Ultrasound Imaging Evaluation of Textural Features in Athletes with Soleus Pathology—A Novel Case-Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Ethical Considerations
2.3. Sample Size Calculation
2.4. Participants
2.5. Clinical Variables
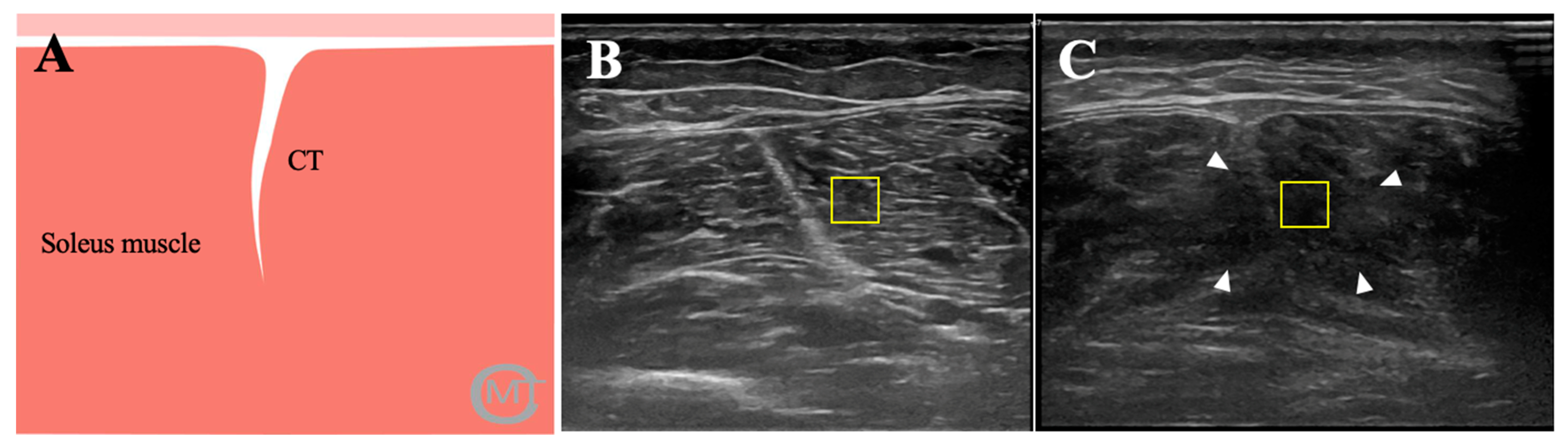
2.6. Ultrasonography
2.7. Image Analysis
2.8. Statistics Analysis
3. Results
4. Discussion
4.1. Clinical Implications
4.2. Limitations and Future Lines
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brukner, P.D.; Connell, D. ‘Serious thigh muscle strains’: Beware the intramuscular tendon which plays an important role in difficult hamstring and quadriceps muscle strains. Br. J. Sports Med. 2015, 50, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Koulouris, G.; Ting, A.Y.I.; Jhamb, A.; Connell, D.; Kavanagh, E.C. Magnetic resonance imaging findings of injuries to the calf muscle complex. Skelet. Radiol. 2007, 36, 921–927. [Google Scholar] [CrossRef]
- Courthaliac, C.; Weilbacher, H. Imaging of painful calf in athletes. J. Radiol. 2007, 88 Pt 2, 200–208. [Google Scholar] [CrossRef]
- Śmigielski, R. Management of Partial Tears of the Gastro-Soleus Complex. Clin. Sports Med. 2008, 27, 219–229. [Google Scholar] [CrossRef]
- Balius, R.; AlOmar, X.; Rodas, G.; Miguel-Pérez, M.; Pedret, C.; Dobado, M.C.; Blasi, J.; Koulouris, G. The soleus muscle: MRI, anatomic and histologic findings in cadavers with clinical correlation of strain injury distribution. Skelet. Radiol. 2012, 42, 521–530. [Google Scholar] [CrossRef]
- Brukner, P.; Cook, J.L.; Purdam, C.R. Does the intramuscular tendon act like a free tendon? Br. J. Sports Med. 2018, 52, 1227–1228. [Google Scholar] [CrossRef] [PubMed]
- Orchard, J.; Best, T.M. The Management of Muscle Strain Injuries: An Early Return Versus the Risk of Recurrence. Clin. J. Sport Med. 2002, 12, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Orchard, J.W.; Best, T.M.; Mueller-Wohlfahrt, H.-W.; Hunter, G.; Hamilton, B.H.; Webborn, A.; Jaques, R.; Kenneally, D.; Budgett, R.; Phillips, N.; et al. The early management of muscle strains in the elite athlete: Best practice in a world with a limited evidence basis. Br. J. Sports Med. 2008, 42, 158–159. [Google Scholar] [CrossRef] [PubMed]
- Balius, R.; Rodas, G.; Pedret, C.; Capdevila, L.; AlOmar, X.; Bong, D.A. Soleus muscle injury: Sensitivity of ultrasound patterns. Skelet. Radiol. 2014, 43, 805–812. [Google Scholar] [CrossRef]
- Loizides, A.; Gruber, H.; Peer, S.; Plaikner, M. Muscular injuries of athletes: Importance of ultrasound. Radiologe 2017, 57, 1019–1028. [Google Scholar] [CrossRef]
- Åström, M.; Gentz, C.-F.; Nilsson, P.; Rausing, A.; Sjöberg, S.; Westlin, N. Imaging in chronic achilles tendinopathy: A comparison of ultrasonography, magnetic resonance imaging and surgical findings in 27 histologically verified cases. Skelet. Radiol. 1996, 25, 615–620. [Google Scholar] [CrossRef]
- Fukumoto, Y.; Ikezoe, T.; Yamada, Y.; Tsukagoshi, R.; Nakamura, M.; Mori, N.; Kimura, M.; Ichihashi, N. Skeletal muscle quality assessed from echo intensity is associated with muscle strength of middle-aged and elderly persons. Graefe’s Arch. Clin. Exp. Ophthalmol. 2012, 112, 1519–1525. [Google Scholar] [CrossRef]
- Wong, V.; Abe, T.; Chatakondi, R.N.; Bell, Z.W.; Spitz, R.W.; Dankel, S.J.; Loenneke, J.P. The influence of biological sex and cuff width on muscle swelling, echo intensity, and the fatigue response to blood flow restricted exercise. J. Sports Sci. 2019, 37, 1865–1873. [Google Scholar] [CrossRef]
- Wong, V.; Spitz, R.W.; Bell, Z.W.; Viana, R.B.; Chatakondi, R.N.; Abe, T.; Loenneke, J.P. Exercise induced changes in echo intensity within the muscle: A brief review. J. Ultrasound 2020, 23, 457–472. [Google Scholar] [CrossRef]
- Medeiros, D.M.; Mantovani, R.F.; Lima, C.S. Effects of low-intensity pulsed ultrasound on muscle thickness and echo intensity of the elbow flexors following exercise-induced muscle damage. Sport Sci. Health 2017, 13, 365–371. [Google Scholar] [CrossRef]
- Yoshiko, A.; Tomita, A.; Ando, R.; Ogawa, M.; Kondo, S.; Saito, A.; Tanaka, N.I.; Koike, T.; Oshida, Y.; Akima, H. Effects of 10-week walking and walking with home-based resistance training on muscle quality, muscle size, and physical functional tests in healthy older individuals. Eur. Rev. Aging Phys. Act. 2018, 15, 1–10. [Google Scholar] [CrossRef]
- Martínez-Payá, J.J.; Ríos-Díaz, J.; Del Baño-Aledo, M.E.; Tembl-Ferrairó, J.I.; Vazquez-Costa, J.F.; Medina-Mirapeix, F. Quantitative Muscle Ultrasonography Using Textural Analysis in Amyotrophic Lateral Sclerosis. Ultrason. Imaging 2017, 39, 357–368. [Google Scholar] [CrossRef]
- Arts, I.M.; Overeem, S.; Pillen, S.; Kleine, B.U.; Boekestein, W.A.; Zwarts, M.J.; Schelhaas, H.J. Muscle ultrasonography: A diagnostic tool for amyotrophic lateral sclerosis. Clin. Neurophysiol. 2012, 123, 1662–1667. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Meschi, T.; Narici, M.V.; Lauretani, F.; Maggio, M. Muscle Ultrasound and Sarcopenia in Older Individuals: A Clinical Perspective. J. Am. Med Dir. Assoc. 2017, 18, 290–300. [Google Scholar] [CrossRef]
- Yitzchaki, N.; Zhu, W.G.; Kuehne, T.E.; Vasenina, E.; Dankel, S.J.; Buckner, S.L. An examination of changes in skeletal muscle thickness, echo intensity, strength and soreness following resistance exercise. Clin. Physiol. Funct. Imaging 2020, 40, 238–244. [Google Scholar] [CrossRef]
- Pillen, S.; Tak, R.O.; Zwarts, M.J.; Lammens, M.M.; Verrijp, K.N.; Arts, I.M.; Van Der Laak, J.A.; Hoogerbrugge, P.M.; Van Engelen, B.G.; Verrips, A. Skeletal Muscle Ultrasound: Correlation Between Fibrous Tissue and Echo Intensity. Ultrasound Med. Biol. 2009, 35, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Payá, J.J.; Ríos-Díaz, J.; Medina-Mirapeix, F.; Vázquez-Costa, J.F.; Del Baño-Aledo, M.E. Monitoring Progression of Amyotrophic Lateral Sclerosis Using Ultrasound Morpho-Textural Muscle Biomarkers: A Pilot Study. Ultrasound. Med. Biol. 2018, 44, 102–109. [Google Scholar] [CrossRef]
- Martínez-Payá, J.J.; Del Baño-Aledo, M.E.; Ríos-Díaz, J.; Tembl-Ferrairó, J.I.; Vázquez-Costa, J.F.; Medina-Mirapeix, F. Muscular Echovariation: A New Biomarker in Amyotrophic Lateral Sclerosis. Ultrasound Med. Biol. 2017, 43, 1153–1162. [Google Scholar] [CrossRef]
- Ríos-Díaz, J.; Martínez-Payá, J.J.; Del Baño-Aledo, M.E.; De Groot-Ferrando, A.; Botía-Castillo, P.; Fernández-Rodríguez, D. Sonoelastography of Plantar Fascia: Reproducibility and Pattern Description in Healthy Subjects and Symptomatic Subjects. Ultrasound Med. Biol. 2015, 41, 2605–2613. [Google Scholar] [CrossRef]
- Almazán-Polo, J.; López-López, D.; Romero-Morales, C.; Rodríguez-Sanz, D.; Becerro-De-Bengoa-Vallejo, R.; Losa-Iglesias, M.E.; Bravo-Aguilar, M.; Calvo-Lobo, C. Quantitative Ultrasound Imaging Differences in Multifidus and Thoracolumbar Fasciae between Athletes with and without Chronic Lumbopelvic Pain: A Case-Control Study. J. Clin. Med. 2020, 9, 2647. [Google Scholar] [CrossRef]
- De-La-Cruz-Torres, B.; Barrera-García-Martín, I.; Almazán-Polo, J.; Jaén-Crespo, G.; Romero-Morales, C. Ultrasound imaging evaluation of structural and textural features in asymptomatic achilles tendons in pre-professional dancers: A cross-sectional study. Phys. Ther. Sport 2020, 44, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Downie, W.W.; Leatham, P.A.; Rhind, V.M.; Wright, V.; Branco, J.A.; Anderson, J.A. Studies with pain rating scales. Ann. Rheum. Dis. 1978, 37, 378–381. [Google Scholar] [CrossRef]
- Caresio, C.; Molinari, F.; Emanuel, G.; Minetto, M.A. Muscle echo intensity: Reliability and conditioning factors. Clin. Physiol. Funct. Imaging 2014, 35, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Heckmatt, J.; Dubowitz, V.; Leeman, S. Detection of pathological change in dystrophic muscle with B-scan ultrasound imaging. Lancet 1980, 315, 1389–1390. [Google Scholar] [CrossRef]
- Giacchino, M.; Caresio, C.; Gorji, N.; Molinari, F.; Massazza, G.; Minetto, M. Quantitative analysis of patellar tendon size and structure in asymptomatic professional players: Sonographic study. Muscle Ligaments Tendons J. 2019, 7, 449. [Google Scholar] [CrossRef]
- Romero-Morales, C.; Calvo-Lobo, C.; Navarro-Flores, E.; Mazoteras-Pardo, V.; García-Bermejo, P.; López-López, D.; Martínez-Jiménez, E.M.; De-La-Cruz-Torres, B. M-mode Ultrasound Examination of Soleus Muscle in Healthy Subjects: Intra- and Inter-Rater Reliability Study. Healthcare 2020, 8, 555. [Google Scholar] [CrossRef] [PubMed]
- De-La-Cruz-Torres, B.; Barrera-García-Martín, I.; Valera-Garrido, F.; Minaya-Muñoz, F.; Romero-Morales, C. Ultrasound-Guided Percutaneous Needle Electrolysis in Dancers with Chronic Soleus Injury: A Randomized Clinical Trial. Evidence-Based Complement. Altern. Med. 2020, 2020, 4156258. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Lobo, C.; Almazán-Polo, J.; Becerro-De-Bengoa-Vallejo, R.; Losa-Iglesias, M.E.; Palomo-López, P.; Rodríguez-Sanz, D.; López-López, D. Ultrasonography comparison of diaphragm thickness and excursion between athletes with and without lumbopelvic pain. Phys. Ther. Sport Off. J. Assoc. Chart. Physiother. Sports Med. 2019, 37, 128–137. [Google Scholar] [CrossRef]
| Data | Total (n = 62) | Healthy Group (n = 31) | Patient Group (n= 31) | p-Value |
|---|---|---|---|---|
| Age, y | 27.00 ± 13.50 † | 27.00 ± 16.00 † | 27.00 ± 12.00 † | 0.994 ‡ |
| Weight, kg | 65.75 ± 16.65 † | 66.00 ± 19.00 † | 65.00 ± 16.00 † | 0.390 ‡ |
| Height, m | 1.69 ± 0.15 * | 1.70 ± 0.09 * | 1.67 ± 0.08 * | 0.095 ** |
| BMI, kg/m2 | 23.26 ± 3.05 † | 23.41 ± 2.89 † | 23.18 ± 3.01 † | 0.938 ‡ |
| NRS | 5.03 ± 1.38 † | n/a | 5.03 ± 1.38 † | n/a |
| NRS at palpation | 5.81 ± 1.53 † | n/a | 5.81 ± 1.53 † | n/a |
| MTh (mm) | 17.94 ± 2.31 * | 17.68 ± 1.77 * | 18.20 ± 2.75 * | 0.381 ** |
| Echointensity (EI) | 50.17 ± 18.94 * | 63.74 ± 12.86 * | 36.71 ± 13.78 * | 0.001 ** |
| Echovariation (EV) | 30.93 ± 12.11 † | 33.07 ± 10.13 † | 28.69 ± 16.75 † | 0.364 ‡ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De-la-Cruz-Torres, B.; Navarro-Flores, E.; López-López, D.; Romero-Morales, C. Ultrasound Imaging Evaluation of Textural Features in Athletes with Soleus Pathology—A Novel Case-Control Study. Int. J. Environ. Res. Public Health 2021, 18, 1983. https://doi.org/10.3390/ijerph18041983
De-la-Cruz-Torres B, Navarro-Flores E, López-López D, Romero-Morales C. Ultrasound Imaging Evaluation of Textural Features in Athletes with Soleus Pathology—A Novel Case-Control Study. International Journal of Environmental Research and Public Health. 2021; 18(4):1983. https://doi.org/10.3390/ijerph18041983
Chicago/Turabian StyleDe-la-Cruz-Torres, Blanca, Emmanuel Navarro-Flores, Daniel López-López, and Carlos Romero-Morales. 2021. "Ultrasound Imaging Evaluation of Textural Features in Athletes with Soleus Pathology—A Novel Case-Control Study" International Journal of Environmental Research and Public Health 18, no. 4: 1983. https://doi.org/10.3390/ijerph18041983
APA StyleDe-la-Cruz-Torres, B., Navarro-Flores, E., López-López, D., & Romero-Morales, C. (2021). Ultrasound Imaging Evaluation of Textural Features in Athletes with Soleus Pathology—A Novel Case-Control Study. International Journal of Environmental Research and Public Health, 18(4), 1983. https://doi.org/10.3390/ijerph18041983









