Vitamin D Levels Are Reduced at the Time of Hospital Admission in Sicilian SARS-CoV-2-Positive Patients
Abstract
1. Introduction
2. Material and Methods
2.1. Data Collection
2.2. Biochemical Analyses
2.3. Statistical Analysis
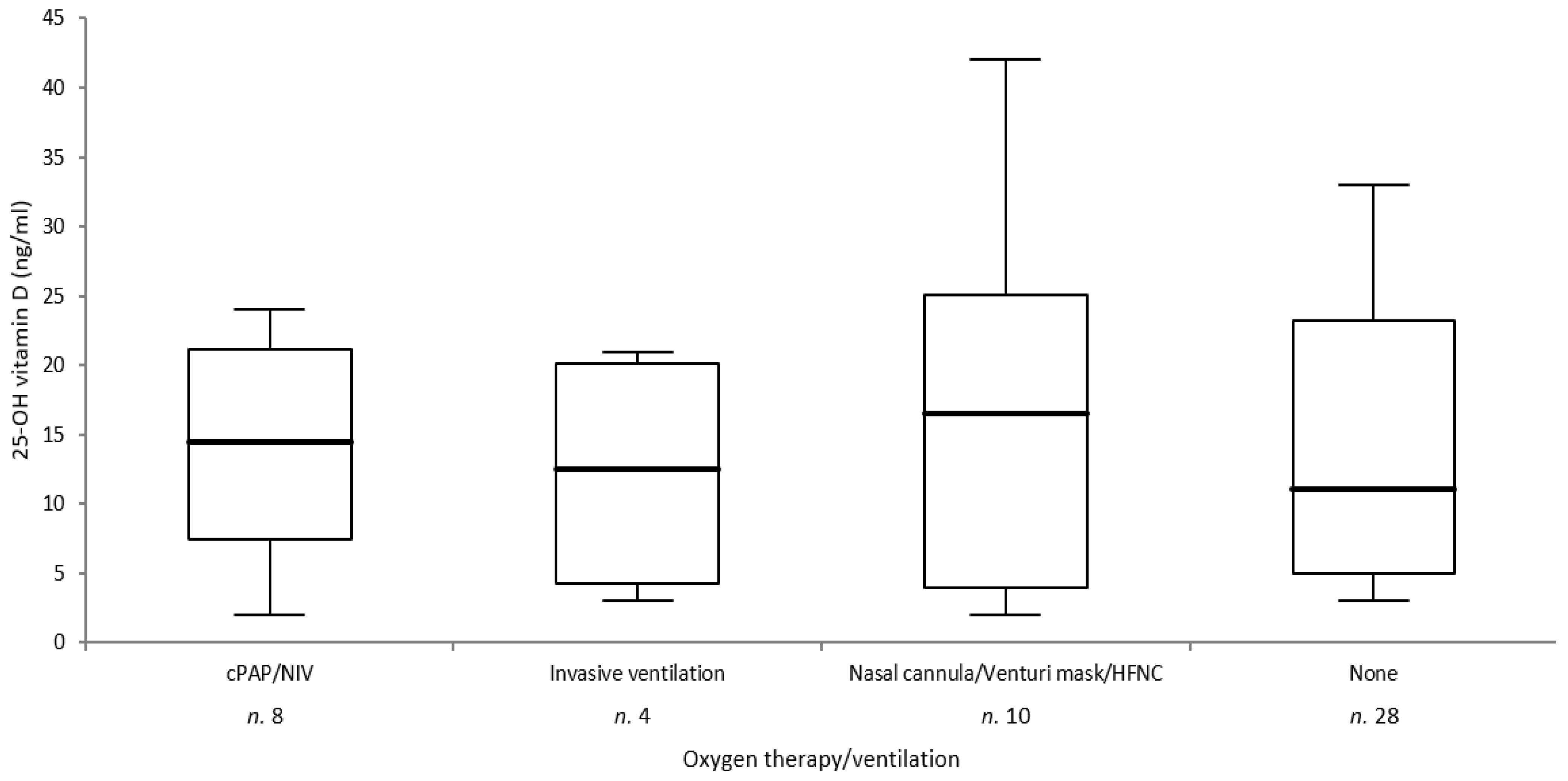
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
- Martina Barchitta, Department of Medical and Surgical Sciences and Advanced Technologies, University of Catania (Italy)
- Niccolò Castellino, Eye Clinic, University of Catania (Italy)
- Martina Di Noto, Department of Clinical and Experimental Medicine—University of Catania (Italy)
- Antonio Longo, Eye Clinic, University of Catania (Italy)
- Paola Magnano San Lio, Department of Clinical and Experimental Medicine—University of Catania (Italy)
- Rosa Manuele, San Marco Hospital, Catania (Italy)
- Elisa Marino, San Marco Hospital, Catania (Italy)
- Salvo Scuto, San Marco Hospital, Catania (Italy)
- Anastasia Xourafa, University Policlinic “G. Rodolico”, Catania (Italy)
- Luca Zanoli, Department of Clinical and Experimental Medicine—University of Catania (Italy)
- Sabrina Zocco, Department of Clinical and Experimental Medicine—University of Catania (Italy).
Conflicts of Interest
References
- Ungogo, M.A.; Mohammed, M.; Umar, B.N.; Bala, A.A.; Khalid, G.M. Review of pharmacologic and immunologic agents in the management of COVID-19. Biosaf. Health 2021. epub, ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Naik, R.R.; Shakya, A.K. Therapeutic Strategies in the Management of COVID-19. Front. Mol. Biosci. 2021, 7, 636738. [Google Scholar] [CrossRef]
- Jan, H.; Faisal, S.; Khan, A.; Khan, S.; Usman, H.; Liaqat, R.; Shah, S.A. COVID-19: Review of Epidemiology and Potential Treatments Against 2019 Novel Coronavirus. Discoveries 2020, 8, e108. [Google Scholar] [CrossRef] [PubMed]
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, Evaluation, and Treatment of Coronavirus. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Beard, J.A.; Bearden, A.; Striker, R. Vitamin D and the anti-viral state. J. Clin. Virol. 2011, 50, 194–200. [Google Scholar] [CrossRef]
- Slominski, R.M.; Stefan, J.; Athar, M.; Holick, M.F.; Jetten, A.M.; Raman, C.; Slominski, A.T. COVID-19 and Vitamin D: A lesson from the skin. Exp. Dermatol. 2020, 29, 885–890. [Google Scholar] [CrossRef]
- Bilezikian, J.P.; Bikle, D.; Hewison, M.; Lazaretti-Castro, M.; Formenti, A.M.; Gupta, A.; Madhavan, M.V.; Nair, N.; Babalyan, V.; Hutchings, N.; et al. Mechanisms in endocrinology: Vitamin D and COVID-19. Eur. J. Endocrinol. 2020, 183, R133–R147. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine Dietary Reference Intakes for Calcium and Vitamin D. Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Napoli, N.; Strollo, R.; Sprini, D.; Maddaloni, E.; Rini, G.B.; Carmina, E. Serum 25-OH Vitamin D in relation to Bone Mineral Density and Bone Turnover. Int. J. Endocrinol. 2014, 2014, 1–5. [Google Scholar] [CrossRef]
- Falchetti, A.; Sferrazza, C.; Cepollaro, C.; Gozzini, A.; Del Monte, F.; Masi, L.; Napoli, N.; Di Fede, G.; Cannone, V.; Cusumano, G.; et al. FokI Polymorphism of the Vitamin D Receptor Gene Correlates with Parameters of Bone Mass and Turnover in a Female Population of the Italian Island of Lampedusa. Calcif. Tissue Int. 2006, 80, 15–20. [Google Scholar] [CrossRef]
- Hope-Simpson, R.E. Epidemic mechanisms of Type A influenza. J. Hyg. 1979, 83, 11–26. [Google Scholar] [CrossRef]
- Cannell, J.J.; Vieth, R.; Umhau, J.C.; Holick, M.F.; Grant, W.B.; Madronich, S.; Garland, C.F.; Giovannucci, E. Epidemic influenza and vitamin D. Epidemiol. Infect. 2006, 134, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Noah, N.D. Cyclical patterns and predictability in infection. Epidemiology Infect. 1989, 102, 175–190. [Google Scholar] [CrossRef]
- Robertsen, S.; Grimnes, G.; Melbye, H. Association between serum 25-hydroxyvitamin D concentration and symptoms of respiratory tract infection in a Norwegian population: The Tromsø Study. Public Health Nutr. 2014, 17, 780–786. [Google Scholar] [CrossRef]
- Hribar, C.A.; Cobbold, P.H.; Church, F.C. Potential Role of Vitamin D in the Elderly to Resist COVID-19 and to Slow Progression of Parkinson’s Disease. Brain Sci. 2020, 10, 284. [Google Scholar] [CrossRef]
- Teymoori-Rad, M.; Shokri, F.; Salimi, V.; Marashi, S.M. The interplay between vitamin D and viral infections. Rev. Med Virol. 2019, 29, e2032. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. An update on vitamin D and human immunity. Clin. Endocrinol. 2012, 76, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Guevara, M.A.; Lu, J.; Moore, R.E.; Chambers, S.A.; Eastman, A.J.; Francis, J.D.; Noble, K.N.; Doster, R.S.; Osteen, K.G.; Damo, S.M.; et al. Vitamin D and Streptococci: The Interface of Nutrition, Host Immune Response, and Antimicrobial Activity in Response to Infection. ACS Infect. Dis. 2020, 6, 3131–3140. [Google Scholar] [CrossRef]
- Siddiqui, M.; Manansala, J.S.; Abdulrahman, H.A.; Nasrallah, G.K.; Smatti, M.K.; Younes, N.; Althani, A.A.; Yassine, H.M. Immune Modulatory Effects of Vitamin D on Viral Infections. Nutrients 2020, 12, 2879. [Google Scholar] [CrossRef]
- Bishop, E.L.; Ismailova, A.; Dimeloe, S.K.; Hewison, M.; White, J.H. Vitamin D and Immune Regulation: Antibacterial, Antiviral, Anti-Inflammatory. JBMR Plus 2021, 5, e10405. [Google Scholar] [CrossRef]
- Gruber–Bzura, B.M. Vitamin D and Influenza—Prevention or Therapy? Int. J. Mol. Sci. 2018, 19, 2419. [Google Scholar] [CrossRef]
- Bergman, P.; Lindh, Å.U.; Björkhem-Bergman, L.; Lindh, J.D. Vitamin D and Respiratory Tract Infections: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. PLoS ONE 2013, 8, e65835. [Google Scholar] [CrossRef]
- Zadshir, A.; Tareen, N.; Pan, D.; Norris, K.C.; Martins, D. The prevalence of hypovitaminosis D among US adults: Data from the NHANES III. Ethn. Dis. 2005, 15 (Suppl. 5), S5–S101. [Google Scholar]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Arboleda, J.F.; Urcuqui-Inchima, S. Vitamin D Supplementation: A Potential Approach for Coronavirus/COVID-19 Therapeutics? Front. Immunol. 2020, 11, 1523. [Google Scholar] [CrossRef]
- Rhodes, J.M.; Subramanian, S.; Laird, E.; Griffin, G.; Kenny, R.A. Perspective: Vitamin D deficiency and COVID-19 severity—plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis. J. Intern. Med. 2021, 289, 97–115. [Google Scholar] [CrossRef]
- Malek Mahdavi, A. A brief review of interplay between vitamin D and angiotensin-converting enzyme 2: Implications for a potential treatment for COVID-19. Rev. Med. Virol. 2020, 30, e2119. [Google Scholar] [CrossRef] [PubMed]
- Marik, P.E.; Kory, P.; Varon, J. Does vitamin D status impact mortality from SARS-CoV-2 infection? Med. Drug Discov. 2020, 6, 100041. [Google Scholar] [CrossRef]
- Rhodes, J.M.; Subramanian, S.; Laird, E.; Kenny, R.A. Editorial: Low population mortality from COVID-19 in countries south of latitude 35 degrees North supports vitamin D as a factor determining severity. Aliment. Pharmacol. Ther. 2020, 51, 1434–1437. [Google Scholar] [CrossRef]
- Hastie, C.E.; Mackay, D.F.; Ho, F.; Celis-Morales, C.A.; Jani, B.D.; Mair, F.S.; Gray, S.R.; O’Donnell, C.A.; Sattar, N.; Pell, J.P.; et al. Vitamin D concentrations and COVID-19 infection in UK Biobank [published correction appears in Diabetes Metab Syndr. 2020, 14, 1315–1316]. Diabetes Metab Syndr. 2020, 14, 561–565. [Google Scholar] [CrossRef]
- Ilie, P.C.; Stefanescu, S.; Smith, L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin. Exp. Res. 2020, 32, 1195–1198. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, D.O.; Best, T.J.; Zhang, H.; Vokes, T.; Arora, V.; Solway, J. Association of Vitamin D Deficiency and Treatment with COVID-19 Incidence. medRxiv. 2020, 20095893, preprint. [Google Scholar]
- D’Avolio, A.; Avataneo, V.; Manca, A.; Cusato, J.; De Nicolò, A.; Lucchini, R.; Keller, F.; Cantù, M. 25-Hydroxyvitamin D Concentrations Are Lower in Patients with Positive PCR for SARS-CoV-2. Nutrients 2020, 12, 1359. [Google Scholar] [CrossRef] [PubMed]
- Carpagnano, G.E.; Di Lecce, V.; Quaranta, V.N.; Zito, A.; Buonamico, E.; Capozza, E.; Palumbo, A.; Di Gioia, G.; Valerio, V.N.; Resta, O. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J. Endocrinol. Investig. 2021, 44, 765–771. [Google Scholar] [CrossRef]
- Munshi, R.; Hussein, M.H.; Toraih, E.A.; Elshazli, R.M.; Jardak, C.; Sultana, N.; Youssef, M.R.; Omar, M.; Attia, A.S.; Fawzy, M.S.; et al. Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J. Med Virol. 2021, 93, 733–740. [Google Scholar] [CrossRef]
- Panagiotou, G.; Tee, S.A.; Ihsan, Y.; Athar, W.; Marchitelli, G.; Kelly, D.; Boot, C.S.; Stock, N.; Macfarlane, J.; Martineau, A.R.; et al. Low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID-19 are associated with greater disease severity. Clin. Endocrinol. 2020, 93, 508–511. [Google Scholar] [CrossRef]
- Merzon, E.; Tworowski, D.; Gorohovski, A.; Vinker, S.; Cohen, A.G.; Green, I.; Frenkel-Morgenstern, M. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: An Israeli population-based study. FEBS J. 2020, 287, 3693–3702. [Google Scholar] [CrossRef]
- Mendy, A.; Apewokin, S.; Wells, A.A.; Morrow, A.L. Factors Associated with Hospitalization and Disease Severity in a Racially and Ethnically Diverse Population of COVID-19 Patients. medRxiv 2020, 20137323, preprint. [Google Scholar]
- Gavioli, E.M.; Miyashita, H.; Hassaneen, O.; Siau, E. An Evaluation of Serum 25-Hydroxy Vitamin D Levels in Patients with COVID-19 in New York City. J. Am. Coll. Nutr. 2021, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Infante, M.; Buoso, A.; Pieri, M.; Lupisella, S.; Nuccetelli, M.; Bernardini, S.; Fabbri, A.; Iannetta, M.; Andreoni, M.; Colizzi, V.; et al. Low Vitamin D Status at Admission as a Risk Factor for Poor Survival in Hospitalized Patients With COVID-19: An Italian Retrospective Study. J. Am. Coll. Nutr. 2021, 18, 1–16. [Google Scholar] [CrossRef]
- Entrenas Castillo, M.; Entrenas Costa, L.M.; Vaquero Barrios, J.M.; Díaz, J.F.A.; Miranda, J.L.; Bouillon, R.; Gomez, J.M.Q. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef]
- Annweiler, G.; Corvaisier, M.; Gautier, J.; Dubée, V.; Legrand, E.; Sacco, G.; Annweiler, C. Vitamin D Supplementation Associated to Better Survival in Hospitalized Frail Elderly COVID-19 Patients: The GERIA-COVID Quasi-Experimental Study. Nutrients 2020, 12, 3377. [Google Scholar] [CrossRef]

| Normal Values | SARS-CoV-2 Patients | Control Group | p | |||
|---|---|---|---|---|---|---|
| n | 50 | 100 | ||||
| Age, years | 65 (24–98) | 61 (22–89) | 0.23 | |||
| Male, n (%) | 26 (52) | 44 (44) | 0.35 | |||
| BMI, Kg/m2 | 18–25 | 27.2 (22.0–38.1) | 26.4 (18.0–39.0) | 0.20 | ||
| Calcium, mg/dl | 8.8–10.6 | 8.9 (6.3–9.9) | 9.5 (7.7–11.0) | <0.001 | ||
| 25-OH vitamin D, ng/ml | ≥20 | 12.5 (2–42) | 20.5 (5–46) | <0.001 | ||
| <12 ng/ml | n (%) | 23 (46) | 15 (15) | <0.001 * | ||
| ≥12, <20 ng/ml | 11 (22) | 32 (32) | 0.20 | |||
| ≥20 ng/ml | 16 (32) | 53 (53) | 0.015 * | |||
| PTH, pg/ml | 12.0–88.0 | 62 (10–215) | 47 (16–183) | 0.03 | ||
| Creatinine, mg/dl | 0.51–1.29 | 1.03 (0.38–3.52) | 0.84 (0.40–3.60) | 0.52 | ||
| Alkaline phosphatase, IU/l | 30–120 | 75 (18–206) | 63 (27–173) § | 0.37 | ||
| Phosphorus, mg/dl | 2.5–4.5 | 3.2 (1.4–5.5) | 3.4 (2.1–5.0) | 0.04 | ||
| Albumin, g/dl | 3.5–5.2 | 3.1 (0.1–4.2) | 2.8 (2.1–4.5) § | 0.34 | ||
| IL-6, pg/ml | 0.0–6.4 | 12.4 (0.9–563.6) | - | |||
| CRP, mg/l | 0.0–5.0 | 10.2 (0.1–254.1) | - | |||
| D-dimer, µg/l | 0–250 | 271 (36–3599) | - | |||
| Discharge (dead/alive) | 5/45 | - | ||||
| Oxygen therapy/ ventilation | None | n (%) | 28 (56) | - | ||
| Nasal cannula/Venturi mask/HFNC | 10 (20) | - | ||||
| cPAP/NIV | 8 (16) | - | ||||
| Invasive ventilation | 4 (8) | - | ||||
| Length of hospitalization, days | 25 (7–94) | - | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaudio, A.; Murabito, A.R.; Agodi, A.; Montineri, A.; Castellino, P.; D.O.CoV Research. Vitamin D Levels Are Reduced at the Time of Hospital Admission in Sicilian SARS-CoV-2-Positive Patients. Int. J. Environ. Res. Public Health 2021, 18, 3491. https://doi.org/10.3390/ijerph18073491
Gaudio A, Murabito AR, Agodi A, Montineri A, Castellino P, D.O.CoV Research. Vitamin D Levels Are Reduced at the Time of Hospital Admission in Sicilian SARS-CoV-2-Positive Patients. International Journal of Environmental Research and Public Health. 2021; 18(7):3491. https://doi.org/10.3390/ijerph18073491
Chicago/Turabian StyleGaudio, Agostino, Andrea Ruben Murabito, Antonella Agodi, Arturo Montineri, Pietro Castellino, and D.O.CoV Research. 2021. "Vitamin D Levels Are Reduced at the Time of Hospital Admission in Sicilian SARS-CoV-2-Positive Patients" International Journal of Environmental Research and Public Health 18, no. 7: 3491. https://doi.org/10.3390/ijerph18073491
APA StyleGaudio, A., Murabito, A. R., Agodi, A., Montineri, A., Castellino, P., & D.O.CoV Research. (2021). Vitamin D Levels Are Reduced at the Time of Hospital Admission in Sicilian SARS-CoV-2-Positive Patients. International Journal of Environmental Research and Public Health, 18(7), 3491. https://doi.org/10.3390/ijerph18073491








