Agriculture Development, Pesticide Application and Its Impact on the Environment
Abstract
1. Introduction
2. Agricultural Development Process
3. Historical Perspectives of Pesticide Usage
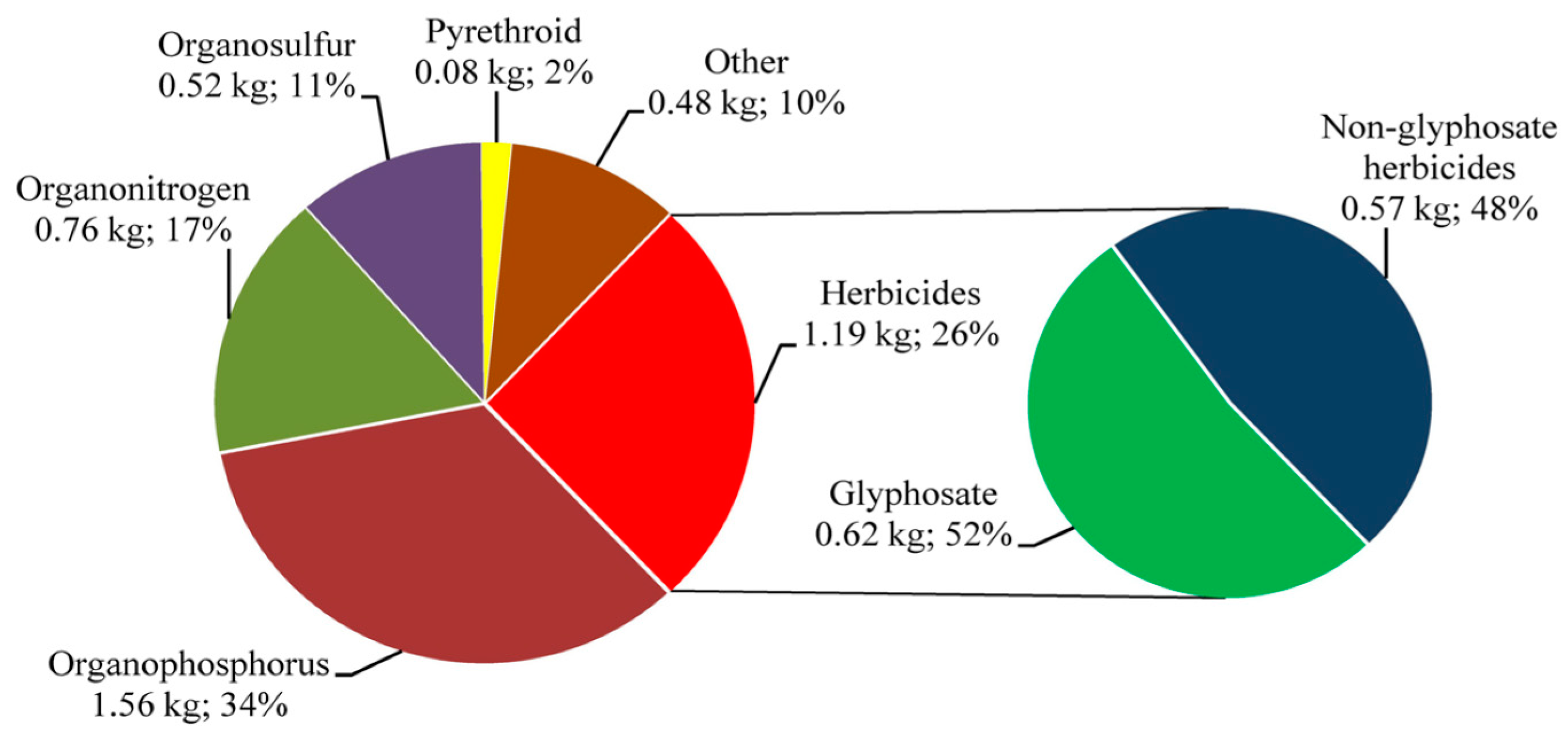
4. Types of Pesticide in Use
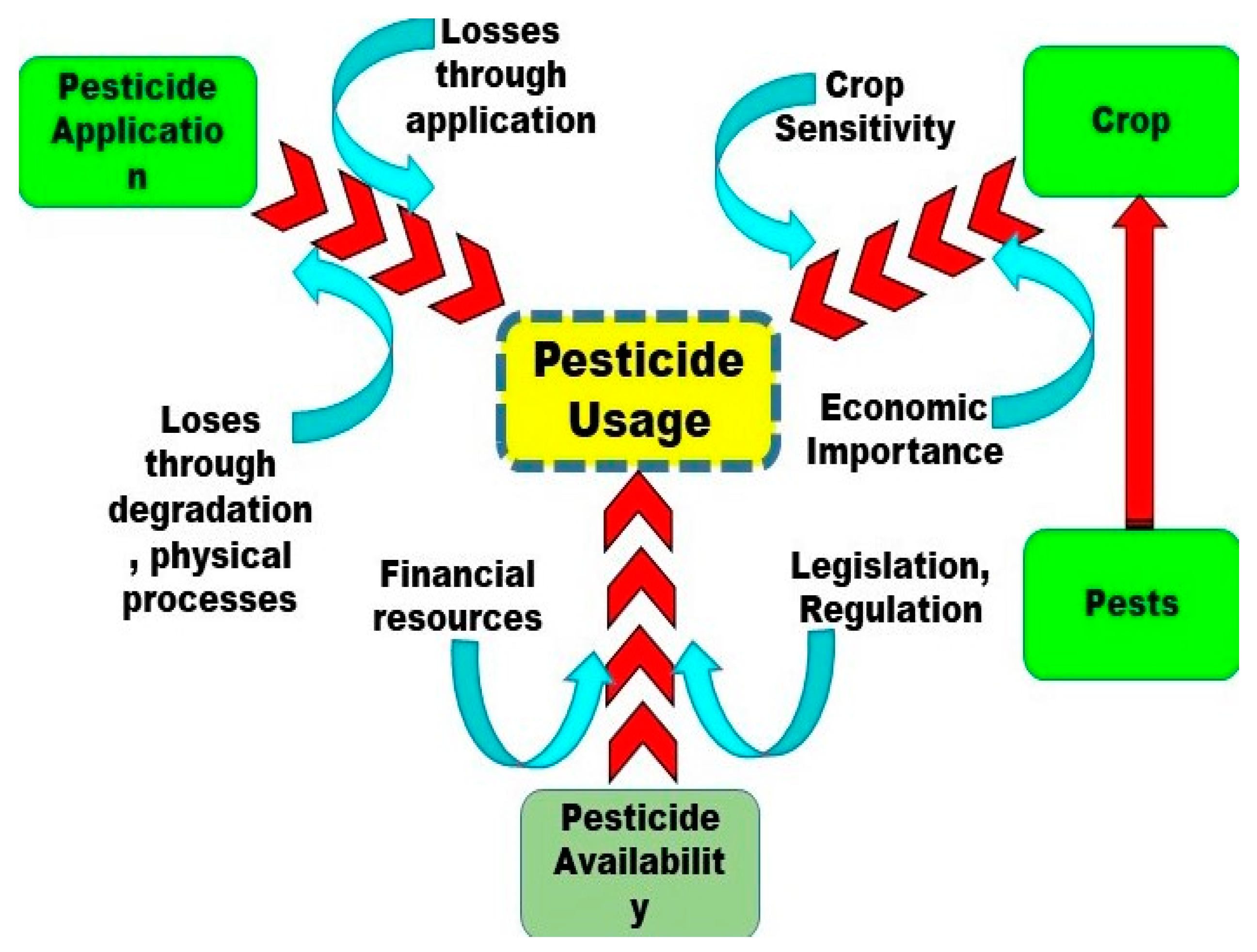
5. The Role of Pesticides
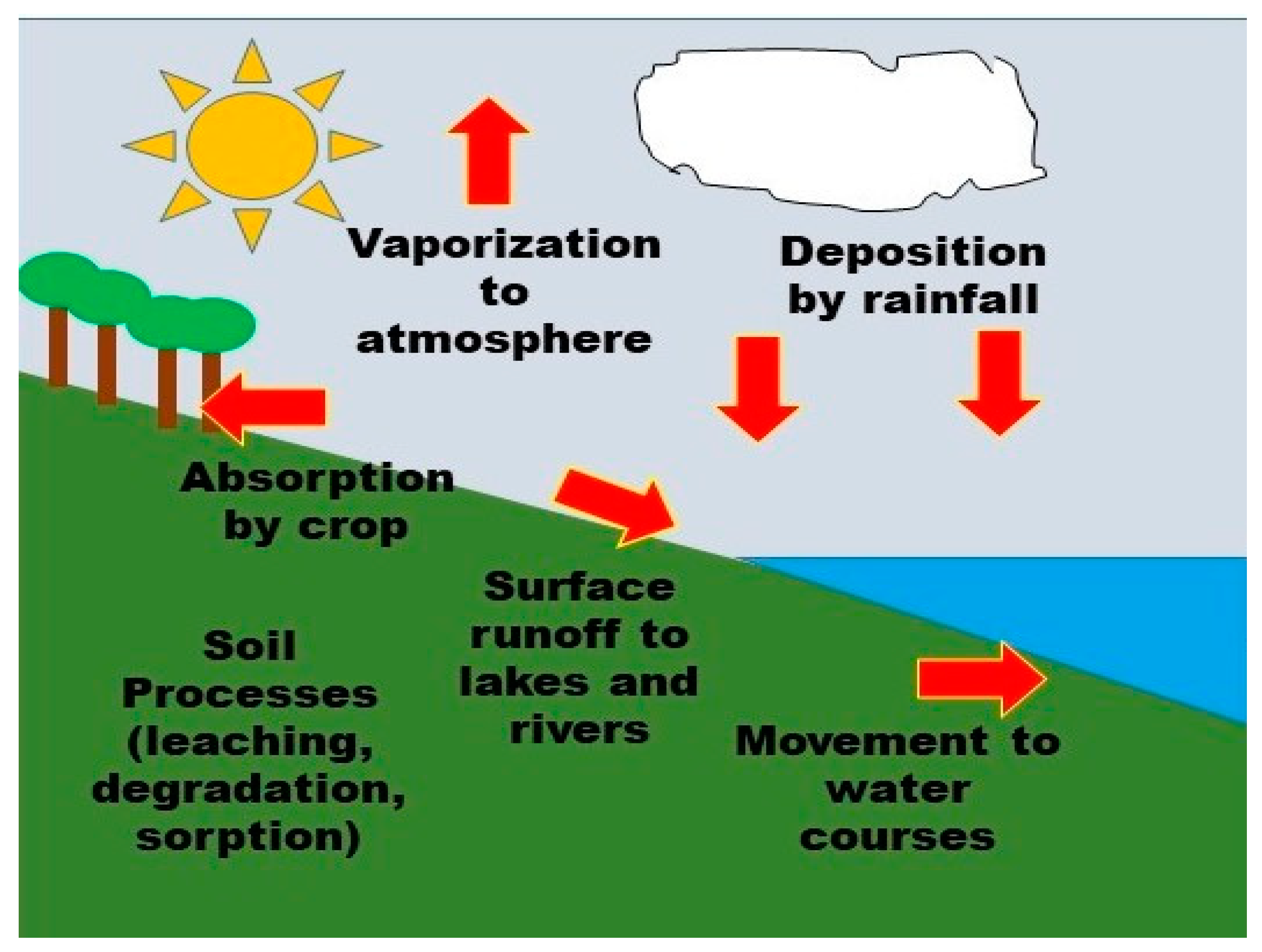
6. Pesticide Behavior in the Environment
6.1. Pesticide Degradation
6.2. Pesticide Migration
6.2.1. Sorption
6.2.2. Leaching
6.2.3. Spray Drift
6.2.4. Volatilization
6.2.5. Surface Runoff
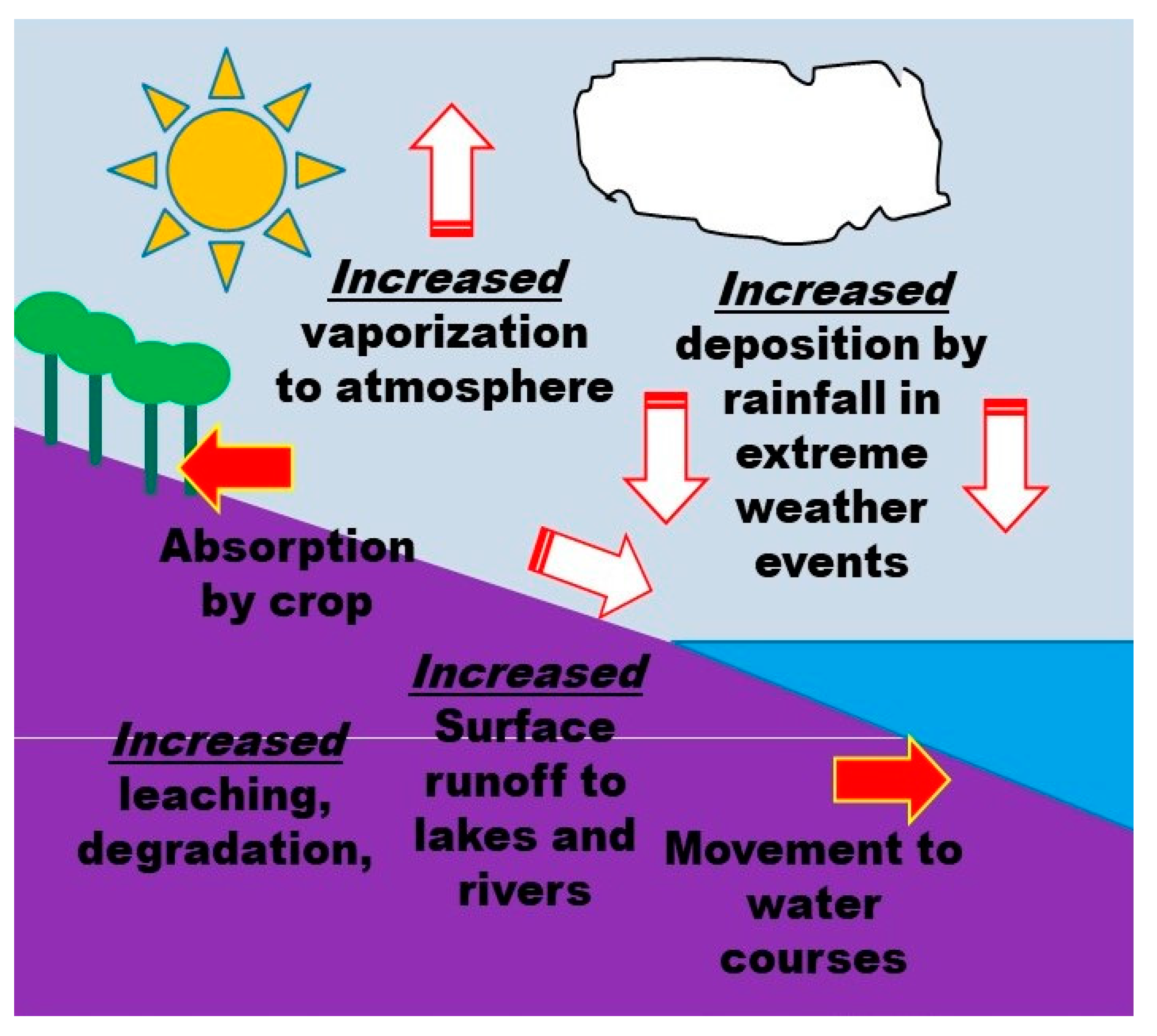
7. Impact of Climate Change-Related Factors on Pesticide Use
7.1. Influence on Soil Condition and Crop Growth
7.2. Influence on Pests, Weeds, and Diseases
7.2.1. Pests in Crops
7.2.2. Determinants of Diseases in Crops
7.2.3. Weed Growth
7.3. Influence on Pesticide Behavior in the Environment
7.3.1. Volatilization
7.3.2. Runoff and Drift
7.3.3. Leaching
7.3.4. Degradation
8. Pesticide Contamination and Its Adverse Effects on the Natural Environment
8.1. Impact on Water
8.2. Impact on Soil
8.3. Impact on Air
8.4. Impact on Food Safety
8.5. Impact on Non-Target Organisms
9. Conclusions and New Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Bernardes, M.F.F.; Pazin, M.; Pereira, L.C.; Dorta, D.J. Impact of Pesticides on Environmental and Human Health. In Toxicology Studies—Cells, Drugs and Environment; IntechOpen: London, UK, 2015; pp. 195–233. [Google Scholar]
- Aktar, W.; Paramasivam, M.; Sengupta, D.; Purkait, S.; Ganguly, M.; Banerjee, S. Impact assessment of pesticide residues in fish of Ganga river around Kolkata in West Bengal. Environ. Monit. Assess. 2008, 157, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Fenik, J.; Tankiewicz, M.; Biziuk, M. Properties and determination of pesticides in fruits and vegetables. TrAC Trends Anal. Chem. 2011, 30, 814–826. [Google Scholar] [CrossRef]
- Strassemeyer, J.; Daehmlow, D.; Dominic, A.; Lorenz, S.; Golla, B. SYNOPS-WEB, an online tool for environmental risk assessment to evaluate pesticide strategies on field level. Crop. Prot. 2017, 97, 28–44. [Google Scholar] [CrossRef]
- Carvalho, F.P. Pesticides, environment, and food safety. Food Energy Secur. 2017, 6, 48–60. [Google Scholar] [CrossRef]
- Hayes, T.B.; Hansen, M.; Kapuscinski, A.R.; Locke, K.A.; Barnosky, A. From silent spring to silent night: Agrochemicals and the anthropocene. Elem Sci Anth 2017, 5, 1–24. [Google Scholar] [CrossRef]
- Hernández, A.F.; Gil, F.; Lacasaña, M.; Rodríguez-Barranco, M.; Tsatsakis, A.M.; Requena, M.; Alarcón, R. Pesticide exposure and genetic variation in xenobiotic-metabolizing enzymes interact to induce biochemical liver damage. Food Chem. Toxicol. 2013, 61, 144–151. [Google Scholar] [CrossRef]
- Unsworth, J. History of Pesticide Use. IUPAC-International Union of Pure and Applied Chemistry, Mai. 2010. Available online: http://agrochemicals.iupac.org/index.php?option=com_sobi2&sobi2Task=sobi2Details&catid=3&sobi2Id=31 (accessed on 10 May 2010).
- Kislev, M.E.; Weiss, E.; Hartmann, A. Impetus for sowing and the beginning of agriculture: Ground collecting of wild cereals. Proc. Natl. Acad. Sci. USA 2004, 101, 2692–2695. [Google Scholar] [CrossRef]
- Davies, O. The Origins of Agriculture in West Africa. Curr. Anthr. 1968, 9, 479–482. [Google Scholar] [CrossRef]
- Lu, T.L.D. The Transition from Foraging to Farming and the Origin of Agriculture in China, British Archaeological Reports Limited. 1999. Available online: https://www.google.com.au/books/edition/The_Transition_from_Foraging_to_Farming/_tkLAAAAYAAJ?hl=en (accessed on 22 August 2007).
- Wilkes, H.G. Mexico and Central America as a Centre for the Origin of Agriculture and the Evolution of Maize. Crop Improvement 1979, 6, 1–18. [Google Scholar]
- Zhang, W.; Jiang, F.; Ou, J. Global pesticide consumption and pollution: With China as a focus. Proc. Int. Acad. Ecol. Environ. Sci. 2011, 1, 125. [Google Scholar]
- Council, N.R. The Future Role of Pesticides in US Agriculture; National Academies Press: Cambridge, MA, USA, 2000. [Google Scholar]
- Sheail, J. The regulation of pesticides use: An historical perspective. In Innovation and Environmental Risks; Belhaven Press: London, UK, 1991; pp. 38–46. [Google Scholar]
- Zhang, Q.; Xia, Z.; Wu, M.; Wang, L.; Yang, H. Human health risk assessment of DDTs and HCHs through dietary exposure in Nanjing, China. Chemosphere 2017, 177, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Ross, G. Risks and benefits of DDT. Lancet 2005, 366, 1771–1772. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, B.-Z.; Li, S.-M.; Zeng, E.Y. Regional dynamics of persistent organic pollutants (POPs) in the Pearl River Delta, China: Implications and perspectives. Environ. Pollut. 2011, 159, 2301–2309. [Google Scholar] [CrossRef]
- Barnhoorn, I.E.J.; Bornman, M.; Van Rensburg, C.J.; Bouwman, H. DDT residues in water, sediment, domestic and indigenous biota from a currently DDT-sprayed area. Chemosphere 2009, 77, 1236–1241. [Google Scholar] [CrossRef]
- Garcia, F.P.; Ascencio, S.Y.C.; Oyarzun, J.C.G.; Hernandez, A.C.; Alavarado, P.V. Pesticides: Classification, uses and toxicity. Measures of exposure and genotoxic risks. Int. J. Environ. Sci. Toxic. Res. 2012, 1, 279–293. [Google Scholar]
- Amaral, A.F.S. Pesticides and Asthma: Challenges for Epidemiology. Front. Public Health 2014, 2, 6. [Google Scholar] [CrossRef]
- Mnif, W.; Hassine, A.I.H.; Bouaziz, A.; Bartegi, A.; Thomas, O.; Roig, B. Effect of Endocrine Disruptor Pesticides: A Review. Int. J. Environ. Res. Public Health 2011, 8, 2265–2303. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Kabir, E.; Jahan, S.A. Exposure to pesticides and the associated human health effects. Sci. Total Environ. 2017, 575, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Sun, Y.; Hu, R.; Huang, J.; Huang, X.; Li, Y.; Yin, Y.; Chen, Z. A comparison of the effects of agricultural pesticide uses on peripheral nerve conduction in China. Sci. Rep. 2018, 8, 9621. [Google Scholar] [CrossRef] [PubMed]
- Aktar, W.; Sengupta, D.; Chowdhury, A. Impact of pesticides use in agriculture: Their benefits and hazards. Interdiscip. Toxicol. 2009, 2, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lawler, S.P. Environmental safety review of methoprene and bacterially-derived pesticides commonly used for sustained mosquito control. Ecotoxicol. Environ. Saf. 2017, 139, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Lamichhane, J.R. Pesticide use and risk reduction in European farming systems with IPM: An introduction to the special issue. Crop. Prot. 2017, 97, 1–6. [Google Scholar] [CrossRef]
- Cooper, J.; Dobson, H. The benefits of pesticides to mankind and the environment. Crop. Prot. 2007, 26, 1337–1348. [Google Scholar] [CrossRef]
- Ridsdill-Smith, T.; Pavri, C. Single Spring Spray Protects Pastures. 2000. Available online: https://www.agric.wa.gov.au/pastures/spray-topping-declared-plants (accessed on 3 August 2017).
- Miller, S.F. The effects of weed control technological change on rural communities. Outlook Agric. 1982, 11, 172–178. [Google Scholar] [CrossRef]
- Singh, D.K. Pesticides and Environment. Pestic. Chem. Toxicol. 2012, 1, 114–122. [Google Scholar]
- Scholtz, M.; Bidleman, T.F. Modelling of the long-term fate of pesticide residues in agricultural soils and their surface exchange with the atmosphere: Part II. Projected long-term fate of pesticide residues. Sci. Total Environ. 2007, 377, 61–80. [Google Scholar] [CrossRef]
- Liu, Y.; Mo, R.; Tang, F.; Fu, Y.; Guo, Y. Influence of different formulations on chlorpyrifos behavior and risk assessment in bamboo forest of China. Environ. Sci. Pollut. Res. 2015, 22, 20245–20254. [Google Scholar] [CrossRef]
- Marie, L.; Payraudeau, S.; Benoit, G.; Maurice, M.; Gwenaël, I. Degradation and Transport of the Chiral Herbicide S-Metolachlor at the Catchment Scale: Combining Observation Scales and Analytical Approaches. Environ. Sci. Technol. 2017, 51, 13231–13240. [Google Scholar] [CrossRef]
- Robinson, D.E.; Mansingh, A.; Dasgupta, T.P. Fate and transport of ethoprophos in the Jamaican environment. Sci. Total Environ. 1999, 238, 373–378. [Google Scholar] [CrossRef]
- Damalas, C.A.; Eleftherohorinos, I. Pesticide Exposure, Safety Issues, and Risk Assessment Indicators. Int. J. Environ. Res. Public Health 2011, 8, 1402–1419. [Google Scholar] [CrossRef] [PubMed]
- Abian, J.; Durand, G.; Barceló, J. Analysis of chlorotriazines and their degradation products in environmental samples by selecting various operating modes in thermospray HPLC/MS/MS. J. Agric. Food Chem. 1993, 41, 1264–1273. [Google Scholar] [CrossRef]
- Wu, L.P.; Chládková, B.; Lechtenfeld, O.J.; Lian, S.; Schindelka, J.; Herrmann, H.; Richnow, H.H. Characterizing chemical transformation of organophosphorus compounds by 13C and 2H stable isotope analysis. Sci. Total Environ. 2018, 615, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Tcaciuc, A.P.; Borrelli, R.; Zaninetta, L.M.; Gschwend, P.M.; Tcaciuc, P. Passive sampling of DDT, DDE and DDD in sediments: Accounting for degradation processes with reaction–diffusion modeling. Environ. Sci. Process. Impacts 2018, 20, 220–231. [Google Scholar] [CrossRef]
- Tariq, S.R.; Nisar, L. Reductive transformation of profenofos with nanoscale Fe/Ni particles. Environ. Monit. Assess. 2018, 190, 123. [Google Scholar] [CrossRef]
- Zhao, Y.; Wendling, L.A.; Wang, C.; Pei, Y. Behavior of chlorpyrifos and its major metabolite TCP (3,5,6-trichloro-2-pyridinol) in agricultural soils amended with drinking water treatment residuals. J. Soils Sediments 2016, 17, 889–900. [Google Scholar] [CrossRef]
- Yue, L.; Ge, C.; Feng, D.; Yu, H.; Deng, H.; Fu, B. Adsorption–desorption behavior of atrazine on agricultural soils in China. J. Environ. Sci. 2017, 57, 180–189. [Google Scholar] [CrossRef]
- Luo, C.; Huang, Y.; Huang, D.; Liu, M.; Xiong, W.; Guo, Q.; Yang, T. Migration and Transformation Characteristics of Niclosamide in a Soil–Plant System. ACS Omega 2018, 3, 2312–2321. [Google Scholar] [CrossRef]
- Su, W.; Hao, H.; Xu, H.; Lu, C.; Wu, R.; Xue, F. Degradation of Mesotrione Affected by Environmental Conditions. Bull. Environ. Contam. Toxicol. 2016, 98, 212–217. [Google Scholar] [CrossRef]
- Han, D.M.; Tong, X.X.; Jin, M.G.; Hepburn, E.; Tong, C.S.; Song, X.F. Evaluation of organic contamination in urban groundwater surrounding a municipal landfill, Zhoukou, China. Environ. Monit. Assess. 2012, 185, 3413–3444. [Google Scholar] [CrossRef]
- Qian, S.; Zhu, H.; Xiong, B.; Zheng, G.; Zhang, J.; Xu, W. Adsorption and desorption characteristics of endosulfan in two typical agricultural soils in Southwest China. Environ. Sci. Pollut. Res. 2017, 24, 11493–11503. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Li, J.; Fang, D.; Yu, Y.; Zhi, J. A disposable biofilm-modified amperometric biosensor for the sensitive determination of pesticide biotoxicity in water. RSC Adv. 2014, 4, 55473–55482. [Google Scholar] [CrossRef]
- Bansal, O.P. Fate of pesticides in the environment. J. Indian Chem. Soc. 2011, 88, 1525–1532. [Google Scholar]
- Quan, G.; Yin, C.; Chen, T.; Yan, J. Degradation of Herbicide Mesotrione in Three Soils with Differing Physicochemical Properties from China. J. Environ. Qual. 2015, 44, 1631–1637. [Google Scholar] [CrossRef]
- Wei, J.; Chen, Y.; Tiemur, A.; Wang, J.-D.; Wu, B. Degradation of pesticide residues by gaseous chlorine dioxide on table grapes. Postharvest Biol. Technol. 2018, 137, 142–148. [Google Scholar] [CrossRef]
- Qin, F.; Gao, Y.X.; Guo, B.Y.; Xu, P.; Li, J.Z.; Wang, H. Environmental behavior of benalaxyl and furalaxyl enantiomers in agricultural soils. J. Environ. Sci. Health Part B 2014, 49, 738–746. [Google Scholar] [CrossRef]
- Xue, N.; Yang, R.; Xu, X.; Seip, H.M.; Zang, Q.; Zeng, Q. Adsorption and Degradation of Benfuracarb in Three Soils in Hunan, People’s Republic of China. Bull. Environ. Contam. Toxicol. 2006, 76, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xu, Z.; Wu, X.; Gui, W.; Zhu, G. Adsorption and desorption behavior of herbicide diuron on various Chinese cultivated soils. J. Hazard. Mater. 2010, 178, 462–468. [Google Scholar] [CrossRef]
- Alvarez, D.O.; Mendes, K.F.; Tosi, M.; De Souza, L.F.; Cedano, J.C.C.; Falcão, N.P.D.S.; Dunfield, K.; Tsai, S.M.; Tornisielo, V.L. Sorption-desorption and biodegradation of sulfometuron-methyl and its effects on the bacterial communities in Amazonian soils amended with aged biochar. Ecotoxicol. Environ. Saf. 2021, 207, 111222. [Google Scholar] [CrossRef]
- Bošković, N.; Brandstätter-Scherr, K.; Sedláček, P.; Bílková, Z.; Bielská, L.; Hofman, J. Adsorption of epoxiconazole and tebuconazole in twenty different agricultural soils in relation to their properties. Chemosphere 2020, 261, 127637. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, J.H.; Guo, Q.N.; Ma, L.Y.; Yang, H. Physiochemical assessment of environmental behaviors of herbicide atrazine in soils associated with its degradation and bioavailability to weeds. Chemosphere 2021, 262, 127830. [Google Scholar] [CrossRef] [PubMed]
- Dong, D.M.; Lan, X.H.; Guo, Z.Y.; HUA, X.Y. Sorption Kinetics of Organochlorine Pesticides on Three Types of Solids in Natural Waters. Chem. J. Chin. Univ.-Chin. 2013, 34, 1180–1186. [Google Scholar]
- Ren, W.; Wang, M.; Zhou, Q. Effect of soil pH and organic matter on desorption hysteresis of chlorimuron-ethyl in two typical Chinese soils. J. Soils Sediments 2011, 11, 552–561. [Google Scholar] [CrossRef]
- Si, Y.; Zhang, J.; Wang, S.; Zhang, L.; Zhou, D. Influence of organic amendment on the adsorption and leaching of ethametsulfuron-methyl in acidic soils in China. Geoderma 2006, 130, 66–76. [Google Scholar] [CrossRef]
- Yuan, G.; Qin, J.-X.; Li, J.; Lang, X.-X.; Wang, G.H. Persistent organic pollutants in soil near the Changwengluozha glacier of the Central Tibetan Plateau, China: Their sorption to clays and implication. Sci. Total Environ. 2014, 472, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Yun, Y.; Jiang, L.; Wu, C. Influence of dissolved organic matter on sorption and desorption of MCPA in ferralsol. Sci. Total Environ. 2018, 616, 1449–1456. [Google Scholar] [CrossRef]
- Wu, P.; Wu, W.Z.; Han, Z.H.; Yang, H. Desorption and mobilization of three strobilurin fungicides in three types of soil. Environ. Monit. Assess. 2016, 188, 363. [Google Scholar] [CrossRef]
- Duan, L.; Wang, W.; Sun, Y. Ammonium Nitrogen Adsorption-Desorption Characteristics and Its Hysteresis of Typical Soils from Guanzhong Basin, China. Asian J. Chem. 2013, 25, 3850–3854. [Google Scholar] [CrossRef]
- Gao, C.; Yang, S.; Wang, W.; Gao, L. Influence of Humic Acid Colloid on Adsorption of DDT in the Riverbed Sediments. Asian J. Chem. 2014, 26, 2637–2642. [Google Scholar] [CrossRef]
- Gao, F.; Jia, J.; Wang, X. Occurrence and Ordination of Dichlorodiphenyltrichloroethane and Hexachlorocyclohexane in Agricultural Soils from Guangzhou, China. Arch. Environ. Contam. Toxicol. 2008, 54, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Zhang, N.; Wang, Y.; Zhang, C.D.; Zhu, L.Y.; Chen, W. Release of hexachlorocyclohexanes from historically and freshly contaminated soils in China: Implications for fate and regulation. Environ. Pollut. 2008, 156, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Al-Wabel, M.I.; El-Saeid, M.H.; El-Naggar, A.H.; Alromian, F.; Osman, K.; Elnazi, K.; Sallam, A.S. Spatial distribution of pesticide residues in the groundwater of a condensed agricultural area. Arab. J. Geosci. 2016, 9, 1–10. [Google Scholar] [CrossRef]
- Yadav, I.C.; Devi, N.L.; Syed, J.H.; Cheng, Z.; Li, J.; Zhang, G.; Jones, K.C. Current status of persistent organic pesticides residues in air, water, and soil, and their possible effect on neighboring countries: A comprehensive review of India. Sci. Total Environ. 2015, 511, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Lozowicka, B.; Abzeitova, E.; Sagitov, A.; Kaczyński, P.; Toleubayev, K.; Li, A. Studies of pesticide residues in tomatoes and cucumbers from Kazakhstan and the associated health risks. Environ. Monit. Assess. 2015, 187, 609. [Google Scholar] [CrossRef]
- Ðurišić-Mladenović, N.; Škrbić, B.; Cvejanov, J. Organochlorine Pesticides in Soil and Sediment from an Urban Zone of Novi Sad, Serbia. In Survival and Sustainability: Environmental Concerns in the 21st Century; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1469–1481. [Google Scholar]
- Fontana, A.; Lana, N.B.; Martínez, L.D.; Altamirano, J.C. Ultrasound-assisted leaching-dispersive solid-phase extraction followed by liquid–liquid microextraction for the determination of polybrominated diphenyl ethers in sediment samples by gas chromatography–tandem mass spectrometry. Talanta 2010, 82, 359–366. [Google Scholar] [CrossRef]
- Connell, D.W. Basic Concepts of Environmental Chemistry; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Geng, Y.; Ma, J.; Zhou, R.; Jia, R.; Li, C. Assessment of insecticide risk to human health in groundwater in Northern China by using the China-PEARL model. Pest Manag. Sci. 2017, 73, 2063–2070. [Google Scholar] [CrossRef]
- Labite, H.; Holden, N.M.; Richards, K.G.; Kramers, G.; Premrov, A.; Coxon, C.; Cummins, E. Comparison of pesticide leaching potential to groundwater under EU FOCUS and site specific conditions. Sci. Total Environ. 2013, 463, 432–441. [Google Scholar] [CrossRef]
- Sijm, D.; Rikken, M.; Rorije, E.; Traas, T.; McLachlan, M.; Peijnenburg, W. Transport, Accumulation and Transformation Processes; Springer: Berlin/Heidelberg, Germany, 2007; pp. 73–158. [Google Scholar]
- Ou, J.; Li, H.; Ou, X.; Yang, Z.; Chen, M.; Liu, K.; Teng, Y.; Xing, B. Degradation, adsorption and leaching of phenazine-1-carboxamide in agricultural soils. Ecotoxicol. Environ. Saf. 2020, 205, 111374. [Google Scholar] [CrossRef]
- Grella, M.; Miranda-Fuentes, A.; Marucco, P.; Balsari, P.; Gioelli, F. Development of Drift-Reducing Spouts For Vineyard Pneumatic Sprayers: Measurement of Droplet Size Spectra Generated and Their Classification. Appl. Sci. 2020, 10, 7826. [Google Scholar] [CrossRef]
- Pourreza, A.; Moghimi, A.; Niederholzer, F.J.A.; Larbi, P.A.; Zuniga-Ramirez, G.; Cheung, K.H.; Khorsandi, F. Spray Backstop: A Method to Reduce Orchard Spray Drift Potential without Limiting the Spray and Air Delivery. Sustainability 2020, 12, 8862. [Google Scholar] [CrossRef]
- Wang, G.; Han, Y.; Li, X.; Andaloro, J.; Chen, P.; Hoffmann, W.C.; Han, X.; Chen, S.; Lan, Y. Field evaluation of spray drift and environmental impact using an agricultural unmanned aerial vehicle (UAV) sprayer. Sci. Total Environ. 2020, 737, 139793. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Qi, L.; Wu, Y.; Musiu, E.M.; Cheng, Z.; Wang, P. Numerical simulation of airflow field from a six–rotor plant protection drone using lattice Boltzmann method. Biosyst. Eng. 2020, 197, 336–351. [Google Scholar] [CrossRef]
- Van Steenwyk, R.A.; Siegel, J.P.; Bisabri, B.; Cabuslay, C.S.; Choi, J.M.; Steggall, J.; Mace, K.C.; Blecker, S.W.; Poe, P.A.; Peters-Collaer, S.R.; et al. Spray drift mitigation using opposing synchronized air-blast sprayers. Pest Manag. Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Grella, M.; Marucco, P.; Balafoutis, A.; Balsari, P. Spray Drift Generated in Vineyard during Under-Row Weed Control and Suckering: Evaluation of Direct and Indirect Drift-Reducing Techniques. Sustainability 2020, 12, 5068. [Google Scholar] [CrossRef]
- Vieira, B.C.; Butts, T.R.; Rodrigues, A.O.; Schleier, J.J.; Fritz, B.K.; Kruger, G. Particle drift potential of glyphosate plus 2,4-D choline pre-mixture formulation in a low-speed wind tunnel. Weed Technol. 2020, 34, 520–527. [Google Scholar] [CrossRef]
- Ghaste, M.; Hayden, N.C.; Osterholt, M.J.; Young, J.; Young, B.; Widhalm, J.R. Evaluation of a Stable Isotope-Based Direct Quantification Method for Dicamba Analysis from Air and Water Using Single-Quadrupole LC–MS. Molecules 2020, 25, 3649. [Google Scholar] [CrossRef]
- Farhan, M.; Wajid, A.; Hussain, T.; Jabeen, F.; Ishaque, U.; Iftikhar, M.; Daim, M.A.; Noureen, A. Investigation of oxidative stress enzymes and histological alterations in tilapia exposed to chlorpyrifos. Environ. Sci. Pollut. Res. 2020, 1–7. [Google Scholar] [CrossRef]
- Zhu, S.; Niu, L.; Aamir, M.; Zhou, Y.; Xu, C.; Liu, W. Spatial and seasonal variations in air-soil exchange, enantiomeric signatures and associated health risks of hexachlorocyclohexanes (HCHs) in a megacity Hangzhou in the Yangtze River Delta region, China. Sci. Total Environ. 2017, 599, 264–272. [Google Scholar] [CrossRef]
- Alamdar, A.; Syed, J.H.; Malik, R.N.; Katsoyiannis, A.; Liu, J.; Li, J.; Zhang, G.; Jones, K.C. Organochlorine pesticides in surface soils from obsolete pesticide dumping ground in Hyderabad City, Pakistan: Contamination levels and their potential for air–soil exchange. Sci. Total Environ. 2014, 470, 733–741. [Google Scholar] [CrossRef]
- Chakraborty, P.; Zhang, G.; Li, J.; Sivakumar, A.; Jones, K.C. Occurrence and sources of selected organochlorine pesticides in the soil of seven major Indian cities: Assessment of air–soil exchange. Environ. Pollut. 2015, 204, 74–80. [Google Scholar] [CrossRef]
- Wong, F.; Alegria, H.A.; Jantunen, L.M.; Bidleman, T.F.; Salvador-Figueroa, M.; Gold-Bouchot, G.; Ceja-Moreno, V.; Waliszewski, S.M.; Infanzon, R. Organochlorine pesticides in soils and air of southern Mexico: Chemical profiles and potential for soil emissions. Atmos. Environ. 2008, 42, 7737–7745. [Google Scholar] [CrossRef]
- Das, S.; Hageman, K.J.; Taylor, M.; Michelsen-Heath, S.; Stewart, I. Fate of the organophosphate insecticide, chlorpyrifos, in leaves, soil, and air following application. Chemosphere 2020, 243, 125194. [Google Scholar] [CrossRef]
- Bloomfield, J.P.; Williams, R.; Gooddy, D.; Cape, J.; Guha, P. Impacts of climate change on the fate and behaviour of pesticides in surface and groundwater—A UK perspective. Sci. Total Environ. 2006, 369, 163–177. [Google Scholar] [CrossRef]
- Sistla, S.A.; Moore, J.C.; Simpson, R.T.; Gough, L.; Shaver, G.R.; Schimel, J.P. Long-term warming restructures Arctic tundra without changing net soil carbon storage. Nat. Cell Biol. 2013, 497, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Delcour, I.; Spanoghe, P.; Uyttendaele, M. Literature review: Impact of climate change on pesticide use. Food Res. Int. 2015, 68, 7–15. [Google Scholar] [CrossRef]
- Lennon, J.J. Potential impacts of climate change on agriculture and food safety within the island of Ireland††This paper is one of a series of reviews on “Climate Change and Food Safety—An Island of Ireland perspective”. Trends Food Sci. Technol. 2015, 44, 1–10. [Google Scholar] [CrossRef]
- Marvin, H.J.; Kleter, G.A.; Van Der Fels-Klerx, H.; Noordam, M.Y.; Franz, E.; Willems, D.J.; Boxall, A. Proactive systems for early warning of potential impacts of natural disasters on food safety: Climate-change-induced extreme events as case in point. Food Control. 2013, 34, 444–456. [Google Scholar] [CrossRef]
- Abass, A.B.; Ndunguru, G.; Mamiro, P.; Alenkhe, B.; Mlingi, N.; Bekunda, M. Post-harvest food losses in a maize-based farming system of semi-arid savannah area of Tanzania. J. Stored Prod. Res. 2014, 57, 49–57. [Google Scholar] [CrossRef]
- Ahmad, W.; Noor, M.A.; Afzal, I.; Bakhtavar, M.A.; Nawaz, M.M.; Sun, X.; Zhou, B.; Ma, W.; Zhao, M. Improvement of Sorghum Crop through Exogenous Application of Natural Growth-Promoting Substances under a Changing Climate. Sustainability 2016, 8, 1330. [Google Scholar] [CrossRef]
- Ainsworth, E.A. Rice production in a changing climate: A meta-analysis of responses to elevated carbon dioxide and elevated ozone concentration. Glob. Chang. Biol. 2008, 14, 1642–1650. [Google Scholar] [CrossRef]
- Akinnuoye-Adelabu, D.B.; Modi, A.T.; Mabhaudhi, T. Potential of producing green mealies in summer and winter at two sites in KwaZulu-Natal, South Africa, considering rainfall, soil moisture and weeding. S. Afr. J. Plant Soil 2017, 34, 211–221. [Google Scholar] [CrossRef]
- Noyes, P.D.; McElwee, M.K.; Miller, H.D.; Clark, B.W.; Van Tiem, L.A.; Walcott, K.C.; Erwin, K.N.; Levin, E.D. The toxicology of climate change: Environmental contaminants in a warming world. Environ. Int. 2009, 35, 971–986. [Google Scholar] [CrossRef] [PubMed]
- Scherm, H. Climate change: Can we predict the impacts on plant pathology and pest management? Can. J. Plant Pathol. 2004, 26, 267–273. [Google Scholar] [CrossRef]
- Meynard, C.N.; Gay, P.-E.; Lecoq, M.; Foucart, A.; Piou, C.; Chapuis, M.-P. Climate-driven geographic distribution of the desert locust during recession periods: Subspecies’ niche differentiation and relative risks under scenarios of climate change. Glob. Chang. Biol. 2017, 23, 4739–4749. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, J.J.; Decker, K.L.; Skagen, S.K.; Van Riper, C. Spatial and temporal variation in climate change: A bird’s eye view. Clim. Chang. 2009, 97, 305–311. [Google Scholar] [CrossRef]
- Jones, L.M.; Koehler, A.K.; Trnka, M.; Balek, J.; Challinor, A.J.; Atkinson, H.J.; Urwin, P.E. Climate change is predicted to alter the current pest status of Globodera pallida and G. rostochiensis in the United Kingdom. Glob. Chang. Biol. 2017, 23, 4497–4507. [Google Scholar] [CrossRef]
- Lesk, C.; Coffel, E.; D’Amato, A.W.; Dodds, K.; Horton, R. Threats to North American forests from southern pine beetle with warming winters. Nat. Clim. Chang. 2017, 7, 713–717. [Google Scholar] [CrossRef]
- López-Blanco, E.; Lund, M.; Williams, M.; Tamstorf, M.P.; Westergaard-Nielsen, A.; Exbrayat, J.-F.; Hansen, B.U.; Christensen, T.R. Exchange of CO2 in Arctic tundra: Impacts of meteorological variations and biological disturbance. Biogeosciences 2017, 14, 4467–4483. [Google Scholar] [CrossRef]
- Olfert, O.; Weiss, R.; Elliott, R.; Soroka, J. Bioclimatic approach to assessing the potential impact of climate change on two flea beetle (Coleoptera: Chrysomelidae) species in Canada. Can. Èntomol. 2017, 149, 616–627. [Google Scholar] [CrossRef]
- Sultana, S.; Baumgartner, J.B.; Dominiak, B.C.; Royer, J.E.; Beaumont, L.J. Potential impacts of climate change on habitat suitability for the Queensland fruit fly. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Guo, H.; Wan, S.; Ge, F. Effect of elevated CO2 and O3 on phytohormone-mediated plant resistance to vector insects and insect-borne plant viruses. Sci. China Life Sci. 2017, 60, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Frew, A.; Allsopp, P.G.; Gherlenda, A.N.; Johnson, S.N. Increased root herbivory under elevated atmospheric carbon dioxide concentrations is reversed by silicon-based plant defences. J. Appl. Ecol. 2016, 54, 1310–1319. [Google Scholar] [CrossRef]
- Patterson, D.T.; Westbrook, J.K.; Joyce†, R.; Lingren, P.D.; Rogasik, J. Weeds, Insects, and Diseases. Clim. Chang. 1999, 43, 711–727. [Google Scholar] [CrossRef]
- Rosenzweig, C.; Iglesias, A.; Yang, X.; Epstein, P.R.; Chivian, E. Climate Change and Extreme Weather Events; Implications for Food Production, Plant Diseases, and Pests. Glob. Chang. Hum. Health 2001, 2, 90–104. [Google Scholar] [CrossRef]
- Caubel, J.; Launay, M.; Ripoche, D.; Gouache, D.; Buis, S.; Huard, F.; Huber, L.; Brun, F.; Bancal, M.O. Climate change effects on leaf rust of wheat: Implementing a coupled crop-disease model in a French regional application. Eur. J. Agron. 2017, 90, 53–66. [Google Scholar] [CrossRef]
- Garrett, K.A.; Andersen, K.F.; Asche, F.; Bowden, R.L.; Forbes, G.A.; Kulakow, P.A.; Zhou, B. Resistance Genes in Global Crop Breeding Networks. Phytopathology 2017, 107, 1268–1278. [Google Scholar] [CrossRef]
- Runion, G.B. Climate change and plant pathosystems—future disease prevention starts here. New Phytol. 2003, 159, 531–533. [Google Scholar] [CrossRef]
- Rydzanicz, K.; Kiewra, D.; Lonc, E. Changes in range of mosquito-borne diseases affected by global climatic fluctuations. Wiad. Parazytol. 2006, 52, 73–83. [Google Scholar]
- Savary, S.; Djurle, A.; Yuen, J.; Ficke, A.; Rossi, V.; Esker, P.D.; Fernandes, J.M.C.; Del Ponte, E.M.; Kumar, J.; Madden, L.V.; et al. A White Paper on Global Wheat Health Based on Scenario Development and Analysis. Phytopathology 2017, 107, 1109–1122. [Google Scholar] [CrossRef]
- Singhvi, N.; Kushwaha, R.; Mathur, S.; Suranarayana, N. Role of weather factors on disease and pest incidence on tasar food plants and silkworms and developing operational disease and pest forewarning system for tasar sericulture. J. Exp. Zool. 2007, 10, 137–143. [Google Scholar]
- Tubby, K.V.; Webber, J.F. Pests and diseases threatening urban trees under a changing climate. Forestry 2010, 83, 451–459. [Google Scholar] [CrossRef]
- Viswanathan, R. Sustainable ecofriendly disease management systems in sugarcane production under the changing climate. J. Mycol. Plant Pathol. 2013, 43, 12–27. [Google Scholar]
- Wei, J.; Hansen, A.; Zhang, Y.; Li, H.; Liu, Q.; Sun, Y.; Xue, S.; Zhao, S.; Bi, P. The Impact of Climate Change on Infectious Disease Transmission: Perceptions of CDC Health Professionals in Shanxi Province, China. PLoS ONE 2014, 9, e109476. [Google Scholar] [CrossRef]
- Yacoub, S.; Kotit, S.; Yacoub, M.H. Disease appearance and evolution against a background of climate change and reduced resources. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2011, 369, 1719–1729. [Google Scholar] [CrossRef]
- Yamamura, K.; Yokozawa, M. Prediction of a geographical shift in the prevalence of rice stripe virus disease transmitted by the small brown planthopper, Laodelphax striatellus(Fallen)(Hemiptera: Delphacidae), under global warming. Appl. Entomol. Zool. 2002, 37, 181–190. [Google Scholar] [CrossRef]
- Anderson, P.K.; Cunningham, A.A.; Patel, N.G.; Morales, F.J.; Epstein, P.R.; Daszak, P. Emerging infectious diseases of plants: Pathogen pollution, climate change and agrotechnology drivers. Trends Ecol. Evol. 2004, 19, 535–544. [Google Scholar] [CrossRef]
- Boland, G.; Melzer, M.; Hopkin, A.; Higgins, V.; Nassuth, A. Climate change and plant diseases in Ontario. Can. J. Plant Pathol. 2004, 26, 335–350. [Google Scholar] [CrossRef]
- Boyd, I.L.; Freer-Smith, P.H.; Gilligan, C.A.; Godfray, H.C.J. The Consequence of Tree Pests and Diseases for Ecosystem Services. Science 2013, 342, 1235773. [Google Scholar] [CrossRef]
- Branco, M.; Bragança, H.; Sousa, E.; Phillips, A.J. Pests and Diseases in Portuguese Forestry: Current and New Threats. World For. 2014, 117–154. [Google Scholar] [CrossRef]
- Carisse, O. Using biovigilance-based information for strategic and tactical disease management decisions. Can. J. Plant Pathol. 2017, 39, 91. [Google Scholar]
- Coakley, S. Projected effects of climate change on plant disease and how plant pathologists can prepare to meet the challenge. Can. J. Plant Pathol.-Rev. Can. Phytopathol. 2012, 34, 328–329. [Google Scholar]
- Colosio, C. Agriculture in Italy nowadays: Ancient risks and emerging diseases. G. Ital. Med. Lav. Ergon. 2013, 35, 293–296. [Google Scholar] [PubMed]
- Davies, S.; Patenaude, G.; Snowdon, P. A new approach to assessing the risk to woodland from pest and diseases. Forestry 2017, 90, 319–331. [Google Scholar] [CrossRef]
- Dereure, J.; Vanwambeke, S.O.; Malé, P.; Martínez, S.; Pratlong, F.; Balard, Y.; Dedet, J.-P. The Potential Effects of Global Warming on Changes in Canine Leishmaniasis in a Focus outside the Classical Area of the Disease in Southern France. Vector-Borne Zoonotic Dis. 2009, 9, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, J.P.; Van Gaest, A.L.; Strickland, S.A.; Arkoosh, M.R. The impact of temperature stress and pesticide exposure on mortality and disease susceptibility of endangered Pacific salmon. Chemosphere 2014, 108, 353–359. [Google Scholar] [CrossRef]
- Evans, N.; Butterworth, M.H.; Baierl, A.; Semenov, M.A.; West, J.S.; Barnes, A.; Moran, D.; Fitt, B.D.L. The impact of climate change on disease constraints on production of oilseed rape. Food Secur. 2010, 2, 143–156. [Google Scholar] [CrossRef]
- Garrett, K.; Dobson, A.; Kroschel, J.; Natarajan, B.; Orlandini, S.; Tonnang, H.; Valdivia, C. The effects of climate variability and the color of weather time series on agricultural diseases and pests, and on decisions for their management. Agric. For. Meteorol. 2013, 170, 216–227. [Google Scholar] [CrossRef]
- Yi, H.; Devkota, B.R.; Yu, J.-S.; Oh, K.-C.; Kim, J.; Kim, H.-J. Effects of global warming on mosquitoes & mosquito-borne diseases and the new strategies for mosquito control. Èntomol. Res. 2014, 44, 215–235. [Google Scholar] [CrossRef]
- Al-Askar, A.A.; Ghoneem, K.M.; Rashad, Y.M.; Abdulkhair, W.M.; Hafez, E.E.; Shabana, Y.M.; Baka, Z.A. Occurrence and distribution of tomato seed-borne mycoflora in S audi A rabia and its correlation with the climatic variables. Microb. Biotechnol. 2014, 7, 556–569. [Google Scholar] [CrossRef]
- Kim, Y.S.; Park, K.H.; Chun, H.S.; Choi, C.; Bahk, G.J. Correlations between climatic conditions and foodborne disease. Food Res. Int. 2015, 68, 24–30. [Google Scholar] [CrossRef]
- Ziska, L.H.; Goins, E.W. Elevated Atmospheric Carbon Dioxide and Weed Populations in Glyphosate Treated Soybean. Crop. Sci. 2006, 46, 1354–1359. [Google Scholar] [CrossRef]
- Ziska, L.H.; Blumenthal, D.M.; Runion, G.B.; Hunt, E.R.; Diaz-Soltero, H. Invasive species and climate change: An agronomic perspective. Clim. Chang. 2010, 105, 13–42. [Google Scholar] [CrossRef]
- Ziska, L.H.; Faulkner, S.; Lydon, J. Changes in biomass and root:shoot ratio of field-grown Canada thistle (Cirsium arvense), a noxious, invasive weed, with elevated CO2: Implications for control with glyphosate. Weed Sci. 2004, 52, 584–588. [Google Scholar] [CrossRef]
- Ramesh, K.; Matloob, A.; Aslam, F.; Florentine, S.; Chauhan, B.S. Weeds in a Changing Climate: Vulnerabilities, Consequences, and Implications for Future Weed Management. Front. Plant Sci. 2017, 8, 95. [Google Scholar] [CrossRef] [PubMed]
- Ziska, L.H.; Teasdale, J.R. Sustained growth and increased tolerance to glyphosate observed in a C3 perennial weed, quackgrass (Elytrigia repens), grown at elevated carbon dioxide. Funct. Plant Biol. 2000, 27, 159. [Google Scholar] [CrossRef]
- Manea, A.; Leishman, M.R.; Downey, P.O. Exotic C4Grasses Have Increased Tolerance to Glyphosate under Elevated Carbon Dioxide. Weed Sci. 2011, 59, 28–36. [Google Scholar] [CrossRef]
- Nowak, K.M.; Girardi, C.; Miltner, A.; Gehre, M.; Schäffer, A.; Kästner, M. Contribution of microorganisms to non-extractable residue formation during biodegradation of ibuprofen in soil. Sci. Total Environ. 2013, 445, 377–384. [Google Scholar] [CrossRef]
- Cavero, J.; Zaragoza, C.; Suso, M.L.; Pardo, A. Competition between maize and Datura stramonium in an irrigated field under semi-arid conditions. Weed Res. 1999, 39, 225–240. [Google Scholar] [CrossRef]
- Lee, J.-S. Combined effect of elevated CO2 and temperature on the growth and phenology of two annual C3 and C4 weedy species. Agric. Ecosyst. Environ. 2011, 140, 484–491. [Google Scholar] [CrossRef]
- Hanzlik, K.; Gerowitt, B. Occurrence and distribution of important weed species in German winter oilseed rape fields. J. Plant Dis. Prot. 2012, 119, 107–120. [Google Scholar] [CrossRef]
- Rodenburg, J.; Meinke, H.; Johnson, D.E. Challenges for weed management in African rice systems in a changing climate. J. Agric. Sci. 2011, 149, 427–435. [Google Scholar] [CrossRef]
- Noroozi, S.; Alizadeh, H.; Mashhadi, H.R. Temperature influences postdispersal predation of weed seeds. Weed Biol. Manag. 2016, 16, 24–33. [Google Scholar] [CrossRef]
- Rodenburg, J.; Riches, C.R.; Kayeke, J.M. Addressing current and future problems of parasitic weeds in rice. Crop. Prot. 2010, 29, 210–221. [Google Scholar] [CrossRef]
- Bailey, S.W. Climate change and decreasing herbicide persistence. Pest Manag. Sci. 2004, 60, 158–162. [Google Scholar] [CrossRef]
- Patterson, D.T. Weeds in a Changing Climate. Weed Sci. 1995, 43, 685–700. [Google Scholar] [CrossRef]
- Froud-Williams, R. Weeds and climate change: Implications for their ecology and control. Asp. Appl. Biol. 1996, 45, 187–196. [Google Scholar]
- Holmsgaard, P.N.; Dealtry, S.; Dunon, V.; Heuer, H.; Hansen, L.H.; Springael, D.; Smalla, K.; Riber, L.; Sørensen, S.J. Response of the bacterial community in an on-farm biopurification system, to which diverse pesticides are introduced over an agricultural season. Environ. Pollut. 2017, 229, 854–862. [Google Scholar] [CrossRef]
- Liu, L.; Tang, J.; Zhong, G.; Zhen, X.; Pan, X.; Tian, C. Spatial distribution and seasonal variation of four current-use pesticides (CUPs) in air and surface water of the Bohai Sea, China. Sci. Total Environ. 2018, 621, 516–523. [Google Scholar] [CrossRef]
- Wang, Q.; Li, C.; Chen, C.; Chen, J.; Zheng, R.; Que, X. Effectiveness of narrow grass hedges in reducing atrazine runoff under different slope gradient conditions. Environ. Sci. Pollut. Res. 2018, 25, 7672–7680. [Google Scholar] [CrossRef]
- Székács, A.; Mörtl, M.; Darvas, B. Monitoring Pesticide Residues in Surface and Ground Water in Hungary: Surveys in 1990–2015. J. Chem. 2015, 2015, 1–15. [Google Scholar] [CrossRef]
- Singh, M.; Pant, G.; Hossain, K.; Bhatia, A.K. Green remediation. Tool for safe and sustainable environment: A review. Appl. Water Sci. 2017, 7, 2629–2635. [Google Scholar] [CrossRef]
- Woodruff, L.G.; Cannon, W.F.; Eberl, D.D.; Smith, D.B.; Kilburn, J.E.; Horton, J.D.; Garrett, R.G.; Klassen, R.A. Continental-scale patterns in soil geochemistry and mineralogy: Results from two transects across the United States and Canada. Appl. Geochem. 2009, 24, 1369–1381. [Google Scholar] [CrossRef]
- Tirado, M.; Clarke, R.; Jaykus, L.; McQuatters-Gollop, A.; Frank, J. Climate change and food safety: A review. Food Res. Int. 2010, 43, 1745–1765. [Google Scholar] [CrossRef]
- Hailu, F. Farmers perception of pesticide use and genetic erosion of landraces of tetraploid wheat (Triticum spp.) in Ethiopia. Genet. Resour. Crop. Evol. 2016, 64, 979–994. [Google Scholar] [CrossRef]
- Miraglia, M.; Marvin, H.; Kleter, G.; Battilani, P.; Brera, C.; Coni, E.; Cubadda, F.; Croci, L.; De Santis, B.; Dekkers, S.; et al. Climate change and food safety: An emerging issue with special focus on Europe. Food Chem. Toxicol. 2009, 47, 1009–1021. [Google Scholar] [CrossRef]
- Elgueta, S.; Moyano, S.; Sepúlveda, P.; Quiroz, C.; Correa, A. Pesticide residues in leafy vegetables and human health risk assessment in North Central agricultural areas of Chile. Food Addit. Contam. Part B 2017, 10, 105–112. [Google Scholar] [CrossRef]
- Mingo, V.; Lötters, S.; Wagner, N. The impact of land use intensity and associated pesticide applications on fitness and enzymatic activity in reptiles—A field study. Sci. Total Environ. 2017, 590, 114–124. [Google Scholar] [CrossRef]
- Škrbic, B.; Cvejanov, J.; Durišić-Mladenović, N. Organochlorine pesticides and polychlorinated biphenyls in surface soils of Novi Sad and bank sediment of the Danube River. J. Environ. Sci. Health Part B 2007, 42, 311–319. [Google Scholar] [CrossRef]
- Kawahara, J.; Horikoshi, R.; Yamaguchi, T.; Kumagai, K.; Yanagisawa, Y. Air pollution and young children’s inhalation exposure to organophosphorus pesticide in an agricultural community in Japan. Environ. Int. 2005, 31, 1123–1132. [Google Scholar] [CrossRef]
- Tuncel, S.G.; Oztas, N.B.; Erduran, M.S. Air and groundwater pollution in an agricultural region of the Turkish Mediterranean coast. J. Air Waste Manag. Assoc. 2008, 58, 1240–1249. [Google Scholar] [CrossRef]
- Baker, L.W.; Fitzell, D.L.; Seiber, J.N.; Parker, T.R.; Shibamoto, T.; Poore, M.W.; Longley, K.E.; Tomlin, R.P.; Propper, R.; Duncan, D.W. Ambient Air Concentrations of Pesticides in California. Environ. Sci. Technol. 1996, 30, 1365–1368. [Google Scholar] [CrossRef]
- Gouin, T.; Shoeib, M.; Harner, T. Atmospheric concentrations of current-use pesticides across south-central Ontario using monthly-resolved passive air samplers. Atmos. Environ. 2008, 42, 8096–8104. [Google Scholar] [CrossRef]
- Fang, Y.; Nie, Z.; Die, Q.; Tian, Y.; Liu, F.; He, J.; Huang, Q. Organochlorine pesticides in soil, air, and vegetation at and around a contaminated site in southwestern China: Concentration, transmission, and risk evaluation. Chemosphere 2017, 178, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Ansara-Ross, T.; Wepener, V.; Brink, P.V.D.; Ross, M. Pesticides in South African fresh waters. Afr. J. Aquat. Sci. 2012, 37, 1–16. [Google Scholar] [CrossRef]
- Wang, D.; Singhasemanon, N.; Goh, K.S. A review of diazinon use, contamination in surface waters, and regulatory actions in California across water years 1992–2014. Environ. Monit. Assess. 2017, 189, 310. [Google Scholar] [CrossRef] [PubMed]
- Ben Salem, F.; Ben Said, O.; Aissa, P.; Mahmoudi, E.; Monperrus, M.; Grunberger, O.; Duran, R. Pesticides in Ichkeul Lake–Bizerta Lagoon Watershed in Tunisia: Use, occurrence, and effects on bacteria and free-living marine nematodes. Environ. Sci. Pollut. Res. 2016, 23, 36–48. [Google Scholar] [CrossRef]
- Adams, R.M.; McAdams, B.C.; Arnold, W.A.; Chin, Y.-P. Transformation of chlorpyrifos and chlorpyrifos-methyl in prairie pothole pore waters. Environ. Sci. Process. Impacts 2016, 18, 1406–1416. [Google Scholar] [CrossRef]
- Mazlan, N.; Ahmed, M.; Muharam, F.M.; Alam, M.A. Status of persistent organic pesticide residues in water and food and their effects on environment and farmers: A comprehensive review in Nigeria. Semin.-Cienc. Agrar. 2017, 38, 2221–2236. [Google Scholar] [CrossRef]
- Kurwadkar, S. Groundwater Pollution and Vulnerability Assessment. Water Environ. Res. 2017, 89, 1561–1577. [Google Scholar] [CrossRef]
- Lai, W.Y. Pesticide use and health outcomes: Evidence from agricultural water pollution in China. J. Environ. Econ. Manag. 2017, 86, 93–120. [Google Scholar] [CrossRef]
- Guo, H.; Wang, Y. Specific vulnerability assessment using the MLPI model in Datong city, Shanxi province, China. Environ. Earth Sci. 2004, 45, 401–407. [Google Scholar] [CrossRef]
- Anderson, B.S.; Phillips, B.M.; Voorhees, J.P.; Deng, X.; Geraci, J.; Worcester, K.; Tjeerdema, R.S. Changing patterns in water toxicity associated with current use pesticides in three California agriculture regions. Integr. Environ. Assess. Manag. 2018, 14, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Brauns, B.; Jakobsen, R.; Song, X.; Bjerg, P.L. Pesticide use in the wheat-maize double cropping systems of the North China Plain: Assessment, field study, and implications. Sci. Total Environ. 2018, 616, 1307–1316. [Google Scholar] [CrossRef] [PubMed]
- Clemow, Y.H.; Manning, G.E.; Breton, R.L.; Winchell, M.F.; Padilla, L.; Rodney, S.I.; Hanzas, J.P.; Estes, T.L.; Budreski, K.; Toth, B.N.; et al. A refined ecological risk assessment for California red-legged frog, Delta smelt, and California tiger salamander exposed to malathion. Integr. Environ. Assess. Manag. 2018, 14, 224–239. [Google Scholar] [CrossRef]
- Otalvaro, J.O.; Brigante, M. Interaction of pesticides with natural and synthetic solids. Evaluation in dynamic and equilibrium conditions. Environ. Sci. Pollut. Res. 2017, 25, 6707–6719. [Google Scholar] [CrossRef]
- Aravinna, P.; Priyantha, N.; Pitawala, A.; Yatigammana, S.K. Use pattern of pesticides and their predicted mobility into shallow groundwater and surface water bodies of paddy lands in Mahaweli river basin in Sri Lanka (vol 52, pg 37, 2016). J. Environ. Sci. Health Part B-Pestic. Food Contam. Agric. Wastes 2018, 53, 95. [Google Scholar]
- Rose, C.E.; Coupe, R.H.; Capel, P.D.; Webb, R.M. Holistic assessment of occurrence and fate of metolachlor within environmental compartments of agricultural watersheds. Sci. Total Environ. 2018, 612, 708–719. [Google Scholar] [CrossRef]
- Kole, R.; Banerjee, H.; Bhattacharyya, A. Monitoring of market fish samples for Endosulfan and Hexachlorocyclohexane residues in and around Calcutta. Bull. Environ. Contam. Toxicol. 2001, 67, 554–559. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, S.; Cui, W.; Meng, X.; Tang, X. Polycyclic aromatic hydrocarbons and organochlorine pesticides in surface water from the Yongding River basin, China: Seasonal distribution, source apportionment, and potential risk assessment. Sci. Total Environ. 2018, 618, 419–429. [Google Scholar] [CrossRef]
- Osten, J.R.-V.; Dzul-Caamal, R. Glyphosate Residues in Groundwater, Drinking Water and Urine of Subsistence Farmers from Intensive Agriculture Localities: A Survey in Hopelchén, Campeche, Mexico. Int. J. Environ. Res. Public Health 2017, 14, 595. [Google Scholar] [CrossRef]
- Donald, D.B.; Syrgiannis, J.; Hunter, F.; Weiss, G. Agricultural pesticides threaten the ecological integrity of northern prairie wetlands. Sci. Total Environ. 1999, 231, 173–181. [Google Scholar] [CrossRef]
- Shakeri, A.; Mehrabi, B. Potentially toxic elements and persistent organic pollutants in water and fish at Shahid Rajaei Dam, north of Iran. Int. J. Environ. Sci. Technol. 2015, 12, 2201–2212. [Google Scholar] [CrossRef]
- Barron, M.; Ashurova, Z.J.; Kukaniev, M.A.; Avloev, H.K.; Khaidarov, K.K.; Jamshedov, J.N.; Rahmatullova, O.S.; Atolikshoeva, S.S.; Mamadshova, S.S.; Manzenyuk, O. Residues of organochlorine pesticides in surface soil and raw foods from rural areas of the Republic of Tajikistan. Environ. Pollut. 2017, 224, 494–502. [Google Scholar] [CrossRef]
- Zhang, F.; He, J.; Yao, Y.; Hou, D.; Jiang, C.; Zhang, X.; Di, C.; Otgonbayar, K. Spatial and seasonal variations of pesticide contamination in agricultural soils and crops sample from an intensive horticulture area of Hohhot, North-West China. Environ. Monit. Assess. 2013, 185, 6893–6908. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Chang, H.; Ma, X. A modified method for pesticide transport and fate in subsurface environment of a winter wheat field of Yangling, China. Sci. Total Environ. 2017, 609, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Andreu, V.; Picó, Y. Determination of pesticides and their degradation products in soil: Critical review and comparison of methods. TrAC Trends Anal. Chem. 2004, 23, 772–789. [Google Scholar] [CrossRef]
- Langenbach, T.; Mano, D.; Campos, M.M.; Cunha, A.L.; De Campos, T.M. Pesticide dispersion by spraying under tropical conditions. J. Environ. Sci. Health Part B 2017, 52, 843–849. [Google Scholar] [CrossRef]
- Lee, S.; McLaughlin, R.; Harnly, M.; Gunier, R.B.; Kreutzer, R. Community exposures to airborne agricultural pesticides in California: Ranking of inhalation risks. Environ. Health Perspect. 2002, 110, 1175–1184. [Google Scholar] [CrossRef]
- de Jong, F.M.; de Snoo, G.R.; van de Zande, J.C. Estimated nationwide effects of pesticide spray drift on terrestrial habitats in the Netherlands. J. Environ. Manag. 2008, 86, 721–730. [Google Scholar] [CrossRef]
- Yera, A.M.B.; Nascimento, M.M.; da Rocha, G.O.; de andrade, J.B.; Vasconcellos, P.C. Occurrence of Pesticides Associated to Atmospheric Aerosols: Hazard and Cancer Risk Assessments. J. Braz. Chem. Soc. 2020, 31, 1317–1326. [Google Scholar] [CrossRef]
- Benka-Coker, W.; Hoskovec, L.; Severson, R.; Balmes, J.; Wilson, A.; Magzamen, S. The joint effect of ambient air pollution and agricultural pesticide exposures on lung function among children with asthma. Environ. Res. 2020, 190, 109903. [Google Scholar] [CrossRef]
- Doan, N.H.; Duong, H.T.; Trinh, H.T.; Tanaka, Y.; Kadokami, K. Comprehensive study of insecticides in atmospheric particulate matter in Hanoi, Vietnam: Occurrences and human risk assessment. Chemosphere 2021, 262, 128028. [Google Scholar] [CrossRef]
- Pan, D.; He, M.; Kong, F. Risk attitude, risk perception, and farmers’ pesticide application behavior in China: A moderation and mediation model. J. Clean. Prod. 2020, 276, 124241. [Google Scholar] [CrossRef]
- Sultana, J.; Syed, J.H.; Mahmood, A.; Ali, U.; Rehman, M.Y.A.; Malik, R.N.; Li, J.; Zhang, G. Investigation of organochlorine pesticides from the Indus Basin, Pakistan: Sources, air–soil exchange fluxes and risk assessment. Sci. Total Environ. 2014, 497, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Colquhoun, J.; Heider, D.J.; Rittmeyer, R.A. Seed Potato Growth and Yield as Affected by Mother Plant Exposure to Herbicides. Weed Technol. 2017, 31, 136–147. [Google Scholar] [CrossRef][Green Version]
- Hoai, P.M.; Sebesvari, Z.; Minh, T.B.; Viet, P.H.; Renaud, F.G. Pesticide pollution in agricultural areas of Northern Vietnam: Case study in Hoang Liet and Minh Dai communes. Environ. Pollut. 2011, 159, 3344–3350. [Google Scholar] [CrossRef] [PubMed]
- Fosu, P.O.; Donkor, A.K.; Ziwu, C.; Dubey, B.; Kingsford-Adaboh, R.; Asante, I.; Nyarko, S.; Tawiah, R.; Nazzah, N. Surveillance of pesticide residues in fruits and vegetables from Accra Metropolis markets, Ghana, 2010–2012: A case study in Sub-Saharan Africa. Environ. Sci. Pollut. Res. 2017, 24, 17187–17205. [Google Scholar] [CrossRef] [PubMed]
- Wanwimolruk, S.; Phopin, K.; Boonpangrak, S.; Prachayasittikul, V. Food safety in Thailand 4: Comparison of pesticide residues found in three commonly consumed vegetables purchased from local markets and supermarkets in Thailand. PeerJ 2016, 4, e2432. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tudi, M.; Daniel Ruan, H.; Wang, L.; Lyu, J.; Sadler, R.; Connell, D.; Chu, C.; Phung, D.T. Agriculture Development, Pesticide Application and Its Impact on the Environment. Int. J. Environ. Res. Public Health 2021, 18, 1112. https://doi.org/10.3390/ijerph18031112
Tudi M, Daniel Ruan H, Wang L, Lyu J, Sadler R, Connell D, Chu C, Phung DT. Agriculture Development, Pesticide Application and Its Impact on the Environment. International Journal of Environmental Research and Public Health. 2021; 18(3):1112. https://doi.org/10.3390/ijerph18031112
Chicago/Turabian StyleTudi, Muyesaier, Huada Daniel Ruan, Li Wang, Jia Lyu, Ross Sadler, Des Connell, Cordia Chu, and Dung Tri Phung. 2021. "Agriculture Development, Pesticide Application and Its Impact on the Environment" International Journal of Environmental Research and Public Health 18, no. 3: 1112. https://doi.org/10.3390/ijerph18031112
APA StyleTudi, M., Daniel Ruan, H., Wang, L., Lyu, J., Sadler, R., Connell, D., Chu, C., & Phung, D. T. (2021). Agriculture Development, Pesticide Application and Its Impact on the Environment. International Journal of Environmental Research and Public Health, 18(3), 1112. https://doi.org/10.3390/ijerph18031112



