Assessment of Selected Aspects of the Quality of Life of Children with Type 1 Diabetes Mellitus in Poland
Abstract
1. Introduction
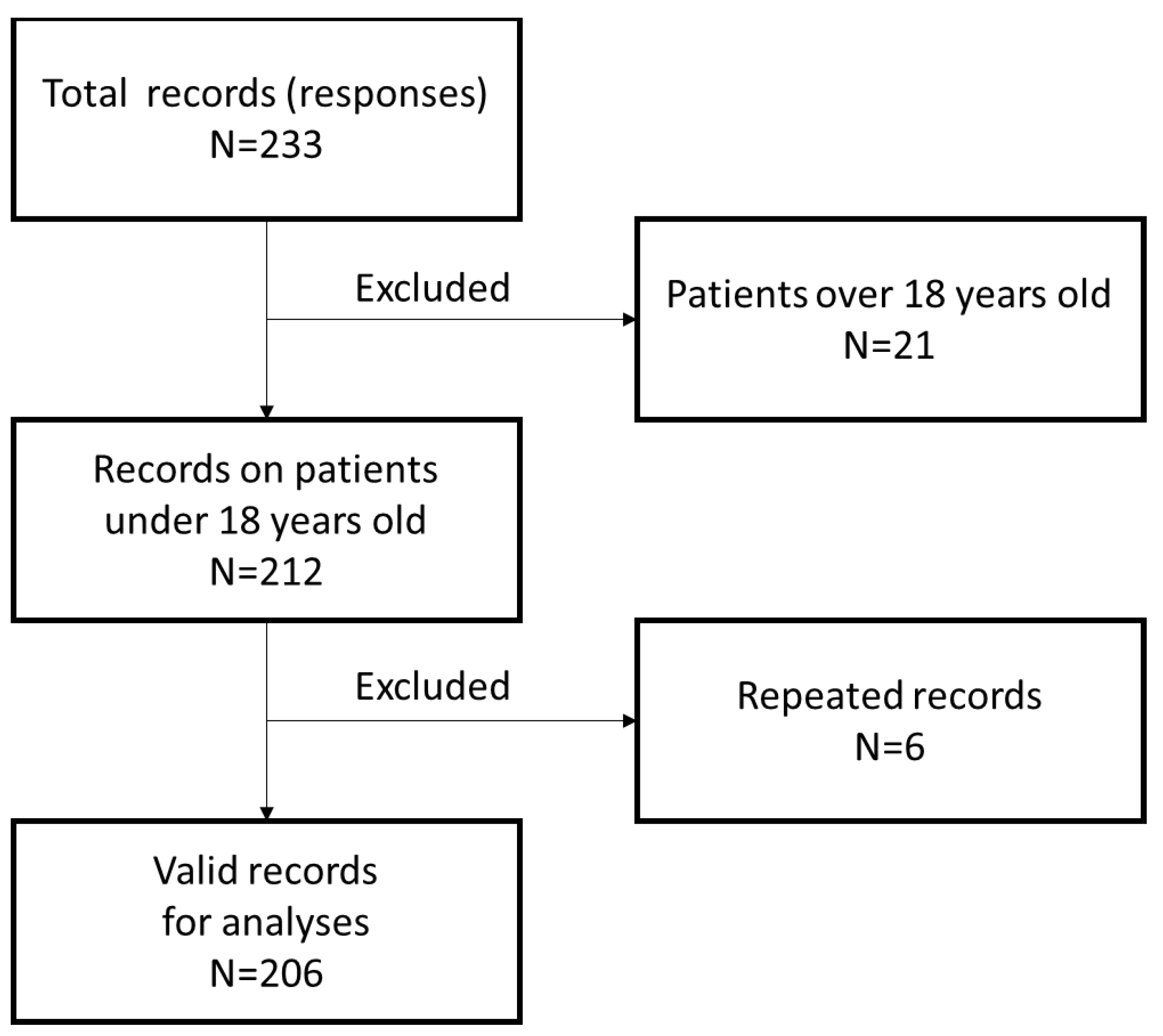
2. Materials and Methods
2.1. Data Collection
2.2. Variables
- Demographic data of the child and parent/caregiver—module I;
- Information on the child’s health and therapy—module II;
- Information about the impact of the disease on family and child life—module III;
- Information on the provision of care for the child by other persons or institutions—module IV.
2.2.1. Demographic Data—Module I
2.2.2. Health Status—Module II
2.2.3. Impact on Child’s and Family’s Life—Module III
2.2.4. Impact on Child’s and Family’s Life—Module III
2.3. Statistical Analysis
3. Results
3.1. Participants Characteristics
3.2. Correlations between Groups of Variables (Modules)
3.2.1. Correlation between Demographic Factors and Health Status
3.2.2. Influence of Demographic Factors
3.3. Influence of Health Status
4. Discussion
5. Study Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knip, M.; Siljander, H. Autoimmune mechanisms in type 1 diabetes. Autoimmun. Rev. 2008, 7, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Banday, M.Z.; Sameer, A.S.; Nissar, S. Pathophysiology of diabetes: An overview. Avicenna J. Med. 2020, 10, 174–188. [Google Scholar] [CrossRef] [PubMed]
- Pociot, F.; Lernmark, Å. Genetic risk factors for type 1 diabetes. Lancet 2016, 387, 2331–2339. [Google Scholar] [CrossRef]
- Redondo, M.J.; Jeffrey, J.; Fain, P.R.; Eisenbarth, G.S.; Orban, T. Concordance for islet autoimmunity among monozygotic twins. N. Engl. J. Med. 2008, 359, 2849–2850. [Google Scholar] [CrossRef]
- Rewers, M.; Ludvigsson, J. Environmental risk factors for type 1 diabetes. Lancet 2016, 387, 2340–2348. [Google Scholar] [CrossRef]
- Streisand, R.; Monaghan, M. Young children with type 1 diabetes: Challenges, research, and future directions. Curr. Diab. Rep. 2014, 14, 520. [Google Scholar] [CrossRef]
- Patterson, C.C.; Dahlquist, G.G.; Gyurus, E.; Green, A.; Soltesz, G. Incidence trends for childhood Type 1 diabetes in Europe during 1989–2003 and predicted new cases 2005–2020: A multicentre prospective registration study. Lancet 2009, 373, 2027–2033. [Google Scholar] [CrossRef]
- Negrato, C.A.; Dias, J.P.; Teixeira, M.F.; Dias, A.; Salgado, M.H.; Lauris, J.R.; Montenegro, R.M., Jr.; Gomes, M.B.; Jovanovic, L. Temporal trends in incidence of Type 1 diabetes between 1986 and 2006 in Brazil. J. Endocrinol. Investig. 2010, 33, 373–377. [Google Scholar] [CrossRef]
- Secrest, A.M.; Becker, D.J.; Kelsey, S.F.; Ronald, L.E.; Trevor, O.J. Cause-specific mortality trends in a large population-based cohort with long-standing childhood-onset type 1 diabetes. Diabetes 2010, 59, 3216–3222. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C.J.; Alves, C. Influence of socioeconomic and psychological factors in glycemic control in young children with type 1 diabetes mellitus. J. Pediatr. 2019, 95, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Healthy Data Portal (Portal Zdrowe Dane). Available online: https://zdrowedane.nfz.gov.pl/pluginfile.php/205/mod_resource/content/4/nfz_o_zdrowiu_cukrzyca.pdf (accessed on 30 December 2020).
- Myśliwiec, M. Cukrzyca u dzieci-etiopatogeneza, diagnostyka i terapia. Forum Med. Rodz. 2007, 1, 125–133. [Google Scholar]
- Brzeziński, M. Is High Inequality an Issue i Poland? IBS Policy Paper 2017. Available online: https://ibs.org.pl/app/uploads/2017/06/IBS_Policy_Paper_01_2017_en.pdf (accessed on 30 December 2020).
- Sochocka, L.; Ledwoń, E.; Rak, S.; Szwamel, K. Self-assessment of teachers’ knowledge regarding their preparation for caring for a child with type 1 diabetes at school. Piel. Zdr. Publ. 2019, 9, 183–190. Available online: http://www.pzp.umed.wroc.pl/pdf/2019/9/3/183.pdf (accessed on 30 December 2020). [CrossRef]
- Sandeep, G.; Bhadada, S.; Kate, N.; Sarkar, S.; Bhansali, A.; Avasthi, A.; Sharma, S.; Goel, R. Coping and caregiving experience of parents of children and adolescents with type-1 diabetes: An exploratory study. Perspect. Clin. Res. 2016, 7, 32–39. [Google Scholar] [CrossRef]
- Starfield, B.; Bergner, M.; Ensminger, M.; Ri-ley, A.; Ryan, S.; Green, B.; McGauhey, P.; Skinner, A.; Kim, S. Adolescent health status measurement: Development of the Child Health and Illness Profile. Pediatrics 1993, 91, 430–435. [Google Scholar]
- Landgraf, J.E.; Ware, J.E., Jr. The Child Health Questionnaire (CHQ): A User’s Manual; The Health Institute: Boston, MA, USA, 1996. [Google Scholar]
- Verrips, E.G.H.; Vogels, T.G.C.; Koopman, H.M.; Theunissen, N.C.M.; Kamphius, R.P.; Fekkes, M. Measuring health-related quality of life in a child population. Eur. J. Public Health 1999, 9, 114–119. [Google Scholar] [CrossRef]
- Seid, M.; Varni, J.W.; Kurtin, P.S. Measuring quality of care for vulnerable children: Challenges and conceptualization of a pediatric outcome measure of quality. Am. J. Med. Qual. 2000, 15, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Ingersoll, G.M.; Marrero, D.G. A modified quality-of-life measure for youths: Psychometric properties. Diabetes Educ. 1991, 17, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Madsen, S.D.; Roisman, G.I.; Collins, W.A. The intersection of adolescent development and intensive intervention: Age-related psychosocial correlates of treatment regimens in the Diabetes Control and Complications Trial. J. Pediatr. Psychol. 2002, 27, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Varni, J.W.; Burwinkle, T.M.; Jacobs, J.R.; Gottschalk, M.; Kaufman, F.; Jones, K.I. The PedsQL in Type 1 and Type 2 Diabetes: Reliability and validity of the Pediatric Quality of Life Inventory Generic Core Scales and Type 1 Diabetes Module. Diabetes Care 2003, 26, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Varni, J.W.; Seid, M.; Rode, C.A. The PedsQL: Measurement model for the pediatric quality of life inventory. Med. Care 1999, 37, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Varni, J.W.; Seid, M.; Kurtin, P.S. PedsQL 4.0: Reliability and validity of the Pediatric Quality of Life Inventory version 4.0 generic core scales in healthy and patient populations. Med. Care 2001, 39, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Barczykowska, E.; Juszczak, A.; Ślusarz, R.; Grabinska, M.; Kurylak, A. Quality of life in children aged 8–12 with diabetes treated with an insulin pump. Med. Sci. Technol. 2011, 52, 57–61. [Google Scholar]
- Caferoğlu, Z.; İnanç, N.; Hatipoğlu, N.; Kurtoğlu, S. Health-Related Quality of Life and Metabolic Control in Children and Adolescents with Type 1 Diabetes Mellitus. J. Clin. Res. Pediatr. Endocrinol. 2016, 8, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Yang, X.; Yin, G.; Lin, S. Diabetes Self-Care Activities and Health-Related Quality-of-Life of individuals with Type 1 Diabetes Mellitus in Shantou, China. J. Int. Med. Res. 2016, 44, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Zrebiec, J.F.; Jacobson, A.M. What attracts patients with diabetes to an internet support group? A 21-month longitudinal website study. Diabetes Med. 2001, 18, 154–158. [Google Scholar] [CrossRef]
- Wu, N.; Brazeau, A.S.; Nakhla, M.; Chan, D.; Da Costa, D.; Mukerji, G.; Butalia, S.; Pacaud, D.; Henderson, M.; Panagiotopoulos, C.; et al. Type 1 Diabetes Mellitus Virtual Patient Network as a Peer Support Community: Protocol for Social Network Analysis and Content Analysis. JMIR Res. Protoc. 2020, 9, 18714. [Google Scholar] [CrossRef]
- Ismail, I.S.; Nazaimoon, W.M.; Mohamad, W.B.; Letchuman, R.; Singaraveloo, M.; Pendek, R. Sociodemographic determinants of glycaemic control in young diabetic patients in peninsular Malaysia. Diabetes Res. Clin. Pract. 2000, 47, 57–69. [Google Scholar] [CrossRef]
- Araujo, M.B.; Mazza, C.S. Assessment of risk factors of poor metabolic control in type 1 diabetic children assisted in a public hospital in Argentina. Pediatr. Diabetes 2008, 9, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Hassan, K.; Loar, R.; Anderson, B.J.; Heptulla, R.A. The role of socioeconomic status, depression, quality of life, and glycemic control in type 1 diabetes mellitus. J. Pediatr. 2006, 149, 526–531. [Google Scholar] [CrossRef]
- Unwin, N.; Binns, D.; Elliott, K.; Kelly, W.F. The relationships between cardiovascular risk factors and socio-economic status in people with diabetes. Diabetes Med. 1996, 13, 72–79. [Google Scholar] [CrossRef]
- Nadas, J.; Putz, Z.; Fovenyi, J.; Gaal, Z.; Gyimesi, A.; Hidvegi, T. Cardiometabolic risk and educational level in adult patients with type 1 diabetes. Acta Diabetol. 2009, 46, 159–162. [Google Scholar] [CrossRef]
- Chaturvedi, N.; Jarrett, J.; Shipley, M.J.; Fuller, J.H. Socioeconomic gradient in morbidity and mortality in people with diabetes: Cohort study findings from the Whitehall Study and the WHO Multinational Study of Vascular Disease in Diabetes. BMJ 1998, 316, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, M.; Charron-Prochownik, D.; Obrosky, D.S. A longitudinal study of biomedical and psychosocial predictors of multiple hospitalizations among young people with insulin-dependent diabetes mellitus. Diabetes Med. 1995, 12, 142–148. [Google Scholar] [CrossRef]
- Secrest, A.M.; Costacou, T.; Gutelius, B.; Miller, R.G.; Songer, T.J.; Orchard, T.J. Association of Socioeconomic Status with Mortality in Type 1 Diabetes: The Pittsburgh Epidemiology of Diabetes Complications (EDC) Study. Ann. Epidemiol. 2011, 21, 367–373. [Google Scholar] [CrossRef]
- Matsushima, M.; Shimizu, K.; Maruyama, M.; Nishimura, R.; LaPorte, R.E.; Tajima, N. Socioeconomic and behavioural risk factors for mortality of individuals with IDDM in Japan: Population-based case-control study. Diabetes Epidemiology Research International (DERI) US-Japan Mortality Study Group. Diabetologia 1996, 39, 710–716. [Google Scholar] [CrossRef]
- Roper, N.A.; Bilous, R.W.; Kelly, W.F.; Unwin, N.C.; Connolly, V.M. Excess mortality in a population with diabetes and the impact of material deprivation: Longitudinal, population based study. BMJ 2001, 322, 1389–1393. [Google Scholar] [CrossRef] [PubMed]
- Forssas, E.; Arffman, M.; Koskinen, S.; Reunanen, A.; Keskimaki, I. Socioeconomic differences in mortality among diabetic people in Finland. Scand. J. Public Health 2010, 38, 691–698. [Google Scholar] [CrossRef]
- Caccavale, L.J.; Weaver, P.; Chen, R.; Streisand, R.; Holmes, C.S. Family Density and SES Related to Diabetes Management and Glycemic Control in Adolescents With Type 1 Diabetes. J. Pediatr. Psychol. 2015, 40, 500–508. [Google Scholar] [CrossRef]
- Mönkemöller, K.; Müller-Godeffroy, E.; Lilienthal, E.; Heidtmann, B.; Becker, M.; Feldhahn, L.; Freff, M.; Hilgard, D.; Krone, B.; Papsch, M.; et al. The association between socio-economic status and diabetes care and outcome in children with diabetes type 1 in Germany: The DIAS study (diabetes and social disparities). Pediatr. Diabetes 2019, 20, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Economic Poverty in Poland in 2019 (Zasięg Ubóstwa Ekonomicznego w Polsce w 2019 Roku). Available online: https://stat.gov.pl/download/gfx/portalinformacyjny/pl/defaultaktualnosci/5487/14/7/1/zasieg_ubostwa_ekonomicznego_w_polsce_w_2019_r.pdf (accessed on 30 December 2020).
- Guimarães, C.; Marra, C.A.; Colley, L.; Gill, S.; Simpson, S.H.; Meneilly, G.S.; Queiroz, R.H.; Lynd, L.D. A valuation of patients’ willingness-to-pay for insulin delivery in diabetes. Int. J. Technol. Assess. Health Care 2009, 25, 359–366. [Google Scholar] [CrossRef]
- Guimarães, C.; Marra, C.A.; Colley, L.; Gill, S.; Simpson, S.; Meneilly, G.; Queiroz, R.H.; Lynd, L.D. Socioeconomic differences in preferences and willingness-to-pay for insulin delivery systems in type 1 and type 2 diabetes. Diabetes Technol. Ther. 2009, 11, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Children and Adolescents. Standards of Medical Care in Diabetes—2021, American Diabetes Association. Diabetes Care 2021, 44, S180–S199. [Google Scholar] [CrossRef] [PubMed]
- Araszkiewicz, A.; Bandurska-Stankiewicz, E.; Budzyński, A.; Cypryk, K.; Czech, A.; Czupryniak, L.; Drzewoski, J.; Dzida, G.; Dziedzic, T.; Franek, E.; et al. Zalecenia Kliniczne Dotyczące Postępowania u Chorych na Cukrzycę 2020—Stanowisko PTD. Diabetol. Prakt. 2020, 6, 1–106. [Google Scholar]
- Gandhi, K.; Vu, B.K.; Eshtehardi, S.S.; Wasserman, R.M.; Hilliard, M.E. Adherence in adolescents with Type 1 diabetes: Strategies and considerations for assessment in research and practice. Diabetes Manag. 2015, 5, 485–498. [Google Scholar] [CrossRef]
- Patton, S.R. Adherence to glycemic monitoring in diabetes. J. Diabetes Sci. Technol. 2015, 9, 668–675. [Google Scholar] [CrossRef]
- OECD/European Observatory on Health Systems and Policies (2017), Poland: Country Health Profile 2017, State of Health in the EU, OECD Publishing, Paris/European Observatory on Health Systems and Policies, Brussels. Available online: http://dx.doi.org/10.1787/9789264283510-en (accessed on 30 December 2020).
- Sowa-Kofta, A. Addressing the Challenges of the Healthcare System in Poland, ESPN Flash Report 2018/14. Available online: https://ec.europa.eu/social/BlobServlet?docId=19206&langId=en (accessed on 30 December 2020).
- Zegarlicka-Poręba, M.; Jarosz-Chobot, P. Problem Cukrzycy i Radzenia Sobie z Chorobą–Ujęcie Psychologiczne, Diabetologii na co Dzień. 2013. Available online: http://diabetologiaonline.pl/pielegniarka_poradnik,info,159.html (accessed on 30 December 2020).
- Borus, J.S.; Laffel, L. Adherence challenges in the management of type 1 diabetes in adolescents: Prevention and intervention. Curr. Opin. Pediatr. 2010, 22, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Siminerio, L.M.; Albanese-O’Neill, A.; Chiang, J.L.; Hathaway, K.; Jackson, C.C.; Weissberg-Benchell, J.; Wright, J.L.; Yatvin, A.L.; Deeb, L.C. Care of Young Children with Diabetes in the Child Care Setting: A Position Statement of the American Diabetes Association. Diabetes Care 2014, 37, 2834–2842. [Google Scholar] [CrossRef]

| Characteristics | Villages and Towns N = 19 | Small Cities (<20,000 Inhabitants) N = 32 | Medium Cities (20,000–100,000 Inhabitants) N = 46 | Big Cities (100,001–500,000 Inhabitants) N = 81 | Largest Cities (>500,000 Inhabitants) N = 28 |
|---|---|---|---|---|---|
| Child’s age (years) | 7.74 (4.68) | 9.22 (4.46) | 8.00 (4.37) | 7.33 (4.48) | 8.07 (3.96) |
| Caregiver’s age (years) | 41.63 (8.30) | 43.31 (9.69) | 41.65 (10.08) | 40.07 (9.90) | 38.71 (8.44) |
| Child’s family situation | |||||
| Two-parent family (cases) | 14 | 24 | 33 | 50 | 16 |
| One-parent family (cases) | 5 | 8 | 13 | 29 | 11 |
| Other (cases) | - | - | - | 2 | 1 |
| Cramer’s V (p-Value)1 | Child’s Age | Caregiver’s Age | Family Type | Main Caregiver | Place of Residence | Material Situation (Self-Assessed) | Material Situation (Objectively) |
|---|---|---|---|---|---|---|---|
| Child’s Health and Therapy—Module II | |||||||
| Frequency of control visits | - | - | - | 0.343 (<0.001) | - | - | 0.251 (0.027) |
| Age at diagnosis | 0.687 (<0.001) | 0.354 (<0.001) | - | 0.354 (<0.001) | - | - | - |
| Insulin delivery method | - | - | - | - | 0.564 (<0.001) | 0.588 (<0.001) | 0.771 (<0.001) |
| Regularity of control visits | - | - | 0.157 (0.038) | - | - | - | - |
| Attending physician | - | - | - | - | - | - | - |
| Consultation (last 12 months) | - | - | - | - | 0.507 (<0.001) | 0.561 (<0.001) | 0.653 (<0.001) |
| Consultation (last 5 years) | - | - | - | 0.274 (0.004) | 0.529 (<0.001) | 0.586 (<0.001) | 0.643 (<0.001) |
| Occurrence of complications | - | - | - | - | - | - | - |
| Cramer’s V (p-Value)1 | Child’s Age | Caregiver’s Age | Family Type | Main Caregiver | Place of Residence | Material Situation (Self-Assessed) | Material Situation (Objectively) |
|---|---|---|---|---|---|---|---|
| Child’s and Family’s Life—Module III | |||||||
| Changes in professional activity | - | - | - | - | - | - | - |
| Limited access to activities | - | - | - | - | 0.224 (0.003) | 0.242 (<0.001) | 0.304 (<0.001) |
| Change of family situation | - | - | - | 0.321 (0.018) | - | - | - |
| Out of pocket health spending | - | - | 0.185 (0.029) | - | 0.682 (<0.001) | 0.737 (<0.001) | 0.493 (<0.001) |
| Caregiver’s concerns | 0.321 (0.007) | 0.240 (0.040) | 0.219 (0.006) | - | 0.239 (0.042) | 0.254 (0.011) | 0.252 (0.001) |
| Child’s concerns | 0.414 (<0.001) | 0.366 (<0.001) | 0.184 (0.030) | - | - | - | - |
| Support Sources | 0.332 (0.006) | - | - | 0.321 (0.018) | - | - | - |
| Provision of Care—Module IV | |||||||
| Day-time care (3rd person) | 0.808 (<0.001) | 0.391 (<0.001) | - | - | - | - | - |
| Night-time care | - | - | - | - | - | - | - |
| Access to institutional care | 0.762 (<0.001) | 0.433 (<0.001) | - | - | 0.314 (0.037) | - | - |
| Cramer’s V (p-Value)1 | Age at Diagnosis | Insulin Delivery Method | Main Doctor | Frequency of Control Visits | Regularity of Control Visits | Consultations (Last 12 Months) | Consultations (Last 5 Years) | Occurrence of Complications | |
|---|---|---|---|---|---|---|---|---|---|
| Child’s and Family’s Life—Module III | |||||||||
| Changes in professional activity | - | 0.141 (0.043) | - | 0.255 (0.004) | - | - | - | - | |
| Limited access to activities | - | - | - | - | - | 0.230 (0.001) | 0.217 (0.003) | - | |
| Change of family Situation | - | - | - | - | - | - | - | 0.430 (0.001) | |
| Out of pocket health spending | - | 0.558 (<0.001) | - | 0.244 (0.044) | 0.261 (0.032) | 0.588 (<0.001) | 0.592 (<0.001) | - | |
| Caregiver’s concerns | 0.235 (0.003) | - | - | - | - | 0.140 0.044) | - | - | |
| Child’s concerns | 0.408 (<0.001) | - | - | - | - | - | - | - | |
| Support sources | 0.349 (<0.001) | 0.258 (0.039) | - | - | - | - | |||
| Provision of Care—Module IV | |||||||||
| Day-time care (3rd person) | 0.592 (<0.001) | - | - | - | - | - | - | - | |
| Night-time care | 0.241 (0.046) | - | 0.259 (0.001) | - | - | - | - | - | |
| Access to institutional care | 0.628 (<0.001) | - | - | - | - | - | - | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grudziąż-Sękowska, J.; Zamarlik, M.; Sękowski, K. Assessment of Selected Aspects of the Quality of Life of Children with Type 1 Diabetes Mellitus in Poland. Int. J. Environ. Res. Public Health 2021, 18, 2107. https://doi.org/10.3390/ijerph18042107
Grudziąż-Sękowska J, Zamarlik M, Sękowski K. Assessment of Selected Aspects of the Quality of Life of Children with Type 1 Diabetes Mellitus in Poland. International Journal of Environmental Research and Public Health. 2021; 18(4):2107. https://doi.org/10.3390/ijerph18042107
Chicago/Turabian StyleGrudziąż-Sękowska, Justyna, Monika Zamarlik, and Kuba Sękowski. 2021. "Assessment of Selected Aspects of the Quality of Life of Children with Type 1 Diabetes Mellitus in Poland" International Journal of Environmental Research and Public Health 18, no. 4: 2107. https://doi.org/10.3390/ijerph18042107
APA StyleGrudziąż-Sękowska, J., Zamarlik, M., & Sękowski, K. (2021). Assessment of Selected Aspects of the Quality of Life of Children with Type 1 Diabetes Mellitus in Poland. International Journal of Environmental Research and Public Health, 18(4), 2107. https://doi.org/10.3390/ijerph18042107








