Prevalence of Fetal Alcohol Spectrum Disorders (FASD) among Children Adopted from Eastern European Countries: Russia and Ukraine
Abstract
1. Introduction
1.1. Alcohol Consumption and Impact on Health and Harm to Others
1.2. Epidemiological Data of Prenatal Exposure to Alcohol (PEA) and FASD
1.3. FASD Prevalence in Adopted Children from Eastern European Countries
2. Patients and Methods
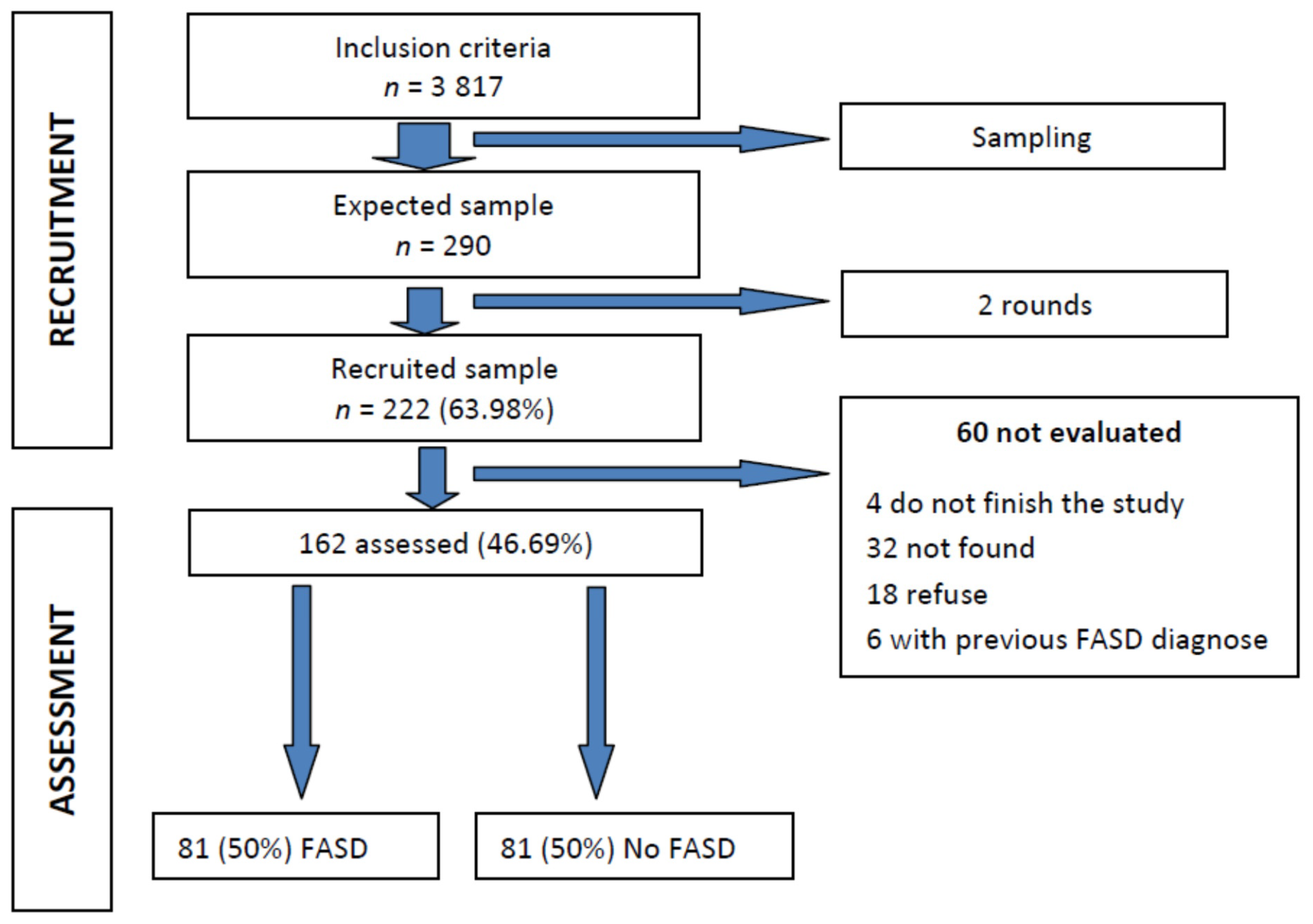
2.1. Sampling Methods and Description
2.2. Recruitment
2.3. Assessment
2.4. Statistical Analysis
2.5. Ethical Aspects
3. Results
General Description of FASD Umbrella Diagnostic Results, Differences by Age, Gender and Year of Adoption
4. Discussion
4.1. Comparison with Other Studies (Strength of the Study)
4.2. Limitations
4.3. Challenges and Opportunities under a Public Health Perspective
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADHD | attention-deficit/hyperactivity disorder |
| ARBD | alcohol-related birth defect |
| ARND | alcohol-related neurodevelopmental disorder |
| FAS | fetal alcohol syndrome |
| FASD | fetal alcohol spectrum disorder |
| IOM | Institute of Medicine |
| OFC | occipitofrontal circumference |
| PEA | prenatal exposure to alcohol |
| pFAS | partial fetal alcohol syndrome |
References
- GBD 206 Alcohol Collaborators. Alcohol use and burden for 195 countries and territories, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2018, 392, e1015–e1035. [Google Scholar] [CrossRef]
- Pichini, S.; Busardò, F.P.; Garcia-Algar, O. Only Total Abstinence From Alcohol Consumption During Pregnancy Guarantees Absolute Absence of Any Deleterious Effect in the Fetus and Child. J. Stud. Alcohol Drugs 2020, 81, 220–221. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Status Report on Alcohol and Health 2018; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Sebastiani, G.; Borrás-Novell, C.; Casanova, M.A.; Pascual, M.; Ferrero, S.; Gómez-Roig, M.D.; García-Algar, O. The Effects of Alcohol and Drugs of Abuse on Maternal Nutritional Profile during Pregnancy. Nutrients 2018, 10, 1008. [Google Scholar] [CrossRef]
- Bastons-Compta, A.; Astals, M.; Andreu-Fernandez, V.; Navarro, E.; García-Algar, Ó. Postnatal nutritional treatment of neurocognitive deficits in fetal alcohol spectrum disorder. Biochem. Cell Biol. 2018, 96, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Hoyme, H.E.; Kalberg, W.O.; Elliott, A.J.; Blankenship, J.; Buckley, D.; Marais, A.-S.; Manning, M.A.; Robinson, L.K.; Adam, M.P.; Abdul-Rahman, O.; et al. Updated Clinical Guidelines for Diagnosing Fetal Alcohol Spectrum Disorders. Pediatrics 2016, 138, e20154256. [Google Scholar] [CrossRef] [PubMed]
- Bower, C.; Elliott, E.J.; Steering Group. Report to the Australian Government Department of Health: Australian Guide to the Diagnosis of Fetal Alcohol Spectrum Disorder (FASD); Australian Government Department of Health: Sydney, Australia, 2020.
- Memo, L.; Gnoato, E.; Caminiti, S.; Pichini, S.; Tarani, L. Fetal alcohol spectrum disorders and fetal alcohol syndrome: The state of the art and new diagnostic tools. Early Hum. Dev. 2013, 89, S40–S43. [Google Scholar] [CrossRef]
- Popova, S.; Lange, S.; Probst, C.; Gmel, G.; Rehm, J. Estimation of national, regional, and global prevalence of alcohol use dur-ing pregnancy and fetal alcohol syndrome: A systematic review and meta-analysis. Lancet Glob. Health 2017, 5, e290–e299. [Google Scholar] [CrossRef]
- Chiandetti, A.; Hernandez, G.; Mercadal-Hally, M.; Alvarez, A.; Andreu-Fernandez, V.; Navarro, E.; Bastons-Compta, A.; Garcia-Algar, O. Prevalence of prenatal exposure to substances of abuse: Questionnaire versus biomarkers. Reprod. Health 2017, 14, 1–12. [Google Scholar] [CrossRef]
- Lange, S.; Shield, K.D.; Koren, G.; Rehm, J.; Popova, S. A comparison of the prevalence of prenatal alcohol exposure obtained via maternal self-reports versus meconium testing: A systematic literature review and meta-analysis. BMC Pregnancy Childbirth 2014, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lange, S.; Probst, C.; Gmel, G.; Rehm, J.; Burd, L.; Popova, S. Global Prevalence of Fetal Alcohol Spectrum Disorder Among Children and Youth: A Systematic Review and Meta-analysis. JAMA Pediatr. 2017, 171, 948–956. [Google Scholar] [CrossRef]
- Messina, M.P.; D’angelo, A.; Battagliese, G.; Coriale, G.; Tarani, L.; Pichini, S.; Rasio, D.; Parlapiano, G.; Fiore, M.; Petrella, C.; et al. Fetal alcohol spectrum disorders awareness in health professionals: Implications for psychiatry. Riv. Psichiatr. 2020, 55, 79–89. [Google Scholar] [PubMed]
- May, P.A.; Fiorentino, D.; Gossage, J.P.; Kalberg, W.O.; Hoyme, H.E.; Robinson, L.K.; Coriale, G.; Jones, K.L.; Del Campo, M.; Tarani, L.; et al. Epidemiology of FASD in a Province in Italy: Prevalence and Characteristics of Children in a Random Sample of Schools. Alcohol. Clin. Exp. Res. 2006, 30, 1562–1575. [Google Scholar] [CrossRef] [PubMed]
- A Quarter of Adopted UK Children Affected by Drinking during Pregnancy. Available online: https://www.theguardian.com/society/2020/sep/29/a-quarter-of-adopted-uk-children-affected-by-drinking-during-pregnancy (accessed on 14 December 2020).
- McQuire, C.; Mukherjee, R.; Hurt, L.; Higgins, A.; Greene, G.; Farewell, D.; Kemp, A.; Paranjothy, S. Screening prevalence of fetal al-cohol spectrum disorders in a region of the United Kingdom: A population-based birth-cohort study. Prev. Med. 2019, 118, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Popova, S.; Lange, S.; Shield, K.D.; Mihic, A.; Chudley, A.E.; Mukherjee, R.A.S.; Bekmuradov, D.; Rehm, J. Comorbidity of fetal alcohol spectrum disorder: A systematic review and meta-analysis. Lancet 2016, 387, 978–987. [Google Scholar] [CrossRef]
- Popova, S.; Lange, S.; Shield, K.; Burd, L.; Rehm, J. Prevalence of fetal alcohol spectrum disorder among special subpopula-tions: A systematic review and meta-analysis. Addiction 2019, 114, 1150–1172. [Google Scholar] [CrossRef]
- Tenenbaum, A.; Mandel, A.; Dor, T.; Sapir, A.; Sapir-Bodnaro, O.; Hertz, P.; Wexler, I.D. Fetal alcohol spectrum disorder among pre-adopted and foster children. BMC Pediatr. 2020, 20, 1–6. [Google Scholar] [CrossRef]
- Nissinen, N.N.; Frederiksen, N. Identifying Use of Alcohol and Other Substances during Pregnancy. A Nordic Overview; Nordic Welfare Centre: Stockholm, Sweden, 2020. [Google Scholar]
- Peñarrubia, M.; Palacios, J.; Román, M. Executive function and early adversity in internationally adopted children. Child. Youth Serv. Rev. 2020, 108, 104587. [Google Scholar] [CrossRef]
- Lange, S.; Shield, K.; Rehm, J.; Anagnostou, E.; Popova, S. Fetal alcohol spectrum disorder: Neurodevelopmentally and be-haviorally indistinguishable from other neurodevelopmental disorders. BMC Psychiatry 2019, 19, 1–10. [Google Scholar] [CrossRef]
- Popova, S.; Yaltonskaya, A.; Yaltonsky, V.; Kolpakov, Y.; Abrosimov, I.; Pervakov, K.; Tanner, V.; Rehm, J. What research is being done on prenatal alcohol exposure and fetal alcohol spectrum disorders in the Russian research community? Alcohol Alcohol. 2013, 49, 84–95. [Google Scholar] [CrossRef]
- Landgren, M.; Svensson, L.; Strömland, K.; Andersson Grönlund, M. Prenatal alcohol exposure and neurodevelopmental dis-orders in children adopted from Eastern Europe. Pediatrics 2010, 125, e1178–e1185. [Google Scholar] [CrossRef]
- Landgren, V.; Svensson, L.; Gyllencreutz, E.; Aring, E.; Grönlund, M.A.; Landgren, M. Fetal alcohol spectrum disorders from childhood to adulthood: A Swedish population-based naturalistic cohort study of adoptees from Eastern Europe. BMJ Open 2019, 9, e032407. [Google Scholar] [CrossRef] [PubMed]
- Institut Català de l’Acolliment i de l’Adopció (ICAA). Departament de Treball, Afers Socials i Famílies. Generalitat de Calaunya. Barcelona. 2020. Available online: https://treballiaferssocials.gencat.cat/ca/ambits_tematics/acolliments_i_adopcions/estadistiques/ (accessed on 14 December 2020).
- GRANMO Software. Available online: https://www.imim.es/ofertadeserveis/software-public/granmo/ (accessed on 14 December 2020).
- Astley, S.J.; Bledsoe, J.M.; Brooks, A.; Davies, J.K.; Jirikowic, T.; Olson, E.; Thorne, J.C. Comparison of the 4-Digit Code, Canadian 2015, Australian 2016 and Hoyme 2016 fetal alcohol spectrum disorder diagnostic guidelines. Adv. Pediatr. Res. 2019, 6, 31. [Google Scholar] [CrossRef]
- Scottish Intercollegiate Guidelines Network (SIGN). Children and Young People Exposed Prenatally to Alcohol; SIGN Publication No. 156; SIGN: Edinburgh, UK, 2019; Available online: http://www.sign.ac.uk (accessed on 14 December 2020).
- Knuiman, S.; Rijk, C.H.A.M.; Hoksbergen, R.A.; Van Baar, A.L. Children adopted from Poland display a high risk of foetal alcohol spectrum disorders and some may go undiagnosed. Acta Paediatr. 2014, 104, 206–211. [Google Scholar] [CrossRef] [PubMed]
| Documented Prenatal Alcohol Exposure | Criteria Required | Features | |
|---|---|---|---|
| FAS | Yes or no | A to D | A. A characteristic pattern of minor facial anomalies (≥2): 1. Short palpebral fissures (≤10th percentile) 2. Thin vermilion border of the upper lip (rank 4 or 5) 3. Smooth philtrum (rank 4 or 5) B. Prenatal and/or postnatal growth deficiency 1. Height and/or weight ≤10th percentile C. Deficient brain growth, abnormal morphogenesis or abnormal neurophysiology (≥1): 1. Head circumference ≤10th percentile 2. Structural brain anomalies 3. Recurrent nonfebrile seizures D. Neurobehavioral impairment 1. For children ≥3 y of age (a or b): a. With cognitive impairment: −Evidence of global impairment, OR −Cognitive deficit in at least 1 neurobehavioral domain b. With behavioral impairment without cognitive impairment: −Evidence of behavioral deficit in at least 1 domain 2. For children <3 y of age: −Evidence of developmental delay |
| pFAS | Yes | A and B | A. A characteristic pattern of minor facial anomalies (≥2): 1. Short palpebral fissures (≤10th percentile) 2. Thin vermilion border of the upper lip (rank 4 or 5) 3. Smooth philtrum (rank 4 or 5) B. Neurobehavioral impairment 1. For children ≥3 y of age (a or b): a. With cognitive impairment: −Evidence of global impairment, OR −Cognitive deficit in at least 1 neurobehavioral domain b. With behavioral impairment without cognitive impairment: −Evidence of behavioral deficit in at least 1 domain 2. For children <3 y of age: −Evidence of developmental delay |
| No | A to C | A. A characteristic pattern of minor facial anomalies (≥2): 1. Short palpebral fissures (≤10th percentile) 2. Thin vermilion border of the upper lip (rank 4 or 5) 3. Smooth philtrum (rank 4 or 5) B. Growth deficiency or deficient brain growth, abnormal morphogenesis or abnormal neurophysiology 1. Height and/or weight ≤10th percentile, OR 2. Deficient brain growth, abnormal morphogenesis or neurophysiology (≥1): a. Head circumference ≤10th percentile b. Structural brain anomalies c. Recurrent nonfebrile seizures C. Neurobehavioral impairment 1. For children ≥3 y of age (a or b): a. With cognitive impairment: −Evidence of global impairment, OR −Cognitive deficit in at least 1 neurobehavioral domain b. With behavioral impairment without cognitive impairment: −Evidence of behavioral deficit in at least 1 domain 2. For children <3 y of age: −Evidence of developmental delay | |
| ARND | Yes | A and B | A. Documented prenatal alcohol exposure B. Neurobehavioral impairment For children ≥3 y of age (a or b): a. With cognitive impairment: −Evidence of global impairment, OR −Cognitive deficit in at least 2 neurobehavioral domains b. With behavioral impairment without cognitive impairment: −Evidence of behavioral deficit in at least 2 domains |
| ARBD | Yes | A and B | A. Documented prenatal alcohol exposure B. One or more specific major malformations demonstrated in animal models and human studies to be the result of prenatal alcohol exposure |
| Letters Sent | Accepted | Refused | Returned by Post | No Response | |
|---|---|---|---|---|---|
| 1st round | 450 | 157 (34.89%) | 25 (5.56%) | 132 (29.33%) | 136 (30.22%) |
| 2nd round | 300 | 65 (21.67%) | 24 (8%) | 100 (33.33%) | 111 (37.00%) |
| Total | 750 | 222 (29.6%) | 49 (6.53%) | 232 (30.93%) | 247 (32.93%) |
| Sex | |||||
|---|---|---|---|---|---|
| Female | Male | Total | |||
| Diagnosis | NO FASD | n | 34 | 47 | 81 |
| % diagnosis | 42.0% | 58.0% | 100.0% | ||
| % by gender | 55.7% | 46.5% | 50.0% | ||
| % total | 21.0% | 29.0% | 50.0% | ||
| ARBD | n | 2 | 0 | 2 | |
| % diagnosis | 100.0% | 0.0% | 100.0% | ||
| % by gender | 3.3% | 0.0% | 1.2% | ||
| % total | 1.2% | 0.0% | 1.2% | ||
| ARND | n | 5 | 13 | 18 | |
| % diagnosis | 27.8% | 72.2% | 100.0% | ||
| % by gender | 8.2% | 12.9% | 11.1% | ||
| % total | 3.1% | 8.0% | 11.1% | ||
| FAS | n | 12 | 21 | 33 | |
| % diagnosis | 36.4% | 63,6% | 100.0% | ||
| % by gender | 19.7% | 20.8% | 20.4% | ||
| % total | 7.4% | 13.0% | 20.4% | ||
| pFAS | n | 8 | 20 | 28 | |
| % diagnosis | 28.6% | 71.4% | 100.0% | ||
| % by gender | 13.1% | 19.8% | 17.3% | ||
| % total | 4.9% | 12.3% | 17.3% | ||
| TOTAL | n | 61 | 101 | 162 | |
| % diagnosis | 37.7% | 62.3% | 100.0% | ||
| % by gender | 100.0% | 100.0% | 100.0% | ||
| % total | 37.7% | 62.3% | 100.0% | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colom, J.; Segura-García, L.; Bastons-Compta, A.; Astals, M.; Andreu-Fernandez, V.; Barcons, N.; Vidal, R.; Ibar, A.I.; Fumadó, V.; Gómez, N.; et al. Prevalence of Fetal Alcohol Spectrum Disorders (FASD) among Children Adopted from Eastern European Countries: Russia and Ukraine. Int. J. Environ. Res. Public Health 2021, 18, 1388. https://doi.org/10.3390/ijerph18041388
Colom J, Segura-García L, Bastons-Compta A, Astals M, Andreu-Fernandez V, Barcons N, Vidal R, Ibar AI, Fumadó V, Gómez N, et al. Prevalence of Fetal Alcohol Spectrum Disorders (FASD) among Children Adopted from Eastern European Countries: Russia and Ukraine. International Journal of Environmental Research and Public Health. 2021; 18(4):1388. https://doi.org/10.3390/ijerph18041388
Chicago/Turabian StyleColom, Joan, Lidia Segura-García, Adriana Bastons-Compta, Marta Astals, Vicente Andreu-Fernandez, Natalia Barcons, Raquel Vidal, Ana I. Ibar, Vicky Fumadó, Nuria Gómez, and et al. 2021. "Prevalence of Fetal Alcohol Spectrum Disorders (FASD) among Children Adopted from Eastern European Countries: Russia and Ukraine" International Journal of Environmental Research and Public Health 18, no. 4: 1388. https://doi.org/10.3390/ijerph18041388
APA StyleColom, J., Segura-García, L., Bastons-Compta, A., Astals, M., Andreu-Fernandez, V., Barcons, N., Vidal, R., Ibar, A. I., Fumadó, V., Gómez, N., Russiñol, A., & Garcia-Algar, O. (2021). Prevalence of Fetal Alcohol Spectrum Disorders (FASD) among Children Adopted from Eastern European Countries: Russia and Ukraine. International Journal of Environmental Research and Public Health, 18(4), 1388. https://doi.org/10.3390/ijerph18041388









