Racial Disparities in Post-Acute Home Health Care Referral and Utilization among Older Adults with Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Data Sources
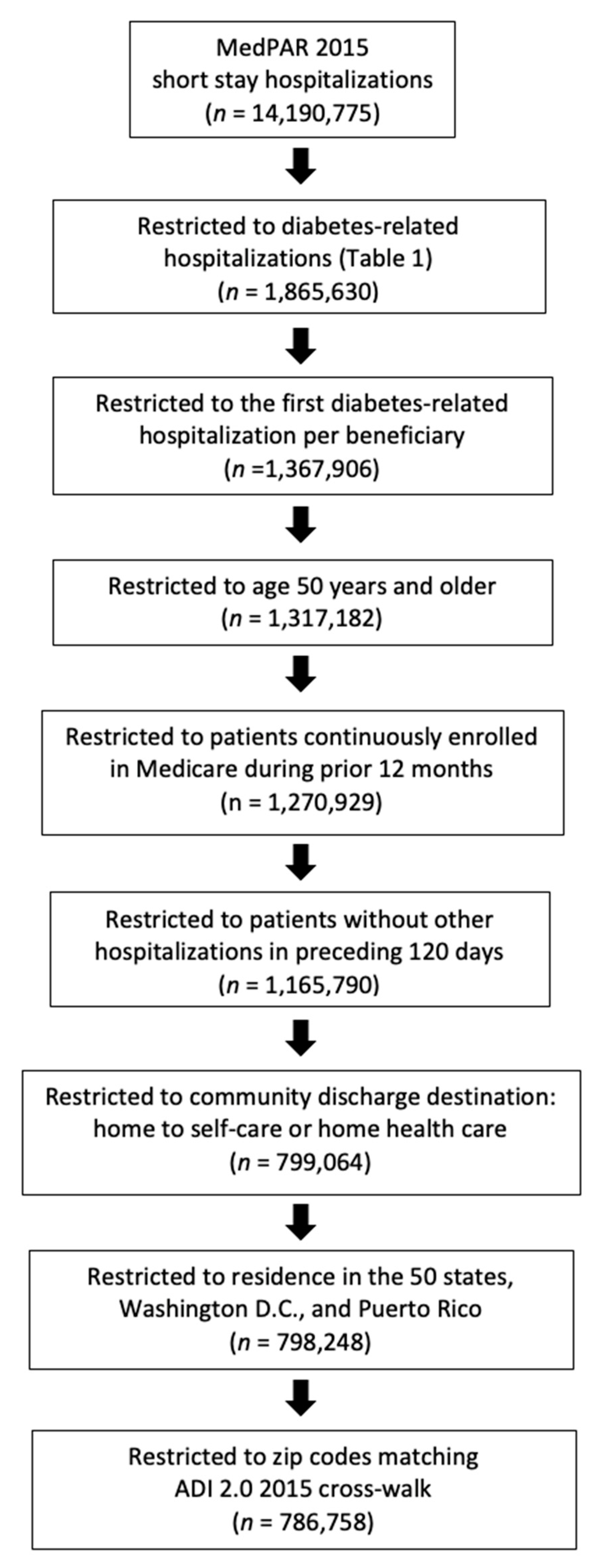
2.2. Study Population
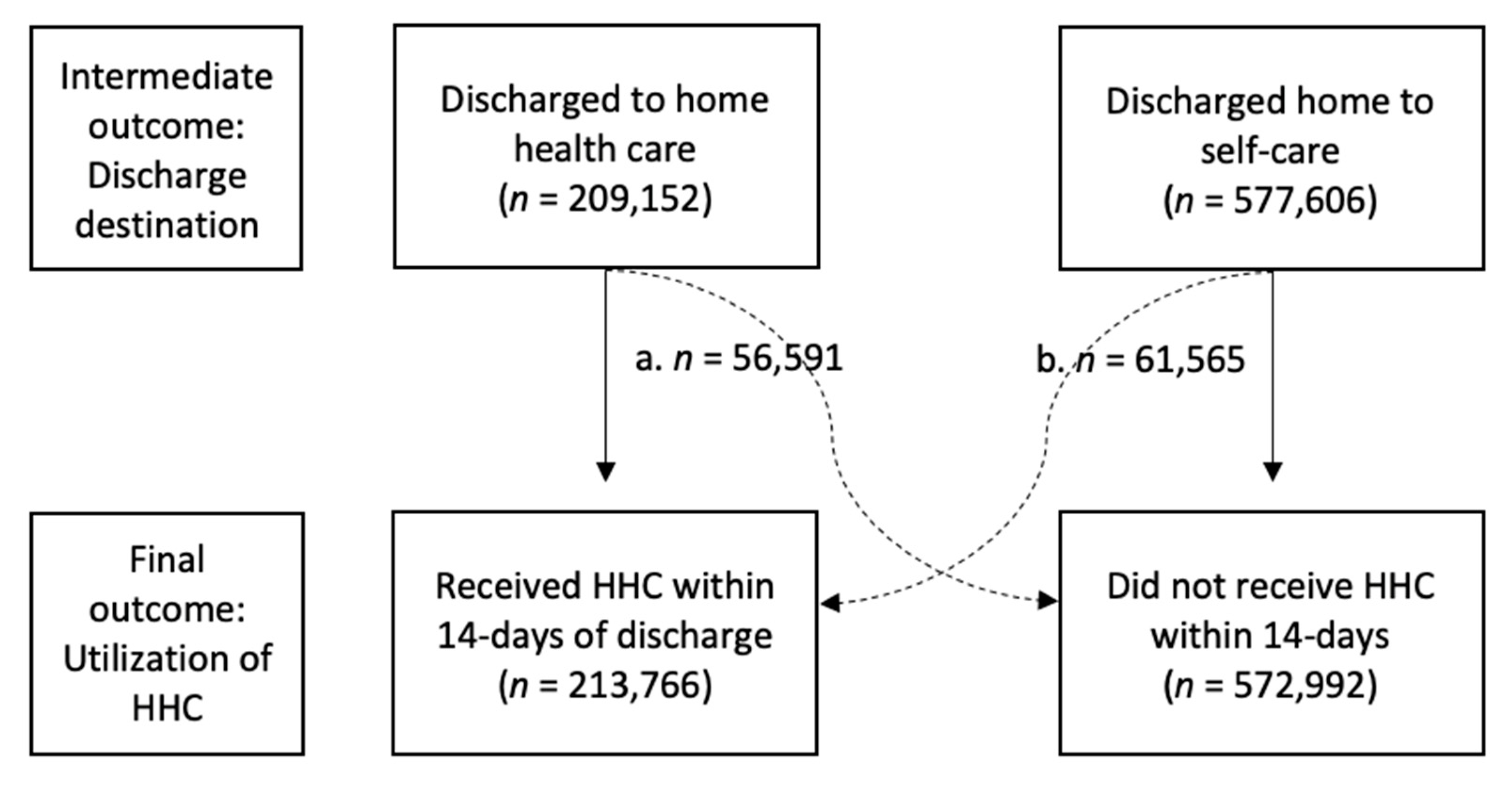
2.3. Outcomes
2.4. Individual-Level Variables
2.5. Geographic-Level Variables
2.6. Analytic Approach
3. Results
3.1. Study Population
3.2. Sample Characteristics Stratified by Race, Neighborhood Profile, and Insurance
3.3. Predictors of Hospital Discharge to Home Health Care
3.4. Predictors of Home Health Care Use after Index Hospital Discharge (First 14-Days)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control. National Diabetes Statistics Report. 2020. Available online: https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf (accessed on 20 December 2020).
- Collins, J.; Abbass, I.M.; Harvey, R.; Suehs, B.; Uribe, C.; Bouchard, J.; Prewitt, R.; DeLuzio, T.; Allen, E. Predictors of all-cause 30 day readmission among Medicare patients with type 2 diabetes. Curr. Med. Res. Opin. 2017, 33, 1517–1523. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, K.N.; McHugh, S.M.; Tracey, M.L.; Fitzgerald, A.P.; Kearney, P.M. Health service utilization and related costs attributable to diabetes. Diabet. Med. 2018, 35, 1727–1734. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, L.M.; Shrestha, D.P.; Rosenthal, M.B.; Hollenbeak, C.S.; Gabbay, R.A. Risk factors associated with 30-day readmission and length of stay in patients with type 2 diabetes. J. Diabetes Complicat. 2017, 31, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Ostling, S.; Wyckoff, J.; Ciarkowski, S.L.; Pai, C.; Choe, H.M.; Bahl, V.; Gianchandani, R. The relationship between diabetes mellitus and 30-day readmission rates. Clin. Diabetes Endocrinol. 2017, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- LaManna, J.B.; Bushy, A.; Norris, A.E.; Chase, S.K. Early and intermediate hospital-to-home transition outcomes of older adults diagnosed with diabetes. Diabetes Educ. 2016, 42, 72–86. [Google Scholar] [CrossRef] [PubMed]
- Lysy, Z.; Fung, K.; Giannakeas, V.; Fischer, H.D.; Bell, C.M.; Lipscombe, L.L. The association between insulin initiation and adverse outcomes after hospital discharge in older adults: A population-based cohort study. J. Gen. Intern. Med. 2019, 34, 575–582. [Google Scholar] [CrossRef]
- Topaz, M.; Trifilio, M.; Maloney, D.; Bar-Bachar, O.; Bowles, K.H. Improving patient prioritization during hospital-homecare transition: A pilot study of a clinical decision support tool. Res. Nurs. Health 2018, 41, 440–447. [Google Scholar] [CrossRef]
- Murtaugh, C.M.; Deb, P.; Zhu, C.; Peng, T.R.; Barrón, Y.; Shah, S.; Moore, S.M.; Bowles, K.H.; Kalman, J.; Feldman, P.H.; et al. Reducing readmissions among heart failure patients discharged to home health care: Effectiveness of early and intensive nursing services and early physician follow-up. Health Serv. Res. 2017, 52, 1445–1472. [Google Scholar] [CrossRef]
- Wang, J.; Liebel, D.V.; Yu, F.; Caprio, T.V.; Shang, J. Inverse dose-response relationship between home health care services and rehospitalization in older adults. J. Am. Med. Dir. Assoc. 2019, 20, 736–742. [Google Scholar] [CrossRef]
- Hildebrand, J.A.; Billimek, J.; Lee, J.A.; Sorkin, D.H.; Olshansky, E.F.; Clancy, S.L.; Evangelista, L.S. Effect of diabetes self-management education on glycemic control in Latino adults with type 2 diabetes: A systematic review and meta-analysis. Patient Educ. Couns. 2020, 103, 266–275. [Google Scholar] [CrossRef]
- Cunningham, A.T.; Crittendon, D.R.; White, N.; Mills, G.D.; Diaz, V.; LaNoue, M.D. The effect of diabetes self-management education on HbA1c and quality of life in African-Americans: A systematic review and meta-analysis. BMC Health Serv. Res. 2018, 18, 367. [Google Scholar] [CrossRef]
- Beck, J.; Greenwood, D.A.; Blanton, L.; Bollinger, S.T.; Butcher, M.K.; Condon, J.E.; Cypress, M.; Faulkner, P.; Fischl, A.H.; Francis, T.; et al. 2017 national standards for diabetes self-management education and support. Diabetes Spectr. 2017, 30, 301–314. [Google Scholar] [CrossRef]
- Bleich, S.N.; Findling, M.G.; Casey, L.S.; Blendon, R.J.; Benson, J.M.; Steelfisher, G.K.; Sayde, J.M.; Miller, C. Discrimination in the United States: Experiences of Black Americans. Health Serv. Res. 2019, 54 (Suppl. 2), 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Findling, M.G.; Casey, L.S.; Fryberg, S.A.; Hafner, S.; Blendon, R.J.; Benson, J.M.; Sayde, J.M.; Miller, C. Discrimination in the United States: Experiences of Native Americans. Health Serv. Res. 2019, 54 (Suppl. 2), 1431–1441. [Google Scholar] [CrossRef] [PubMed]
- Findling, M.G.; Bleich, S.N.; Casey, L.S.; Blendon, R.J.; Benson, J.M.; Sayde, J.M.; Miller, C. Discrimination in the United States: Experiences of Latinos. Health Serv. Res. 2019, 54 (Suppl. 2), 1409–1418. [Google Scholar] [CrossRef]
- McMurtry, C.L.; Findling, M.G.; Casey, L.S.; Blendon, R.J.; Benson, J.M.; Sayde, J.M.; Miller, C. Discrimination in the United States: Experiences of Asian Americans. Health Serv. Res. 2019, 54 (Suppl. 2), 1419–1430. [Google Scholar] [CrossRef] [PubMed]
- Sefcik, J.S.; Nock, R.H.; Flores, E.J.; Chase, J.D.; Bradway, C.; Potashnik, S.; Bowles, K.H. Patient preferences for information on post-acute care services. Res. Gerontol. Nurs. 2016, 9, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Sefcik, J.S.; Ritter, A.Z.; Flores, E.J.; Nock, R.H.; Chase, J.D.; Bradway, C.; Potashnik, S.; Bowles, K.H. Why older adults may decline offers of post-acute care services: A qualitative descriptive study. Geriatr. Nurs. 2017, 38, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Bowles, K.H.; Ratcliffe, S.J.; Holmes, J.H.; Keim, S.; Potashnik, S.; Flores, E.; Humbrecht, D.; Whitehouse, C.R.; Naylor, M.D. Using a decision support algorithm for referrals to post-acute care. J. Am. Med. Dir. Assoc. 2019, 20, 408–413. [Google Scholar] [CrossRef]
- Gaskin, D.J.; Roberts, E.T.; Chan, K.S.; McCleary, R.; Buttorff, C.; Delarmente, B.A. No man is an island: The impact of neighborhood disadvantage on mortality. Int. J. Environ. Res. Public Health 2019, 16, 1265. [Google Scholar] [CrossRef]
- Tsui, J.; Hirsch, J.A.; Bayer, F.J.; Quinn, J.W.; Cahill, J.; Siscovick, D.; Lovasi, G.S. Patterns in geographic access to health care facilities across neighborhoods in the United States based on data from the National Establishment Time-Series between 2000 and 2014. JAMA Netw. Open 2020, 3, e205105. [Google Scholar] [CrossRef]
- Kind, A.J.H.; Golden, R.N. Social determinants of health: Fundamental drivers of health inequity. WMJ 2018, 117, 231–232. [Google Scholar]
- Kind, A.J.H.; Jencks, S.; Brock, J.; Yu, M.; Bartels, C.; Ehlenbach, W.; Greenberg, C.; Smith, M. Neighborhood socioeconomic disadvantage and 30-day rehospitalization: A retrospective cohort study. Ann. Intern. Med. 2014, 161, 765–774. [Google Scholar] [CrossRef]
- Joynt Maddox, K.E.; Chen, L.M.; Zuckerman, R.; Epstein, A.M. Association between race, neighborhood, and Medicaid enrollment and outcomes in Medicare home health care. J. Am. Geriatr. Soc. 2018, 66, 239–246. [Google Scholar] [CrossRef]
- Durfey, S.N.M.; Kind, A.J.H.; Buckingham, W.R.; DuGoff, E.H.; Trivedi, A.N. Neighborhood disadvantage and chronic disease management. Health Serv. Res. 2019, 54, 206–216. [Google Scholar] [CrossRef]
- Medicare Payment Advisory Commission. Report to the Congress: Medicare Payment Policy; Chapter 13 The Medicare Advantage Program: Status Report; MedPAC: Washington, DC, USA, 2020; pp. 365–401. Available online: http://medpac.gov/docs/default-source/reports/mar20_entirereport_sec.pdf (accessed on 21 December 2020).
- Andersen, R.; Newman, J.F. Societal and individual determinants of medical care utilization in the United States. Milbank Meml. Fund Q. Health Soc. 1973, 51, 95–124. [Google Scholar] [CrossRef]
- Jiang, H.J.; Andrews, R.; Stryer, D.; Friedman, B. Racial/ethnic disparities in potentially preventable readmissions: The case of diabetes. Am. J. Public Health 2005, 95, 1561–1567. [Google Scholar] [CrossRef] [PubMed]
- Ogunwole, S.M.; Golden, S.H. Social determinants of health and structural inequities- Root causes of diabetes disparities. Diabetes Care 2021, 44, 11–13. [Google Scholar] [CrossRef] [PubMed]
- Moore, B.J.; White, S.; Washington, R.; Coenen, N.; Elixhauser, A. Identifying increased risk of readmission and in-hospital mortality using hospital administrative data: The AHRQ Elixhauser Comorbidity Index. Med. Care 2017, 55, 698–705. [Google Scholar] [CrossRef] [PubMed]
- Jarrín, O.F.; Nyandege, A.N.; Grafova, I.B.; Dong, X.; Lin, H. Validity of race and ethnicity codes in Medicare administrative data compared with gold-standard self-reported race collected during routine home health care visits. Med. Care 2020, 58, e1–e8. [Google Scholar] [CrossRef]
- Jones, C.D.; Wald, H.L.; Boxer, R.S.; Masoudi, F.A.; Burke, R.E.; Capp, R.; Coleman, E.A.; Ginde, A.A. Characteristics associated with home health care referrals at hospital discharge: Results from the 2012 National Inpatient Sample. Health Serv. Res. 2017, 52, 879–894. [Google Scholar] [CrossRef]
- University of Wisconsin School of Medicine and Public Health. Neighborhood Atlas. 2020. Available online: https://www.neighborhoodatlas.medicine.wisc.edu/ (accessed on 22 December 2020).
- U.S. Department of Agriculture. Rural-Urban Continuum Codes Documentation. 2013. Available online: https://www.ers.usda.gov/data-products/rural-urban-continuum-codes/documentation/ (accessed on 22 December 2020).
- Hosmer, D.W.; Lemeshow, S.; Sturdivant, R.X. Applied Logistic Regression, 3rd ed.; Wiley Series in Probability and Statistics; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013. [Google Scholar]
- Towne, S.D.; Bolin, J.; Ferdinand, A.; Nicklett, E.J.; Smith, M.L.; Ory, M.G. Assessing diabetes and factors associated with foregoing medical care among persons with diabetes: Disparities facing American Indian/Alaska Native, Black, Hispanic, low income, and Southern adults in the U.S. (2011–2015). Int. J. Environ. Res. Public Health 2017, 14, 464. [Google Scholar] [CrossRef]
- Adakai, M.; Sandoval-Rosario, M.; Xu, F.; Aseret-Manygoats, T.; Allison, M.; Greenlund, K.J.; Barbour, K.E. Health disparities among American Indians/Alaska Natives—Arizona, 2017. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 1314–1318. [Google Scholar] [CrossRef] [PubMed]
- King, C.; Atwood, S.; Lozada, M.; Nelson, A.K.; Brown, C.; Sabo, S.; Curley, C.; Muskett, O.; Orav, E.J.; Shin, S. Identifying risk factors for 30-day readmission events among American Indian patients with diabetes in the Four Corners region of the southwest from 2009 to 2016. PLoS ONE. 2018, 13, e0195476. [Google Scholar] [CrossRef]
- Franz, C.; Atwood, S.; Orav, E.J.; Curley, C.; Brown, C.; Trevisi, L.; Nelson, A.K.; Begay, M.; Shin, S. Community-based outreach associated with increased health utilization among Navajo individuals living with diabetes: A matched cohort study. BMC Health Serv. Res. 2020, 20, 460. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, K.; Lim, E.; Davis, J.; Chen, J.J. Racial disparities in health service utilization among Medicare fee-for-service beneficiaries adjusting for multiple chronic conditions. J. Aging Health 2018, 30, 1224–1243. [Google Scholar] [CrossRef]
- Li, J.; Qi, M.; Werner, R.M. Assessment of receipt of the first home health care visit after hospital discharge among older adults. JAMA Netw. Open 2020, 3, e2015470. [Google Scholar] [CrossRef] [PubMed]
- Hazel-Fernandez, L.; Li, Y.; Nero, D.; Moretz, C.; Slabaugh, L.; Meah, Y.; Baltz, J.; Costantino, M.; Patel, N.C.; Bouchard, J. Racial/ethnic and gender differences in severity of diabetes-related complications, health care resource use, and costs in a Medicare population. Popul. Health Manag. 2015, 18, 115–122. [Google Scholar] [CrossRef]
- Li, Q.; Rahman, M.; Gozalo, P.; Keohane, L.M.; Gold, M.R.; Trivedi, A.N. Regional variations: The use of hospitals, home health, and skilled nursing in traditional Medicare and Medicare Advantage. Health Aff. 2018, 37, 1274–1281. [Google Scholar] [CrossRef]
- Skopec, L.; Huckfeldt, P.J.; Wissoker, D.; Aarons, J.; Dey, J.; Oliveira, I.; Zuckerman, S. Home health and postacute care use in Medicare Advantage and traditional Medicare. Health Aff. 2020, 39, 837–842. [Google Scholar] [CrossRef]
- Skopec, L.; Zuckerman, S.; Aarons, J.; Wissoker, D.; Huckfeldt, P.J.; Feder, J.; Berenson, R.A.; Dey, J.; Oliveira, I. Home health use in Medicare Advantage compared to use in traditional Medicare. Health Aff. 2020, 39, 1072–1079. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Tarraf, W.; Maroukis, B.L.; Levy, H.G. Does Medicare Managed Care reduce racial/ethnic disparities in diabetes preventive care and healthcare expenditures? Am. J. Manag. Care 2016, 22, e360–e367. [Google Scholar]
- Li, Q.; Keohane, L.M.; Thomas, K.; Lee, Y.; Trivedi, A.N. Association of cost sharing with use of home health services among Medicare Advantage enrollees. JAMA Intern. Med. 2017, 177, 1012–1018. [Google Scholar] [CrossRef][Green Version]
- Freed, M.; Damico, A.; Neuman, T. A Dozen Facts about Medicare Advantage in 2020. Kaiser Family Foundation. 2020. Available online: https://www.kff.org/medicare/issue-brief/a-dozen-facts-about-medicare-advantage-in-2020/ (accessed on 21 December 2020).
- Jones, C.P. Confronting institutionalized racism. Phylon 2002, 50, 7–22. [Google Scholar] [CrossRef]
- Golden, S.H.; Joseph, J.J.; Hill-Briggs, F. Casting a health equity lens on endocrinology and diabetes. J. Clin. Endocrinol. Metab. 2021. pre-publication. [Google Scholar] [CrossRef]
- Davitt, J.K.; Bourjolly, J.; Frasso, R. Understanding inequities in home health care outcomes staff views on agency and system factors. Res. Gerontol. Nurs. 2015, 8, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; DuGoff, E.H.; Novak, P.; Wang, M.Q. Variation of hospital-based adoption of care coordination services by community-level social determinants of health. Health Care Manag. Rev. 2020, 45, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Kaiser State Health Facts. Medicaid & CHIP. 2020. Available online: https://www.kff.org/state-category/medicaid-chip/ (accessed on 18 December 2020).
- Center for Medicare and Medicaid Services. Important Message about the Jimmo Settlement. 2019. Available online: https://www.cms.gov/Center/Special-Topic/Jimmo-Center (accessed on 20 December 2020).
- Jones, C.; Bowles, K. Emerging challenges and opportunities for home health care in the time of COVID-19. J. Am. Med. Dir. Assoc. 2020, 21, 1517–1518. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Chastain, A.M.; Perera, U.G.E.; Quigley, D.D.; Fu, C.J.; Dick, A.W.; Pogorzelska-Maziarz, M.; Stone, P.W. COVID-19 preparedness in US home health care agencies. J. Am. Med. Dir. Assoc. 2020, 21, 924–927. [Google Scholar] [CrossRef] [PubMed]
| Ninth Revision, Clinical Modification (ICD-9-CM) | Tenth Revision, Clinical Modification (ICD-10-CM) |
|---|---|
| Cardiovascular diseases: 410–414, 3,9891, 426–428, 7850, 7851, 430–438, 401–405, 4372, 415–417, 429, 440–444, 446–448, 451–453, 458–459, 557, 7859, 7865, 7943, 7962 | Cardiovascular diseases: G45, 58; I09–13,15, 20–21, 23–28, 44–45, 47–51, 62–63, 65–67, 70–75, 77–78, 80–82, 87, 69, 95, 97, 99; K55; M30–31; R00, 03, 07, 09, 58, 94 |
| Diabetes mellitus: 250xx | Diabetes mellitus: E11xx |
| Renal diseases: 580–586, 590, 595, 597, 5,9800, 5,9801, 5990 | Renal diseases: N00–05, 08, 10, 11, 16–19, 30, 34–35, 37, 39 |
| Lower extremity diseases and complications: 0201, 0210, 0220, 0311, 03285, 035, 0390, 337, 342–344, 354, 355, 3568, 3569, 3572, 3581, 4402, 4423, 4438, 4439, 44422, 44502, 4510, 4512, 454, 680–682, 684–686, 690, 694–698, 700–703, 707, 709, 711, 7184, 7271, 730, 735, 736, 7396, 7854 | Lower extremity diseases and complications: A20, 21, 31, 36; E08–11, 13, 83; G56–60, 73, 80–83, 90; I83, 96; L00–03, 05, 08–13, 20–21, 26, 28–30, 40, 42–44, 49, 51–53, 57, 60, 66, 71, 80–85, 87, 89–95, 97–98; M00–02, 20–21, 24, 46, 57, 66, 83, 85–87, 89–92 |
| Eye diseases and vision defects: 361, 362, 365–369 | Eye diseases and vision defects: E113, 09–11, 13; H26, 28, 36, 250–252, 258–262, 311, 330–334, 338, 340–2, 348–359, 400–406, 408–9, 520, 523–527, 530–536, 538–548; Q150 |
| Mycoses: 110–112, 1141, 1143, 1149, 115–118 | Mycoses: B35–49 |
| Fluid and electrolytes: 276 | Fluid and electrolytes: E86–87 |
| White | Black | Hispanic | AAPI | AIAN | |
|---|---|---|---|---|---|
| n = 534,733 | n = 134,250 | n = 86,834 | n = 19,888 | n = 5859 | |
| Sex, Male % | 53.8 | 41.7 | 49.1 | 50.1 | 48.0 |
| Age 1, % | |||||
| 50–65 | 17.6 | 34.9 | 26.5 | 18.9 | 32.8 |
| 66–75 | 39.3 | 34.5 | 35.6 | 34.7 | 37.1 |
| 76–85 | 31.0 | 22.7 | 28.0 | 31.9 | 23.8 |
| 86+ | 12.1 | 8.0 | 9.9 | 14.5 | 6.3 |
| Insurance, % | |||||
| Medicare FFS | 54.6 | 32.1 | 20.2 | 27.1 | 47.0 |
| FFS + Medicaid | 13.7 | 27.3 | 27.6 | 34.0 | 35.8 |
| Medicare Advantage (MA) | 25.0 | 20.8 | 25.5 | 19.2 | 10.1 |
| MA + Medicaid | 6.7 | 19.8 | 26.7 | 19.7 | 7.1 |
| RUCC-ADI 2.0, % | |||||
| Rural-Advantaged | 17.4 | 6.0 | 3.6 | 2.9 | 26.8 |
| Rural-Disadvantaged | 4.0 | 5.4 | 2.5 | 0.4 | 17.7 |
| Urban-Advantaged | 71.1 | 61.8 | 70.7 | 89.9 | 44.3 |
| Urban-Disadvantaged | 7.5 | 26.7 | 23.2 | 6.8 | 11.2 |
| Elixhauser score 2 (mean) | 27.4 | 30.7 | 27.0 | 29.7 | 29.1 |
| Common Comorbidities, % | |||||
| Chronic pulmonary | 28.4 | 24.6 | 18.2 | 18.2 | 25.8 |
| Congestive heart | 37.8 | 40.3 | 31.5 | 35.8 | 35.0 |
| Dementia | 15.8 | 18.0 | 17.5 | 19.1 | 15.7 |
| Depression | 13.0 | 7.8 | 9.1 | 5.7 | 11.3 |
| Diabetes complication | 27.5 | 32.6 | 35.0 | 34.0 | 35.7 |
| Fluid/electrolyte | 33.6 | 38.3 | 34.9 | 42.6 | 38.6 |
| Hypertension | 88.3 | 94.3 | 91.4 | 92.4 | 88.0 |
| Hypothyroidism | 20.4 | 10.0 | 16.2 | 14.0 | 18.3 |
| Peripheral vascular | 17.7 | 15.7 | 17.6 | 15.4 | 16.3 |
| Renal failure | 34.6 | 45.9 | 38.5 | 45.6 | 42.4 |
| Prior HHC (120-days), % | 14.6 | 18.7 | 16.5 | 12.6 | 13.2 |
| Hospital LoS 3 (mean) | 3.8 | 4.0 | 4.2 | 3.9 | 3.9 |
| Discharge to HHC | HHC Use within 14-Days | |||
|---|---|---|---|---|
| Unadjusted | Model 1 | Model 2a Discharged to Home Health Care | Model 2b Discharged to Self-Care | |
| c = 0.754 | c = 0.674 | c = 0.797 | ||
| OR, 95% CI | OR, 95% CI | OR, 95% CI | OR, 95% CI | |
| Race (reference group = white) | ||||
| Black | 1.1, 1.1–1.1 * | 1.0, 1.0–1.0 * | 0.9, 0.8–0.9 * | 1.2, 1.2–1.2 * |
| Hispanic | 0.8, 0.8–0.8 * | 0.8, 0.8–0.8 * | 0.7, 0.6–0.7 * | 1.0, 1.0–1.0 |
| AAPI | 1.0, 1.0–1.0 | 0.9, 0.9–1.0 | 0.8, 0.8–0.9 * | 1.0, 0.9–1.0 |
| AIAN | 0.7, 0.6–0.7 * | 0.8, 0.7–0.9 * | 0.8, 0.7–0.9 * | 0.8, 0.7–0.9 * |
| Sex (reference = female) | ||||
| Male | 0.8, 0.8–0.8 * | 0.9, 0.9–0.9 * | 1.0, 0.9–1.0 * | 0.8, 0.8–0.9 * |
| Age (ref = 50–65 years) | ||||
| 66–75 | 1.0, 1.0–1.0 * | 1.2, 1.2–1.2 * | 1.0, 1.0–1.1 * | 1.2, 1.1–1.2 * |
| 76–85 | 1.5, 1.5–1.5 * | 1.7, 1.7–1.7 * | 1.3, 1.2–1.3 * | 1.7, 1.7–1.8 * |
| 86+ | 2.4, 2.4–2.5 * | 2.6, 2.6–2.7 * | 1.4, 1.3–1.4 * | 2.5, 2.4–2.6 * |
| Insurance (reference = FFS only) | ||||
| Medicare FFS + Medicaid | 1.3, 1.3–1.4 * | 1.2, 1.2–1.2 * | 1.0, 1.0–1.0 | 1.4, 1.4–1.4 * |
| Medicare Advantage (MA) | 1.0, 1.0–1.1 * | 1.2, 1.2–1.2 * | 0.5, 0.5–0.5 * | 1.1, 1.0–1.1 * |
| MA + Medicaid | 1.3, 1.3–1.3 * | 1.3, 1.3–1.3 * | 0.5, 0.5–0.5 * | 1.1, 1.1–1.2 * |
| Prior HHC (ref = no) | ||||
| HHC in prior 120 days | 3.7, 3.7–3.8 * | 3.1, 3.1–3.2 * | 2.5, 2.4–2.5 * | 9.0, 8.8–9.2 * |
| RUCC-ADI 2.0 (reference = Rural-Advantaged) | ||||
| Rural-Disadvantaged | 1.2, 1.2–1.2 * | 1.1, 1.1–1.1 * | 1.1, 1.0–1.1 | 1.2, 1.1–1.2 * |
| Urban-Advantaged | 1.1, 1.0–1.1 * | 1.1, 1.0–1.1 * | 0.9, 0.8–0.9 * | 1.1, 1.0–1.1 * |
| Urban-Disadvantaged | 1.2, 1.2–1.2 * | 1.1, 1.1–1.2 * | 0.9, 0.8–0.9 * | 1.1, 1.1–1.2 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, J.M.; Jarrín, O.F.; Lin, H.; Tsui, J.; Dharamdasani, T.; Thomas-Hawkins, C. Racial Disparities in Post-Acute Home Health Care Referral and Utilization among Older Adults with Diabetes. Int. J. Environ. Res. Public Health 2021, 18, 3196. https://doi.org/10.3390/ijerph18063196
Smith JM, Jarrín OF, Lin H, Tsui J, Dharamdasani T, Thomas-Hawkins C. Racial Disparities in Post-Acute Home Health Care Referral and Utilization among Older Adults with Diabetes. International Journal of Environmental Research and Public Health. 2021; 18(6):3196. https://doi.org/10.3390/ijerph18063196
Chicago/Turabian StyleSmith, Jamie M., Olga F. Jarrín, Haiqun Lin, Jennifer Tsui, Tina Dharamdasani, and Charlotte Thomas-Hawkins. 2021. "Racial Disparities in Post-Acute Home Health Care Referral and Utilization among Older Adults with Diabetes" International Journal of Environmental Research and Public Health 18, no. 6: 3196. https://doi.org/10.3390/ijerph18063196
APA StyleSmith, J. M., Jarrín, O. F., Lin, H., Tsui, J., Dharamdasani, T., & Thomas-Hawkins, C. (2021). Racial Disparities in Post-Acute Home Health Care Referral and Utilization among Older Adults with Diabetes. International Journal of Environmental Research and Public Health, 18(6), 3196. https://doi.org/10.3390/ijerph18063196







