Fecal Indicator Bacteria Data to Characterize Drinking Water Quality in Low-Resource Settings: Summary of Current Practices and Recommendations for Improving Validity
Abstract
1. Introduction
2. Materials and Methods
2.1. Systematic Review of FIB Data Reporting
2.2. Search Strategy
2.3. Inclusion Criteria
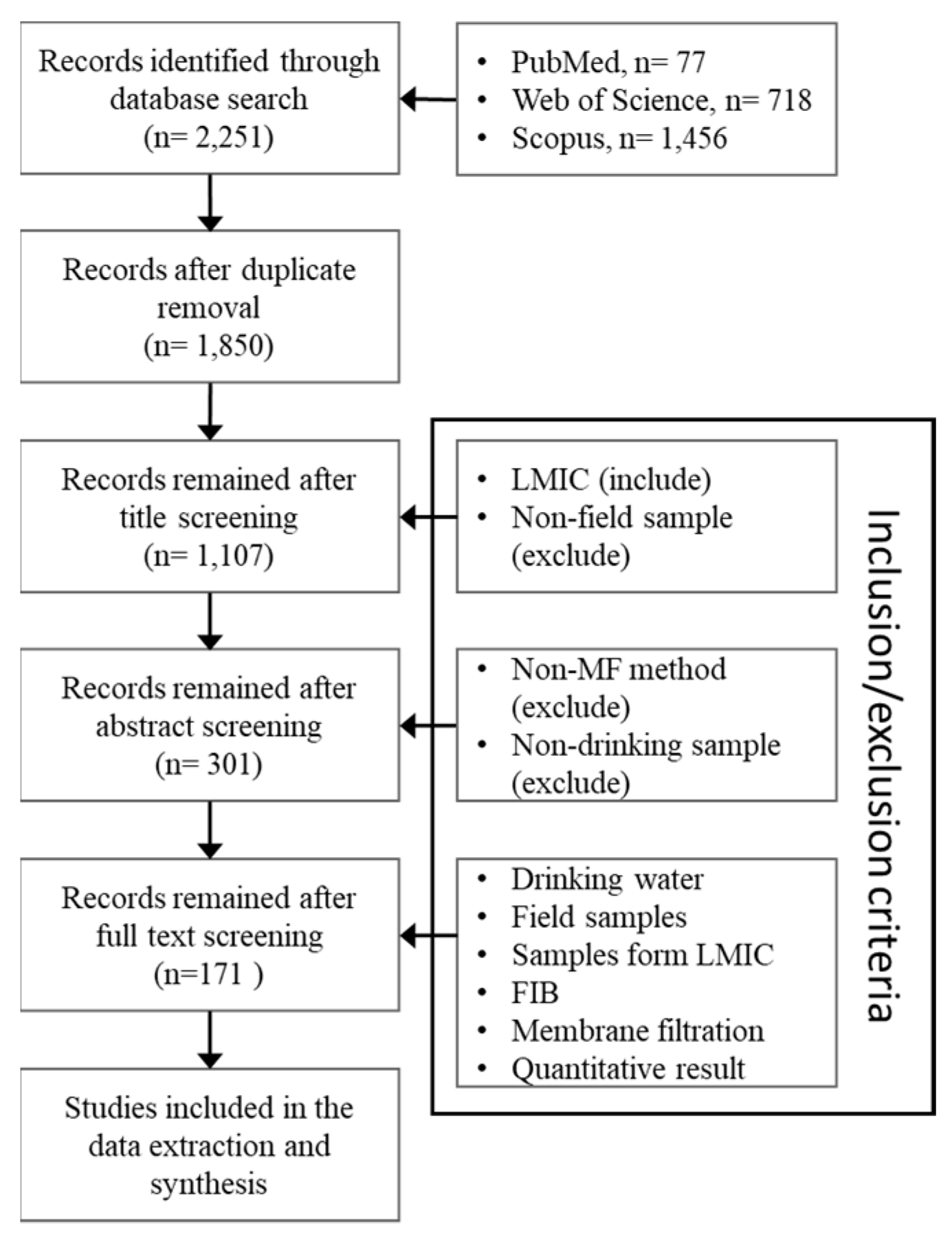
2.4. Selection and Processing
2.5. Result Synthesis
2.6. Analysis of Selected Example FIB Data Sets
3. Results
3.1. Systematic Review Results
3.1.1. Sample Collection and Processing
3.1.2. Data Preparation and Analysis
3.2. Analyses Using Example Dataset
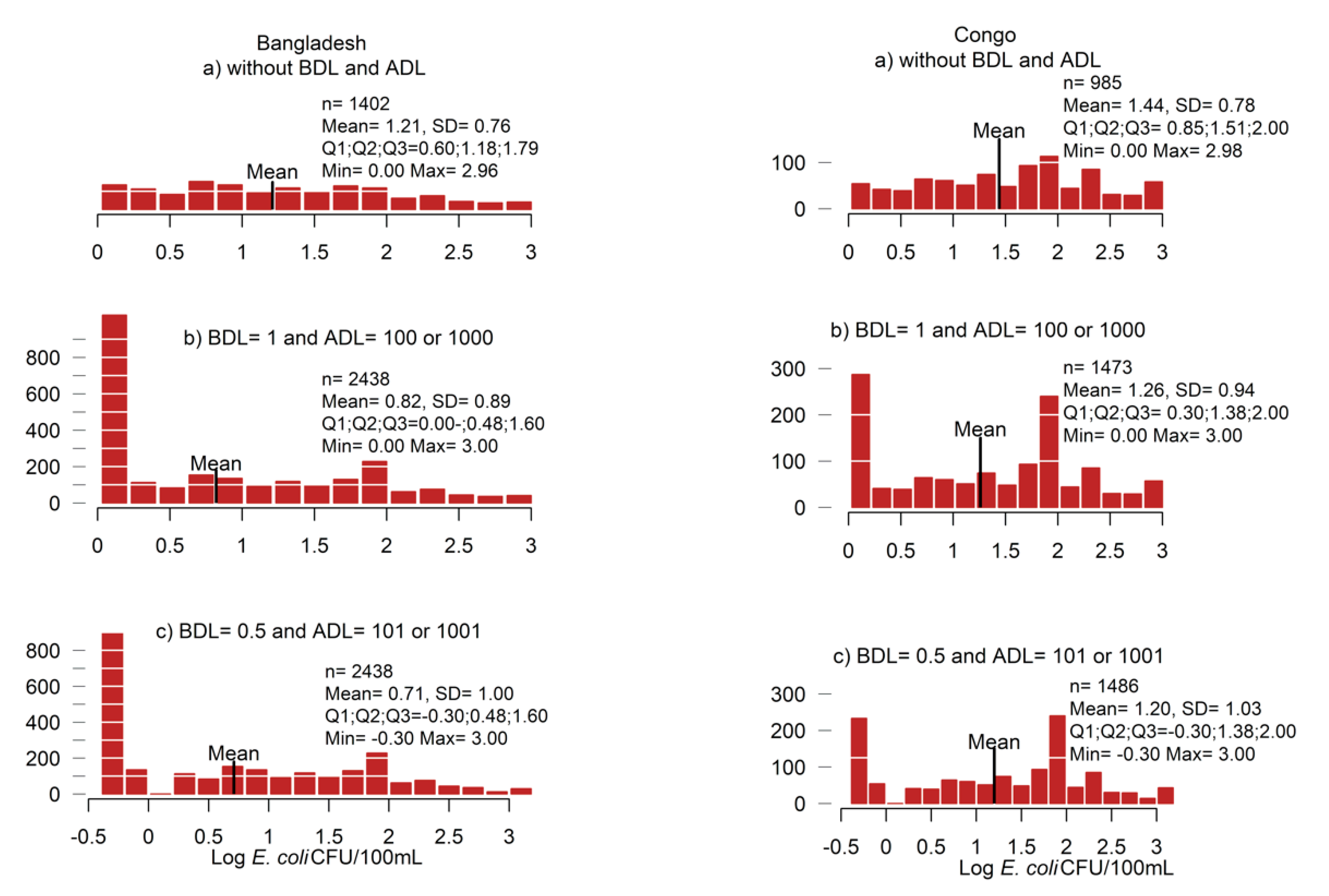
3.2.1. Data Replacement Methods
3.2.2. Descriptive Statistics
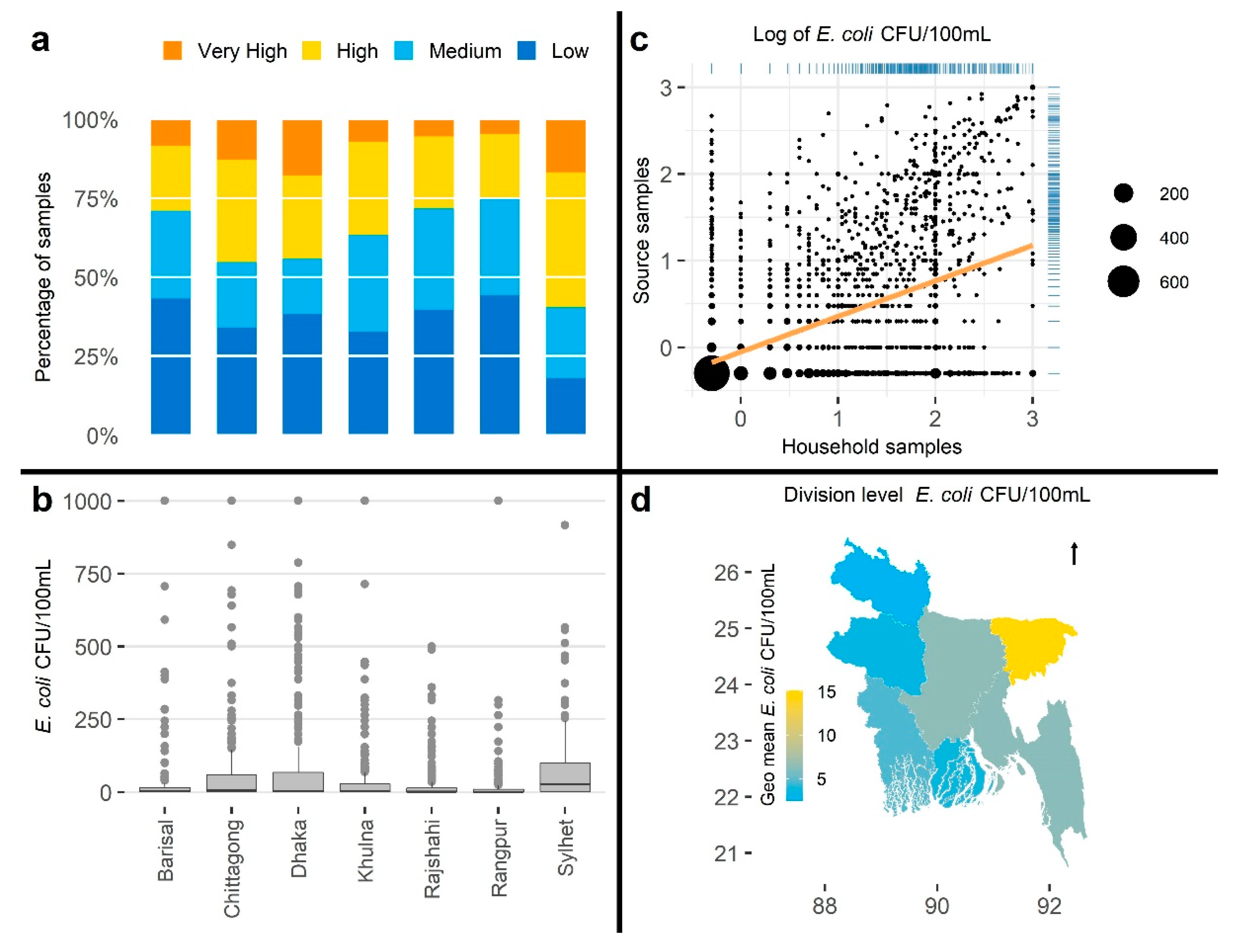
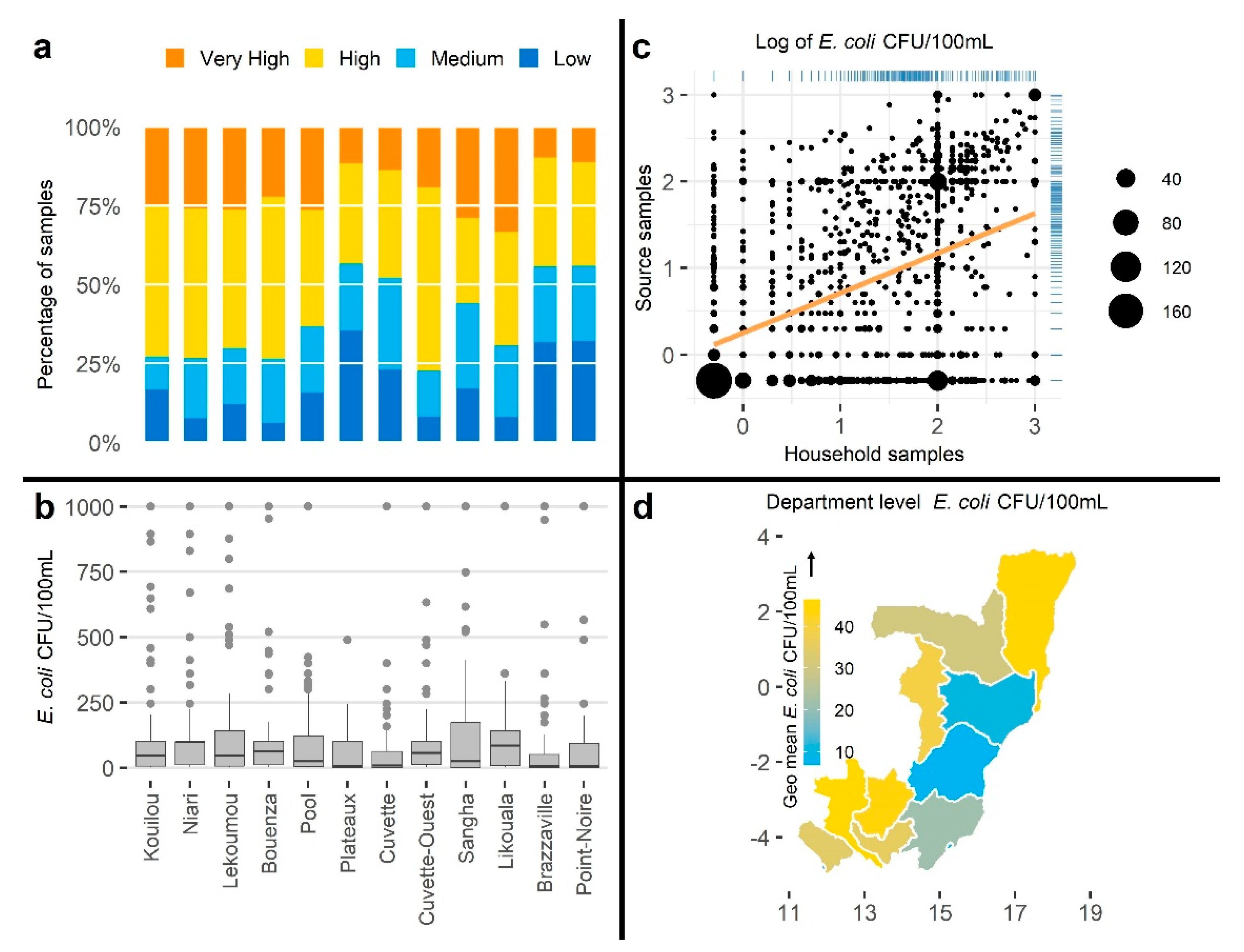
3.2.3. Visualizations
3.2.4. Hypothesis/Comparison Testing
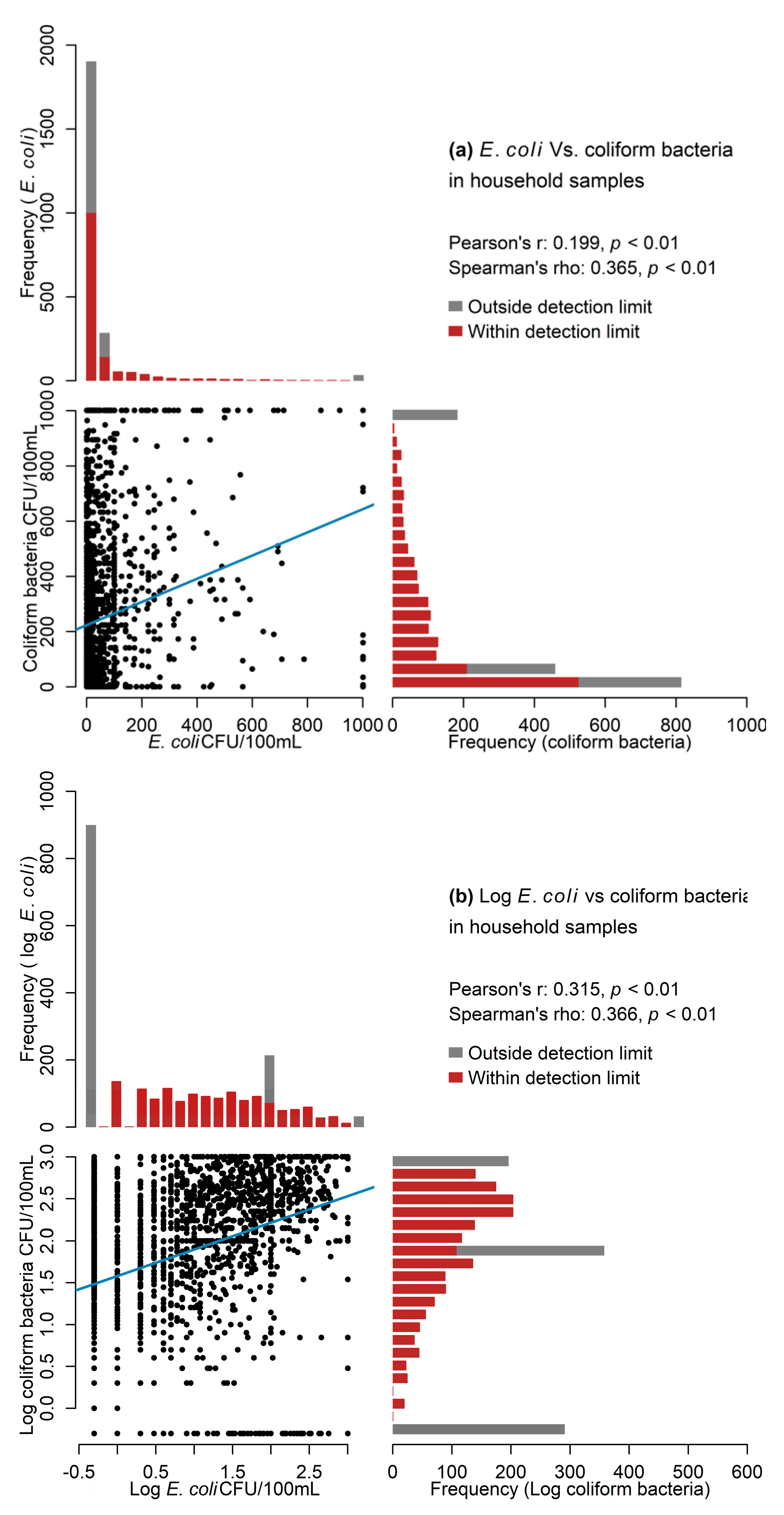
3.2.5. Associations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating The First Addendum, 4th ed.; World Health Organization: Geneva, Switzerland, 2017; ISBN 9789241549950. [Google Scholar]
- Bain, R.; Bartram, J.; Elliott, M.; Matthews, R.; Mcmahan, L.; Tung, R.; Chuang, P.; Gundry, S. A summary catalogue of microbial drinking water tests for low and medium resource settings. Int. J. Environ. Res. Public Health 2012, 9, 1609–1625. [Google Scholar] [CrossRef]
- Bain, R.; Cronk, R.; Hossain, R.; Bonjour, S.; Onda, K.; Wright, J.; Yang, H.; Slaymaker, T.; Hunter, P.; Prüss-Ustün, A.; et al. Global assessment of exposure to faecal contamination through drinking water based on a systematic review. Trop. Med. Int. Health 2014, 19, 917–927. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Microbiological Indicator Testing in Developing Countries: A Fact Sheet for the Field Practitioner; CDC: Atlanta, GA, USA, 2010. [Google Scholar]
- WHO. Indicators of microbial water quality. In Water Quality: Guidelines, Standards and Health; Fewtrell, L., Bartram, J., Eds.; IWA Publishing: London, UK, 2001; pp. 289–316. ISBN 1900222280. [Google Scholar]
- APHA/AWWA/WEF 9222 Membrane Filter Technique for Members of the Coliform Group (2017). In Standard Methods for the Examination of Water and Wastewater; American Public Health Association, American Water Works Association, Water Environment Federation: Denver, CO, USA, 2017.
- Environmental Protection Agency, Office of Water. Method 1604: Total Coliforms and Escherichia coli in Water by Membrane Filtration Using a Simultaneous Detection Technique (MI Medium); EPA: Washington, DC, USA, 2002. [Google Scholar]
- APHA/AWWA/WEF 9020 Quality Assurance/Quality Control (2017). In Standard Methods for the Examination of Water and Wastewater; American Public Health Association, American Water Works Association, Water Environment Federation: Denver, CO, USA, 2018.
- Hach Analytical Procedures. m-ColiBlue24 Broth Procedure for Membrane Filtration; Hach: Loveland, CO, USA, 1999. [Google Scholar]
- Haas, C.N. How to average microbial densities to characterize risk. Water Res. 1996, 30, 1036–1038. [Google Scholar] [CrossRef]
- Haas, C.N.; Scheff, P.A. Estimation of averages in truncated samples. Environ. Sci. Technol. 1990, 24, 912–919. [Google Scholar] [CrossRef]
- Alexander, N. Analysis of Parasite and Other Skewed Counts. Trop. Med. Int. Health 2012, 17, 684–693. [Google Scholar] [CrossRef]
- Clasen, T.F.; Alexander, K.T.; Sinclair, D.; Boisson, S.; Peletz, R.; Chang, H.H.; Majorin, F.; Cairncross, S. Interventions to improve water quality for preventing diarrhoea. Cochrane Database Syst. Rev. 2015, 2015, CD004794. [Google Scholar] [CrossRef]
- Gruber, J.S.; Ercumen, A.; Colford, J.M. Coliform bacteria as indicators of diarrheal risk in household drinking water: Systematic review and meta-analysis. PLoS ONE 2014, 9, e107429. [Google Scholar] [CrossRef] [PubMed]
- Bain, R.; Cronk, R.; Wright, J.; Yang, H.; Slaymaker, T.; Bartram, J. Fecal Contamination of Drinking-Water in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis. PLoS Med. 2014, 11, e1001644. [Google Scholar] [CrossRef] [PubMed]
- Hunter, P.R. Drinking water and diarrhoeal disease due to Escherichia coli. J. Water Health 2003, 1, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.R.; Bain, R.E.S.; Fisher, M.B.; Cronk, R.; Kelly, E.R.; Bartram, J. A systematic review and meta-analysis of fecal contamination and inadequate treatment of packaged water. PLoS ONE 2015, 10, e0140899. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.; Gundry, S.; Conroy, R. Household drinking water in developing countries: A systematic review of microbiological contamination between source and point-of-use. Trop. Med. Int. Health 2004, 9, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Hamzah, L.; Boehm, A.B.; Davis, J.; Pickering, A.J.; Wolfe, M.; Mureithi, M.; Harris, A. Ruminant fecal contamination of drinking water introduced post-collection in rural kenyan households. Int. J. Environ. Res. Public Health 2020, 17, 608. [Google Scholar] [CrossRef] [PubMed]
- Ercumen, A.; Naser, A.M.; Unicomb, L.; Arnold, B.F.; Colford, J.M.; Luby, S.P. Effects of source-versus household contamination of tubewell water on child diarrhea in Rural Bangladesh: A randomized controlled trial. PLoS ONE 2015, 10, e0121907. [Google Scholar] [CrossRef] [PubMed]
- Sikder, M.; Mirindi, P.; String, G.; Lantagne, D. Delivering Drinking Water by Truck in Humanitarian Contexts: Results from Mixed-Methods Evaluations in the Democratic Republic of the Congo and Bangladesh. Environ. Sci. Technol. 2020, 54, 5041–5050. [Google Scholar] [CrossRef]
- Sikder, M.; String, G.; Kamal, Y.; Farrington, M.; Rahman, A.S.; Lantagne, D. Effectiveness of water chlorination programs along the emergency-transition-post-emergency continuum: Evaluations of bucket, in-line, and piped water chlorination programs in Cox’s Bazar. Water Res. 2020, 178, 115854. [Google Scholar] [CrossRef]
- Singh, A.K.; Das, S.; Singh, S.; Pradhan, N.; Gajamer, V.R.; Kumar, S.; Lepcha, Y.D.; Tiwari, H.K. Physicochemical parameters and alarming coliform count of the potable water of Eastern Himalayan state Sikkim: An indication of severe fecal contamination and immediate health risk. Front. Cell Dev. Biol. 2019, 7, 174. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.B.; Williams, A.R.; Jalloh, M.F.; Saquee, G.; Bain, R.E.S.; Bartram, J.K. Microbiological and chemical quality of packaged sachet water and household stored drinking water in Freetown, Sierra Leone. PLoS ONE 2015, 10, e0131772. [Google Scholar] [CrossRef] [PubMed]
- Affum, A.O.; Osae, S.D.; Nyarko, B.J.B.; Afful, S.; Fianko, J.R.; Akiti, T.T.; Adomako, D.; Acquaah, S.O.; Dorleku, M.; Antoh, E.; et al. Total coliforms, arsenic and cadmium exposure through drinking water in the Western Region of Ghana: Application of multivariate statistical technique to groundwater quality. Environ. Monit. Assess. 2015, 187, 1. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Long, S.C.; Das, D.; Dorner, S.M. Are microbial indicators and pathogens correlated? A statistical analysis of 40 years of research. J. Water Health 2011, 9, 265–278. [Google Scholar] [CrossRef]
- Luby, S.P.; Halder, A.K.; Huda, T.M.; Unicomb, L.; Islam, M.S.; Arnold, B.F.; Johnston, R.B. Microbiological contamination of drinking water associated with subsequent child diarrhea. Am. J. Trop. Med. Hyg. 2015, 93, 904–911. [Google Scholar] [CrossRef] [PubMed]
- George, C.M.; Jung, D.S.; Saif-Ur-Rahman, K.M.; Monira, S.; Sack, D.A.; Rashid, M.U.; Toslim Mahmud, M.; Mustafiz, M.; Rahman, Z.; Bhuyian, S.I.; et al. Sustained uptake of a hospital-based handwashing with soap and water treatment intervention (cholera-hospital-based intervention for 7 days [CHoBI7]): A randomized controlled trial. Am. J. Trop. Med. Hyg. 2016, 94, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, I.F.; Hoover, C.M.; Remais, J.V.; Monaghan, A.; Celada, M.; Carlton, E.J. Estimating the risk of domestic water source contamination following precipitation events. Am. J. Trop. Med. Hyg. 2016, 94, 1403–1406. [Google Scholar] [CrossRef]
- Exum, N.G.; Olórtegui, M.P.; Yori, P.P.; Davis, M.F.; Heaney, C.D.; Kosek, M.; Schwab, K.J. Floors and Toilets: Association of Floors and Sanitation Practices with Fecal Contamination in Peruvian Amazon Peri-Urban Households. Environ. Sci. Technol. 2016, 50, 7373–7381. [Google Scholar] [CrossRef] [PubMed]
- Heitzinger, K.; Rocha, C.A.; Quick, R.E.; Montano, S.M.; Tilley, D.H.; Mock, C.N.; Jannet Carrasco, A.; Cabrera, R.M.; Hawes, S.E. Improved but not necessarily safe: An assessment of fecal contamination of household drinking water in rural Peru. Am. J. Trop. Med. Hyg. 2015, 93, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Reygadas, F.; Gruber, J.S.; Ray, I.; Nelson, K.L. Field efficacy evaluation and post-treatment contamination risk assessment of an ultraviolet disinfection and safe storage system. Water Res. 2015, 85, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Benwic, A.; Kim, E.; Khema, C.; Phanna, C.; Sophary, P.; Cantwell, R.E. Factors associated with post-treatment E. coli contamination in households practising water treatment: A study of rural Cambodia. Int. J. Environ. Health Res. 2018, 28, 178–191. [Google Scholar] [CrossRef]
- Chik, A.H.S.; Schmidt, P.J.; Emelko, M.B. Learning something from nothing: The critical importance of rethinking microbial non-detects. Front. Microbiol. 2018, 9, 2304. [Google Scholar] [CrossRef]
- El-Shaarawi, A.H.; Esterby, S.R.; Dutka, B.J. Bacterial density in water determined by poisson or negative binomial distributions. Appl. Environ. Microbiol. 1981, 41, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Silvestri, E.E.; Yund, C.; Taft, S.; Bowling, C.Y.; Chappie, D.; Garrahan, K.; Brady-Roberts, E.; Stone, H.; Nichols, T.L. Considerations for estimating microbial environmental data concentrations collected from a field setting. J. Expo. Sci. Environ. Epidemiol. 2017, 27, 141–151. [Google Scholar] [CrossRef] [PubMed]
- CRD. Systematic Reviews—CRD’s Guidance for Undertaking Reviews in Health Care; CRD, University of York: York, UK, 2009; ISBN 9781900640473. [Google Scholar]
- World Bank. World Bank Analytical Classifications; World Bank: Washington, DC, USA, 2019. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Institut National de la Statistique; UNICEF. Enquête par Grappes à Indicateurs Multiples (MICS5 2014–2015), Rapport Final; Institut National de la Statistique: Brazzaville, Congo, 2015. [Google Scholar]
- Bangladesh Bureau of Statistics; UNICEF. Bangladesh Multiple Indicator Cluster Survey 2012–2013, ProgotirPathey: Final Report; Bangladesh Bureau of Statistics: Dhaka, Bangladesh, 2014. [Google Scholar]
- UNICEF. Manual for Water Quality; UNICEF: New York, NY, USA, 2016. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- APHA/AWWA/WEF Part 9000 Microbiological Examination. In Standard Methods for the Examination of Water and Wastewater; American Public Health Association, American Water Works Association, Water Environment Federation: Denver, CO, USA, 1999; ISBN 0-87553-235-7.
- Chui, K.K.H.; Wenger, J.B.; Cohen, S.A.; Naumova, E.N. Visual Analytics for Epidemiologists: Understanding the Interactions between Age, Time, and Disease with Multi-Panel Graphs. PLoS ONE 2011, 6, e14683. [Google Scholar] [CrossRef]
- Pope, M.L.; Bussen, M.; Feige, M.A.; Shadix, L.; Gonder, S.; Rodgers, C.; Chambers, Y.; Pulz, J.; Miller, K.; Connell, K.; et al. Assessment of the Effects of Holding Time and Temperature on Escherichia coli Densities in Surface Water Samples. Appl. Environ. Microbiol. 2003, 69, 6201–6207. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Stauber, C.; Murphy, J.L.; Khan, A.; Mu, T.; Elliott, M.; Sobsey, M.D. Ambient-temperature incubation for the field detection of Escherichia coli in drinking water. J. Appl. Microbiol. 2011, 110, 915–923. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.L.; Kumpel, E.; Peletz, R.; Khush, R.S.; Lantagne, D.S. The effect of sodium thiosulfate dechlorination on fecal indicator bacteria enumeration: Laboratory and field data. J. Water Health 2018, 16, 44–48. [Google Scholar] [CrossRef]
- Kulinkina, A.V.; Mohan, V.R.; Francis, M.R.; Kattula, D.; Sarkar, R.; Plummer, J.D.; Ward, H.; Kang, G.; Balraj, V.; Naumova, E.N. Seasonality of water quality and diarrheal disease counts in urban and rural settings in south India. Sci. Rep. 2016, 6, 20521. [Google Scholar] [CrossRef]
- Alarcon Falconi, T.M.; Kulinkina, A.V.; Mohan, V.R.; Francis, M.R.; Kattula, D.; Sarkar, R.; Ward, H.; Kang, G.; Balraj, V.; Naumova, E.N. Quantifying tap-to-household water quality deterioration in urban communities in Vellore, India: The impact of spatial assumptions. Int. J. Hyg. Environ. Health 2017, 220, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, E.L.; Meier, P. Nonparametric Estimation from Incomplete Observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- El-Shaarawi, A.H.; Esterby, S.R. Replacement of censored observations by a constant: An evaluation. Water Res. 1992, 26, 835–844. [Google Scholar] [CrossRef]
- Helsel, D.R. More than obvious: Better methods for interpreting nondetect data. Environ. Sci. Technol. 2005, 39, 419–423. [Google Scholar] [CrossRef]
- Helsel, D.R. Fabricating data: How substituting values for nondetects can ruin results, and what can be done about it. Chemosphere 2006, 65, 2434–2439. [Google Scholar] [CrossRef]
- Cha, S.M.; Lee, S.W.; Park, Y.E.; Cho, K.H.; Lee, S.; Kim, J.H. Spatial and temporal variability of fecal indicator bacteria in an urban stream under different meteorological regimes. Water Sci. Technol. 2010, 61, 3102–3108. [Google Scholar] [CrossRef]
- Ferguson, A.S.; Layton, A.C.; Mailloux, B.J.; Culligan, P.J.; Williams, D.E.; Smartt, A.E.; Sayler, G.S.; Feighery, J.; McKay, L.D.; Knappett, P.S.K.; et al. Comparison of fecal indicators with pathogenic bacteria and rotavirus in groundwater. Sci. Total Environ. 2012, 431, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Korajkic, A.; McMinn, B.R.; Harwood, V.J. Relationships between microbial indicators and pathogens in recreational water settings. Int. J. Environ. Res. Public Health 2018, 15, 2842. [Google Scholar] [CrossRef] [PubMed]
- APHA/AWWA/WEF 9223 Enzyme Substrate Coliform Test. In Standard Methods for the Examination of Water and Wastewater; American Public Health Association, American Water Works Association, Water Environment Federation: Denver, CO, USA, 2018.
- Aquagenx Aquagenx ® CBT EC+TC (Compartment Bag Test) Most Probable Number (MPN) Kit Instructions for Use: Drinking Water.
- Michel, M.C.; Murphy, T.J.; Motulsky, H.J. New author guidelines for displaying data and reporting data analysis and statistical methods in experimental biology. J. Pharmacol. Exp. Ther. 2020, 372, 136–147. [Google Scholar] [CrossRef] [PubMed]

| Reported Topics | N = 171 |
|---|---|
| Collection | |
| Included a reference to standard method | 95 (56%) |
| Included any sample collection sterility information | 66 (39%) |
| Used sodium thiosulfate | 34 (20%) |
| Reported storing the sample in “low” temperature (range: 2–8 °C) | 102 (60%) |
| Reported time between sample collection and membrane filtration (range: 2–48 h) | 94 (55%) |
| Reported starting the test in 6 h and/or completed in 8 h | 50 (29%) |
| Analysis | |
| Reported using blank samples (negative controls) to check sterile procedures | 31 (18%) |
| Reported using duplicate samples to check the precision of the analysis | 44 (26%) |
| Reported using multiple appropriate dilutions based on water source | 41 (24%) |
| Reported the volume of filtered sample water | 80 (47%) |
| Reported the name/type of the growth media | 126 (74%) |
| Reported incubation temperature | 112 (65%) |
| Reported incubation time | 110 (64%) |
| Processing | |
| Reported how values from multiple dilutions were aggregated | 35 (20%) |
| Reported the percent of BDL and ADL samples | 84 (49%) |
| Reported how BDL results were handled | 46 (27%) |
| Reported how ADL results were handled | 49 (29%) |
| Reported log transforming data | 73 (43%) |
| Bangladesh | Congo | |||
|---|---|---|---|---|
| Category | Urban (n = 390) | Rural (n = 2048) | Urban (n = 503) | Rural (n = 970) |
| Arithmetic mean | 63.0 | 51.7 | 66.5 | 124.0 |
| Geometric mean | 4.9 | 5.2 | 8.0 | 22.7 |
| Standard deviation (SD) | 168.0 | 136.0 | 160.8 | 221.9 |
| Geometric SD | 10.8 | 9.8 | 10.4 | 9.9 |
| Median | 3 | 3 | 9 | 42 |
| 25th and 75th percentiles | 0.5–33.8 | 0.5–40.0 | 0.5–76.7 | 0.5–100.0 |
| Interquartile range (IQR) | 35.3 | 39.5 | 76.2 | 96.0 |
| 10th and 90th percentiles | 0.5–141.4 | 0.5–114.0 | 0.5–141.4 | 0.5–316.2 |
| Minimum and maximum | 0.5–1001.0 | 0.5–1001.0 | 0.5–1001.0 | 0.5–1001.0 |
| Skewness | 4.1 | 4.9 | 4.5 | 2.9 |
| Kurtosis | 17.6 | 27.6 | 21.7 | 7.9 |
| Data Type | Test | Null H0 | Statistic | p-Value |
|---|---|---|---|---|
| Bangladesh | ||||
| Continuous | Wilcoxon rank sum test | Medians are equal in both divisions | W = 68,041 | 0.025 * |
| Binary (cutoff ≥ 1) | Pearson’s chi-squared test | Proportions are equal in both divisions | χ2 = 3.82 | 0.051 |
| Binary (cutoff ≥ 10) | χ2 = 3.79 | 0.052 | ||
| Congo | ||||
| Continuous | Wilcoxon rank sum test | Medians are equal in both departments | W = 5952 | <0.001 * |
| Binary (cutoff ≥ 1) | Pearson’s chi-squared test | Proportions are equal in both departments | χ2 = 3.34 | 0.068 |
| Binary (cutoff ≥ 10) | χ2 = 13.32 | <0.001 * |
| Section/Topic | Checklist Item |
|---|---|
| Sample collection | |
| 1 | Report sample collection equipment and supplies (e.g., sterile bottle/bag/vial) |
| 2 | Report if sodium thiosulfate (or equivalent) was used (if chlorinated sample) |
| 3 | Report if aseptic procedure was maintained to prevent contamination |
| 4 | If not analyzed in one hour, report if <10 °C was maintained |
| 5 | If not analyzed immediately, report the time between collection and analysis |
| Membrane filtration | |
| 6 | Report if positive and negative controls were checked |
| 7 | Report the volume of sample filtered |
| 8 | Report number, dilution, and/or volume of serial dilutions |
| 9 | What diluent was used if any |
| 10 | Report the selective growth media |
| 11 | Report the incubation time and temperature |
| Enumeration | |
| 12 | Report the detection minimum/maximum range for enumeration |
| 13 | Report aggregation method for serial dilutions |
| 14 | Report the number of BDL and ADL samples |
| 15 | If BDL/ADL samples were included in analysis, report how values were replaced |
| 16 | Report if any data were dropped due to positive/negative controls |
| 17 | If a subset of enumerations were verified by a second person |
| Statistical analysis | |
| 18 | Report if the data were transformed |
| 19 | Report if the data were analyzed as count, continuous, categorical, and binary |
| 20 | Describe dataset using parameters that justify any following statistical analysis |
| 21 | For data visualization, ensure proper tool was selected to aid information communication |
| 22 | Provide rationales for the choice of statistical method |
| 23 | Report if the data met the assumptions of the selected statistical test |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sikder, M.; Naumova, E.N.; Ogudipe, A.O.; Gomez, M.; Lantagne, D. Fecal Indicator Bacteria Data to Characterize Drinking Water Quality in Low-Resource Settings: Summary of Current Practices and Recommendations for Improving Validity. Int. J. Environ. Res. Public Health 2021, 18, 2353. https://doi.org/10.3390/ijerph18052353
Sikder M, Naumova EN, Ogudipe AO, Gomez M, Lantagne D. Fecal Indicator Bacteria Data to Characterize Drinking Water Quality in Low-Resource Settings: Summary of Current Practices and Recommendations for Improving Validity. International Journal of Environmental Research and Public Health. 2021; 18(5):2353. https://doi.org/10.3390/ijerph18052353
Chicago/Turabian StyleSikder, Mustafa, Elena N. Naumova, Anthonia O. Ogudipe, Mateo Gomez, and Daniele Lantagne. 2021. "Fecal Indicator Bacteria Data to Characterize Drinking Water Quality in Low-Resource Settings: Summary of Current Practices and Recommendations for Improving Validity" International Journal of Environmental Research and Public Health 18, no. 5: 2353. https://doi.org/10.3390/ijerph18052353
APA StyleSikder, M., Naumova, E. N., Ogudipe, A. O., Gomez, M., & Lantagne, D. (2021). Fecal Indicator Bacteria Data to Characterize Drinking Water Quality in Low-Resource Settings: Summary of Current Practices and Recommendations for Improving Validity. International Journal of Environmental Research and Public Health, 18(5), 2353. https://doi.org/10.3390/ijerph18052353






