caused by Heated Tobacco Products and Electronic Nicotine Delivery Systems: A Systematic Review
Abstract
1. Introduction
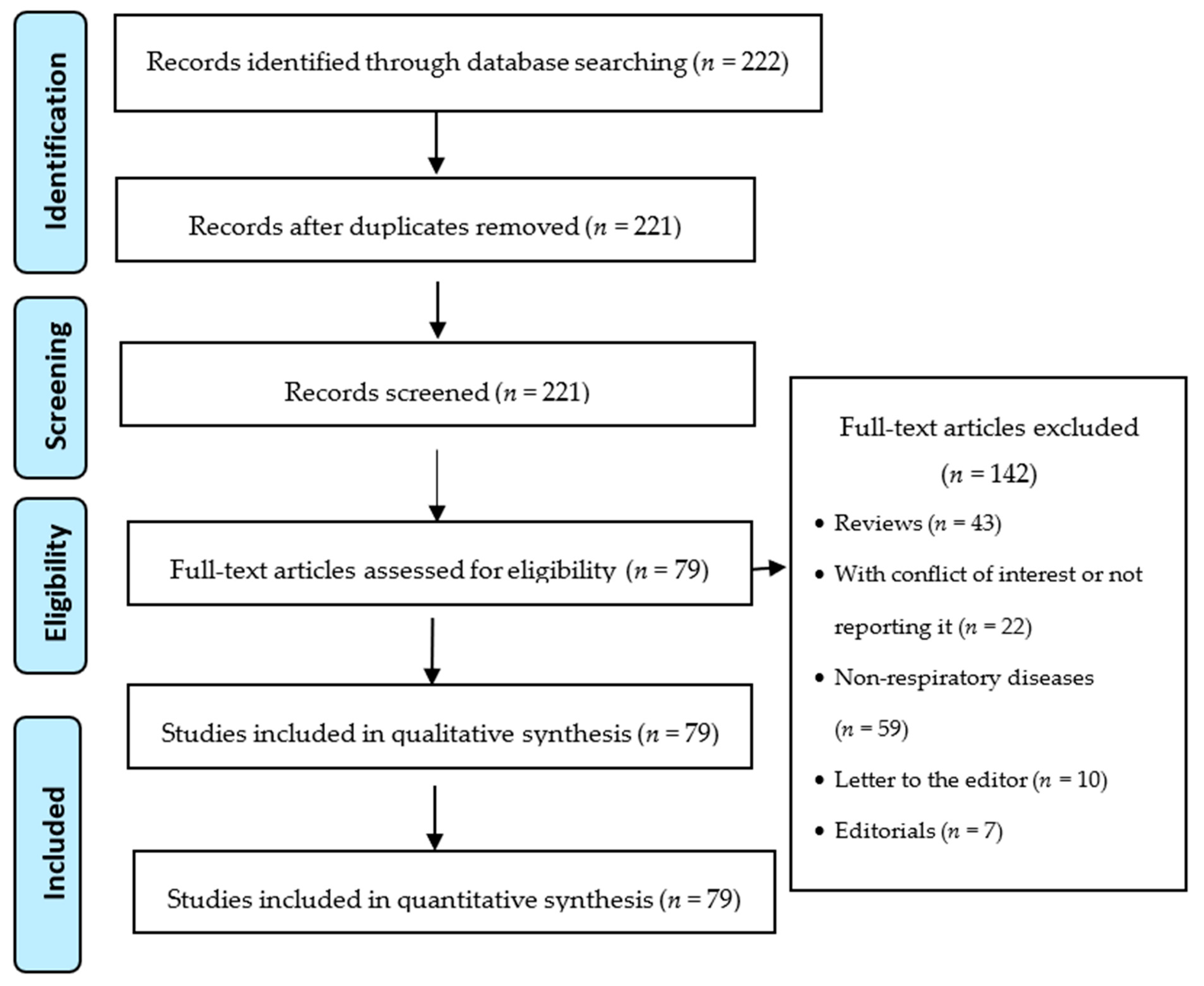
2. Materials and Methods
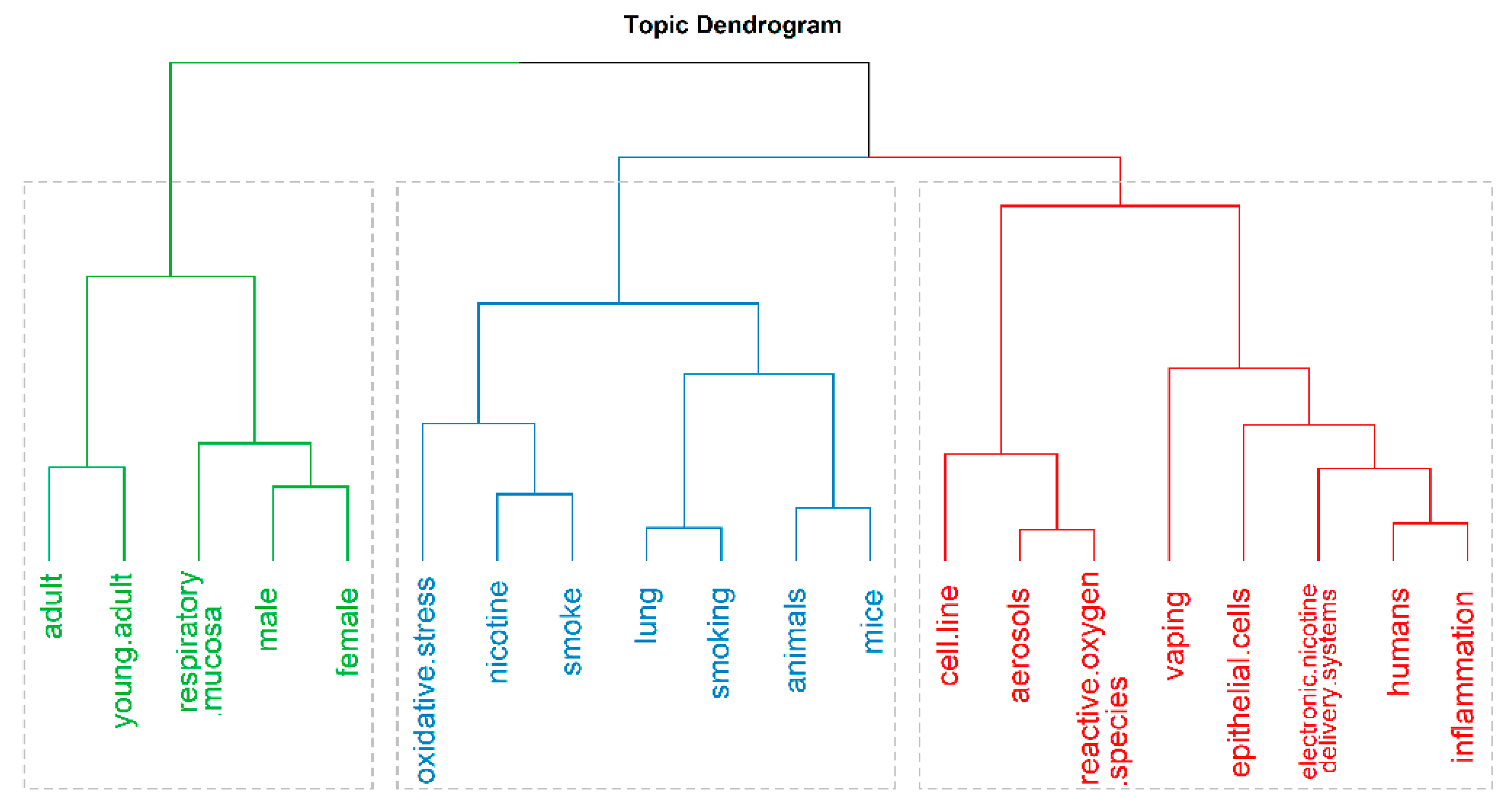
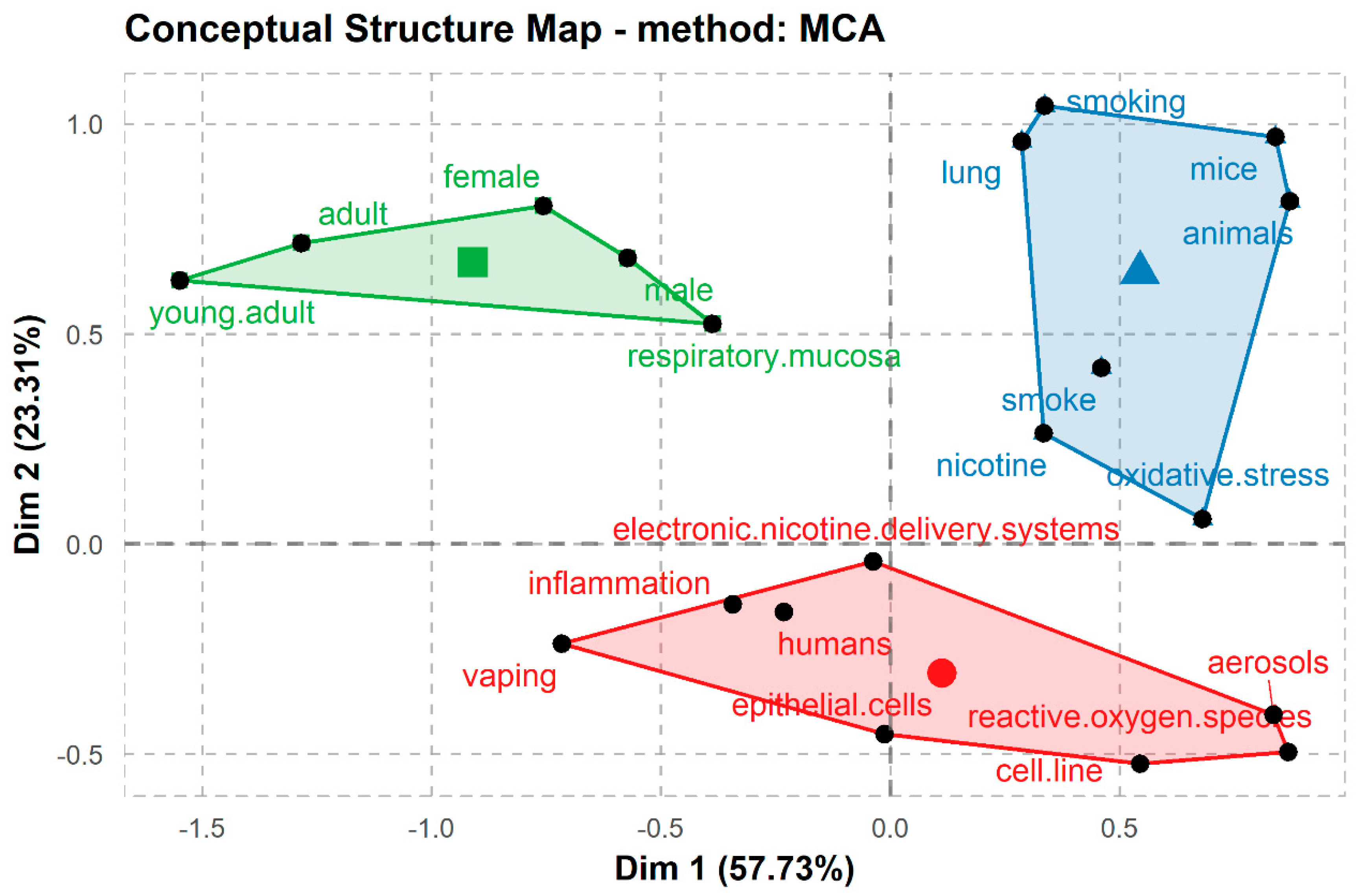
3. Results
3.1. In Vitro Models
3.1.1. Cytotoxicity in in vitro Models
3.1.2. Inflammation
3.1.3. Airway Infections
3.2. Animal Models
3.2.1. Impaired Lung
3.2.2. Lung Disease Associated with Consumption of ENDS
3.3. Human Studies
3.3.1. Damage in Human Lungs
3.3.2. Pulmonary Pathology
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zavala-Arciniega, L.; Reynales-Shigematsu, L.M.; Levy, D.T.; Lau, Y.K.; Meza, R.; Gutiérrez-Torres, D.S.; Arillo-Santillán, E.; Fleischer, N.L.; Thrasher, J. Smoking trends in Mexico, 2002–2016: Before and after the ratification of the WHO’s Framework Convention on Tobacco Control. Tob. Control 2020, 29, 687–691. [Google Scholar] [CrossRef]
- World Health Organization. WHO|Global Strategy to Accelerate Tobacco Control: Advancing Sustainable Development through the Implementation of the WHO FCTC 2019–2025; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- WHO. World Health Organization Study Group on Tobacco Product Regulation Report on the Scientific Basic of Tobacco Product Regulation: Seventh Report of a WHO Study Group; WHO Technical Report Series; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Cirillo, S.; Urena, J.F.; Lambert, J.D.; Vivarelli, F.; Canistro, D.; Paolini, M.; Cardenia, V.; Rodriguez-Estrada, M.T.; Richie, J.P.; Elias, R.J. Impact of electronic cigarette heating coil resistance on the production of reactive carbonyls, reactive oxygen species and induction of cytotoxicity in human lung cancer cells in vitro. Regul. Toxicol. Pharmacol. 2019, 109, 104500. [Google Scholar] [CrossRef] [PubMed]
- The Lancet. E-cigarettes: Public Health England’s evidence-based confusion. Lancet 2015, 386, 829. [Google Scholar] [CrossRef]
- Organización Mundial de la Salud Sistemas electrónicos de administración de nicotina y sistemas similares sin nicotina. In Proceedings of the Conferencia de las Partes en el Convenio Marco la OMS para el Control del Tabaco, Delhi, India, 7–12 November 2016; pp. 1–13.
- World Health Organization Tobacco: E-Cigarettes. Available online: https://www.who.int/news-room/q-a-detail/e-cigarettes-how-risky-are-they (accessed on 12 August 2020).
- Center for Disease Control & Prevention Outbreak of Lung Injury Associated with the Use of E-Cigarette, or Vaping, Products. Available online: https://www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease.html (accessed on 25 November 2020).
- Convenio Marco de la OMS para el Control del Tabaco. Rev. Esp. Salud Publica 2003, 77, 475–496. Available online: http://scielo.isciii.es/scielo.php?script=sci_arttext&pid=S1135-57272003000400005&lng=es&nrm=iso (accessed on 17 December 2020). [CrossRef][Green Version]
- Instituto Nacional de Desarrollo Social Ley General para el Control del Tabaco. Available online: https://www.gob.mx/indesol/documentos/ley-general-para-el-control-del-tabaco (accessed on 1 February 2021).
- Shamah-Levy, T.; Vielma-Orozco, E.; Heredia-Hernández, O.; Romero-Martínez, M.; Mojica-Cuevas, J.; Cuevas-Nasu, L.; Santaella-Castell, J.; Rivera-Dommarco, J. Encuesta Nacional de Salud y Nutrición 2018–19: Resultados Nacionales; Instituto Nacional de Salud Pública: Cuernavaca, México, 2020. [Google Scholar]
- Dusautoir, R.; Zarcone, G.; Verriele, M.; Garçon, G.; Fronval, I.; Beauval, N.; Allorge, D.; Riffault, V.; Locoge, N.; Lo-Guidice, J.M.; et al. Comparison of the chemical composition of aerosols from heated tobacco products, electronic cigarettes and tobacco cigarettes and their toxic impacts on the human bronchial epithelial BEAS-2B cells. J. Hazard. Mater. 2021, 401, 123417. [Google Scholar] [CrossRef]
- WHO Statement on Heated Tobacco Products and the US FDA Decision Regarding IQOS. Available online: https://www.who.int/news/item/27-07-2020-who-statement-on-heated-tobacco-products-and-the-us-fda-decision-regarding-iqos (accessed on 19 October 2020).
- Noguez, R. Philip Morris Celebra Fallo de la Corte Para Venta de IQOS en Sanborns. Available online: https://www.forbes.com.mx/negocios-philip-morris-fallo-corte-venta-iqos-sanborns/ (accessed on 1 February 2021).
- Rodríguez, L.S. Philip Morris Aprovecha Rezago Regulatorio para Vender IQOS en México. Available online: https://aristeguinoticias.com/2705/mexico/philip-morris-aprovecha-rezago-regulatorio-para-vender-iqos-en-mexico/ (accessed on 1 February 2021).
- Gobierno de México Secretaria de Gobernación Decreto por el que se Modifica la Tarifa de la Ley de los Impuestos Generales de Importación y de Exportación. Available online: http://www.dof.gob.mx/nota_detalle.php?codigo=5586899&fecha=19/02/2020 (accessed on 22 September 2020).
- Suprema Corte de Justicia de la Nación La comercialización de los aparatos denominados “cigarros electrónicos” es legalmente posible únicamente cuando su funcionamiento sea para el uso de productos del tabaco. Segunda Sala 2020.
- PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/ (accessed on 25 November 2020).
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Aria, M.; Cuccurullo, C. bibliometrix: An R-tool for comprehensive science mapping analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- RStudio Team. RStudio: Integrated Development Environment for R 2021. Available online: https://www.rstudio.com/products/team/ (accessed on 17 December 2020).
- Herr, C.; Tsitouras, K.; Niederstraßer, J.; Backes, C.; Beisswenger, C.; Dong, L.; Guillot, L.; Keller, A.; Bals, R. Cigarette smoke and electronic cigarettes differentially activate bronchial epithelial cells. Respir. Res. 2020, 21, 67. [Google Scholar] [CrossRef]
- Higham, A.; Bostock, D.; Booth, G.; Dungwa, J.V.; Singh, D. The effect of electronic cigarette and tobacco smoke exposure on COPD bronchial epithelial cell inflammatory responses. Int. J. COPD 2018, 13, 989–1000. [Google Scholar] [CrossRef]
- Ganapathy, V.; Manyanga, J.; Brame, L.; McGuire, D.; Sadhasivam, B.; Floyd, E.; Rubenstein, D.A.; Ramachandran, I.; Wagener, T.; Queimado, L. Electronic cigarette aerosols suppress cellular antioxidant defenses and induce significant oxidative DNA damage. PLoS ONE 2017, 12, e0177780. [Google Scholar] [CrossRef]
- Gerloff, J.; Sundar, I.K.; Freter, R.; Sekera, E.R.; Friedman, A.E.; Robinson, R.; Pagano, T.; Rahman, I. Inflammatory Response and Barrier Dysfunction by Different e-Cigarette Flavoring Chemicals Identified by Gas Chromatography–Mass Spectrometry in e-Liquids and e-Vapors on Human Lung Epithelial Cells and Fibroblasts. Appl. Vitr. Toxicol. 2017, 3, 28–40. [Google Scholar] [CrossRef]
- Crotty Alexander, L.E.; Drummond, C.A.; Hepokoski, M.; Mathew, D.; Moshensky, A.; Willeford, A.; Das, S.; Singh, P.; Yong, Z.; Lee, J.H.; et al. Chronic inhalation of e-cigarette vapor containing nicotine disrupts airway barrier function and induces systemic inflammation and multiorgan fibrosis in mice. Am. J. Physiol-Regul. Integr. Comp. Physiol. 2018, 314, R834–R847. [Google Scholar] [CrossRef]
- Wu, Q.; Jiang, D.; Minor, M.; Chu, H.W. Electronic cigarette liquid increases inflammation and virus infection in primary human airway epithelial cells. PLoS ONE 2014, 9, e108342. [Google Scholar] [CrossRef]
- Rowell, T.R.; Keating, J.E.; Zorn, B.T.; Glish, G.L.; Shears, S.B.; Tarran, R. Flavored e-liquids increase cytoplasmic Ca 2+ levels in airway epithelia. Am. J. Physiol. Cell. Mol. Physiol. 2020, 318, L226–L241. [Google Scholar] [CrossRef]
- Vasanthi Bathrinarayanan, P.; Brown, J.E.P.; Marshall, L.J.; Leslie, L.J. An investigation into E-cigarette cytotoxicity in-vitro using a novel 3D differentiated co-culture model of human airways. Toxicol. Vitr. 2018, 52, 255–264. [Google Scholar] [CrossRef]
- Miyashita, L.; Suri, R.; Dearing, E.; Mudway, I.; Dove, R.E.; Neill, D.R.; Van Zyl-Smit, R.; Kadioglu, A.; Grigg, J. E-cigarette vapour enhances pneumococcal adherence to airway epithelial cells. Eur. Respir. J. 2018, 51, 1701592. [Google Scholar] [CrossRef]
- Tayyarah, R. E-cigarretes task force Technical report: 2014 Electronic Cigarette Aerosol Parameters Study. Coop. Cent. Sci. Res. Relat. Tob. 2015. Available online: https://www.coresta.org/2014-electronic-cigarette-aerosol-parameters-study-29232.html (accessed on 17 December 2020).
- Rankin, G.D.; Wingfors, H.; Uski, O.; Hedman, L.; Ekstrand-Hammarström, B.; Bosson, J.; Lundbäck, M. The toxic potential of a fourth-generation E-cigarette on human lung cell lines and tissue explants. J. Appl. Toxicol. 2019, 39, 1143–1154. [Google Scholar] [CrossRef]
- Hung, P.; Savidge, M.; De, M.; Kang, J.; Healy, S.M.; Valerio, L.G. In vitro and in silico genetic toxicity screening of flavor compounds and other ingredients in tobacco products with emphasis on ENDS. J. Appl. Toxicol. 2020. [Google Scholar] [CrossRef]
- Clapp, P.W.; Pawlak, E.A.; Lackey, J.T.; Keating, J.E.; Reeber, S.L.; Glish, G.L.; Jaspers, I. Flavored e-cigarette liquids and cinnamaldehyde impair respiratory innate immune cell function. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2017, 313, L278–L292. [Google Scholar] [CrossRef]
- Bitzer, Z.T.; Goel, R.; Reilly, S.M.; Elias, R.J.; Silakov, A.; Foulds, J.; Muscat, J.; Richie, J.P. Effect of flavoring chemicals on free radical formation in electronic cigarette aerosols. Free Radic. Biol. Med. 2018, 120, 72–79. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, Y.; Sisler, J.D.; Shaffer, J.; Leonard, S.S.; Morris, A.M.; Qian, Y.; Bello, D.; Demokritou, P. Assessment of reactive oxygen species generated by electronic cigarettes using acellular and cellular approaches. J. Hazard. Mater. 2018, 344, 549–557. [Google Scholar] [CrossRef]
- Reilly, S.M.; Bitzer, Z.T.; Goel, R.; Trushin, N.; Richie, J.P. Free Radical, Carbonyl, and Nicotine Levels Produced by Juul Electronic Cigarettes. Nicotine Tob. Res. 2019, 21, 1274–1278. [Google Scholar] [CrossRef]
- Hwang, J.H.; Lyes, M.; Sladewski, K.; Enany, S.; McEachern, E.; Mathew, D.P.; Das, S.; Moshensky, A.; Bapat, S.; Pride, D.T.; et al. Electronic cigarette inhalation alters innate immunity and airway cytokines while increasing the virulence of colonizing bacteria. J. Mol. Med. 2016, 94, 667–679. [Google Scholar] [CrossRef]
- Higham, A.; Rattray, N.J.W.; Dewhurst, J.A.; Trivedi, D.K.; Fowler, S.J.; Goodacre, R.; Singh, D. Electronic cigarette exposure triggers neutrophil inflammatory responses. Respir. Res. 2016, 17, 56. [Google Scholar] [CrossRef]
- Gellatly, S.; Pavelka, N.; Crue, T.; Schweitzer, K.S.; Day, B.J.; Min, E.; Numata, M.; Voelker, D.R.; Scruggs, A.; Petrache, I.; et al. Nicotine-Free e-Cigarette Vapor Exposure Stimulates IL6 and Mucin Production in Human Primary Small Airway Epithelial Cells. J. Inflamm. Res. 2020, 13, 175–185. [Google Scholar] [CrossRef]
- Son, Y.; Mishin, V.; Laskin, J.D.; Mainelis, G.; Wackowski, O.A.; Delnevo, C.; Schwander, S.; Khlystov, A.; Samburova, V.; Meng, Q. Hydroxyl Radicals in E-Cigarette Vapor and E-Vapor Oxidative Potentials under Different Vaping Patterns. Chem. Res. Toxicol. 2019, 32, 1087–1095. [Google Scholar] [CrossRef]
- Husari, A.; Shihadeh, A.; Talih, S.; Hashem, Y.; El Sabban, M.; Zaatari, G. Acute Exposure to Electronic and Combustible Cigarette Aerosols: Effects in an Animal Model and in Human Alveolar Cells. Nicotine Tob. Res. 2016, 18, 613–619. [Google Scholar] [CrossRef]
- Scott, A.; Lugg, S.T.; Aldridge, K.; Lewis, K.E.; Bowden, A.; Mahida, R.Y.; Grudzinska, F.S.; Dosanjh, D.; Parekh, D.; Foronjy, R.; et al. Pro-inflammatory effects of e-cigarette vapour condensate on human alveolar macrophages. Thorax 2018, 73, 1161–1169. [Google Scholar] [CrossRef]
- Yu, V.; Rahimy, M.; Korrapati, A.; Xuan, Y.; Zou, A.E.; Krishnan, A.R.; Tsui, T.; Aguilera, J.A.; Advani, S.; Crotty Alexander, L.E.; et al. Electronic cigarettes induce DNA strand breaks and cell death independently of nicotine in cell lines. Oral Oncol. 2016, 52, 58–65. [Google Scholar] [CrossRef]
- Bahmed, K.; Lin, C.R.; Simborio, H.; Karim, L.; Aksoy, M.; Kelsen, S.; Tomar, D.; Madesh, M.; Elrod, J.; Messier, E.; et al. The role of DJ-1 in human primary alveolar type II cell injury induced by e-cigarette aerosol. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 317, L475–L485. [Google Scholar] [CrossRef]
- Smith, K.R.; Hayat, F.; Andrews, J.F.; Migaud, M.E.; Gassman, N.R. Dihydroxyacetone Exposure Alters NAD(P)H and Induces Mitochondrial Stress and Autophagy in HEK293T Cells. Chem. Res. Toxicol. 2019, 32, 1722–1731. [Google Scholar] [CrossRef]
- Sussan, T.E.; Gajghate, S.; Thimmulappa, R.K.; Ma, J.; Kim, J.H.; Sudini, K.; Consolini, N.; Cormier, S.A.; Lomnicki, S.; Hasan, F.; et al. Exposure to electronic cigarettes impairs pulmonary anti-bacterial and anti-viral defenses in a mouse model. PLoS ONE 2015, 10, e0116861. [Google Scholar] [CrossRef] [PubMed]
- Poulianiti, K.; Karatzaferi, C.; Flouris, A.D.; Fatouros, I.G.; Koutedakis, Y.; Jamurtas, A.Z. Antioxidant responses following active and passive smoking of tobacco and electronic cigarettes. Toxicol. Mech. Methods 2016, 26, 446–452. [Google Scholar] [CrossRef]
- Salama, S.A.; Arab, H.H.; Omar, H.A.; Maghrabi, I.A.; Snapka, R.M. Nicotine mediates hypochlorous acid-induced nuclear protein damage in mammalian cells. Inflammation 2014, 37, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, A.; Thompson, P.R.; Segal, B.H.; Urban, C.F. Nicotine induces neutrophil extracellular traps. J. Leukoc. Biol. 2016, 100, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Shivalingappa, P.C.; Hole, R.; Van Westphal, C.; Vij, N. Airway exposure to e-cigarette vapors impairs autophagy and induces aggresome formation. Antioxid. Redox Signal. 2016, 24, 186–204. [Google Scholar] [CrossRef]
- Bharadwaj, S.; Mitchell, R.J.; Qureshi, A.; Niazi, J.H. Toxicity evaluation of e-juice and its soluble aerosols generated by electronic cigarettes using recombinant bioluminescent bacteria responsive to specific cellular damages. Biosens. Bioelectron. 2017, 90, 53–60. [Google Scholar] [CrossRef]
- Gilpin, D.F.; McGown, K.-A.; Gallagher, K.; Bengoechea, J.; Dumigan, A.; Einarsson, G.; Elborn, J.S.; Tunney, M.M. Electronic cigarette vapour increases virulence and inflammatory potential of respiratory pathogens. Respir. Res. 2019, 20, 267. [Google Scholar] [CrossRef]
- Lerner, C.A.; Sundar, I.K.; Yao, H.; Gerloff, J.; Ossip, D.J.; McIntosh, S.; Robinson, R.; Rahman, I. Vapors produced by electronic cigarettes and E-juices with flavorings induce toxicity, oxidative stress, and inflammatory response in lung epithelial cells and in mouse lung. PLoS ONE 2015, 10, e0116732. [Google Scholar] [CrossRef]
- Ween, M.P.; Whittall, J.J.; Hamon, R.; Reynolds, P.N.; Hodge, S.J. Phagocytosis and Inflammation: Exploring the effects of the components of E-cigarette vapor on macrophages. Physiol. Rep. 2017, 5, e13370. [Google Scholar] [CrossRef] [PubMed]
- Lerner, C.A.; Rutagarama, P.; Ahmad, T.; Sundar, I.K.; Elder, A.; Rahman, I. Electronic cigarette aerosols and copper nanoparticles induce mitochondrial stress and promote DNA fragmentation in lung fibroblasts. Biochem. Biophys. Res. Commun. 2016, 477, 620–625. [Google Scholar] [CrossRef] [PubMed]
- Scheffler, S.; Dieken, H.; Krischenowski, O.; Förster, C.; Branscheid, D.; Aufderheide, M. Evaluation of e-cigarette liquid vapor and mainstream cigarette smoke after direct exposure of primary human bronchial epithelial cells. Int. J. Environ. Res. Public Health 2015, 12, 3915–3925. [Google Scholar] [CrossRef] [PubMed]
- Gómez, A.-C.C.; Rodríguez-Fernández, P.; Villar-Hernández, R.; Gibert, I.; Muriel-Moreno, B.; Lacoma, A.; Prat-Aymerich, C.; Domínguez, J. E-cigarettes: Effects in phagocytosis and cytokines response against Mycobacterium tuberculosis. PLoS ONE 2020, 15, 1–15. [Google Scholar] [CrossRef]
- Lee, H.W.; Park, S.H.; Weng, M.-W.; Wang, H.T.; Huang, W.C.; Lepor, H.; Wu, X.R.; Chen, L.C.; Tang, M.-S. E-cigarette smoke damages DNA and reduces repair activity in mouse lung, heart, and bladder as well as in human lung and bladder cells. Proc. Natl. Acad. Sci. USA 2018, 115, E1560–E1569. [Google Scholar] [CrossRef]
- Wang, Q.; Ahmad Khan, N.; Muthumalage, T.; Lawyer, G.R.; McDonough, S.R.; Chuang, T.; Gong, M.; Sundar, I.K.; Rehan, V.K.; Rahman, I. Dysregulated repair and inflammatory responses by e-cigarette-derived inhaled nicotine and humectant propylene glycol in a sex-dependent manner in mouse lung. FASEB BioAdvances 2019, 1, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.B.; Kim, S.H. Inhallation of e-Cigarette Cartridge Solution Aggravates Allergen-induced Airway Inflammation and Hyper-responsiveness in Mice. Toxicol. Res. 2014, 30, 13–18. [Google Scholar] [CrossRef]
- Wang, Q.; Sundar, I.K.; Li, D.; Lucas, J.H.; Muthumalage, T.; McDonough, S.R.; Rahman, I. E-cigarette-induced pulmonary inflammation and dysregulated repair are mediated by nAChR α7 receptor: Role of nAChR α7 in SARS-CoV-2 Covid-19 ACE2 receptor regulation. Respir. Res. 2020, 21, 154. [Google Scholar] [CrossRef]
- Madison, M.C.; Landers, C.T.; Gu, B.H.; Chang, C.Y.; Tung, H.Y.; You, R.; Hong, M.J.; Baghaei, N.; Song, L.Z.; Porter, P.; et al. Electronic cigarettes disrupt lung lipid homeostasis and innate immunity independent of nicotine. J. Clin. Investig. 2019, 129, 4290–4304. [Google Scholar] [CrossRef]
- Cirillo, S.; Vivarelli, F.; Turrini, E.; Fimognari, C.; Burattini, S.; Falcieri, E.; Rocchi, M.B.L.; Cardenia, V.; Rodriguez-Estrada, M.T.; Paolini, M.; et al. The customizable e-cigarette resistance influences toxicological outcomes: Lung degeneration, inflammation and oxidative stress-induced in a rat model. Toxicol. Sci. 2019, 172, 132–145. [Google Scholar] [CrossRef]
- Canistro, D.; Vivarelli, F.; Cirillo, S.; Marquillas, C.B.; Buschini, A.; Lazzaretti, M.; Marchi, L.; Cardenia, V.; Rodriguez-Estrada, M.T.; Lodovici, M.; et al. E-cigarettes induce toxicological effects that can raise the cancer risk. Sci. Rep. 2017, 7, 2028. [Google Scholar] [CrossRef] [PubMed]
- Larcombe, A.N.; Janka, M.A.; Mullins, B.J.; Berry, L.J.; Bredin, A.; Franklin, P.J. The effects of electronic cigarette aerosol exposure on inflammation and lung function in mice. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2017, 313, L67–L79. [Google Scholar] [CrossRef]
- Taha, H.R.; Al-Sawalha, N.A.; Alzoubi, K.H.; Khabour, O.F. Effect of E-Cigarette aerosol exposure on airway inflammation in a murine model of asthma. Inhal. Toxicol. 2020, 32, 503–511. [Google Scholar] [CrossRef]
- Bhat, T.A.; Kalathil, S.G.; Bogner, P.N.; Blount, B.C.; Goniewicz, M.L.; Thanavala, Y.M. An Animal Model of Inhaled Vitamin E Acetate and EVALI-like Lung Injury. N. Engl. J. Med. 2020, 382, 1175–1177. [Google Scholar] [CrossRef]
- Voos, N.; Smith, D.; Kaiser, L.; Mahoney, M.C.; Bradizza, C.M.; Kozlowski, L.T.; Benowitz, N.L.; O’Connor, R.J.; Goniewicz, M.L. Effect of e-cigarette flavors on nicotine delivery and puffing topography: Results from a randomized clinical trial of daily smokers. Psychopharmacology 2020, 237, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.; To, V.; Talbot, P. Comparison of cytotoxicity of IQOS aerosols to smoke from Marlboro Red and 3R4F reference cigarettes. Toxicol. Vitr. 2019, 61, 104652. [Google Scholar] [CrossRef]
- Mikheev, V.B.; Buehler, S.S.; Brinkman, M.C.; Granville, C.A.; Lane, T.E.; Ivanov, A.; Cross, K.M.; Clark, P.I. The Application of Commercially Available Mobile Cigarette Topography Devices for E-cigarette Vaping Behavior Measurements. Nicotine Tob. Res. 2020, 22, 681–688. [Google Scholar] [CrossRef]
- Singh, K.P.; Lawyer, G.; Muthumalage, T.; Maremanda, K.P.; Khan, N.A.; McDonough, S.R.; Ye, D.; McIntosh, S.; Rahman, I. Systemic biomarkers in electronic cigarette users: Implications for noninvasive assessment of vaping-associated pulmonary injuries. ERJ Open Res. 2019, 5, 00182–02019. [Google Scholar] [CrossRef]
- Song, M.-A.; Reisinger, S.A.; Freudenheim, J.L.; Brasky, T.M.; Mathé, E.A.; McElroy, J.P.; Nickerson, Q.A.; Weng, D.Y.; Wewers, M.D.; Shields, P.G. Effects of Electronic Cigarette Constituents on the Human Lung: A Pilot Clinical Trial. Cancer Prev. Res. 2020, 13, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.C.; Chakladar, J.; Li, W.T.; Chen, C.; Chang, E.Y.; Wang-Rodriguez, J.; Ongkeko, W.M. Tobacco, but Not Nicotine and Flavor-Less Electronic Cigarettes, Induces ACE2 and Immune Dysregulation. Int. J. Mol. Sci. 2020, 21, 5513. [Google Scholar] [CrossRef]
- Reidel, B.; Radicioni, G.; Clapp, P.W.; Ford, A.A.; Abdelwahab, S.; Rebuli, M.E.; Haridass, P.; Alexis, N.E.; Jaspers, I.; Kesimer, M. E-Cigarette Use Causes a Unique Innate Immune Response in the Lung, Involving Increased Neutrophilic Activation and Altered Mucin Secretion. ATS J. 2018, 197, 492–501. [Google Scholar] [CrossRef]
- Carnevale, R.; Sciarretta, S.; Violi, F.; Nocella, C.; Loffredo, L.; Perri, L.; Peruzzi, M.; Marullo, A.G.M.; De Falco, E.; Chimenti, I.; et al. Acute Impact of Tobacco vs Electronic Cigarette Smoking on Oxidative Stress and Vascular Function. Chest 2016, 150, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Sakamaki-Ching, S.; Williams, M.; Hua, M.; Li, J.; Bates, S.M.; Robinson, A.N.; Lyons, T.W.; Goniewicz, M.L.; Talbot, P. Correlation between biomarkers of exposure, effect and potential harm in the urine of electronic cigarette users. BMJ Open Respir. Res. 2020, 7, e000452. [Google Scholar] [CrossRef] [PubMed]
- Mittal, A.; Baig, A.; Zulfikar, R.; Sharma, S. Chronic Vaping Related Tracheomalacia (TM): A Case of Vaping Induced Altered Innate Immunity that Culminated in Severe TM. Cureus 2020, 12, e7571. [Google Scholar] [CrossRef] [PubMed]
- Madsen, L.R.; Krarup, N.H.V.; Bergmann, T.K.; Brentzen, S.; Neghabat, S.; Duval, L.; Knudsen, S.T. A cancer that went up in smoke pulmonary reaction to e-cigarettes imitating metastatic cancer. Chest 2016, 149, e65–e67. [Google Scholar] [CrossRef]
- Suhling, H.; Welte, T.; Fuehner, T. Three Patients with Acute Pulmonary Damage Following the Use of E-Cigarettes-A Case Series. Dtsch. Arztebl. Int. 2020, 117, 177–182. [Google Scholar] [CrossRef]
- Chen, L.; Arens, R.; Chidambaram, A.G.; Capponi, S.; Alshawa, L.; Claeys, T.A.; Hayes, D.; Robinson, R.T. Vaping Associated Pulmonary Nontuberculous Mycobacteria. Lung 2021, 199, 21–27. [Google Scholar] [CrossRef]
- Aberegg, S.K.; Cirulis, M.M.; Maddock, S.D.; Freeman, A.; Keenan, L.M.; Pirozzi, C.S.; Raman, S.M.; Schroeder, J.; Mann, H.; Callahan, S.J. Clinical, Bronchoscopic, and Imaging Findings of e-Cigarette, or Vaping, Product Use-Associated Lung Injury Among Patients Treated at an Academic Medical Center. JAMA Netw. Open 2020, 3, e2019176. [Google Scholar] [CrossRef]
- Blount, B.C.; Karwowski, M.P.; Shields, P.G.; Morel-Espinosa, M.; Valentin-Blasini, L.; Gardner, M.; Braselton, M.; Brosius, C.R.; Caron, K.T.; Chambers, D.; et al. Vitamin E Acetate in Bronchoalveolar-Lavage Fluid Associated with EVALI. N. Engl. J. Med. 2020, 382, 697–705. [Google Scholar] [CrossRef]
- Layden, J.E.; Ghinai, I.; Pray, I.; Kimball, A.; Layer, M.; Tenforde, M.W.; Navon, L.; Hoots, B.; Salvatore, P.P.; Elderbrook, M.; et al. Pulmonary Illness Related to E-Cigarette Use in Illinois and Wisconsin—Final Report. N. Engl. J. Med. 2020, 382, 903–916. [Google Scholar] [CrossRef]
- Messina, M.D.; Levin, T.L.; Blumfield, E. Cytotoxic lesion of the splenium of the corpus callosum in a patient with EVALI. Clin. Imaging 2020, 66, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Chaumont, M.; Tagliatti, V.; Channan, E.M.; Colet, J.M.; Bernard, A.; Morra, S.; Deprez, G.; van Muylem, A.; Debbas, N.; Schaefer, T.; et al. Short halt in vaping modifies cardiorespiratory parameters and urine metabolome: A randomized trial. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 318, L331–L344. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bravo-Gutiérrez, O.A.; Falfán-Valencia, R.; Ramírez-Venegas, A.; Sansores, R.H.; Ponciano-Rodríguez, G.; Pérez-Rubio, G. Lung Damage Caused by Heated Tobacco Products and Electronic Nicotine Delivery Systems: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 4079. https://doi.org/10.3390/ijerph18084079
Bravo-Gutiérrez OA, Falfán-Valencia R, Ramírez-Venegas A, Sansores RH, Ponciano-Rodríguez G, Pérez-Rubio G. Lung Damage Caused by Heated Tobacco Products and Electronic Nicotine Delivery Systems: A Systematic Review. International Journal of Environmental Research and Public Health. 2021; 18(8):4079. https://doi.org/10.3390/ijerph18084079
Chicago/Turabian StyleBravo-Gutiérrez, Omar Andrés, Ramcés Falfán-Valencia, Alejandra Ramírez-Venegas, Raúl H. Sansores, Guadalupe Ponciano-Rodríguez, and Gloria Pérez-Rubio. 2021. "Lung Damage Caused by Heated Tobacco Products and Electronic Nicotine Delivery Systems: A Systematic Review" International Journal of Environmental Research and Public Health 18, no. 8: 4079. https://doi.org/10.3390/ijerph18084079
APA StyleBravo-Gutiérrez, O. A., Falfán-Valencia, R., Ramírez-Venegas, A., Sansores, R. H., Ponciano-Rodríguez, G., & Pérez-Rubio, G. (2021). Lung Damage Caused by Heated Tobacco Products and Electronic Nicotine Delivery Systems: A Systematic Review. International Journal of Environmental Research and Public Health, 18(8), 4079. https://doi.org/10.3390/ijerph18084079